Biocompatible nano hydroxyapatite – curcumin bi-coated antibacterial activated carbon for water purification†
Abstract
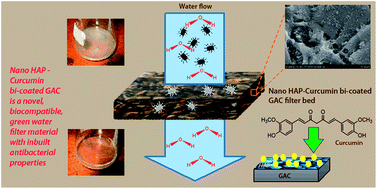
Activated carbon has been used for water purification since ancient times due to its well-known sorption properties. However it is not capable of disinfecting water borne pathogens such as bacteria. The main objective of this study was to incorporate antibacterial properties while maintaining the existing properties of Granular Activated Carbon (GAC). This was achieved by a biocompatible double coating on to GAC which consists of hydroxyapatite (HAP) nanoparticles and on top of those curcumin molecules. Coating of GAC with HAP was carried out using in situ precipitation of HAP under basic conditions. A layer of curcumin molecules was then attached on top of the HAP coating in order to obtain HAP-curcumin bi-coated GAC (HAP/C/GAC). Synthesized HAP/GAC and HAP/C/GAC were characterized using FT-IR spectroscopy, scanning electron microscopy (SEM), powder X-ray diffractometry (PXRD) and thermogravimetry (TGA). Characterization revealed that needle shaped HAP nanoparticles (50–100 nm in width and approximately 200–500 nm in length) can be anchored and immobilized successfully on GAC which in turn enhances the adhesion of curcumin on it. Antibacterial properties of pure GAC, HAP/GAC and HAP/C/GAC were then investigated using both Gram negative (Escherichia coli) and Gram positive (Staphylococcus aureus) bacteria. The results show that the antibacterial properties of HAP/C/GAC are remarkably higher than that of HAP/GAC and the antibacterial activity of pure GAC is negligible.

 Please wait while we load your content...
Please wait while we load your content...