Simultaneous electrochemical determination of acetaminophen, caffeine and ascorbic acid using a new electrochemical sensor based on CuO–graphene nanocomposite†
Abstract
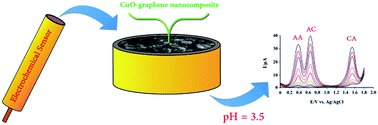
A new nanocomposite based on CuO nanoparticles/graphene nanosheets was prepared and used as a new electrode material for the simultaneous determination of acetaminophen, caffeine and ascorbic acid. CuO nanoparticles were supported on graphene nanosheets by a simple method. This nanostructure was characterized by different techniques including scanning electron microscopy, X-ray diffraction, energy dispersive X-ray spectroscopy and Fourier transform infrared spectroscopy. The high electrochemical activity, fast electron transfer rate, high surface area and good antifouling properties of the synthesized nanostructure enhanced the oxidation peak currents and reduced the peak potentials of acetaminophen, caffeine and ascorbic acid at the surface of the proposed sensor. Simultaneous determination of analytes was explored using differential pulse voltammetry. A linear range of 0.025–5.3 μmol L−1 was achieved for acetaminophen, caffeine and ascorbic acid with detection limits of 0.008, 0.010 and 0.011 μmol L−1, respectively. Finally, the proposed method was used for their determination in blood serum, urine and pharmaceutical samples.

 Please wait while we load your content...
Please wait while we load your content...