Theoretical perspective of the lone pair activity influence on band gap and SHG response of lead borates†
Danni Liab,
Qun Jinga,
Chen Leiab,
Shilie Pana,
Bingbing Zhangab and
Zhihua Yang*a
aKey Laboratory of Functional Materials and Devices for Special Environments of CAS, Xinjiang Technical Institute of Physics & Chemistry of CAS, Xinjiang Key Laboratory of Electronic Information Materials and Devices, 40-1 South Beijing Road, Urumqi 830011, China. E-mail: zhyang@ms.xjb.ac.cn; Fax: +86-991-3835096; Tel: +86-991-3816801
bUniversity of Chinese Academy of Sciences, Beijing 100049, China
First published on 11th September 2015
Abstract
Metal lone pairs play an important role in determining the SHG enhancement and bandgap red shift. In this work, the electronic structures and optical properties of a class of lead borates Pb2B5O9Cl, BaPb[B5O9(OH)]·H2O, and Ba2Pb(B3O6)2 have been investigated to uncover the influence of the lead atom on the band gap and SHG response. It is found that BaPb[B5O9(OH)]·H2O and Ba2Pb(B3O6)2 have little bandgap redshift due to their weak distortion and stereochemical activity. The intensity of the lead lone pair stereochemical activity plays an important role in determining the reduction of the band-gap.
Introduction
The prerequisite for good ultraviolet (UV) or deep-ultraviolet (deep-UV) nonlinear optical (NLO) materials follows at least the following four criteria: wide band gap, high second-harmonic generation (SHG) response, appropriate large birefringence to meet the phase matching condition, good chemical stability and mechanical properties.1,2 Due to the rich chemical structures and good performances, borates have long been prominent sources of nonlinear optical (NLO) materials, such as β-BaB2O4 (BBO),3 LiB3O5 (LBO),4 KBe2BO3F2 (KBBF),5 CsLiB6O10 (CLBO),6 K3B6O10Cl (KBOC),7 Li4Sr(BO3)2.8 In order to obtain borates with large SHG intensity, a general strategy is to introduce asymmetric structural units such as the d0 metal centered distorted polyhedra or stereochemically active lone-pair anions.9–15 Inspired by this idea, lead borates have long been attracting increasing attentions and becoming one kind of important NLO materials with large SHG responses. However, it is found that the cost of good SHG performance is the narrow band gap, which limits the transmittance in the UV region. Typical examples possessing remarkable strong SHG responses are Pb2B5O9I (3.1 eV, 13.5 KDP),11 Pb4O(BO3)2 (3.3 eV, 3.0 KDP),16,17 Pb2(BO3)(NO3) (3.6 eV, 9 KDP),18 CsPbCO3F (4.12 eV, 13.4 KDP).19 Therefore, the key point to obtain lead borates used in UV region is getting the subtle balance between the SHG response and bandgap.In this paper, in order to clarify how to keep balance on the seesaw of the energy band gap and SHG coefficients, a class of lead borates Pb2B5O9Cl,20–22 Ba2Pb(B3O6)2 (ref. 23) and BaPb[B5O9(OH)]·H2O24 are studied. For comparison, the electronic structures of Ba2B5O9Cl,25 the isostructural virtual compound Ba2[B5O9(OH)]·H2O and Ba3(B3O6)2 were also investigated. It is found that two of these lead borates, Ba2Pb(B3O6)2 and BaPb[B5O9(OH)]·H2O have relative large band gaps and relative small band gap shift. The influence of the lead lone pair on the bandgap and SHG response will be discussed.
Numerical calculation details
Geometry optimization, electronic structures and optical properties were carried out by the density functional theory (DFT) method within the generalized gradient approximation (GGA) in the scheme of Perdew–Burke–Ernzerhof (PBE) functional,26 which was implemented in the CASTEP package.27–30 And the norm-conserving pseudopotential (NCP) was adopted,27 the following orbital electrons were treated as valence electrons: Pb 5s25p65d106s26p2, Cl 3s23p5, Ba 5s25p66s2, B 2s22p1, O 2s22p4, H 1s1. The number of plane waves included in the basis was determined by a cutoff energy of 910 eV (Pb2B5O9Cl, BaPb[B5O9(OH)]·H2O, Ba2B5O9Cl), and 820 eV (Ba2Pb(B3O6)2), throughout the calculations. Meanwhile, the Monk-horst–Pack k-point scheme of Pb2B5O9Cl, BaPb[B5O9(OH)]·H2O and Ba2Pb(B3O6)2 were set as 4 × 4 × 2, 4 × 4 × 2, 4 × 4 × 4 in the irreducible Brillouin zone. The optimized parameters are given in the ESI.†The length-gauge formalism derived by Aversa and Sipe was used to calculated the SHG coefficients at a zero frequency limit.31 The static second order coefficients χ(2)αβγ can be written as:32
| χ(2)αβγ = χ(2)αβγ(VE) + χ(2)αβγ(VH) + χ(2)αβγ(two-bands) | (1) |
In this sum-over-states type formalism, the total SHG coefficient χ(2) is divided into virtual-electron (VE) process, virtual-hole (VH) process and two-bands. And it should also be noted that the contribution of two-band process has been proved strictly to be zero.33 Hence, the new formalisms are represented as:
| χ(2)αβγ = χ(2)αβγ(VE) + χ(2)αβγ(VH) | (2) |
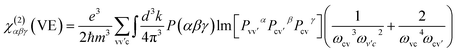 | (3) |
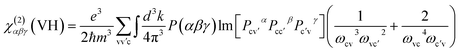 | (4) |
To show the distribution of the quantum states relevant to SHG, the SHG-density technique was implemented by using the effective SHG of each band (occupied and unoccupied) as weighting coefficient (after normalized with total VE or VH χ(2) value) to sum the probability densities of all occupied or unoccupied states. The effective SHG of each band was obtained by partially summing over two out of the three band indices for χ(2)αβγ (VE) or χ(2)αβγ (VH), and two kinds of summing sequences induce a consequent decomposition of the SHG strength into occupied and unoccupied band representations of orbital contributions. Therefore, the resulting distribution of SHG density represents a highlight of the origin of SHG optical nonlinearity in real space.
Results and discussion
Electronic structure and band gap
The crystal data of Pb2B5O9Cl, Ba2Pb(B3O6)2, BaPb[B5O9(OH)]·H2O and Ba2B5O9Cl are shown in Table S1 in the ESI.† Pb2B5O9Cl, Ba2B5O9Cl and BaPb[B5O9(OH)]·H2O crystallize in the acentric space group Pnn2, Pnn2 and Cc respectively. Pb2B5O9Cl and its isostructural Ba2B5O9Cl exhibit three-dimensional borate frameworks. The boron–oxygen groups of BaPb[B5O9(OH)]·H2O form a 2-D layer with 9 member ring. The Ba2Pb(B3O6)2 crystallizes into a trigonal system with centric space group R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) .
.
Using the method described above, the band structures of Pb2B5O9Cl, BaPb[B5O9(OH)]·H2O and Ba2Pb(B3O6)2 are obtained, and the calculated band gaps are 3.359 eV, 4.749 eV, 4.573 eV respectively. The calculated result shows that Ba2Pb(B3O6)2 and BaPb[B5O9(OH)]·H2O have relatively large band gaps. The tendency of calculated bandgaps is consistent well with the experimental values. The experimental values of Ba2Pb(B3O6)2 and BaPb[B5O9(OH)]·H2O are 5.17 eV and 6.20 eV, respectively. It is well known that introduction of the lead into alkaline-earth borates usually leads to a red-shift of the UV cutoff edge. Curiously, relative large bandgap was found in BaPb[B5O9(OH)]·H2O. Why?
In order to investigate the influence of the lead cations on the bandgap, the electronic structures of their isostructural compound Ba2B5O9Cl, Ba2[B5O9(OH)]·H2O and Ba3(B3O6)2 are studied for comparison. The virtual structures are obtained by substituting Pb atoms with Ba atoms and the electronic structure of these virtual structures are investigated after the geometry optimization. The convergence tolerance of Ba2[B5O9(OH)]·H2O and Ba3(B3O6)2 are shown in ESI.† The calculated band gaps of Ba2B5O9Cl, Ba2[B5O9(OH)]·H2O and Ba3(B3O6)2 are 5.315 eV, 4.958 eV and 4.549 eV, respectively. It is found that the gap difference between Pb2B5O9Cl and Ba2B5O9Cl is 1.956 eV. While the calculated band gaps of compounds Ba3(B3O6)2 and Ba2[B5O9(OH)]·H2O are close to that of Ba2Pb(B3O6)2 and BaPb[B5O9(OH)]·H2O, respectively. Furthermore the bandgap of Ga2[B5O9(OH)]·H2O,34 the isostructure of BaPb[B5O9(OH)]·H2O, with an experimental band gap about 6.2 eV also can indirectly show the little redshift of BaPb[B5O9(OH)]·H2O. This phenomenon indicates that introducing of lead in borates will not always reduce the band gap like most of lead borates do. To visually show the redshift of the cutoff edge, a factor Δλ = λ(I) − λ(II) is defined to clarify the redshift of the wavelength in which λ(I), and λ(II) are the wavelength cutoff edge of the one containing lead atoms (compound II) and its isostructural alkaline-earth borates (compound I). The results show that the shift Δλ from BaPb[B5O9(OH)]·H2O to Ba2[B5O9(OH)]·H2O is 12.36 nm (GGA), and the redshift from Ba2Pb(B3O6)2 to Ba3(B3O6)2 is almost zero. While, for Pb2B5O9Cl (3.359 eV), and its isostructural Ba2B5O9Cl (5.315 eV),25,35 the Δλ is as large as 135.79 nm. It is well known that the GGA-PBE in the standard DFT always underestimates the band gap.36 To get reliable results, the bandgaps of BaPb[B5O9(OH)]·H2O and the virtual compound Ba2[B5O9(OH)]·H2O were recalculated using the hybrid XC functional PBE0 again. And the bandgaps of BaPb[B5O9(OH)]·H2O and Ba2[B5O9(OH)]·H2O calculated by PBE0 are 6.896 eV, 7.353 eV, respectively, and Δλ from BaPb[B5O9(OH)]·H2O to Ba2[B5O9(OH)]·H2O is 11.17 nm, which has a similar tendency in band gap changes with GGA calculations. The calculated band gap for BaPb[B5O9(OH)]·H2O by PBE0 is in accord with the experimental value.24 The values of Δλ indicate that BaPb[B5O9(OH)]·H2O and Ba2Pb(B3O6)2 have less red-shift than Pb2B5O9Cl when barium is replaced by lead atoms in the compounds.
As BaPb[B5O9(OH)]·H2O owns an acentric structure and large band gap, more attention will be paid on it and its isostructural Ba2[B5O9(OH)]·H2O. In order to further observe the influence of lead cation on the band gap, the HOMO (highest occupied molecular orbital) and LUMO (lowest unoccupied molecular orbital) (shown in Fig. 1 and 2) are analyzed. Fig. 1 shows that the oxygen atoms surround the lead atom are crucial for the HOMO of BaPb[B5O9(OH)]·H2O, whereas, the contribution of the LUMO mainly comes from the hybrid orbitals of Pb, O9 and O11. Noting that O9 is terminal oxygen of BO3 group, and O11 belongs to water molecules. Comparatively, in Ba2[B5O9(OH)]·H2O, the oxygen atoms at the LUMO are not only the O9, O11 but also O10, and the metallic cation barium is not the decisive element for LUMO. As for HOMO, compared with BaPb[B5O9(OH)]·H2O, when the lead atom was substituted by the barium, it is observed that there is no contribution from the oxygen atoms O2, O7, O11 in Ba2[B5O9(OH)]·H2O. Thus, one can find that the Pb atoms affect the band gap, but such influence does not make the band gap of BaPb[B5O9(OH)]·H2O red-shift too much. So what does this phenomenon happen? To address this issue, the structure and density of states (DOS) of three lead containing compounds are analyzed.
Generally speaking, microscopic structures influence the macroscopic properties. To better understand the emergence of a large band gap, the coordination environment of the lead atom is firstly considered. Fig. 3 shows the coordination environment of Pb atom with the bond length less than 3.1 Å. The Pb1 and Pb2 of compound Pb2B5O9Cl are both surrounded by seven oxygen atoms, which distributes similarly as a plane. And five of the oxygen atoms around Pb1 show obvious directivity by one side. The PbO8 polyhedron of BaPb[B5O9(OH)]·H2O is falling with the sphere around lead. And the hexa-coordinated lead of compound Ba2Pb(B3O6)2 exhibits a symmetric distribution with no stereochemical active. It is known that stereochemical active cause distortion, to quantitatively analyze, the distortion of lead–oxygen polyhedron in compounds Pb2B5O9Cl, Ba2Pb(B3O6)2, and BaPb[B5O9(OH)]·H2O are calculated. The distortion value of two different lead atoms Pb1 and Pb2 of compound Pb2B5O9Cl are 4.023 and 2.683. The distortion value of Ba2Pb(B3O6)2, BaPb[B5O9(OH)]·H2O are 0.002, 0.982 respectively. The results show that the distortion of Ba2Pb(B3O6)2, BaPb[B5O9(OH)]·H2O are much smaller than Pb2B5O9Cl. The degree of the distortion of the lead–oxygen polyhedral in these lead borates is shown in Fig. 4, along with the redshift of the bandgaps of these compounds. As shown in Fig. 4, similar tendency can be found for the distortion of lead–oxygen polyhedron and the redshift of bandgap, indicating the more distortion value the lead–oxygen polyhedral are, the larger redshift the lead borate owns.
 | ||
| Fig. 3 The coordination of lead atom (the bond length of Pb–O is in the range less than 3.1 Å are drawn). (a) Pb1 and (b) Pb2 in Pb2B5O9Cl, (c) BaPb[B5O9(OH)]·H2O, (d) Ba2Pb(B3O6)2. | ||
 | ||
| Fig. 4 The relationship between the distortion of Pb–O polyhedron and the redshift of band gaps of lead borates. | ||
To better understand the internal mechanism of how the more largely distorted Pb2+ cause smaller gaps for lead borates, the electronic structures of these lead borates were then calculated. The calculated project density of states (PDOS) of Pb2B5O9Cl, Ba2Pb(B3O6)2, and BaPb[B5O9(OH)]·H2O are shown in Fig. 5. It is clearly shown that the bands spanning from about −9 eV to the Fermi level of these compounds are mainly derived from O 2p with small B 2p, B s components, and there are also some contributions from Pb s, Pb p states at the top of the valance band (VB) near the Fermi level. The states at the bottom of the conduction band (CB) are mainly the 2p states of lead. An overlap between Pb p and O 2p states was found at the energy region close to the Fermi level. It is clearly shown that the Pb atom plays a decisive role in determining the band gap of these three compounds.
On the other hand, according to the lone pair model,37,38 due to the interaction of Pb and O, an mixture between Pb 6s and O 2p states exists, producing a filled antibonding state with some Pb 6s character at the top of the VB. Further mixing between the antibonding state and empty Pb 6p states is responsible for the distortion of Pb–O group. Therefore, the asymmetric electron density for a distorted Pb–O group arises predominantly from a mix of O 2p and Pb 6p orbitals at the top of the VB. At the top of the VB of BaPb[B5O9(OH)]·H2O there is a small mixing between O 2p and Pb 6p orbitals which indicates a weak interaction between lead and oxygen. The PDOS of Ba2Pb(B3O6)2 at the top of VB has a similar tendency with BaPb[B5O9(OH)]·H2O, there is also no obvious Pb 6p at the top of the VB in Fig. 5(b). In contrast, it shows that there is an apparent Pb p state with Pb s state at the top VB of Pb2B5O9Cl (Fig. 5(c)). The strength of stereochemical lone-pair activity was then calculated by the contribution of Pb p state. The contribution ratio39 of BaPb[B5O9(OH)]·H2O, Ba2Pb(B3O6)2 and Pb2B5O9Cl are 0.0399, 0.0277 and 0.1348 respectively, which indicates that compound Pb2B5O9Cl has a stronger stereochemical activity. In addition, the electron localization function (ELF) of BaPb[B5O9(OH)]·H2O are also calculated, Fig. 6 shows that the electron localization are non-spherical distributed around Pb but no asymmetric lobes on it, which proves that there is no obvious stereochemical lone-pair activity of BaPb[B5O9(OH)]·H2O. Comparatively, compound Pb2B5O9Cl has a larger lead–oxygen polyhedron distortion and distribution of O 2p state, but a smaller band gap. Comparing with the project density of states and distortion values, one can deduce that the larger the distortion of lead–oxygen polyhedral owns, the stronger interaction between lead and oxygen has. And because the bandgap is determined by the lead–oxygen states, the relatively strong interaction makes lead borates own smaller gaps. In a word the intensity of stereochemical activity is one reason leading to reduction of the band-gap.
SHG effect
As BaPb[B5O9(OH)]·H2O has a little redshift of UV cutoff edge due to the weak stereochemical activity of lead lone pair, the influence of lead to SHG response is investigated. Here the SHG coefficients are calculated firstly, the result shows 3.2 × KDP for the largest component, which is almost in accord with experimental value of 2.2 × KDP. To observe the SHG coefficient difference between BaPb[B5O9(OH)]·H2O and Ba2[B5O9(OH)]·H2O, the SHG coefficient tensors of Ba2[B5O9(OH)]·H2O are also calculated, the maximal tensor element is 1.7 × KDP. It is also found that all the SHG coefficients are decreased when the lead atom is substituted by the barium atom (Table 1).| d11 | d15 | d12 | d13 | d24 | d33 | |
|---|---|---|---|---|---|---|
| I | −1.26 | −0.76 | 0.96 | −0.66 | −0.52 | 0.86 |
| II | −0.34 | −0.45 | 0.56 | −0.01 | −0.38 | 0.67 |
The contribution of total SHG coefficients can be divided into two processes, virtual-electron and virtual-hole. The virtual-electron contribution to the total SHG coefficients of BaPb[B5O9(OH)]·H2O are 76.91% (d11), 73.38% (d15), 84.87% (d12), 58.13% (d13), 84.51% (d24), 114.8% (d33). Here we will just show the SHG-density for virtual-electron process of the largest SHG tensors d11 to discuss the original of the SHG. Fig. 7 illustrate that the oxygen atoms have a dominant contribution to the SHG coefficients for both virtual-electron occupied states and unoccupied states. And the Pb atom takes an important role in the virtual-electron contribution for unoccupied states. In Fig. 7(a), it is observed that the SHG density of the oxygen atom on occupied states concentrate on about 9.151 × 10−3, and the density of unoccupied states (Fig. 7(b)) shows that the density of Pb cations is approximate 2.231 × 10−3. Hence, the Pb cations contribute to SHG response but not the dominator.
 | ||
| Fig. 7 The SHG-density of the virtual-electron of the largest SHG tensors of BaPb[B5O9(OH)]·H2O. (a) Occupied process (b) unoccupied process. | ||
Based on the experimental values, it is suggested that the BaPb[B5O9(OH)]·H2O has a relatively large band gap that can reach UV region. The SHG response of BaPb[B5O9(OH)]·H2O also can meet the need of the UV application. Through theoretical calculation and simulation, the introduction of lead atom makes a little redshift of the cutoff edge of BaPb[B5O9(OH)]·H2O but indirectly increases to the intensity of SHG response. This balance of band gap and SHG is related to the little stereochemical activity of the lead atom.
Conclusions
In conclusion, BaPb[B5O9(OH)]·H2O, Pb2B5O9Cl, Ba2Pb(B3O6)2, and their isostructual compounds “Ba2[B5O9(OH)]·H2O”, Ba2B5O9Cl and “Ba3(B3O6)2” are studied mainly via DFT calculation. Analyzing the structure and electronic structure of these compounds, we found that the lead containing borates with little red-shift also has a small distortion and weak stereochemical activity. Futhermore, the SHG-density of BaPb[B5O9(OH)]·H2O also shows that the lead atom has a contribution to its SHG response. Therefore, by controlling the intensity of stereochemical activity, one may obtain lead borates with large SHG response and wide transmittance or lead-free borates with moderate SHG response and wide transmittance. This balance of the band gap and SHG response can offer guidance to material design and experimental synthesis.Acknowledgements
This work is supported by the National Key Basic Research Program of China (Grant No. 2014CB648400), the National Natural Science Foundation of China (Grant No. 11104344, 51425206, U1129301), the Western Light Joint Scholar Foundation Program of Chinese Academy of Sciences (Grant No. LHXZ201101), and the High-level Professional and Technical Personnel of Autonomous region.Notes and references
- C. T. Chen, T. Sasaki, R. K. Li, Y. C Wu, Z. S. Lin, Y. Mori, Z. G. Hu and J. Y. Wang, Nonlinear Optical Borate Crystals, Principles and Applications, WILEY-VCH, 2012 Search PubMed.
- J. F. Nye, Physical Properties of Crystals, Oxford University Press, United States, 1957 Search PubMed.
- C. T. Chen, B. C. Wu, A. D. Jiang and G. M. You, Sci. Sin., Ser. B, 1985, 28, 235–243 Search PubMed.
- C. T. Chen, Y. C. Wu, A. D. Jiang, B. C. Wu, G. M. You, R. K. Li and S. J. Lin, J. Opt. Soc. Am. B, 1989, 6, 616–621 CrossRef CAS.
- C. T. Chen, Y. B. Wang, B. C. Wu, K. C. Wu, W. L. Zeng and L. H. Yu, Nature, 1995, 373, 322–324 CrossRef CAS PubMed.
- Y. Mori, I. Kuroda, S. Nakajima, T. Sasaki and S. Nakai, Appl. Phys. Lett., 1995, 67, 1818–1820 CrossRef CAS PubMed.
- H. P. Wu, S. L. Pan, K. R. Poeppelmeier, H. Y. Li, D. Z. Jia, Z. H. Chen, X. Y. Fan, Y. Yang, J. M. Rondinelli and H. S. Luo, J. Am. Chem. Soc., 2011, 133, 7786–7790 CrossRef CAS PubMed.
- S. G. Zhao, P. F. Gong, L. Bai, X. Xu, S. Q. Zhang, Z. H. Sun, Z. S. Lin, M. C. Hong, C. T. Chen and J. H. Luo, Nat. Commun., 2014, 5, 4019 CAS.
- W. L. Zhang, W. D. Cheng, H. Zhang, L. Geng, C. S. Lin and Z. Z. He, J. Am. Chem. Soc., 2010, 132, 1508–1509 CrossRef CAS PubMed.
- R. E. Sykora, K. M. Ok, P. S. Halasyamani and T. E. Albrecht-Schmitt, J. Am. Chem. Soc., 2002, 124, 1951–1957 CrossRef CAS PubMed.
- Y. Z. Huang, L. M. Wu, X. T. Wu, L. H. Li, L. Chen and Y. F. Zhang, J. Am. Chem. Soc., 2010, 132, 12788–12789 CrossRef CAS PubMed.
- E. O. Chi, K. M. Ok, Y. Porter and P. S. Halasyamani, Chem. Mater., 2006, 18, 2070–2074 CrossRef CAS.
- D. Phanon and I. Gautier-Luneau, Angew. Chem., Int. Ed., 2007, 46, 8488–8491 CrossRef CAS PubMed.
- H. S. Ra, K. M. Ok and P. S. Halasyamani, J. Am. Chem. Soc., 2003, 125, 7764–7765 CrossRef CAS PubMed.
- H. Y. Chang, S. H. Kim, P. S. Halasyamani and K. M. Ok, J. Am. Chem. Soc., 2009, 131, 2426–2427 CrossRef CAS PubMed.
- H. W. Yu, S. L. Pan, H. P. Wu, W. W. Zhao, F. F. Zhang, H. Y. Li and Z. H. Yang, J. Mater. Chem., 2012, 22, 2105–2110 RSC.
- Z. H. Yang, S. L. Pan, H. W. Yu and M. H. Lee, J. Solid State Chem., 2013, 198, 77–80 CrossRef CAS PubMed.
- J. L. Song, C. L. Hu, X. Xu, F. Kong and J. G. Mao, Angew. Chem., Int. Ed., 2015, 54, 3679–3682 CrossRef CAS PubMed.
- G. H. Zou, L. Huang, N. Ye, C. S. Lin, W. D. Cheng and H. Huang, J. Am. Chem. Soc., 2013, 135, 18560–18566 CrossRef CAS PubMed.
- B. V. Egorova, A. V. Olenev, P. S. Berdonosov, A. N. Kuznetsov, S. Y. Stefanovich, V. A. Dolgikh, T. Mahenthirarajah and P. Lightfoot, J. Solid State Chem., 2008, 181, 1891–1898 CrossRef CAS PubMed.
- Q. Jing, X. Y. Dong, X. L. Chen, Z. H. Yang, S. L. Pan and C. Lei, Chem. Phys., 2015, 453, 42–46 CrossRef PubMed.
- Z. H. Chen, S. L. Pan, Z. H. Yang, X. Y. Dong, X. Su and Y. Yang, J. Mater. Sci., 2013, 48, 2590–2596 CrossRef CAS.
- H. Y. Li, L. Y. Dong, Y. Lu, S. L. Pan, X. Q. Lu, H. W. Yu, H. P. Wu, X. Su and Z. H. Yang, J. Alloys Compd., 2014, 615, 561–565 CrossRef CAS PubMed.
- H. Q. Wu, H. He, B. F. Yang and G. Y. Yang, Inorg. Chem. Commun., 2013, 37, 77–79 CrossRef PubMed.
- P. Held, J. Liebertz and L. Bohaty, Z. Kristallogr.–New Cryst. Struct., 2002, 217, 463–464 CAS.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865–3868 CrossRef CAS.
- A. M. Rappe, K. M. Rabe, E. Kaxiras and J. D. Joannopoulos, Phys. Rev. B: Condens. Matter Mater. Phys., 1990, 41, 1227–1230 CrossRef.
- S. J. Clark, M. D. Segall, C. J. Pickard, P. J. Hasnip, M. J. Probert, K. Refson and M. C. Payne, Z. Kristallogr., 2005, 220, 567–570 CrossRef CAS.
- B. G. Pfrommer, M. Cote, S. G. Louie and M. L. Cohen, J. Comput. Phys., 1997, 131, 233–240 CrossRef CAS.
- J. S. Lin, A. Qteish, M. C. Payne and V. Heine, Phys. Rev. B: Condens. Matter Mater. Phys., 1993, 47, 4174–4180 CrossRef.
- C. Aversa and J. E. Sipe, Phys. Rev. B: Condens. Matter Mater. Phys., 1995, 52, 14636–14645 CrossRef CAS.
- J. Lin, M. H. Lee, Z. P. Liu, C. T. Chen and C. J. Pickard, Phys. Rev. B: Condens. Matter Mater. Phys., 1999, 60, 13380–13389 CrossRef CAS.
- B. B. Zhang, M. H. Lee, Z. H. Yang, Q. Jing, S. L. Pan, M. Zhang, H. P. Wu, X. Su and C. S. Li, Appl. Phys. Lett., 2015, 106, 031906 CrossRef PubMed.
- Q. Wei, J. W. Cheng, C. He and G. Y. Yang, Inorg. Chem., 2014, 53, 11757–11763 CrossRef CAS PubMed.
- Z. H. Chen, S. L. Pan, W. W. Zhao, Y. Yang, H. P. Wu and F. F. Zhang, J. Synth. Cryst., 2011, 40, 1394–1398 CAS.
- M. K. Y. Chan and G. Ceder, Phys. Rev. Lett., 2010, 105, 196403 CrossRef CAS.
- D. J. Payne, R. G. Egdell, A. Walsh, G. W. Watson, J. Guo, P.-A. Glans, T. Learmonth and K. E. Smith, Phys. Rev. Lett., 2006, 96, 157403 CrossRef CAS.
- A. Walsh, D. J. Payne, R. G. Egdell and G. W. Watson, Chem. Soc. Rev., 2011, 40, 4455–4463 RSC.
- Q. Jing, Z. H. Yang, S. L. Pan and D. F. Xue, Phys. Chem. Chem. Phys., 2015, 17, 21968–21973 RSC.
Footnote |
| † Electronic supplementary information (ESI) available: The convergence tolerance, calculated band structure, lattice parameters and optimized parameter. See DOI: 10.1039/c5ra13647d |
| This journal is © The Royal Society of Chemistry 2015 |




