DBU-promoted cyclization of vinyl isocyanides with ethers via the functionalization of a C(sp3)–H bond for the synthesis of isoquinolines†
Ping Qiana,
Bingnan Dua,
Jie Zhoua,
Haibo Meia,
Jianlin Han*a and
Yi Pan*ab
aSchool of Chemistry and Chemical Engineering, Nanjing University, Nanjing, 210093, China. E-mail: hanjl@nju.edu.cn; Fax: +86-25-83686133; Tel: +86-25-83686133
bState Key Laboratory of Coordination Chemistry, Nanjing University, Nanjing, 210093, China. E-mail: yipan@nju.edu.cn; Fax: +86-25-83592846; Tel: +86-25-83592846
First published on 13th July 2015
Abstract
A DBU-promoted cascade functionalization of a C(sp3)–H bond adjacent to oxygen and a radical cyclization reaction of vinyl isocyanides were developed. The reaction was carried out without the use of any metal catalysts or photoredox catalysis, which provides easy access to multi-functionalized isoquinolines.
Selective C–H bond functionalization has become an active research topic in organic chemistry and emerged as a powerful tool to construct complex molecules due to high atom-economy and alleviating the need for prefunctionalized substrates.1 Direct functionalization of relatively unreactive C(sp3)–H bonds,2 especially the C(sp3)–H bonds adjacent to a heteroatom, is of high value in organic synthesis as functionalized ethers and amines widely exist in natural products and bioactive compounds.3 In recent years, a number of C(sp3)–H bond functionalization and cross-dehydrogenative coupling (CDC) reactions of ethers and alcohols have been developed to construct ethers and alcohol derivatives.4 Tu and other groups have developed a cascade C(sp3)–H bond functionalization and addition reaction of several alkenes5 and alkynes,6 affording hydroalkylation products. Recently, Ji’s group explored a metal-free functionalization of the C(sp3)–H bond of ethers and a 1,2-aryl migration cascade process resulting in α-aryl-β-oxyalkylated carbonyl ketones with good yields.7 However, the metal-free functionalization of the C(sp3)–H bond of ethers followed by intermolecular or intramolecular cyclization reactions remains a great challenge and is less explored.8
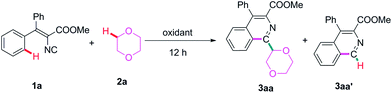
Isonitriles belong to an important class of organic intermediates, which could directly construct heterocycles with high efficiency.9,10 For example, the cyclization of 2-alkenylphenylisocyanide has been used as a key step for the total synthesis of aspidophytine.11 Recently, the radical initiated cyclization of aryl isonitriles has been well developed and several radical precursors,12 including ethers and alcohols,13 have been developed to construct substituted phenanthridines. However, the cyclization of vinyl isonitriles to synthesize substituted isoquinolines has been less explored and only a few examples have been reported. Recently, Xu’s group developed a manganese-catalyzed oxidative radical cascade reaction of vinyl isocyanides for the synthesis of aryl isoquinolines with boronic acids as aryl radical precursors.14 Yu’s group reported the light-promoted cyclization reactions of vinyl isocyanides with diaryliodonium salts or the Umemoto’s trifluoromethylation reagent.15 The Studer group developed a method for the synthesis of 1-trifluoromethylated isoquinolines via a radical trifluoromethylation of isonitriles.16 However, pre-functionalized substrates, transition metals and light conditions are usually needed for these transformations. Furthermore, the functionalization of the C(sp3)–H bond adjacent to an oxygen atom has never been explored to trigger the cyclization of vinyl isocyanides. So, the development of a simple metal-free radical cyclization of vinyl isocyanides with ethers and alcohols becomes highly desirable. Herein, we would like to report a facile metal-free functionalization of a C(sp3)–H bond adjacent to oxygen and the subsequent cyclization reaction of vinyl isocyanides affording 1-ether group substituted isoquinolines (Scheme 1).
The initial reaction of methyl 2-isocyano-3,3-diphenylacrylate (1a) was carried out using 2.0 equiv. of DTBP as the oxidant in 1,4-dioxane (2a) under a nitrogen atmosphere at 120 °C affording the expected product 3aa in very poor yield after 12 h (36%, entry 1, Table 1). Then, 30% H2O2 in water was tried for this reaction, and no obvious improvement was found (entry 2). It was noticeable that the reactions with other oxidants, such as BPO, TBPA and TBPB, gave dramatically increased chemical yields (entries 3–5). TBPB was the best oxidant and 65% yield was found. Running the reaction in air gave a lower yield of 3aa, which disclosed that the reaction has to be done under an inert atmosphere (48% yield, entry 6). The by-product methyl 4-phenylisoquinoline-3-carboxylate (3aa′) was also detected from these reactions. To improve the chemoselectivity, several metal catalysts were tried in the reaction. However, even worse chemoselectivity and lower chemical yields were found (entries 7–11). Interestingly, when some organic bases, including DMAP (entry 14) and DBU (entry 15), as well as the inorganic bases t-BuOK (entry 12) and K2CO3 (entry 13) were used as catalysts for this reaction, excellent chemoselectivities were obtained. In particular, the reaction in the presence of DBU only gave the desired product 3aa with a higher yield (72%, entry 15). Increasing the temperature to 130 °C or decreasing the temperature to 90 °C resulted in slightly lower chemical yields (entries 16 and 17). Finally, the loading amount of the oxidant and DBU was investigated, which showed that 1.0 equiv. of TBPB and 30 mol% of DBU were the best reaction conditions (entries 18–21).
| Entry | Oxidant (equiv.) | Catalyst (mol%) | Temp (°C) | Yieldd (%) (3aa/3aa′) |
|---|---|---|---|---|
| a Reaction conditions: 1a (0.2 mmol), 1,4-dioxane 2a (2.0 mL), oxidant, 12 h, N2 atmosphere.b Yield determined using1H NMR with CH2Br2 as the internal label.c Yield determined using 1H NMR after column chromatography isolation.d Isolated yield based on 1a.e DTBP: di-tert-butyl peroxide.f 30% H2O2 in water.g BPO: benzoyl peroxide.h TBPA: tert-butyl peracetate.i TBPB: tert-butyl peroxybenzoate.j Under air. | ||||
| 1 | DTBP (2.0)e | — | 120 | 36/1b |
| 2 | H2O2 (2.0)f | — | 120 | 31/24b |
| 3 | BPO (2.0)g | — | 120 | 63/15b |
| 4 | TBPA (2.0)h | — | 120 | 41/3b |
| 5 | TBPB (2.0)i | — | 120 | 65/2b |
| 6 | TBPB (2.0) | — | 120 | 48/2j,b |
| 7 | TBPB (2.0) | CuBr (20) | 120 | 30/25c |
| 8 | TBPB (2.0) | Cu2O (20) | 120 | 55/5c |
| 9 | TBPB (2.0) | Cu(OAc)2 (20) | 120 | 32/11c |
| 10 | TBPB (2.0) | Mn(OAc)3·2H2O (20) | 120 | 42/40c |
| 11 | TBPB (2.0) | MnCl2 (20) | 120 | 40/27c |
| 12 | TBPB (2.0) | t-BuOK (30) | 120 | 68/1c |
| 13 | TBPB (2.0) | K2CO3 (30) | 120 | 46/1c |
| 14 | TBPB (2.0) | DMAP (30) | 120 | 54/0c |
| 15 | TBPB (2.0) | DBU (30) | 120 | 72/0c |
| 16 | TBPB (2.0) | DBU (30) | 90 | 70 |
| 17 | TBPB (2.0) | DBU (30) | 130 | 64 |
| 18 | TBPB (1.0) | DBU (30) | 120 | 82 |
| 19 | TBPB (3.0) | DBU (30) | 120 | 53 |
| 20 | TBPB (1.0) | DBU (10) | 120 | 75 |
| 21 | TBPB (1.0) | DBU (50) | 120 | 80 |
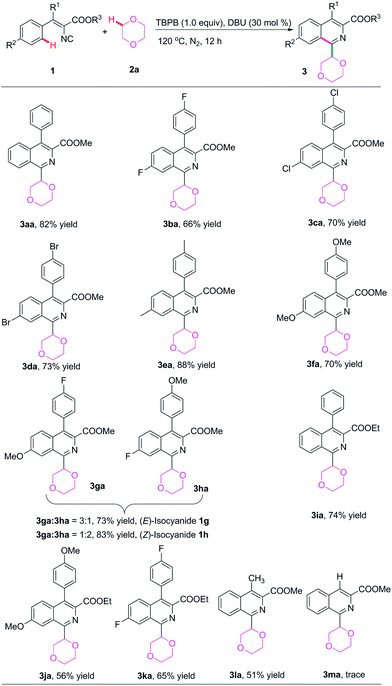
Then, the scope of this radical cyclization reaction with vinyl isocyanides was examined, and the results are summarized in Scheme 2. Several substituted groups on the aromatic ring, including fluoro (3ba), chloro (3ca), bromo (3da), methyl (3ea), and methoxyl (3fa) were tolerated in this reaction, and afforded the corresponding 1-ether group substituted isoquinolines in moderate to good yields. It was noticed that the reactions of the substrates with different substituted aromatic rings (1g and 1h) also proceeded smoothly, resulting in the expected product in good chemical yields but with moderate regioselectivities (3ga and 3ha). The substrates with the ethyl ester group at the terminal position of the vinyl group also worked well in the reaction (3ia–3ka), resulting in the product with slightly lower yields compared to those substrates with the methyl ester group (3aa, 3fa and 3ba). Finally, when the methyl group was used as the substituent on the vinyl moiety instead of a phenyl ring, it also worked well giving the expected product in 51% yield (3la). However, almost no product was observed if a hydrogen atom existed at this position on the vinyl group (3ma).
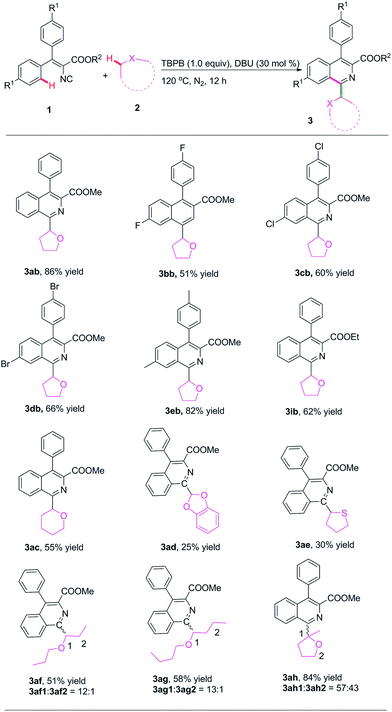
Then, various ethers were investigated in this radical cyclization reaction of 2-isocyano-3,3-diphenylacrylate (1) (Scheme 3). As shown in Scheme 3, tetrahydrofuran (2b) could react well with methyl and ethyl 2-isocyano-3,3-diphenylacrylate (1a and 1i) resulting in the desired products 3ab and 3ib (86% and 62%, respectively). Fortunately, several substituted vinyl isocyanides could also react well with tetrahydrofuran giving the corresponding product (3bb–3eb). Tetrahydropyran and benzo[d][1,3]dioxole were also tolerated in this reaction affording the expected substituted isoquinolines with 55% and 25% yield, respectively (3ac and 3ad). Tetrahydrothiophene was a suitable substrate for this reaction resulting in the expected 1-tetrahydrothiophene substituted product with 30% chemical yield (3ae). The reactions with these two linear ethers also proceeded well, affording the corresponding products with 51% and 58% yields respectively. The reactions also gave excellent regioselectivities, with ratios of 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 13
1 and 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 for products 3af and 3ag, respectively. 2-MeTHF also worked well in this reaction, affording the desired product with 84% chemical yield and 57
1 for products 3af and 3ag, respectively. 2-MeTHF also worked well in this reaction, affording the desired product with 84% chemical yield and 57![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 43 regioselectivity.
43 regioselectivity.
Finally, studies on the investigation of the reaction mechanism were carried out. When a radical scavenger 2,2,6,6-tetramethyl-1-piperidinyloxy (TEMPO) was added to the reaction of 1,4-dioxane, the reaction was completely inhibited and no desired cyclic product was observed (Scheme 4a). Only the coupling product (4) with 1,4-dioxane was detected using MS. This result discloses that the reaction proceeds through a radical process. Also, an intermolecular competing kinetic isotope effect (KIE) experiment was conducted with tetrahydrofuran and [D]-tetrahydrofuran as starting materials (Scheme 4b). An obvious KIE was found with the ratio of 4.35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (kH
1 (kH![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) kD), which shows that the cleavage of the C(sp3)–H bond to form the ether radical may be involved in the rate-determining steps of this procedure.
kD), which shows that the cleavage of the C(sp3)–H bond to form the ether radical may be involved in the rate-determining steps of this procedure.
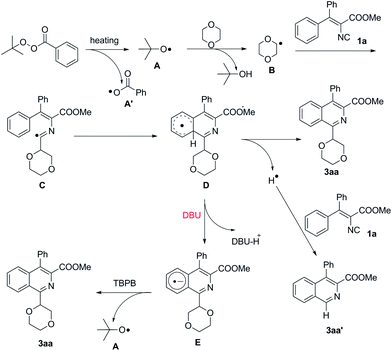
Based on the above results and previous reports,13 a plausible reaction mechanism for this cyclization reaction was supposed (Scheme 5). Firstly, the decomposition of TBPB generates a tert-butoxy radical intermediate A and a benzoate radical A′ under heating, the former reacts with 1,4-dioxane to give the radical B through C(sp3)–H bond cleavage. The intermediate B adds to isonitrile 1a and affords the intermediate C. Subsequently, the intramolecular radical cyclization of intermediate C generates the cyclohexadienyl radical D. In the presence of DBU, intermediate D undergoes deprotonation to give a radical anion E. Finally, the radical anion E gets oxidized by TBPB to afford the product 3aa along with a tert-butoxy radical A and a benzoate anion.13b,13c,17 If no DBU is added, intermediate D undergoes a radical process to give the final product 3aa along with a hydrogen radical. The hydrogen radical reacts with 1a to give the by-product, methyl 4-phenylisoquinoline-3-carboxylate (3aa′).
Conclusions
In summary, a DBU-promoted cascade C(sp3)–H bond functionalization of ethers and a radical cyclization reaction of vinyl isocyanide were reported. The reaction tolerated a wide range of substrates and could be performed under simple conditions. This reaction involves new C(sp3)–C(sp2) and C(sp2)–C(sp2) bond formations, which provide straightforward and atom-economical access to multi-functionalized isoquinolines from readily available starting materials. The current reaction has the limitation of an ether solvent, and only THF and 1,4-dioxane afforded synthetically good yields. This will be further investigated in our future study on this reaction.Acknowledgements
We gratefully acknowledge the financial support from the National Natural Science Foundation of China (No. 21102071 and 21472082).Notes and references
- For selected reviews on C–H bond functionalization, see: (a) C. S. Yeung and V. M. Dong, Chem. Rev., 2011, 111, 1215 CrossRef CAS PubMed; (b) C. Liu, H. Zhang, W. Shi and A. Lei, Chem. Rev., 2011, 111, 1780 CrossRef CAS PubMed; (c) J. Yamaguchi, A. D. Yamaguchi and K. Itami, Angew. Chem., Int. Ed., 2012, 51, 8960 CrossRef CAS PubMed; (d) B. J. Li and Z. J. Shi, Chem. Soc. Rev., 2012, 41, 5588 RSC.
- For selected reviews on C(sp3)–H bond functionalization, see (a) K. Godula and D. Sames, Science, 2006, 312, 67 CrossRef CAS PubMed; (b) H. M. L. Davies and D. Morton, Chem. Soc. Rev., 2011, 40, 1857 RSC; (c) K. M. Engle, T. S. Mei, M. Wasa and J. Q. Yu, Acc. Chem. Res., 2012, 45, 788 CrossRef CAS PubMed; (d) T. Newhouse and P. S. Baran, Angew. Chem., Int. Ed., 2011, 50, 3362 CrossRef CAS PubMed; (e) J. L. Roizen, M. E. Harvey and B. J. Du, Acc. Chem. Res., 2012, 45, 911 CrossRef CAS PubMed; (f) G. Rouquet and N. Chatani, Angew. Chem., Int. Ed., 2013, 52, 11726 CrossRef CAS PubMed.
- For selected reviews on C(sp3)–H bond functionalization adjacent to heteroatoms, see: S. Y. Zhang, F. M. Zhang and Y. Q. Tu, Chem. Soc. Rev., 2011, 40, 1937 RSC.
- For selected recent examples of CDC reaction of C(sp3)–H bonds adjacent to heteroatoms, see: (a) Z. P. Li, R. Yu and J. H. Li, Angew. Chem., Int. Ed., 2008, 47, 7497 CrossRef CAS PubMed; (b) D. Liu, C. Liu, H. Li and A. Lei, Chem. Commun., 2014, 50, 3623 RSC; (c) A. Tanoue, W. J. Yoo and S. Kobayashi, Org. Lett., 2014, 16, 2346 CrossRef CAS PubMed; (d) R. P. Pandit and Y. R. Lee, Adv. Synth. Catal., 2014, 356, 3171 CrossRef CAS PubMed; (e) J. C. Zhao, H. Fang, W. Zhou, J. L. Han and Y. Pan, J. Org. Chem., 2014, 79, 3847 CrossRef CAS PubMed; (f) S. R. Guo, Y. Q. Yuan and J. N. Xiang, Org. Lett., 2013, 15, 4654 CrossRef CAS PubMed; (g) D. Liu, C. Liu, H. Li and A. Lei, Angew. Chem., Int. Ed., 2013, 52, 4453 CrossRef CAS PubMed; (h) J. C. Zhao, W. Zhou, J. L. Han, G. Li and Y. Pan, Tetrahedron Lett., 2013, 54, 6507 CrossRef CAS PubMed; (i) L. Chen, E. Shi, Z. Liu, S. Chen, W. Wei, H. Li, K. Xu and X. Wan, Chem.–Eur. J., 2011, 17, 4085 CrossRef CAS PubMed; (j) Z. Wu, C. Pi, X. Cui, J. Bai and Y. Wu, Adv. Synth. Catal., 2013, 355, 1971 CrossRef CAS PubMed; (k) Z. Cui, X. Shang, X. F. Shao and Z. Q. Liu, Chem. Sci., 2012, 3, 2853 RSC.
- (a) L. Shi, Y. Q. Tu, M. Wang, F. M. Zhang, C. A. Fan, Y. M. Zhao and W. J. Xia, J. Am. Chem. Soc., 2005, 127, 10836 CrossRef CAS PubMed; (b) J. K. Cheng and T. Loh, J. Am. Chem. Soc., 2015, 137, 42 CrossRef CAS PubMed; (c) Y. J. Jiang, Y. Q. Tu, E. Zhang, S. Y. Zhang, K. Cao and L. Shi, Adv. Synth. Catal., 2008, 350, 552 CrossRef CAS PubMed; (d) W. Zhou, P. Qian, J. C. Zhao, H. Fang, J. L. Han and Y. Pan, Org. Lett., 2015, 17, 1160 CrossRef CAS PubMed; (e) S. Y. Zhang, Y. Q. Tu, C. A. Fan, F. M. Zhang and L. Shi, Angew. Chem., Int. Ed., 2009, 48, 8761 CrossRef CAS PubMed.
- Z. Q. Liu, L. Sun, J. G. Wang, J. Han, Y. K. Zhao and B. Zhou, Org. Lett., 2009, 11, 1437 CrossRef CAS PubMed.
- X. Q. Chu, H. Meng, Y. Zi, X. P. Xu and S. J. Ji, Chem. Commun., 2014, 50, 9718 RSC.
- (a) W. T. Wei, M. B. Zhou, J. H. Fan, W. Liu, R. J. Song, Y. Liu, M. Hu, P. Xie and J. H. Li, Angew. Chem., Int. Ed., 2013, 52, 3638 CrossRef CAS PubMed; (b) K. M. McQuaid and D. Sames, J. Am. Chem. Soc., 2009, 131, 402 CrossRef CAS PubMed; (c) Y. Meng, L. N. Guo, H. Wang and X. H. Duan, Chem. Commun., 2013, 49, 7540 RSC.
- For reviews on isocyanides, see (a) A. V. Lygin and A. de Meijere, Angew. Chem., Int. Ed., 2010, 49, 9094 CrossRef CAS PubMed; (b) B. Zhang and A. Studer, Chem. Soc. Rev., 2015, 44, 3505 RSC.
- For recent examples on constructions of heterocycles from isocyanides, see (a) T. Mitamura, K. Iwata and A. Ogawa, J. Org. Chem., 2011, 76, 3880 CrossRef CAS PubMed; (b) T. Mitamura and A. Ogawa, J. Org. Chem., 2011, 76, 1163 CrossRef CAS PubMed; (c) T. Mitamura, K. Iwata and A. Ogawa, Org. Lett., 2009, 11, 3422 CrossRef CAS PubMed; (d) M. Tobisu, H. Fujihara, K. Koh and N. Chatani, J. Org. Chem., 2010, 75, 4841 CrossRef CAS PubMed; (e) X. Lu, J. L. Petersen and K. K. Wang, Org. Lett., 2003, 5, 3277 CrossRef CAS PubMed.
- S. Sumi, K. Matsumoto, H. Tokuyama and T. Fukuyama, Org. Lett., 2003, 5, 1891 CrossRef CAS PubMed.
- For selected examples, see (a) M. Tobisu, K. Koh, T. Furukawa and N. Chatani, Angew. Chem., Int. Ed., 2012, 51, 11363 CrossRef CAS PubMed; (b) B. Zhang, C. Muck-Lichtenfeld, C. G. Daniliuc and A. Studer, Angew. Chem., Int. Ed., 2013, 52, 10792 CrossRef CAS PubMed; (c) B. Zhang and A. Studer, Org. Lett., 2014, 16, 3990 CrossRef CAS PubMed; (d) Q. Wang, X. Dong, T. Xiao and L. Zhou, Org. Lett., 2013, 15, 4846 CrossRef CAS PubMed; (e) D. Leifert, C. G. Daniliuc and A. Studer, Org. Lett., 2013, 15, 6286 CrossRef CAS PubMed; (f) H. Fang, J. C. Zhao, P. Qian, J. L. Han and Y. Pan, Asian J. Org. Chem., 2014, 3, 1266 CrossRef CAS PubMed; (g) J. Liu, C. Fan, H. Yin, C. Qin, G. Zhang, X. Zhang, H. Yi and A. Lei, Chem. Commun., 2014, 50, 2145 RSC; (h) X. Li, M. Fang, P. Hu, G. Hong, Y. Tang and X. Xu, Adv. Synth. Catal., 2014, 356, 2103 CrossRef CAS PubMed; (i) H. Fang, J. C. Zhao, S. Y. Ni, H. B. Mei, J. L. Han and Y. Pan, J. Org. Chem., 2015, 80, 3151 CrossRef CAS PubMed; (j) L. Gu, C. Jin, J. Liu, H. Ding and B. Fan, Chem. Commun., 2014, 50, 4643 RSC.
- (a) L. Wang, W. X. Sha, Q. Dai, X. M. Feng, W. T. Wu, H. B. Peng, B. Chen and J. Cheng, Org. Lett., 2014, 16, 2088 CrossRef CAS PubMed; (b) J. Cao, T. Zhu, S. Wang, Z. Gu, X. Wang and S. Ji, Chem. Commun., 2014, 50, 6439 RSC; (c) Z. Li, F. Fan, J. Yang and Z. Q. Liu, Org. Lett., 2014, 16, 3396 CrossRef CAS PubMed.
- H. Wang, Y. Yu, X. Hong and B. Xu, Chem. Commun., 2014, 50, 13485 RSC.
- H. Jiang, Y. Cheng, R. Wang, Y. Zhang and S. Yu, Chem. Commun., 2014, 50, 6164 RSC.
- B. Zhang and A. Studer, Org. Biomol. Chem., 2014, 12, 9895 CAS.
- W. Wang, Q. Zhang, T. Zhang, Z. Li, W. Zhang and W. Yu, Adv. Synth. Catal., 2015, 357, 221 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Experimental procedures, full spectroscopic data for compound 3 and copies of 1H and 13C NMR spectra. See DOI: 10.1039/c5ra11530b |
| This journal is © The Royal Society of Chemistry 2015 |