Microwave-assisted, ruthenium-catalyzed intramolecular amide-alkyne annulation for the rapid synthesis of fused tricyclic isoquinolinones†
T. Swamyab,
B. Maheshwar Raoa,
J. S. Yadava,
V. Ravinderb,
B. Sridhara and
B. V. Subba Reddy*a
aNatural Product Chemistry, Laboratory of X-ray Crystallography, Indian Institute of Chemical Technology, Tarnaka, 500 007, Hyderabad, India. E-mail: basireddy@iict.res.in; Web: http://www.iictindia.org Fax: +91-40-27160512
bDepartment of Organic Chemistry, Kakatiya University, Warangal, 506009, India
First published on 6th August 2015
Abstract
Microwave irradiation strongly accelerates the ruthenium(II)-catalyzed intramolecular annulation of alkyne appended benzamides to generate the fused tricyclic isoquinolinone scaffolds through a domino C–C and C–N bond formation by C–H and N–H activation, which is very useful for the synthesis of several analogues of tricyclic isoquinolinone alkaloids. The reaction proceeds with high functional group tolerance to furnish the products in good to high yields.
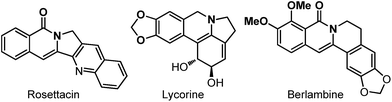
Tricyclic isoquinolinone core is frequently found in many natural products such as Rosettacin, Berlambine, and Lycorine (Fig. 1).1
Consequently, various approaches have been reported for the synthesis of isoquinolinones.2 Among them, oxidative annulation of alkynes with aryl or alkenyl amides is one of the most elegant strategies for the synthesis of isoquinolinone derivatives.3 Recently, rhodium-catalyzed intermolecular annulation of benzamides has been reported for the synthesis of isoquinolines, which obviously avoids the use of an external oxidant and also enhances the reactivity and selectivity under mild conditions.4,5 The products thus obtained, were readily transformed into indolizidine scaffolds. More recently, an intramolecular annulation of tethered alkynes has been reported for the synthesis of tricyclic isoquinolinones.6,7 Therefore, C–H annulation provides a direct access to a wide range of valuable heterocycles without a pre-functionalization of the substrate.8
The efficiency of C–H activation strategy generally depends upon the coordination ability of the metal to the heteroatom, which directs the site selective C–H functionalization through a five- or six-membered metallacycle.9
Due to high cost of the rhodium catalyst and prolonged reaction times, we were interested to develop a rapid alkyne-amide annulation process using a low cost Ru catalyst under microwave irradiation. In recent years, ruthenium(II)-catalyzed oxidative C–H activation strategy has received significant attention because of its high efficiency, selectivity, and functional-group tolerance.10 However, many of these reactions require stoichiometric amounts of external oxidants and extended reaction times. In the past decade, microwave irradiation has become an alternative heating source for the rapid synthesis of heterocycles.11 In deed, a few microwave assisted reactions are known in literature for a faster and efficient C–H activation.12 Based on intermolecular alkyne annulation,8a we designed ruthenium(II)-catalyzed intramolecular annulation reactions.
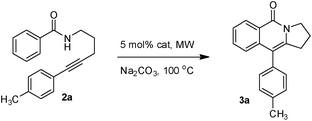
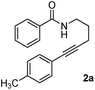
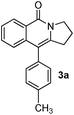
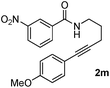
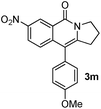
As part of our continuing interest on metal-catalyzed C–H activation,13 we herein report a rapid intramolecular alkyne-amide annulation using Ru(II)/Cu(II) complex under microwave irradiation. To optimize the reaction conditions, the annulation of 2a was performed in different solvents using various catalysts under diverse conditions to produce the fused tricyclic isoquinolinone 3a (Table 1). Various catalysts such as Pd(OAc)2, PdCl2, [Ru(p-cymene)Cl2]2, Rh, Ir, and Fe complexes were screened under microwave irradiation. The reaction was highly efficient, when Ru(II) was used as catalyst and Cu(OAc)2·H2O as co-oxidant and toluene as solvent (entry c, Table 1). Under optimized conditions, the required product 3a was isolated in 85% yield. Other catalysts such as Pd, Ni and Fe were found to be ineffective. Although rhodium and ruthenium complexes were proved to be efficient for the oxidative annulation of benzamides,4–7 the ruthenium complexes are attractive due to their low cost and ready accessibility compared to rhodium complexes.10
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a
a
| Entry | Catalyst | Oxidant | Solvent | Time (min) | Yieldb |
|---|---|---|---|---|---|
| a Reactions were performed with alkynylbenzamide (0.1 mmol), [Ru(p-cymene)Cl2]2 (5 mol%), oxidant (2.1 equiv.), Na2CO3 (2.2 equiv.) and solvent (2 mL) at 100 °C for 15 min under microwaves.b Yield refers to pure products after column chromatography.c Reaction was performed conventionally at 100 °C in toluene. | |||||
| a | Pd(OAc)2 | Cu(OAc)2·H2O | Toluene | 20 | 0 |
| b | PdCl2 | Cu(OAc)2·H2O | Dioxane | 20 | 0 |
| c | [{RuCl2(p-cymene)}2] | Cu(OAc)2·H2O | Toluene | 15 | 85 |
| d | [{RuCl2(p-cymene)}2] | Cu(OAc)2·H2O | t-AmOH | 10 | 70 |
| e | [{RuCl2(p-cymene)}2] | Cu(OAc)2·H2O | Dioxane | 10 | 65 |
| f | [{RuCl2(p-cymene)}2] | — | Toluene | 20 | 0 |
| g | [{RhCp*Cl2}2] | Cu(OAc)2·H2O | Toluene | 10 | 68 |
| h | Ni(acac)2 | Cu(OAc)2·H2O | Toluene | 20 | 0 |
| i | Fe(acac)2 | Cu(OAc)2·H2O | Toluene | 20 | 0 |
| j | [{RuCl2(p-cymene)}2] | Cu(OAc)2·H2O | Toluene | 15 h | 60c |
To know the effect of microwave, we performed the annulation of 2a under both conventional heating conditions and microwave irradiation. The desired tricyclic isoquinolinone 3a was obtained in 70% yields over 12 h under conventional conditions, whereas 3a was isolated in 85% yield after 15 min under microwave irradiation. Though, the reaction was faster under microwave (10 min) in polar solvents such as dioxane and t-amyl alcohol, the corresponding product was isolated in low yields (entries d and e, Table 1).
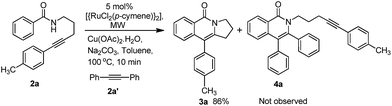
In order to know the reactivity of a tethered alkyne, we carried out the annulation of 2a in the presence of an external alkyne 2a′. Interestingly, the expected product 3a was obtained exclusively without the formation of 4a. This clearly indicates that intramolecular annulation is much faster than the intermolecular annulation (Scheme 1).
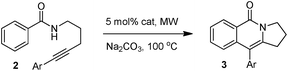
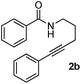
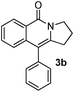
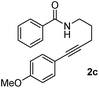
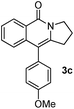
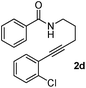
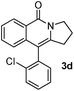
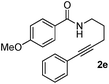
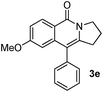
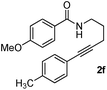
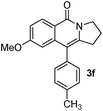
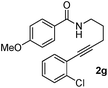
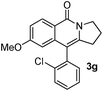
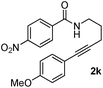
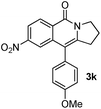
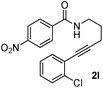
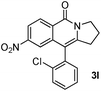
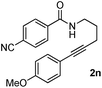
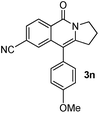
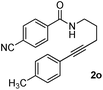
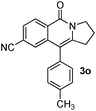
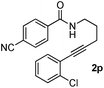
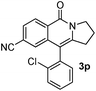
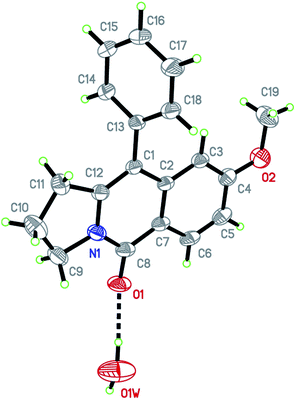
Having the optimized conditions in hand, we further examined the substrate scope using different alkyne appended benzamides. For example, an intramolecular annulation of N-(5-phenylpent-4-ynyl)benzamide 2b in the presence [Ru(p-cymene)Cl2]2, Cu(OAc)2·H2O and Na2CO3 in toluene under microwave conditions afforded the desired tricyclic product 3b in 84% yield (entry b, Table 2). Similarly, N-(5-(4-methoxyphenyl)pent-4-ynyl)benzamide 2c underwent a smooth intramolecular cyclization to afford the corresponding 10-(4-methoxyphenyl)-2,3-dihydropyrrolo[1,2-b]isoquinolin-5(1H)-one 3c in 87% yield (entry c, Table 2). The scope of this method is further exemplified with N-(5-(2-chlorophenyl)pent-4-ynyl)benzamide 2d to produce the desired product 3d in 85% yield (entry d, Table 2). Next, we extended this approach to 4-methoxy-N-(5-phenylpent-4-ynyl)benzamide 2e. Interestingly, the corresponding tricyclic benzamide 3e was obtained in good yield (Table 2). The structure of 3e was established by X-ray crystallography (Fig. 2).14
The substituent present on aromatic ring of the amide functionality had shown some effect on conversion. It was observed that the methoxy substituted benzamide gave the product in higher yields than nitro- and cyano-substituted substrates. The results presented in Table 2 clearly indicate that the reactions are faster with electron rich benzamides. However, there was no considerable effect of the substituents that are present on aromatic ring of the alkyne moiety. The reaction was quite successful under microwave irradiation, hence this method is useful for the synthesis of a diverse range of tricyclic isoquinolinones in a simple way.
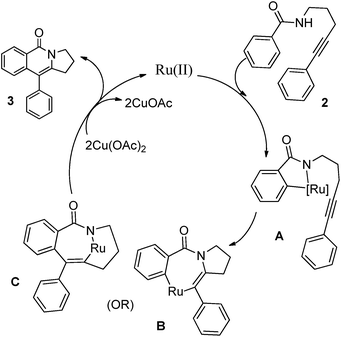
Based on our previous observation,13 we proposed a possible reaction mechanism, which is shown in Scheme 2. The reaction proceeds likely through the formation of a five-membered transition state (A).3b Ruthenium is known to activate the ortho-position of the benzamide by an oxidative insertion into aromatic C–H bond.8a Thus formed metallacycle (A) reacts with tethered alkyne to facilitate the C–N bond formation (B) or ortho-functionalization (C). A subsequent reductive elimination of Ru(0) would give the desired product. Finally, Ru(0) could be reoxidized to Ru(II) by Cu(OAc)2 to regenerate the catalyst.8h
In summary, we have developed a one-pot strategy for the synthesis of biologically relevant tricyclic isoquinolinone derivatives. This reaction proceeds through a domino C–C and C–N bond formation. The use of microwave irradiation provides the desired products with improved yields and enhanced reaction rates over conventional methods.
Experimental
IR spectra were recorded on FT-IR spectrometer (KBr) and reported in reciprocal centimetres (cm−1). 1H NMR spectra were recorded at 500 MHz, 300 MHz and 13C NMR at 125 MHz, 75 MHz. For 1H NMR, tetramethylsilane (TMS) was used as internal standard (δ = 0) and the values are reported as follows: chemical shift, integration, multiplicity (s = singlet, d = doublet, t = triplet, q = quartet, m = multiplet, br = broad), and the coupling constants in Hz. For 13C NMR, CDCl3 (δ = 77.27) was used as internal standard and spectra were obtained with complete proton decoupling. Low-resolution MS and HRMS data were obtained using ESI ionization. Melting points were measured on micro melting point apparatus. Reaction progress was monitored by analytic HPLC and also by using analytical thin layer chromatography (TLC) on precoated silica gel GF254 plates and the spots were detected under UV light (254 nm).General procedure for the annulation of alkyne-amide using Ru catalyst under microwave irradiation (3)
An oven-dried 10 mL microwave reaction vessel containing a stir bar was charged with alkyne-amide (0.1 mmol), [Ru(p-cymene)Cl2]2 (5 mol%), Na2CO3 (2.2 equiv.), Cu(OAc)2·H2O (2.1 equiv.) and 2 mL toluene. The vessel was sealed with a microwave septum and placed into the CEM Discover SP system under the following conditions: 1–2 min ramp time, pre-stirring 30 s, the stirring was set at high. Power max was kept On. The maximum power and pressure were set at 250 W and 250 psi respectively, with a set temperature of 100 °C for 15 min (hold time). After irradiation was complete, the mixture was cooled to room temperature and then diluted with ethyl acetate and filtered through celite. The filtrate was concentrated and the resulting product was purified by column chromatography using silica gel, eluting with ethyl acetate/hexane mixtures.Acknowledgements
T. S and B. M thanks CSIR, New Delhi and DST-SERB New Delhi for the financial support.Notes and references
- (a) K. I. Yamada, M. Yamashita, T. Sumiyoshi, K. Nishimura and K. Tomioka, Org. Lett., 2009, 11, 1631 CrossRef CAS PubMed; (b) M. Cushman and F. W. Dekow, J. Org. Chem., 1979, 44, 407 CrossRef CAS; (c) F. Pin, S. Comesse, M. Sanselme and A. Daïch, J. Org. Chem., 2008, 73, 1975 CrossRef CAS PubMed.
- (a) Z.-W. Chen, Y.-Z. Zhu, J.-W. Ou, Y.-P. Wang and J.-Y. Zheng, J. Org. Chem., 2014, 79, 10988 CrossRef CAS PubMed; (b) Y. Kajita, S. Matsubara and T. Kurahashi, J. Am. Chem. Soc., 2008, 130, 6058 CrossRef CAS PubMed; (c) C.-C. Liu, K. Parthasarathy and C.-H. Cheng, Org. Lett., 2010, 12, 3518 CrossRef CAS PubMed; (d) H. Shiota, Y. Ano, Y. Aihara, Y. Fukumoto and N. Chatani, J. Am. Chem. Soc., 2011, 133, 14952 CrossRef CAS PubMed.
- (a) T. Satoh and M. Miura, Chem.–Eur. J., 2010, 16, 11212 CrossRef CAS PubMed; (b) L. Ackermann and S. Fenner, Org. Lett., 2011, 13, 6548 CrossRef CAS PubMed.
- (a) T. K. Hyster and T. Rovis, J. Am. Chem. Soc., 2010, 132, 10565 CrossRef CAS PubMed; (b) G. Song, D. Chen, C.-L. Pan, R. H. Crabtree and X. Li, J. Org. Chem., 2010, 75, 7487 CrossRef CAS PubMed; (c) F. W. Patureau and F. Glorius, Angew. Chem., Int. Ed., 2011, 50, 1977 CrossRef CAS PubMed.
- (a) N. Guimond, S. I. Gorelsky and K. Fagnou, J. Am. Chem. Soc., 2011, 133, 6449 CrossRef CAS PubMed; (b) N. Guimond, C. Gouliaras and K. Fagnou, J. Am. Chem. Soc., 2010, 132, 6908 CrossRef CAS PubMed.
- (a) X. Xu, Y. Liu and C.-M. Park, Angew. Chem., Int. Ed., 2012, 51, 9372 CrossRef CAS PubMed; (b) N. Quinones, A. Seoane, R. G. Fandino, J. L. Mascarenas and M. Gulıas, Chem. Sci., 2013, 4, 2874 RSC.
- (a) P. Tao and Y. Jia, Chem. Commun., 2014, 50, 7367 RSC; (b) X. Zhang, Y. Li, H. Shi, L. Zhang, S. Zhang, X. Xu and Q. Liu, Chem. Commun., 2014, 50, 7306 RSC.
- (a) L. Ackermann, A. V. Lygin and N. Hofmann, Angew. Chem., Int. Ed., 2011, 50, 6379 CrossRef CAS PubMed; (b) X. Xu, Y. Liu and C. M. Park, Angew. Chem., Int. Ed., 2012, 51, 9372 CrossRef CAS PubMed; (c) D. J. Burns and H. W. Lam, Angew. Chem., Int. Ed., 2014, 53, 9931 CrossRef CAS PubMed; (d) L. Ackermann, J. Pospech, K. Graczyk and K. Rauch, Org. Lett., 2012, 14, 930 CrossRef CAS PubMed; (e) S. Ding, Z. Shi and N. Jiao, Org. Lett., 2010, 12, 1540 CrossRef CAS PubMed; (f) Z. Shi, Y. Cui and N. Jiao, Org. Lett., 2010, 12, 2908 CrossRef CAS PubMed; (g) J. Chen, Q. Pang, Y. Sun and X. Li, J. Org. Chem., 2011, 76, 3523 CrossRef CAS PubMed; (h) L. Ackermann, L. Wang and A. V. Lygin, Chem. Sci., 2012, 3, 177 RSC; (i) L. Ackermann and A. V. Lygin, Org. Lett., 2012, 14, 764 CrossRef CAS PubMed; (j) X. Zhang, W. Si, M. Bao, N. Asao, Y. Yamamoto and T. Jin, Org. Lett., 2014, 16, 4830 CrossRef CAS PubMed.
- (a) C. S. Yeung and V. M. Dong, Chem. Rev., 2011, 111, 1215 CrossRef CAS PubMed; (b) J. Wencel-Delord, T. Droge, F. Liu and F. Glorius, Chem. Soc. Rev., 2011, 40, 4740 RSC; (c) L. McMurray, F. O'Hara and M. J. Gaunt, Chem. Soc. Rev., 2011, 40, 1885 RSC; (d) L. Ackermann, Chem. Rev., 2011, 111, 1315 CrossRef CAS PubMed; (e) D. A. Colby, R. G. Bergman and J. A. Ellman, Chem. Rev., 2010, 110, 624 CrossRef CAS PubMed.
- (a) L. Ackermann and R. Vicente, Top. Curr. Chem., 2010, 292, 211 CrossRef CAS; (b) L. Ackermann, Chem. Commun., 2010, 46, 4866 RSCH. Weissman, X. Song and D. Milstein, J. Am. Chem. Soc., 2001, 123, 337 CrossRef CAS; (c) T. Ueyama, S. Mochida, T. Fukutani, K. Hirano, T. Satoh and M. Miura, Org. Lett., 2011, 13, 706 CrossRef CAS PubMed.
- (a) P. Lidstrom, J. Tierney, B. Wathey and J. Westman, Tetrahedron, 2001, 57, 9225 CrossRef CAS; (b) N. Kuhnert, Angew. Chem., Int. Ed., 2002, 41, 1863 CrossRef CAS; (c) M. Larhed, C. Moberg and A. Hallberg, Acc. Chem. Res., 2002, 35, 717 CrossRef CAS PubMed; (d) B. Wathey, J. Tierney, P. Lidstrom and J. Westman, Drug Discovery Today, 2002, 7, 373 CrossRef CAS; (e) A. Lew, P. O. Krutzik, M. E. Hart and A. R. Chamberlin, J. Comb. Chem., 2002, 4, 95 CrossRef CAS.
- (a) K. L. Tan, A. Vasudevan, R. G. Bergman, J. A. Ellman and A. J. Souers, Org. Lett., 2003, 5, 2131 CrossRef CAS PubMed; (b) K. L. Hull, W. Q. Anani and M. S. Sanford, J. Am. Chem. Soc., 2006, 128, 7134 CrossRef CAS PubMed; (c) M. N. Bagra and J. Rahul, J. Org. Chem., 2013, 78, 10954 CrossRef PubMed.
- (a) B. V. S. Reddy, N. Umadevi, G. Narasimulu and J. S. Yadav, Tetrahedron Lett., 2012, 53, 6091 CrossRef PubMed; (b) B. V. S. Reddy, G. Narasimhulu, N. Umadevi and J. S. Yadav, Synlett, 2012, 9, 1364 CrossRef; (c) B. V. S. Reddy, G. Revanthi, A. S. Reddy and J. S. Yadav, Tetrahedron Lett., 2011, 52, 5926 CrossRef CAS PubMed; (d) B. V. S. Reddy, K. Ramesh and J. S. Yadav, Synlett, 2011, 8, 169 CrossRef.
- ESI†..
Footnote |
| † Electronic supplementary information (ESI) available: Detailed procedures and spectroscopic data for novel compounds. Copies of 1H NMR, 13C NMR spectra of novel compounds, are available. CCDC 1061398. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c5ra11133a |
| This journal is © The Royal Society of Chemistry 2015 |