Metal-free C–N cross-coupling of electrophilic compounds and N-haloimides†
Luyan Zhanga,
Yanru Lia,
Long-Yi Jinc and
Fushun Liang*ab
aDepartment of Chemistry, Northeast Normal University, Changchun 130024, China. E-mail: liangfs112@nenu.edu.cn; Fax: +86 431 85099759
bKey Laboratory for UV-Emitting Materials and Technology of Ministry of Education, Northeast Normal University, Changchun 130024, China
cFaculty of Chemistry, Yanbian University, China
First published on 27th July 2015
Abstract
When DBU is added, the cross-coupling reaction between alkyl halides (halogen = Cl, Br and I) and N-haloimides (halogen = Cl, Br) occurs, resulting in the formation of aminated products. A halogen bond activated nucleophilic substitution mechanism was proposed. The methodology represents an elegant example of applying the halogen bond activation strategy in an organic transformation.
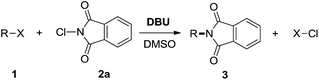
Amine is an important raw material and intermediate in medicines, pesticides, as well as liquid crystal materials. Phthalimides are versatile synthetic tools that are primarily employed in the preparation of amines via the Gabriel reaction.1 The Gabriel reaction has proven to be a very effective means of forming N-alkylphthalimides. Starting from N-substituted phthalimides, for example, and using Nefkens' reagent, N-alkylphthalimides can also be prepared.2 Adimurthy et al. found an L-proline catalyzed transamidation method of phthalimide with amines, to achieve new amines.3 A. K. Yadav and L. D. S. Yadav used N-tosylhydrazone and phthalimides to prepare amines.4 Additionally, transition metal-catalyzed C–N coupling has been developed as a powerful method.5 Herein, we would like to report a new type of cross-coupling reaction6 between alkyl halides7 and N-haloimides, which provides an efficient and straightforward method for the synthesis of amines and functionalized amines.
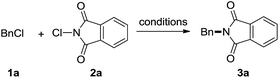
In our recent research, we have developed N-haloimides to be a versatile amination reagent used in organic transformation.8 In further work, we found that, with organobases like DBU as the activator,9 benzyl chloride can react with N-chlorophthalimide (NCP, 1.2 equiv.), giving the corresponding imidation product in 91% yield. It was not surprising that no reaction can occur in the absence of DBU (eqn (1)). The possible mechanism for the imidation is depicted in Scheme 1.10 The interaction between NCP and DBU gives halogen bond adduct I,11 which delivers an ion pair intermediate II containing highly electrophilic chlorine cation and potentially nucleophilic phthalimide anion.12 The nucleophilic substitution of alkyl halides by phthalimide anion affords the imidation product.13 We think that the halogen bond between 1-chloro-2,3,4,6,7,8,9,10-octahydropyrimido[1,2-a]azepinium (III) and alkyl halide may facilitate the nucleophilic substitution reaction.9 The mutual activation induced by halogen bonding makes otherwise inert reaction to take place.14
Our initial optimization of the model reaction commences from benzyl chloride (1a). The impact of various parameters on the efficiency of the reaction of 1a with NCP (2a) is summarized in Table 1. In the presence of DBU (1.2 equiv.), the reaction mixture conducted in DCE at room temperature, afforded product 3a in 31% isolated yield (entry 1). Other solvents screened include MeCN, DMF and DMSO (entries 2–4). DMSO proved to be the most efficient (85% yield) (entry 4).15 The reaction mixture performed at 60 °C gave product 3a in improved yield (91%) and shortened reaction time (5 min) (entry 5). Besides DBU, NaOH, t-BuONa, PPh3, DBN, MTBD, and DABCO were selected as the activators. MTBD gave similar reactivity and DBN exhibits relatively lower efficiency (entries 6 and 7). However, NaOH, t-BuONa, PPh3 and DABCO appear to be inert (entries 8–11). Catalytic amount of DBU, e.g., 0.2 equiv., could not drive the reaction to completion (entry 12), which may support the presence of the halogen bonding interaction between 1-chloro-2,3,4,6,7,8,9,10-octahydropyrimido[1,2-a]azepinium and alkyl halide, i.e., adduct III in Scheme 1.16
| Entry | Temp (°C) | Activator | Solventb | Time (min) | Yieldc (%) |
|---|---|---|---|---|---|
| a Reactions were carried out with 1a (1.0 mmol), 2a (1.2 equiv.) and activator (1.2 equiv.) in solvent (2.0 mL).b Dry solvents were used in all cases to prevent the hydrolysis of benzyl chloride.c Isolated yield. | |||||
| 1 | rt | DBU | DCE | 180 | 31 |
| 2 | rt | DBU | MeCN | 120 | 56 |
| 3 | rt | DBU | DMF | 120 | 57 |
| 4 | rt | DBU | DMSO | 30 | 85 |
| 5 | 60 | DBU | DMSO | 5 | 91 |
| 6 | 60 | DBN | DMSO | 180 | 61 |
| 7 | 60 | MTBD | DMSO | 6 | 88 |
| 8 | 60 | NaOH | DMSO | 180 | n.r |
| 9 | 60 | t-BuONa | DMSO | 180 | n.r |
| 10 | 60 | Ph3P | DMSO | 180 | n.r |
| 11 | 60 | DABCO | DMSO | 180 | n.r |
| 12 | 60 | DBU (0.2 equiv.) | DMSO | 180 | 16 |
With optimized reaction conditions in hand (Table 1, entry 5), we investigated this methodology by reacting various alkyl halides with NCP (Table 2).17 The scope of this new coupling reaction is fairly broad. Alkyl chlorides examined includes benzyl chloride, n-butyl chloride, α-chlorocyclopentanone and α-chloro N,N-dimethyl acetamine. Benzyl chloride is highly efficient, while n-butyl chloride exhibits relatively lower efficiency. α-Chlorocyclopentanone and α-chloro N,N-dimethylacetamine gave satisfactory yields. Primary alkyl bromides containing various substituents like benzyl, allyl,18 ethyl, homobenzyl, isobutyl are suitable substrates. A lot of functional groups are compatible with the reaction conditions, such as methoxy, dimethoxyacetal, carbonyl and ester. Secondary alkyl bromides like isopropyl bromide and diphenylmethyl bromide can also afford the corresponding aminated product, albeit in low yields. However, cyclic alkyl bromide, i.e., cyclohexyl bromide, was inefficient. Alkyl iodide like methyl iodide worked very well. In one word, alkyl halides RX (X = Cl, Br and I) exhibit increased reactivity in the order of RCl, RBr and RI. The gram-scale synthesis of 3a (1.51 g) was also achieved in 92% yield by DBU-mediated reaction of benzyl bromide and NCP.
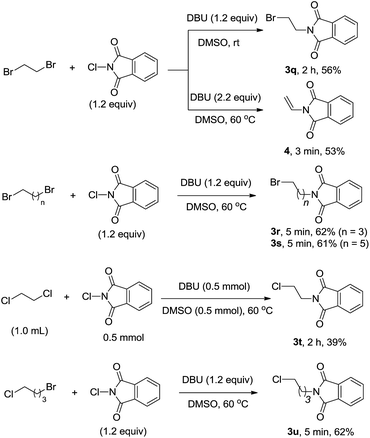
Interestingly, we found that dihalides are also suitable for the cross-coupling reaction (Scheme 2). In the reaction of 1,2-dibromoethane with NCP (1.2 equiv.), two types of products could be prepared, depending the feed ratio of DBU employed. When 1.2 equiv. of DBU was used, the mixture afforded monobromo substituted amine 3q in 56% yield. In the case of 2.2 equiv. of DBU, further elimination of HBr occurs, giving rise to enamine 4 in 53% yield. However, no elimination of HBr or HCl was observed for 1,4-dibromobutane, 1,6-dibromooctane and 1,2-dichloroethane substrates, even though excess amount of DBU was used. The corresponding imidation products 3r–t with a pendant bromine or chlorine atom were attained in low to moderate yields, which may be potentially useful in further transformation.19 Mixed dihalides were also subjected to the reaction sequence. For example, 1-bromo-4-chlorobutane gave chlorine atom-tolerant imidation product 3u as the only product, due to the different reactivity between bromine and chlorine atoms.
We investigated the efficacy of N-haloimides by reacting them with benzyl bromide (Scheme 3). Different N-haloimide exhibits different reactivity. NBP is less efficient than NCP (65%). Similar trend was observed for N-halosuccinimides. NCS gave product 7 in 51% yield while NBS and NIS could not react. From Scheme 1 one can see there are two types of halogen bonding interaction, N-haoimide and DBU (adduct I), and the resulting chlorine cation in 1-chloro-2,3,4,6,7,8,9,10-octahydropyrimido[1,2-a]azepinium with alkyl halide RX (adduct III). For the latter, chlorine atom gives stronger halogen bonding ability than bromine atom, due to the larger electron negativity, thus leading to R–X bond cleavage easier (nucleophilic substitution is the rate-determining step). N-Bromosaccharin was a competent reagent, affording 2-benzylbenzo[d]isothiazol-3(2H)-one 1,1-dioxide 8 in 89% yield. Other N-haloimides like 1,3-dibromo-5,5-dimethyl hydantoin and N-bromoacetamide proved to be inefficient. All the above reactions indicated relatively high efficiency and broad scope for both alkyl halides and N-haloimides.
In summary, we have developed a highly efficient and metal-free cross-coupling reaction of alkyl halides and N-haloimides. With this strategy, various alkyl- and functionalized alkylamines were synthesized. The scope of the reaction is broad in terms of both alkyl halides and N-haloimides. A halogen bond activated nucleophilic substitution mechanism was proposed. This work demonstrated the feasibility and potential of halogen bonding activation in organic transformation.
Acknowledgements
Financial support from the National Natural Science Foundation of China (21172034 and 21372039), and the 111 Project is gratefully acknowledged.Notes and references
- (a) M. S. Gibson and R. W. Bradshaw, Angew. Chem., Int. Ed., 1968, 7, 919 CrossRef CAS; (b) J. C. Sheehan and W. A. Bolhofer, J. Am. Chem. Soc., 1950, 72, 2786 CrossRef.
- (a) G. H. L. Nefkens, Nature, 1960, 185, 309 CrossRef CAS PubMed; (b) G. H. L. Nefkens and G. I. Tesser, J. Am. Chem. Soc., 1961, 83, 1263 CrossRef CAS; (c) P. M. Worster, C. C. Leznoff and C. R. McArthur, J. Org. Chem., 1980, 45, 174 CrossRef CAS; (d) E. G. Bowen and D. J. Wardrop, J. Am. Chem. Soc., 2009, 131, 6062 CrossRef CAS PubMed.
- S. N. Rao, D. C. Mohan and S. Adimurthy, Org. Lett., 2013, 15, 1496 CrossRef CAS PubMed.
- A. K. Yadav and L. D. S. Yadav, RSC Adv., 2014, 4, 34764 RSC.
- Selected reviews: (a) G. Evano, N. Blanchard and M. Toumi, Chem. Rev., 2008, 108, 3054 CrossRef CAS PubMed; (b) J. Bariwal and E. an der Eycken, Chem. Soc. Rev., 2013, 42, 9283 RSC; (c) J. F. Hartwig, Acc. Chem. Res., 2008, 41, 1534 CrossRef CAS PubMed; (d) J. P. Wolfe, S. Wagaw, J.-F. Marcoux and S. Buchwald, Acc. Chem. Res., 1998, 31, 805 CrossRef CAS; (e) D. S. Surry and S. L. Buchwald, Angew. Chem., Int. Ed., 2008, 47, 6338 CrossRef CAS PubMed.
- Cross-coupling of halides: (a) J. Zhou and G. C. Fu, J. Am. Chem. Soc., 2004, 126, 1340 CrossRef CAS PubMed; (b) X. Wu, J. W. T. See, K. Xu, H. Hirao, J. Roger, J.-C. Hierso and J. Zhou, Angew. Chem., Int. Ed., 2014, 53, 13573 CrossRef CAS PubMed; (c) O. Gutierrez, J. C. Tellis, D. N. Primer, G. A. Molander and M. C. Kozlowski, J. Am. Chem. Soc., 2015, 137, 4896 CrossRef CAS PubMed; (d) N. A. Owston and G. C. Fu, J. Am. Chem. Soc., 2010, 132, 11908 CrossRef CAS PubMed; (e) Z. Lu and G. C. Fu, Angew. Chem., Int. Ed., 2010, 49, 6676 CrossRef CAS PubMed; (f) H. Xu, C. Zhao, Q. Qian, W. Deng and H. Gong, Chem. Sci., 2013, 4, 4022 RSC; (g) C. Zhao, X. Jia, X. Wang and H. Gong, J. Am. Chem. Soc., 2014, 136(50), 17645 CrossRef CAS PubMed; (h) C.-T. Yang, Z.-Q. Zhang, J. Liang, J.-H. Liu, X.-Y. Lu, H.-H. Chen and L. Liu, J. Am. Chem. Soc., 2012, 134, 11124 CrossRef CAS PubMed.
- Alkyl halides are ubiquitous building blocks throughout organic chemistry owing to their participation in radical reactions, nucleophilic substitution reactions and organometallic cross-coupling reactions: (a) H. Togo, Advanced Free Radical Reactions for Organic Synthesis, Elsevier, 2004 Search PubMed; (b) F. A. Carey and R. J. Sundberg, Advanced Organic Chemistry, Springer, 5th edn, 2007 Search PubMed; (c) A. Rudolph and M. Lautens, Angew. Chem., Int. Ed., 2009, 48, 2656 CrossRef CAS PubMed; (d) J. Terao and N. Kambe, Acc. Chem. Res., 2008, 41, 1545 CrossRef CAS PubMed; (e) N. A. Owston and G. C. Fu, J. Am. Chem. Soc., 2010, 132, 11908 CrossRef CAS PubMed.
- (a) H. Tan, M. Li and F. Liang, RSC Adv., 2014, 4, 33765 RSC; (b) Y. Wei, S. Lin and F. Liang, Org. Lett., 2012, 14, 4202 CrossRef CAS PubMed; (c) Y. Wei, S. Lin, F. Liang and J. Zhang, Org. Lett., 2013, 15, 852 CrossRef CAS PubMed; (d) Y. Wei, F. Liang and X. Zhang, Org. Lett., 2013, 15, 5186 CrossRef CAS PubMed; (e) Y. Li, L. Zhang, H. Yuan, F. Liang and J. Zhang, Synlett, 2015, 26, 116 CAS.
- For a review of NBS activation by Lewis base, see: (a) S. E. Denmark, W. E. Kuester and M. T. Burk, Angew. Chem., Int. Ed., 2012, 51, 10938 CrossRef CAS PubMed ; Selected papers; (b) A. Sakakura, A. Ukai and K. Ishihara, Nature, 2007, 445, 900 CrossRef CAS PubMed; (c) S. A. Snyder and D. S. Treitler, Angew. Chem., Int. Ed., 2009, 48, 7899 CrossRef CAS PubMed; (d) X. L. Cui and R. S. Brown, J. Org. Chem., 2000, 65, 5653 CrossRef CAS PubMed; (e) X. Huang, M. Seid and J. W. Keillor, J. Org. Chem., 1997, 62, 7495 CrossRef CAS PubMed.
- A possible radical pathway was excluded via a control experiment with TEMPO as the radical trapping reagent. Also, the model reaction worked well under dark.
- For reviews on halogen bonding, see: (a) P. Metrangolo, H. Neukirch, T. Pilati and G. Resnati, Acc. Chem. Res., 2005, 38, 386 CrossRef CAS PubMed; (b) P. Auffinger, F. A. Hays, E. Westhof and P. S. Ho, Proc. Natl. Acad. Sci. U. S. A., 2004, 101, 16789 CrossRef CAS PubMed; (c) P. Metrangolo and G. Resnati, Chem.–Eur. J., 2001, 7, 2511 CrossRef CAS PubMed ; Selected papers on halogen bonding; (d) F. Sladojevich, E. McNeill, J. Börgel, S.-L. Zheng and T. Ritter, Angew. Chem., Int. Ed., 2015, 54, 3712 CrossRef CAS PubMed; (e) I. Castellote, M. Moron, C. Burgos, J. Alvarez-Builla, A. Martin, P. Gomez-Sal and J. J. Vaquero, Chem. Commun., 2007, 43, 1281 RSC; (f) K. Raatikainen and K. Rissanen, Chem. Sci., 2012, 3, 1235 RSC; (g) Y.-M. Wang, J. Wu, C. Hoong, V. Rauniyar and F. D. Toste, J. Am. Chem. Soc., 2012, 134, 12928 CrossRef CAS PubMed; (h) P. Metrangolo, F. Meyer, T. Pilati, G. Resnati and G. Terraneo, Angew. Chem., Int. Ed., 2008, 47, 6114 CrossRef CAS PubMed; (i) C. K. Tan, L. Zhou and Y.-Y. Yeung, Synlett, 2011, 10, 1335 Search PubMed; (j) F. Chen, C. K. Tan and Y.-Y. Yeung, J. Am. Chem. Soc., 2013, 135, 1232 CrossRef CAS PubMed.
- For an excellent review on halogen-bonded adducts are pre-reactive complexes, see: (a) A. C. Legon, Angew. Chem., Int. Ed., 1999, 38, 2686 CrossRef . Selected papers; (b) S. M. Walter, F. Kniep, E. Herdtweck and S. M. Huber, Angew. Chem., Int. Ed., 2011, 50, 7187 CrossRef CAS PubMed; (c) R. Castelli, S. Schindler, S. M. Walter, F. Kniep, H. S. Overkleeft, G. A. van der Marel, S. M. Huber and J. D. C. Codée, Chem.–Asian J., 2014, 9, 2095 CrossRef CAS PubMed; (d) F. Kniep, L. Rout, S. M. Walter, H. K. V. Bensch, S. H. Jungbauer, E. Herdtweck and S. M. Huber, Chem. Commun., 2012, 48, 9299 RSC.
- Both SN1 and SN2 pathway are possible, depending on different structure of alkyl halides employed.
- Halogen bonding has received relatively little attention from synthetic organic chemists. Early report: (a) A. Bruckmann, M. A. Pena and C. Bolm, Synlett, 2008, 06, 900 Search PubMed; (b) O. Coulembier, F. Meyer and P. Dubois, Polym. Chem., 2010, 1, 434 RSC ; Huber's work; (c) F. Kniep, S. M. Walter, E. Herdtweck and S. M. Huber, Chem.–Eur. J., 2012, 18, 1306 CrossRef CAS PubMed; (d) F. Kniep, S. H. Jungbauer, Q. Zhang, S. M. Walter, S. Schindler, I. Schnapperelle, E. Herdtweck and S. M. Huber, Angew. Chem., Int. Ed., 2013, 52, 7028 CrossRef CAS PubMed; (e) S. H. Jungbauer, S. M. Walter, S. Schindler, L. Rout, F. Kniep and S. M. Huber, Chem. Commun., 2014, 50, 6281 RSC; (f) S. H. Jungbauer, S. Schindler, F. Kniep, S. M. Walter, L. Rout and S. M. Huber, Synlett, 2013, 24, 2624 CrossRef CAS.
- For halogen cation stabilized by DMSO, see: (a) Y. Ashikari, A. Shimizu, T. Nokami and J. Yoshida, J. Am. Chem. Soc., 2013, 135, 16070 CrossRef CAS PubMed; (b) A. Shimizu, R. Hayashi, Y. Ashikari, T. Nokami and J. Yoshida, Beilstein J. Org. Chem., 2015, 11, 242 CrossRef PubMed; (c) S. Song, X. Sun, X. Li, Y. Yuan and N. Jiao, Org. Lett., 2015, 17, 2886 CrossRef CAS PubMed.
- That DBU binding to the halogen in any step will help us to understand why stoichiometric amount of DBU is required in the reaction.
- When KI-starch test paper was dipped into the reaction system, it turns blue after water treatment. This indicates the formation of dihalogen.
- The C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bond is tolerant under such conditions. One possible reason is no reaction occurs on the C
C double bond is tolerant under such conditions. One possible reason is no reaction occurs on the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bond due to the low concentration of the dihalogen. The other reason is halogenation takes place, followed by a rapid elimination. Thus, the net result is the C
C double bond due to the low concentration of the dihalogen. The other reason is halogenation takes place, followed by a rapid elimination. Thus, the net result is the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bond is intact.
C double bond is intact. - No second coupling (imidation) takes place.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra13708j |
| This journal is © The Royal Society of Chemistry 2015 |