A green route for preparation of low surface area SiO2 microspheres from wheat straw ash with activated carbon and NPK compound fertilizer as by-products†
Jinlong Cui,
Liying Cui,
Fupeng Cheng,
Lingjuan Liu,
Hongliang Sun,
Song Li,
Zhongsheng Wen and
Juncai Sun*
Institute of Materials and Technology, Dalian Maritime University, Dalian 116026, China. E-mail: sunjc@dlmu.edu.cn; Fax: +86-411-84725960; Tel: +86-411-84727959
First published on 16th September 2015
Abstract
In order to make full use of wheat straw ash which is abundant and causes some serious environmental problems in the north of China, an environmentally friendly route is proposed to convert the wheat straw ash to useful materials in this paper. In this route, low surface area SiO2 microspheres are obtained from wheat straw ash, with the activated carbon and Nitrogen–Phosphorus–Potassium (NPK) compound fertilizer as by-products. KOH is used as the silica extraction reagent and H3PO4 is the precipitator. The resulting SiO2 microspheres with narrow size distribution have an amorphous structure with diameters of approximately 480 nm and a specific surface area of 47.79 m2 g−1 under the optimized synthesis conditions with a volume ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.5
1.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 for extract/water/ethanol, a pH value of 8 and H3PO4 solution dripping rate of 3 mL min−1. In this optimized route, the yields of SiO2 and activated carbon reach 96.77% and 71.89%, respectively. The waste water produced in this process is used to produce NPK compound fertilizer. The whole route is an environmentally friendly process without any emission of pollution.
1 for extract/water/ethanol, a pH value of 8 and H3PO4 solution dripping rate of 3 mL min−1. In this optimized route, the yields of SiO2 and activated carbon reach 96.77% and 71.89%, respectively. The waste water produced in this process is used to produce NPK compound fertilizer. The whole route is an environmentally friendly process without any emission of pollution.
1. Introduction
Wheat straw (WS), as the main agricultural by-products in the wheat producing process, is generated at about 529 million tons worldwide every year.1 In the north of China, about 110 million tons of wheat straw is produced annually. Some of the wheat straw is used as feedstock for the paper industry, fuel of biomass power plants and cattle food;2 the other wheat straw is burnt out directly in the farmland after the wheat is harvested. With the development of the biomass power plant, a large amount of wheat straw ash (WSA) is produced every year in China, which will become a serious environmental issue. If the wheat straw ash is not treated and recovered, the WHA will floats around in the air, influencing the traffic safety, polluting the surface water and causing some problems to people's respiratory. Considering that the WSA is mainly composed of silica and carbon, there is a great potentiality to recover WSA to task-specific products, such as silica powder and activated carbon adsorbent.3Silica is a very important inorganic oxide material that has been extensively utilized in sorts of industries.4 Porous silica with high surface area and light weight could be used in polymer filler, adsorption, bio-imaging, optical devices, catalyst, drug delivery, and biomedical applications. In the previous published works, porous silica with high surface area (330–995 m2 g−1) has already been prepared using rice husk ash, grass ash or rice straw ash as the Si source.5,6 Almost all of these researches use NaOH solution to dissolve the silica from the raw materials and then precipitate the silica again from the extract with HCl or H2SO4 solution using polyethylene glycol (PEG), pluronic P123 or CTAB (C16H33N(CH3)3Br) as the surfactant.
However, when the porous silica is used as a filler of hydrophobic materials, such as ceramics and dental materials, the higher surface area will increase the uptake of water and the resultant degradation of the filler/matrix interface. Furthermore, the higher surface area also causes the decrease of the filler loading and the reduction of hydrophobic materials' mechanical properties. To overcome these defects stated above, hydrophobic materials could use a combination of high surface area silica and low surface area silica as their filler.7,8 Therefore, the porous silica microspheres with low surface area are also an important industrial raw materials and worth us to study deeply. In our previous work, SiO2 microspheres with specific surface area below 100 m2 g−1 have been synthesized using the wheat husk ash as raw material.8 In that work, NaOH solution is used to extract the silica from the wheat husk ash to form sodium silicate solution, and then HCl solution is used to precipitate the silica again from the diluted extract without any surfactant. However, the waste water produced in the synthesis process of silica and the carbon residue of the wheat husk ash were not used effectively.
As is known to all, activated carbons, a kind of amorphous carbonaceous material with high porosity and surface area, have been applied in various industries for several decades owing to its wonderful structures and easy availability. Specifically, due to its high adsorption capacity towards pollutants such as phenolic compound, methylene blue, metallic ions and some volatile organic compounds,9 activated carbons have been extensively studied for the water treatment, solvent recovery, air purification and gas separation and so on.10 In addition, N, P and K are the three main nutrients which influence the growth, yield, quality and leaf mineral composition of crops, vegetables and fruits.11 Considering that NPK compound fertilizer contains K, P and N, it is very beneficial for the healthy growth and development of many species.12
Here, in order to make full use of the wheat straw ash and achieve the purpose of zero emission in the synthesis process, an environmentally friendly route is proposed to convert the wheat straw ash to low surface area SiO2 microspheres, with activated carbon and Nitrogen–Phosphorus–Potassium (NPK) compound fertilizer as by-products. In this route, we use KOH solution to extract the silica from the WSA which is washed with HNO3 solution in the beginning of the process. After the dilution of potassium silicate extract (WSA-extract) with distilled water and ethanol, we precipitate the silica again from the diluted WSA-extract with H3PO4 solution. The carbon reside is dried directly after the WSA was extracted by KOH solution and then activated to obtain activated carbon. The waste water obtained in this route is used to produce NPK compound fertilizer. Every components of the wheat straw ash and all the chemicals involved in the proposed process have been effectively used. This work would supply a new method for the recovery of useful materials from the wheat straw ash.
2. Experimental section
2.1 Material and reagents
Wheat straw ash (WSA) was obtained from a biomass power plant of Henan Province in China, in the summer of 2014. The chemical compositions of the WSA used in this study are given in Table 1. The WSA was soaked for 4 h in 0.5 M HNO3 with a ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 (w/v, g mL−1) at ambient temperature to remove metal impurities which were mainly iron filings introduced from wheat harvesting process. After the soaking processes, the acid liquor which was separated from WSA solid by filtration was returned to the soaking processes. After washed to neutrality with distilled water, the WSA was dried overnight at 105 °C. The cleaned WSA was porphyrized to 60 mesh and stored in double plastic bags for later experimental use. The waste acid liquor (washing solution) was collected to produce fertilizer.
7 (w/v, g mL−1) at ambient temperature to remove metal impurities which were mainly iron filings introduced from wheat harvesting process. After the soaking processes, the acid liquor which was separated from WSA solid by filtration was returned to the soaking processes. After washed to neutrality with distilled water, the WSA was dried overnight at 105 °C. The cleaned WSA was porphyrized to 60 mesh and stored in double plastic bags for later experimental use. The waste acid liquor (washing solution) was collected to produce fertilizer.
| Component | Amount, wt% | Component | Amount, wt% |
|---|---|---|---|
| C | 49.97 | Al | 0.12 |
| SiO2 | 46.23 | Na | 0.24 |
| K | 1.28 | Mg | 0.17 |
| Ca | 0.81 | Zn | 0.07 |
| Fe | 0.95 |
All chemicals used in the synthetic procedures of this work were purchased from Tianjin Kermel Co. Ltd, which were of analytical grade. Distilled water was employed in all synthesis and treatment processes.
2.2 Preparation of activated carbon
The cleaned WSA powder and 2.0 M KOH were mixed at a ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 (w/v, g mL−1) in a 500 mL three-neck round-bottom flask equipped with a thermometer and a magnetic stirrer and heated to 90 °C for 3 h. The extract (WSA-extract) containing potassium silicate was separated from WSA solid reside by vacuum-assisted filtration for the preparation of SiO2 microspheres. The WSA solid reside was dried at 120 °C overnight directly and then activated under argon atmosphere in a horizontal pyrolysis reactor at 750 °C for 1.5 h to obtain activated carbon. The yield of activated carbon was calculated as the following equation:
6 (w/v, g mL−1) in a 500 mL three-neck round-bottom flask equipped with a thermometer and a magnetic stirrer and heated to 90 °C for 3 h. The extract (WSA-extract) containing potassium silicate was separated from WSA solid reside by vacuum-assisted filtration for the preparation of SiO2 microspheres. The WSA solid reside was dried at 120 °C overnight directly and then activated under argon atmosphere in a horizontal pyrolysis reactor at 750 °C for 1.5 h to obtain activated carbon. The yield of activated carbon was calculated as the following equation:
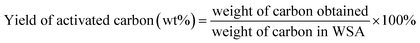 | (1) |
The obtained activated carbon was washed to neutrality with distilled water. The waste alkali liquor (washing solution) mainly containing KOH and K2CO3 was separated from activated carbon by vacuum-assisted filtration. The waste alkali liquor was collected and named filtrate-n according the washing times (n represents the washing times). For example, after the first washing and filtration, the collected liquid product was named filtrate-1. The filtrate containing a high concentration of alkali was returned to the activation processes. The filtrate containing a low concentration of alkali was used to produce fertilizer.
2.3 Preparation of SiO2 microspheres
20 mL of WSA-extract was diluted with different volume ratios of water and ethanol at ambient temperature. Different amounts of 2 M H3PO4 were dripped into the diluted extract with a buret to achieve a pH of 6–9 for silica precipitation under constantly stirring. The dripping rate of H3PO4 solution is controlled at 0.5 mL min−1, 1 mL min−1, 2 mL min−1 and 3 mL min−1, respectively. Then, the suspension liquids were further stirred for 1.5 h. The precipitate was separated from the suspension liquids by vacuum-assisted filtration. The filtrate was stored in reagent bottles for the preparation of NPK compound fertilizers. The precipitate was washed several times with distilled water and centrifuged at 9000 rpm for 5 min with each wash. The waste liquor (washing solution) mainly containing phosphate was also used to prepare NPK compound fertilizers. The cleaned precipitate was dried at 60 °C in a vacuum drying oven for 12 h and calcined at 550 °C for 1 h in a muffle furnace to obtain SiO2 microspheres. The yield of SiO2 was calculated as the following equation:
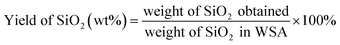 | (2) |
2.4 Preparation of NPK compound fertilizer
The ethanol was separated from the above filtrate via distillation. The No-ethanol filtrate was mixed together with the waste liquor produced in the washing process of SiO2 microspheres, the filtrate-n (n > 1) and the above waste acid produced in the washing process of WSA. Then, the mixed liquor was distilled to obtain NPK compound fertilizers. The ethanol and the water obtained in the distillation process were returned to the dilution process of the WSA-extract. Fig. 1 shows the schematic diagram of the production processes of SiO2 microspheres, activated carbon and NPK compound fertilizer. | ||
| Fig. 1 The schematic diagram of the production processes of SiO2 microspheres, activated carbon and NPK compound fertilizer. | ||
2.5 Characterizations
The contents of silica and carbon in the WSA were measured according to the China National Standards GB/T 1873-1980 and GB/T 223.71-1991, respectively. The contents of metal elements in the WSA were determined by an inductively coupled plasma (ICP)-optical emission spectrometer (Optima 2000 DV, Perkin Elmer, USA).The particle size distributions of the SiO2 microspheres were characterized by a laser particle-size analyzer (LPSA) (Zetasizer nano ZS, MALVERN, UK). The specific surface area and pore size of the SiO2 microspheres and the obtained activated carbon were measured by the Brunauer–Emmett–Teller (BET) method with an automated chemisorption/physisorption surface area and pore size analyzer (Tristar II 3020, Micromeritics Instrument Corporation, USA), respectively. The microstructure and morphology of the typical SiO2 microspheres were examined using a scanning electron microscope (SEM) (SUPRA 55 SAPPHIRE, CARL ZEISS, Germany) with metallization by Pt-spraying on surface. TEM images of the typical SiO2 microspheres and the obtained activated carbon were received by a transmission electron microscope (TEM) (JEM-2100, JEOL, Japan). The elements in the typical SiO2 microspheres were determined using an energy-dispersive X-ray analysis spectrometer (EDS) (X-Max, OXFORD, UK) adjunct to the SEM. Fourier transform infrared (FTIR) spectra of the typical SiO2 microspheres was acquired with an FTIR instrument (Spectrum One-B, PE, USA) at a wavenumber range of 400–4000 cm−1. X-ray diffraction (XRD) (RigakuD/MAX-3A, Rigaku Corporation, Japan) pattern of the typical SiO2 microspheres was obtained using a Cu Kα radiation (λ = 1.5406 Å) at operating voltage of 40 kV and a filament current of 40 mA. The diffraction angle was scanned from 10° to 80°, at a rate of 6° min−1. The contents of NO3−, Cl− in the obtained NPK compound fertilizers were measured using an ion chromatography (IC) (ICS-5000, DIONEX, USA). Other macronutrients (K, P, S, Ca and Mg) and micronutrients (Fe, Zn, Mn and Cu) in the obtained NPK compound fertilizers were determined by the inductively coupled plasma (ICP)-optical emission spectrometer.
3. Results and discussion
3.1 Factors affecting size distribution of SiO2 microspheres
The volume ratio of WSA-extract/water/ethanol is one more important factor that influences the formation and the particle size distribution of silica microspheres. Without the additions of ethanol, no microspheres can be obtained. Without distilled water, the particles exhibit a large size distribution.8,13 Fig. 2a shows that the particle size distribution curves of silica microspheres synthesized at pH 8 at different volume ratios of WSA-extract/water/ethanol using H3PO4 solution dripping rate of 3 mL min−1. Obviously, when the volume ratio of WSA-extract/water/ethanol was 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.5
1.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, the particle size distribution was more narrower; while the volume ratio was 1
1, the particle size distribution was more narrower; while the volume ratio was 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.5, it was much broader. The reason for this phenomenon is that the high miscibility of ethanol accelerates the penetration into the silicate precursor. This behavior of ethanol facilitates the formation of
1.5, it was much broader. The reason for this phenomenon is that the high miscibility of ethanol accelerates the penetration into the silicate precursor. This behavior of ethanol facilitates the formation of ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Si–OH and
Si–OH and ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Si–O–Si
Si–O–Si![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) that produces more spherical and larger particles during the aging process.14,15 Moreover, water is necessary to decrease the concentration of potassium silicate, and ethanol is required to lower the solubility of potassium silicate. Appropriate amount of water and ethanol would have a synergistic effect on the super-saturation of potassium silicate, thus creating a positive condition for the formation of primary particles with relatively uniform size distribution.8,13
that produces more spherical and larger particles during the aging process.14,15 Moreover, water is necessary to decrease the concentration of potassium silicate, and ethanol is required to lower the solubility of potassium silicate. Appropriate amount of water and ethanol would have a synergistic effect on the super-saturation of potassium silicate, thus creating a positive condition for the formation of primary particles with relatively uniform size distribution.8,13
The particle size distribution curves of silica microspheres synthesized under different pH conditions are shown in Fig. 2b, when the volume ratio of WSA-extract/water/ethanol was 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.5
1.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and the dripping rate of H3PO4 solution was 3 mL min−1. Obviously, when the pH value was 8, the particle size distribution was more narrower. The particle size of silica microspheres increased as the pH increased. A higher pH results in higher hydrolysis and condensation rates (reactions (3) and (4)) that promote the growth of spherical particles. At pH > 9, spherical particles is formed immediately after the addition of phosphoric acid.15 Therefore, heterogeneous silica microspheres with a broad size distribution are formed under constantly stirring.
1 and the dripping rate of H3PO4 solution was 3 mL min−1. Obviously, when the pH value was 8, the particle size distribution was more narrower. The particle size of silica microspheres increased as the pH increased. A higher pH results in higher hydrolysis and condensation rates (reactions (3) and (4)) that promote the growth of spherical particles. At pH > 9, spherical particles is formed immediately after the addition of phosphoric acid.15 Therefore, heterogeneous silica microspheres with a broad size distribution are formed under constantly stirring.
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Si–OH + OH− → Si–OH + OH− → ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Si–O− + H2O Si–O− + H2O
| (3) |
![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Si–OH + Si–OH + ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Si–O− → Si–O− → ![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) Si–O–Si Si–O–Si![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) + OH− + OH−
| (4) |
Furthermore, the level of protonation of Si–O– groups are dependent on the solution pH, and more Si–OH groups are formed leading to the generation of more minisize silica microspheres as the pH decreases.6,13,16 When the pH value is 8, the hydrolysis and condensation rates of Si–OH groups and the protonation of Si–O– groups have a cooperative effect on the formation of silica microspheres with relatively uniform diameters. However, the mechanism is worthy of further investigation.
The dripping rate of reactant has an significant effect on the particle size, size distribution and yield of the silica spheres.8,17,18 Fig. 2c shows that the particle size distribution curves of silica microspheres prepared at pH 8 at the volume ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.5
1.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 of WSA-extract/water/ethanol using different H3PO4 solution dripping rate. The particle size of silica microspheres decreased with the increase of the dripping rate of H3PO4 solution. That may be because a slow dripping rate probably allows more time for the primary particles to aggregate, resulting in larger particles.17
1 of WSA-extract/water/ethanol using different H3PO4 solution dripping rate. The particle size of silica microspheres decreased with the increase of the dripping rate of H3PO4 solution. That may be because a slow dripping rate probably allows more time for the primary particles to aggregate, resulting in larger particles.17
3.2 Surface area and porosity of the silica microspheres
Nitrogen adsorption and desorption isotherms were measured at 77 K in order to characterize the pore structure of the silica microspheres. The specific surface area was analyzed by the BET method, and the pore volume distribution was analyzed by Barrett–Joyner–Halenda (BJH) method using desorption isotherm data. In the BJH method, the pore shape was assumed to be cylindrical and the measured desorption was assumed to be made up of both condensed liquid lost from pores and adsorbate lost from multilayers of adsorbed molecule.19The surface area of the silica microspheres prepared at different experimental conditions is shown in Table 2. The silica microspheres prepared at the volume ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1.5
1.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 of WSA-extract/water/ethanol using H3PO4 solution dripping rate of 3 mL min−1 with pH value of 6, 7, 8 and 9 were designated as Se, Sf, Sg, and Sh, respectively. The results obtained show that the surface area of the silica microspheres increased markedly as the pH decreased. It may be because the protonation of Si–O– groups promotes the generation of more minisize silica microspheres as the pH decreases.
1 of WSA-extract/water/ethanol using H3PO4 solution dripping rate of 3 mL min−1 with pH value of 6, 7, 8 and 9 were designated as Se, Sf, Sg, and Sh, respectively. The results obtained show that the surface area of the silica microspheres increased markedly as the pH decreased. It may be because the protonation of Si–O– groups promotes the generation of more minisize silica microspheres as the pH decreases.
| Sample | Specific surface area (m2 g−1) | Total pore volume (cm3 g−1) |
|---|---|---|
| Se | 214.36 | 0.176 |
| Sf | 152.15 | 0.161 |
| Sg | 47.79 | 0.133 |
| Sh | 44.43 | 0.128 |
Fig. 3 and ESI 1S† show that the pore size distribution curves of Se, Sg, Sf, and Sh are similar. The pores of the four silica samples are mainly distributed in 2.0–40 nm, so the four silica samples could be regarded as mesoporous materials. Obviously, they all exhibited a point B and a hysteresis loop appearing between their adsorption and desorption branches in their monolithic isotherms (Fig. 3 and ESI 1S),† indicating the typical capillary condensation of the mesoporous structure.20 All the isotherms belonged to the type IV category based on the IUPAC classification, with H3 hysteresis loop. Moreover, in the isotherms of Se, Sg, Sf, and Sh, the amount adsorbed all increased rapidly near the relative pressure (P/Po) of 1.0 (Fig. 3 and ESI 1S),† indicating that the larger mesopores may company with slit-shaped pores in an aggregation of silica microspheres.20 However, the shapes of the monolithic isotherms were different from each other. In the isotherms of Se and Sf, the amount adsorbed was much higher at P/Po 0.2–0.8 (Fig. 3a and ESI 1S(as)),† where pore condensation took place, indicating the large quantity of mesopores involved in Se and Sf.21 While there was lower amount adsorbed at P/Po 0.2–0.8 in the isotherms of Sg and Sh (Fig. 3b and ESI 1S(bs)),† it demonstrates that the quantity of mesopores contained inside Sg and Sh is lower. This is another reason why the surface area of the silica microspheres increased markedly as the pH decreased. Although the surface area of Sg is a little bigger than Sh (Table 2), the particle size distribution of Sg is much narrower than Sh (Fig. 2b), so 8 is the optimum pH value to prepare low surface area SiO2 microspheres with relatively uniform size distribution.
3.3 Characteristics of the typical silica microspheres
Fig. 4 shows the SEM micrograph and TEM image of Sg. As shown in Fig. 4a, Sg was composed of microsphere particles homogeneously. The interconnection among the micro-spheres was also observed in the SEM images (Fig. 4a). The interconnection could be due to the desiccation and calcination of the silica precipitate. No obvious porous structure on the surface of these spheres was found. The silica microspheres were nearly solid, as shown in TEM image (Fig. 4b). The chemical composition of Sg was confirmed by EDS (Fig. 5a) that showed O and Si at a calculated 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 atomic ratio. Except K, other elements contained in raw WSA materials, e.g. Ca, Na, Mg, Fe, and Al were not found, confirming that washing WSA with 0.5 M HNO3 solution is an effective way to remove these earth metals. Fig. 5b shows the FTIR spectrum of Sg. The strong absorption peak at 1089 cm−1 is designated to the asymmetric stretching vibration of the Si–O–Si bonds. The peaks at 469 cm−1 and 801 cm−1 are derived from the plane swing vibration and the bending vibration of the Si–O bonds, respectively. The absorption peaks at 3428 cm−1 and 1658 cm−1 are designated to the H–O–H stretching vibration and bending vibration modes of the adsorbed water, respectively.13 There were no other absorption bands (Fig. 5b). Therefore, the EDS and FTIR data clearly demonstrated that Sg was silica. The X-ray diffraction pattern of Sg showed a broad peak between 15° and 38°, centered at 22°, typical for amorphous silica (Fig. 5c). Furthermore, the absence of peaks of possible impurities such as potassium phosphate and other alkaline or alkaline earth metals affirmed the purity of the obtained silica.22
1 atomic ratio. Except K, other elements contained in raw WSA materials, e.g. Ca, Na, Mg, Fe, and Al were not found, confirming that washing WSA with 0.5 M HNO3 solution is an effective way to remove these earth metals. Fig. 5b shows the FTIR spectrum of Sg. The strong absorption peak at 1089 cm−1 is designated to the asymmetric stretching vibration of the Si–O–Si bonds. The peaks at 469 cm−1 and 801 cm−1 are derived from the plane swing vibration and the bending vibration of the Si–O bonds, respectively. The absorption peaks at 3428 cm−1 and 1658 cm−1 are designated to the H–O–H stretching vibration and bending vibration modes of the adsorbed water, respectively.13 There were no other absorption bands (Fig. 5b). Therefore, the EDS and FTIR data clearly demonstrated that Sg was silica. The X-ray diffraction pattern of Sg showed a broad peak between 15° and 38°, centered at 22°, typical for amorphous silica (Fig. 5c). Furthermore, the absence of peaks of possible impurities such as potassium phosphate and other alkaline or alkaline earth metals affirmed the purity of the obtained silica.22
3.4 Characteristics of the activated carbon and the components of NPK compound fertilizer
Fig. 6a shows that the N2 adsorption/desorption isotherms and pore size distribution of the activated carbon produced during the synthesis process. The isotherm belonged to the type IV category based on the IUPAC classification, with H1 hysteresis loop.20 The pores of the activated carbon were mainly distributed in 2.0–20 nm, so the activated carbon could be regarded as mesoporous materials. The specific surface area of the activated carbon reached 1933 m2 g−1 (Table 3), so the activated carbon could be used as adsorbent or electrode materials of super-capacitors.23 Fig. 6b shows that the pores on the surface of activated carbon were mainly micropores and mesopores. The opening of the pores in the surface of activated carbon is due to the extraction of some component during the activation process. | ||
| Fig. 6 N2 adsorption/desorption isotherms and pore size distribution (a) and TEM image (b) of the activated carbon produced during the synthesis process. | ||
| Specific surface area (m2 g−1) | Total pore volume (cm3 g−1) | Average pore diameter (nm) |
|---|---|---|
| 1933 | 1.28 | 2.60 |
The main components of NPK compound fertilizer which was produced during the preparation process of Sg are listed in Table 4. Except C, H and O, it contained macronutrients N, K, P, Ca and Mg, micronutrients Fe and some other micronutrients less than 0.01%. The obtained NPK compound fertilizer could completely dissolve in water, which is beneficial for the healthy growth and development of many species, particularly dioscoreaceae plants and some fabaceae plants.24 If the obtained NPK compound fertilizer is used as the fertilizer of the green vegetables, such as Chinese cabbage or crown daisy, we only need to add a certain amount of ammonium nitrate to increase the content of nitrogen.25
| Composition | K | P | N | Ca | Fe | Mg |
|---|---|---|---|---|---|---|
| Amount, wt% | 41.18 | 20.93 | 3.23 | 0.07 | 0.04 | 0.03 |
3.5 The yield of the products
In the preparation process of Sg, the yield of SiO2 and activated carbon reach 96.77% and 71.89%, respectively. The losing SiO2 gets into NPK compound fertilizer eventually. During the activation process, about 27% of carbon losses with the formation of the pores on the surface of the activated carbon. The metal elements in the WSA also get into NPK compound fertilizer finally. Except ethanol, all the chemicals involved in the proposed process are changed into NPK compound fertilizer eventually. About 98% of ethanol is returned to the dilution process of the WSA-extract. Except the first feeding, 20 g of WSA (on dry basis) consumes approximately 13.4 g of KOH, 2.1 mL ethanol, 8.4 mL of 85% H3PO4 and 0.52 mL of 98% HNO3, and produces about 8.9 g of low surface area SiO2, 7.2 g of activated carbon and 21.1 g of NPK compound fertilizer finally. Except a small amount of water and ethanol, there is no other chemicals loss. Therefore, the optimized route is an economical route.4. Conclusions
In this paper, a green approach to make full use of every components of wheat straw ash is introduced. Low surface area SO2 microspheres with activated carbon as by-product are synthesized from the WSA. The waste water produced in this process is all converted into NPK compound fertilizer. For the synthesis of silica microspheres, the particle size distribution is affected dramatically by the volume ratio of WSA-extract/water/ethanol and the pH of the diluted extract, while the size of silica microspheres can be controlled by the dripping rate of H3PO4 solution. Moreover, the specific surface area of silica microspheres is also controlled by the pH of the diluted extract obviously. All the chemicals involved in the proposed process have been effectively used. The whole route is an environmentally friendly process without any emission of pollution.Acknowledgements
This research is financially supported by the Natural Science Foundation of China (No. 21176034, 51479019, 21476035 and 21506198) and the Fundamental Research Funds for the Central Universities (3132014323).Notes and references
- S. P. Govumoni, S. Koti, S. Y. Kothagouni, S. Venkateshwar and V. R. Linga, Carbohydr. Polym., 2013, 91, 646 CrossRef CAS PubMed; X. Y. Liu, S. M. Zicari, G. Q. Liu, Y. Q. Li and R. H. Zhang, Bioresour. Technol., 2015, 185, 150 CrossRef PubMed.
- J. Giuntoli, A. K. Boulamanti, S. Corrado, M. Motegh, A. Agostini and D. Baxter, GCB Bioenergy, 2013, 5, 497 CrossRef PubMed.
- H. Biricik, F. Aköz, Ì. Berktay and A. N. Tulgar, Cem. Concr. Res., 1999, 29, 637 CrossRef CAS.
- N. Pijarn, A. Jaroenworaluck, W. Sunsaneeyametha and R. Stevens, Powder Technol., 2010, 203, 462 CrossRef CAS PubMed.
- M. Chareonpanich, A. Nanta-ngern and J. Limtrakul, Mater. Lett., 2007, 61, 5153 CrossRef CAS PubMed; F. Ghorbani, H. Younesi, Z. Mehraban, M. S. Celik, A. A. Ghoreyshi and M. Anbia, J. Taiwan Inst. Chem. Eng., 2013, 44, 821 CrossRef PubMed; O. Jullaphan, T. Witoon and M. Chareonpanich, Mater. Lett., 2009, 63, 1303 CrossRef PubMed; D. W. Li and X. F. Zhu, Mater. Lett., 2011, 65, 1528 CrossRef PubMed; P. Lu and Y. L. Hsieh, Powder Technol., 2012, 225, 149 CrossRef PubMed; Y. Yang and T. Coradin, Green Chem., 2008, 10, 183 RSC.
- D. W. Li, D. Y. Chen and X. F. Zhu, Bioresour. Technol., 2011, 102, 7001 CrossRef CAS PubMed.
- A. R. Curtis, A. C. Shortall, P. M. Marquis and W. M. Palin, J. Dent., 2008, 36, 186 CrossRef CAS PubMed; S. P. Samuel, S. X. Li, I. Mukherjee, Y. Guo, A. C. Patel, G. Baran and Y. Wei, Dent. Mater., 2009, 25, 296 CrossRef PubMed.
- J. L. Cui, H. L. Sun, Z. L. Luo, J. C. Sun and Z. S. Wen, Mater. Lett., 2015, 156, 42 CrossRef CAS PubMed.
- K. L. Chiu and D. H. L. Ng, Biomass Bioenergy, 2012, 46, 102 CrossRef CAS PubMed; D. Liu, P. Yuan, D. Y. Tan, H. M. Liu, T. Wang, M. D. Fan, J. X. Zhu and H. P. He, J. Colloid Interface Sci., 2012, 388, 176 CrossRef PubMed; Y. C. Sharma and Uma, J. Chem. Eng. Data, 2010, 55, 435 CrossRef; Z. G. Liu and F. S. Zhang, Desalination, 2011, 267, 101 CrossRef PubMed.
- N. V. Sych, S. I. Trofymenko, O. I. Poddubnaya, M. M. Tsyba, V. I. Sapsay, D. O. Klymchuk and A. M. Puziy, Appl. Surf. Sci., 2012, 261, 75 CrossRef CAS PubMed; N. Bagheri and J. Abedi, Chem. Eng. Res. Des., 2009, 87, 1059 CrossRef PubMed.
- T. Milošević, N. Milošević and I. Glišić, Sci. Hortic., 2013, 156, 131 CrossRef CAS PubMed; B. C. Xue, Q. Hao, T. Liu and E. B. Liu, Powder Technol., 2013, 247, 8 CrossRef PubMed; A. M. Senna, J. B. do Carmo, J. M. S. da Silva and V. R. Botaro, J. Environ. Chem. Eng., 2015, 3, 996 CrossRef PubMed.
- L. L. Ding, B. Zou, L. Shen, C. Zhao, Z. C. Wang, Y. P. Guo, X. F. Wang and Y. H. Liu, Colloids Surf., A, 2014, 446, 90 CrossRef CAS PubMed.
- L. Lin, S. R. Zhai, Z. Y. Xiao, N. Liu, Y. Song, B. Zhai and Q. D. An, Bioresour. Technol., 2012, 125, 172 CrossRef CAS PubMed.
- C. Y. Jung, J. S. Kim, T. S. Chang, S. T. Kim, H. J. Lim and S. M. Koo, Langmuir, 2010, 26, 5456 CrossRef CAS PubMed.
- N. S. C. Zulkifli, I. A. Rahman, D. Mohamad and A. Husein, Ceram. Int., 2013, 39, 4559 CrossRef CAS PubMed.
- T. Yokoi, J. Wakabayashi, Y. Otsuka, W. Fan, M. Iwama, R. Watanabe, K. Aramaki, A. Shimojima, T. Tatsumi and T. Okubo, Chem. Mater., 2009, 21, 3719 CrossRef CAS.
- S. K. Park, K. D. Kim and H. T. Kim, Colloids Surf., A, 2002, 197, 7 CrossRef CAS.
- I. A. Rahman, P. Vejayakumaran, C. S. Sipaut, J. Ismail, M. Abu Bakar, R. Adnan and C. K. Chee, Colloids Surf., A, 2007, 294, 102 CrossRef CAS PubMed.
- J. L. Cui, H. L. Sun, X. Wang, J. C. Sun, M. H. Niu and Z. S. Wen, Ind. Crops Prod., 2015, 74, 689 CrossRef CAS PubMed; Y. N. Qu, Y. M. Tian, B. Zou, J. Zhang, Y. H. Zheng, L. L. Wang, Y. Li, C. G. Rong and Z. C. Wang, Bioresour. Technol., 2010, 101, 8402 CrossRef PubMed.
- K. S. W. Sing, D. H. Everett, R. A. W. Haul, L. Moscou, R. A. Pierotti, J. Rouquérol and T. Siemieniewska, Pure Appl. Chem., 1985, 57(4), 603 CrossRef CAS.
- H. X. Zhang, X. Zhao, X. F. Ding, H. Lei, X. Chen, D. M. An, Y. L. Li and Z. C. Wang, Bioresour. Technol., 2010, 101, 1263 CrossRef CAS PubMed.
- K. F. Yu, Y. P. Guo, X. F. Ding, J. Z. Zhao and Z. C. Wang, Mater. Lett., 2005, 59, 4013 CrossRef CAS PubMed.
- Y. Chen, S. R. Zhai, N. Liu, Y. Song, Q. D. An and X. W. Song, Bioresour. Technol., 2013, 144, 401 CrossRef CAS PubMed; S. H. Li, D. K. Huang, J. C. Yang, B. Y. Zhang, X. F. Zhang, G. Yang, M. K. Wang and Y. Shen, Nano Energy, 2014, 9, 309 CrossRef PubMed.
- A. Ierna, G. Pandino, S. Lombardo and G. Mauromicale, Agr. Water Manag., 2011, 101, 35 CrossRef PubMed; K. C. Kaizzi, J. Byalebeka, O. Semalulu, I. N. Alou, W. Zimwanguyizza, A. Nansamba, E. Odama, P. Musinguzi, P. Ebanyat, T. Hyuha, A. K. Kasharu and C. S. Wortmann, Field Crops Research, 2012, 127, 109 CrossRef PubMed.
- M. Eksi, D. B. Rowe, R. Fernández–Cañero and B. M. Cregg, Urban Forestry and Urban Greening, 2015, 14, 315 CrossRef PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra14622d |
| This journal is © The Royal Society of Chemistry 2015 |




