Aggregation induced emission and mechanochromism in tetraphenylethene substituted pyrazabole†
Thaksen Jadhav,
Bhausaheb Dhokale,
Yuvraj patil and
Rajneesh Misra*
Department of Chemistry, Indian Institute of Technology, Indore 452 017, India. E-mail: rajneeshmisra@iiti.ac.in
First published on 3rd August 2015
Abstract
Tetraphenylethene (TPE) substituted pyrazabole 2 was designed and synthesized by the Pd-catalyzed Suzuki cross-coupling reaction. The pyrazabole exhibits strong aggregation induced emission (AIE) and reversible mechanochromic behavior with high color contrast between blue and green. The powder XRD studies show that destruction of crystalline state into amorphous state is responsible for mechanochromism.
Introduction
In recent years research on design and synthesis of organic molecules exhibiting AIE and mechanochromism has gained momentum due to their applications in mechano-sensors, optical data storage, and security papers.1 Molecular systems exhibiting strong solid state fluorescence and high color contrast are essential for mechanochromism.2 The conventional organic fluorescent dyes are non-fluorescent in solid state due to the notorious effect called aggregation caused quenching (ACQ).3 Tang et al. has introduced the concept of aggregation induced emission (AIE) to overcome the ACQ.4 Generally, the propeller-like conformation of AIE active molecules restrict tight packing in the aggregated state, which minimizes the chance of intermolecular π–π stacking and results in highly fluorescent solid.4The tetraphenylethene is AIE active molecule and widely studied for numerous applications.5 However, there is huge space for developing TPE-based functional materials. The TPE derivatives, containing electron donor as well as acceptor moieties have been used to develop piezochromic, mechanochromic, vapochromic, thermochromic, and solvatochromic materials.1,5,6 Our group is involved in design and synthesis of mechanochromic materials.7 Recently we have shown, that the TPE substituted phenanthroimidazole and benzothiazole can be used as mechanochromic materials. The literature reveals that, variety of weak acceptor units (phenanthroimidazole, benzothiazole, aldehyde and triazolyl units) have been attached to weak donor TPE for synthesizing mechanochromic materials.6–8 We were interested to see the effect of TPE substitution on the pyrazabole.
The organoboron compounds are of great interest due to their potential applications in OLEDs, organic photovoltaics (OPVs), organic field effect transistor (OFET), and NLO materials.9 Pyrazabole is a class of organoboron compound, which is synthesized by condensation of two pyrazole units with triabutylboranes. The pyrazabole act as a weak acceptor and has been explored for variety of applications such as liquid crystalline material, multiphoton absorption, and luminescent polymers.10 Recently we have reported high two photon absorption (2PA) and three photon absorption (3PA) in ferrocenyl substituted pyrazaboles.11
Herein, we have attached TPE unit on the pyrazabole through single bond and explored its AIE and mechanochromic properties. The TPE substituted pyrazabole exhibits strong blue colored emission upon aggregation, and highly reversible mechanochromism between blue and green color.
Result and discussion
The TPE substituted pyrazabole 2 was synthesized by the Pd-catalyzed Suzuki cross-coupling reaction of 2,6-diiodopyrazabole with the 4-(1,2,2-triphenylvinyl)phenylboronic acid pinacol ester in 45% yield (Scheme 1). The precursors 2,6-diiodopyrazabole and 4-(1,2,2-triphenylvinyl)phenylboronic acid pinacol ester were synthesized by reported procedures.11,12 The TPE substituted pyrazabole was well characterized by usual spectroscopic techniques. Thermo gravimetric analysis (TGA) of TPE substituted pyrazabole show good thermal stability with thermal decomposition temperature (Td) at 307 °C corresponding to 5% weight loss under nitrogen atmosphere (Fig. S1†).In order to understand the geometrical structure of the pyrazabole 2, computational calculation was performed using density functional theory (DFT) at the B3LYP/6-31G(d) level using the Gaussian 09 program.13 The energy minimized structure of the pyrazabole 2 show twisted geometry for TPE unit, while pyrazabole core show planar geometry (Fig. 1). The central core of pyrazabole show chair conformation. The pyrazabole and TPE units are non-planar with dihedral angle of 27° between pyrazabole and phenyl of TPE.
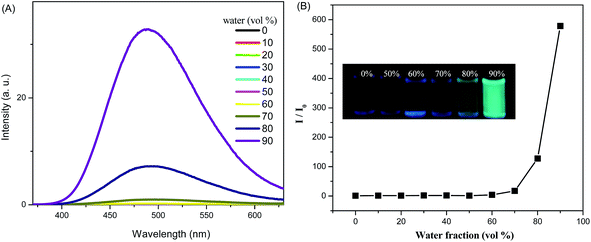
The electronic absorption spectra of the TPE substituted pyrazabole 2 exhibit absorption band between 250–380 nm. The TPE substituted pyrazabole 2 is weakly fluorescent in solution, due to free molecular rotations of phenyl rings of tetraphenylethylene unit. The AIE property of TPE substituted pyrazabole was studied by absorption and fluorescence spectroscopy (Fig. 2 and S2†). The TPE substituted pyrazabole is highly soluble in tetrahydrofuran (THF) and insoluble in water, therefore the small aggregate particles were prepared by increasing the water fraction in the THF–water mixture. In pure THF solution the TPE substituted pyrazabole 2 exhibit poor fluorescence quantum yield, which remains constant until the molecules start aggregating significantly (AIE effect). The fluorescence of TPE substituted pyrazabole in THF–water mixture remains constant upto the 60% water fraction (fw) and above 60% fw molecules started aggregating with enhanced fluorescence. At 90% water fraction the fluorescence intensity was enhanced by 578 fold (Fig. 2). The photograph of 2 in THF–water mixtures with different water fractions under 365 nm UV illumination shows the AIE behavior (Fig. 2). The study of absorption behavior of AIE show noticeable change after 60% water fraction, and started to show light scattering of the nanoaggregate suspension in the THF–water mixtures (Fig. S2†).
In order to determine the dimensions of the nano-aggregates formed, the nanoaggregates at 90% water fraction (fw) were studied with the help of dynamic light scattering (DLS) and scanning electron microscopy (SEM). The DLS study shows the average hydrodynamic diameter of 180 nm for nano-aggregates (Fig. 3). The SEM study reveals rectangular sized nanoaggregates were formed with average size of ∼115 nm. These studies confirm the formation of nanoaggregates at 90% (fw).
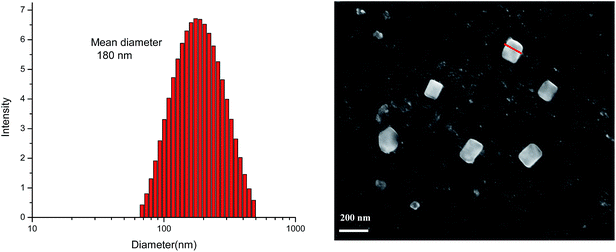 | ||
Fig. 3 The particle size distributions of 2 in THF–water mixture (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 90, v 90, v![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) v) (left) and SEM image (right). v) (left) and SEM image (right). | ||
The mechanochromic properties of TPE substituted pyrazabole 2 was explored by the emission studies. The pristine form of 2 emits blue light at 453 nm which upon grinding using a spatula or a pestle exhibits drastic 44 nm change in the emission behavior and emits green light at 497 nm (Fig. 4). This color contrast mechanochromic effect of TPE substituted pyrazabole 2 can be reverted to its original color either by annealing or fuming with dichloromethane vapor. The grinded form of 2 upon annealing at 150 °C for 5 min or fuming with dichloromethane vapor for 4 min restored the original blue emission. This reversible mechanochromic behavior can be repeated between blue and green emission and proves these stimuli does not cause any chemical change (Fig. S3†).
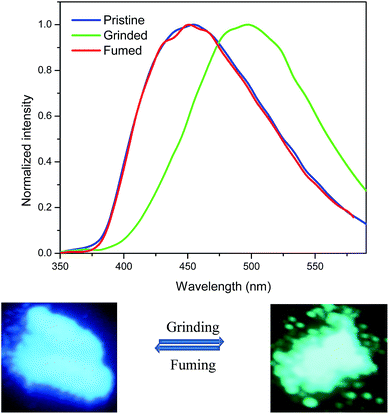 | ||
| Fig. 4 Emission spectra of 2 as pristine, grinded and fumed solids and photograph taken under 365 nm UV illumination. | ||
In order to understand the mechanism of mechanochromism in TPE substituted pyrazabole 2 powder X-ray diffraction (PXRD) analysis was performed (Fig. S4†). The pristine form of pyrazabole 2 exhibit intense and sharp diffraction peaks reflecting the crystalline behavior. Upon grinding pyrazabole 2 show weak and diffused diffraction peaks suggesting the transition to amorphous state. The grinded form of pyrazabole 2 when subjected to heating or fuming, the sharp diffraction peaks were observed again indicating the transformation of amorphous state of sample to crystalline state. This result concludes that the mechanochromism in pyrazabole 2 is associated with the morphological change from the crystalline state to the amorphous state and vice versa.
Conclusion
In summary, we have designed and synthesized AIE active mechanochromic TPE substituted pyrazabole 2 by the Pd-catalyzed Suzuki cross-coupling reaction. The geometry optimized structure shows twisted geometry of TPE unit. The AIE study reveals remarkable 578 folds enhancement in the emission intensity at 90% water fraction. The TPE substituted pyrazabole 2 show highly reversible mechanochromism between blue and green light with 44 nm grinding induced spectral shift. The powder XRD results indicate the transformation from crystalline to amorphous state is responsible for mechanochromism. The results obtained here will help in design of new mechanochromic molecules for various optoelectronic applications.Experimental details
General methods
Chemicals were used as received unless otherwise indicated. All oxygen or moisture sensitive reactions were performed under nitrogen/argon atmosphere. 1H NMR (400 MHz), and 13C NMR (100 MHz) spectra were recorded on the Bruker Avance (III) 400 MHz instrument by using CDCl3. 1H NMR chemical shifts are reported in parts per million (ppm) relative to the solvent residual peak (CDCl3, 7.26 ppm). Multiplicities are given as: s (singlet), d (doublet), t (triplet), m (multiplet), and the coupling constants J are given in Hz. 13C NMR chemical shifts are reported relative to the solvent residual peak (CDCl3, 77.0 ppm). Thermogravimetric analyses were performed on the Mettler Toledo Thermal Analysis system. UV-visible absorption spectra were recorded on a Cary-100 Bio UV-visible Spectrophotometer. Emission spectra were taken in a fluoromax-4p fluorimeter from HoribaYovin (model: FM-100). The excitation and emission slits were 2/2 nm for the emission measurements. All of the measurements were done at 25 °C.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethanol (8
ethanol (8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) mixtures. Yield: 42.0%. 1H NMR (400 MHz, CDCl3, 25 °C): δ 7.75 (s, 4H), 7.30 (d, 4H, J = 4 Hz), 7.02–7.17 (m, 34H), 1.13–1.19 (m, 8H), 0.65–0.82 (m, 28H) ppm; 13C NMR (100 MHz, CDCl3, 25 °C): δ 143.7, 143.7, 143.6, 142.7, 141.1, 140.3, 132.0, 131.4, 131.3, 130.1, 129.2, 127.8, 127.7, 127.6, 126.5, 126.5, 126.4, 124.8, 122.7, 27.5, 26.2, 14.1, 0.00 ppm.
2) mixtures. Yield: 42.0%. 1H NMR (400 MHz, CDCl3, 25 °C): δ 7.75 (s, 4H), 7.30 (d, 4H, J = 4 Hz), 7.02–7.17 (m, 34H), 1.13–1.19 (m, 8H), 0.65–0.82 (m, 28H) ppm; 13C NMR (100 MHz, CDCl3, 25 °C): δ 143.7, 143.7, 143.6, 142.7, 141.1, 140.3, 132.0, 131.4, 131.3, 130.1, 129.2, 127.8, 127.7, 127.6, 126.5, 126.5, 126.4, 124.8, 122.7, 27.5, 26.2, 14.1, 0.00 ppm.Acknowledgements
We acknowledge the support by DST, and CSIR Govt. of India, New Delhi. T. J. thanks UGC, B. D. thanks to CSIR and Y. P. thanks MHRD New Delhi for their fellowships. We acknowledge Sophisticated Instrumentation Centre (SIC) facility, IIT Indore.References
- (a) Z. Chi, X. Zhang, B. Xu, X. Zhou, C. Ma, Y. Zhang, S. Liu and J. Xu, Chem. Soc. Rev., 2012, 41, 3878–3896 RSC; (b) Y. Sagara and T. Kato, Nat. Chem., 2009, 1, 605 CrossRef CAS PubMed; (c) D. A. Davis, A. Hamilton, J. L. Yang, L. D. Cremar, D. van Gough, S. L. Potisek, M. T. Ong, P. V. Braun, T. J. Martinez, S. R. White, J. S. Moore and N. R. Sottos, Nature, 2009, 459, 68 CrossRef CAS PubMed; (d) Y. J. Dong, B. Xu, J. B. Zhang, X. Tan, L. J. Wang, J. L. Chen, H. G. Lv, S. P. Wen, B. Li, L. Ye, B. Zou and W. J. Tian, Angew. Chem., Int. Ed., 2012, 51, 10782 CrossRef CAS PubMed; (e) X. L. Luo, J. N. Li, C. H. Li, L. P. Heng, Y. Q. Dong, Z. P. Liu, Z. S. Bo and B. Z. Tang, Adv. Mater., 2011, 23, 3261 CrossRef CAS PubMed; (f) B. Dhokale, T. Jadhav, S. M. Mobin and R. Misra, J. Org. Chem. DOI:10.1021/acs.joc.5b01117.
- M. S. Kwon, J. Gierschner, S. J. Yoon and S. Y. Park, Adv. Mater., 2012, 24, 5487 CrossRef CAS PubMed.
- (a) J. B. Birks, Photophysics of Aromatic Molecules, Wiley, London, U.K, 1970 Search PubMed; (b) S. Jayanty and T. P. Radhakrishnan, Chem.–Eur. J., 2004, 10, 791 CrossRef CAS PubMed; (c) H. J. Tracy, J. L. Mullin, W. T. Klooster, J. A. Martin, J. Haug, S. Wallace, I. Rudloe and K. Watts, Inorg. Chem., 2005, 44, 2003 CrossRef CAS PubMed.
- (a) J. D. Luo, Z. L. Xie, J. W. Y. Lam, L. Cheng, H. Y. Chen, C. F. Qiu, H. S. Kwok, X. W. Zhan, Y. Q. Liu, D. B. Zhu and B. Z. Tang, Chem. Commun., 2001, 1740 RSC; (b) Y. Li, T. Liu, H. Liu, M. Z. Tian and Y. Li, Acc. Chem. Res., 2014, 47, 1186 CrossRef CAS PubMed; (c) H. Liu, J. Xu and Y. Li, Acc. Chem. Res., 2010, 43, 1496–1508 CrossRef CAS PubMed; (d) Y. Hong, J. W. Y. Lam and B. Z. Tang, Chem. Soc. Rev., 2011, 40, 5361 RSC.
- (a) Y. Dong, J. W. Y. Lam, A. Qin, J. Liu, Z. Li, B. Z. Tang, J. Sun and H. S. Kwok, Appl. Phys. Lett., 2007, 91, 011111 CrossRef PubMed; (b) J. Huang, X. Yang, J. Wang, C. Zhong, L. Wang, J. Qin and Z. Li, J. Mater. Chem., 2012, 22, 2478 RSC; (c) N. Zhao, Z. Y. Yang, J. W. Y. Lam, H. H. Y. Sung, N. Xie, S. J. Chen, H. M. Su, M. Gao, I. D. Williams, K. S. Wong and B. Z. Tang, Chem. Commun., 2012, 48, 8637 RSC; (d) N. Zhao, M. Li, Y. L. Yan, J. W. Y. Lam, Y. L. Zhang, Y. S. Zhao, K. S. Wong and B. Z. Tang, J. Mater. Chem. C, 2013, 1, 4640 RSC; (e) B. Xu, M. Xie, J. He, B. Xu, Z. Chi, W. Tian, L. Jiang, F. Zhao, S. Liu, Y. Zhang, Z. Xu and J. Xu, Chem. Commun., 2013, 49, 273 RSC; (f) B. Xu, Z. Chi, X. Zhang, H. Li, C. Chen, S. Liu, Y. Zhang and J. Xu, Chem. Commun., 2011, 47, 11080 RSC.
- (a) X. Y. Shen, Y. J. Wang, E. Zhao, W. Z. Yuan, Y. Liu, P. Lu, A. Qin, Y. Ma, J. Z. Sun and B. Z. Tang, J. Phys. Chem. C, 2013, 117, 7334–7347 CrossRef CAS; (b) B. Xu, J. He, Y. Mu, Q. Zhu, S. Wu, Y. Wang, Y. Zhang, C. Jin, C. Lo, Z. Chi, A. Lien, S. Liu and J. Xu, Chem. Sci., 2015, 6, 3236–3241 RSC.
- (a) R. Misra, T. Jadhav, B. Dhokale and S. M. Mobin, Chem. Commun., 2014, 50, 9076–9078 RSC; (b) T. Jadhav, B. Dhokale, S. M. Mobin and R. Misra, RSC Adv., 2015, 5, 29878–29884 RSC; (c) P. Gautam, R. Maragani, S. M. Mobin and R. Misra, RSC Adv., 2014, 4, 52526 RSC.
- J. Wang, J. Mei, R. Hu, J. Z. Sun, A. Qin and B. Z. Tang, J. Am. Chem. Soc., 2012, 134, 9956 CrossRef CAS PubMed.
- (a) C. D. Entwistle and T. B. Marder, Angew. Chem., Int. Ed., 2002, 41, 2927 CrossRef CAS; (b) C. D. Entwistle and T. B. Marder, Chem. Mater., 2004, 16, 4574 CrossRef CAS; (c) F. Jäkle, Coord. Chem. Rev., 2006, 250, 1107 CrossRef PubMed; (d) S. Yamaguchi and A. Wakamiya, Pure Appl. Chem., 2006, 78, 1413 CAS; (e) S. Saito, K. Matsuo and S. Yamaguchi, J. Am. Chem. Soc., 2012, 134, 9130 CrossRef CAS PubMed; (f) Y.-L. Rao and S. Wang, Inorg. Chem., 2011, 50, 12263 CrossRef CAS PubMed; (g) Z. M. Hudson and S. Wang, Acc. Chem. Res., 2009, 42, 1584 CrossRef CAS PubMed; (h) F. Jakle, Chem. Rev., 2010, 110, 3985 CrossRef CAS PubMed.
- (a) J. Barberá, R. Giménez and J. L. Serrano, Adv. Mater., 1994, 6, 470 CrossRef PubMed; (b) J. Barberá, R. Giménez and J. L. Serrano, Chem. Mater., 2000, 12, 481 CrossRef; (c) E. Cavero, D. P. Lydon, S. Uriel, M. R. de la Fuente, J. L. Serrano and R. Giménez, Angew. Chem., Int. Ed., 2007, 119, 5267–5269 CrossRef PubMed; (d) E. Cavero, M. R. de la Fuente, E. Beltrán, P. Romero, J. L. Serrano and R. Giménez, Chem. Mater., 2007, 19, 6230–6239 CrossRef CAS; (e) F. Jakle, T. Priermeier and M. Wagner, Organometallics, 1996, 15, 2033 CrossRef; (f) E. Herdtweck, F. Jakle, G. Opromolla, M. Spiegler, M. Wagner and P. Zanello, Organometallics, 1996, 15, 5524 CrossRef CAS.
- (a) T. Jadhav, R. Maragani, R. Misra, V. Sreeramulu, D. N. Rao and S. M. Mobin, Dalton Trans., 2013, 4340–4342 RSC; (b) R. Misra, T. Jadhav and S. M. Mobin, Dalton Trans., 2014, 2014 Search PubMed.
- R. R. Hu, J. L. Maldonado, M. Rodriguez, C. M. Deng, C. K. W. Jim, J. W. Y. Lam, M. M. F. Yuen, G. R. Ortiz and B. Z. Tang, J. Mater. Chem., 2012, 22, 232–240 RSC.
- (a) R. G. Parr and W. Yang, Annu. Rev. Phys. Chem., 1995, 46, 701 CrossRef CAS PubMed; (b) C. Lee, W. Yang and R. G. Parr, Phys. Rev., 1988, 37, 785 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: Thermogravimetric analysis, mechanochromic effect and copies of 1H and 13C NMR spectra. See DOI: 10.1039/c5ra12697e |
| This journal is © The Royal Society of Chemistry 2015 |