Influence of temperature, added electrolyte, and polymer molecular weight on the counterion-condensation phenomenon in aqueous solution of sodium polystyrenesulfonate: a scaling theory approach†
Ranjit Dea,
Dhiman Rayb and
Bijan Das*b
aAdvanced Photonics Research Institute (APRI), Gwangju Institute of Science and Technology (GIST), 261 Cheomdan-gwagiro (Oryng-dong), Buk-gu, Gwangju 500-712, Republic of Korea
bDepartment of Chemistry, Presidency University, 86/1, College Street, Kolkata 700 073, India. E-mail: bijan.chem@presiuniv.ac.in; Fax: +91 33 2257 2444; Tel: +91 33 2241 3893
First published on 15th June 2015
Abstract
Interactions between a polyion and its counterions in aqueous solutions of an anionic polyelectrolyte sodium polystyrenesulfonate in the presence of sodium chloride were investigated using electrical conductivity as the probe. The specific conductivity vs. polyelectrolyte concentration data were analyzed with an equation recently developed by us considering the scaling description for the conformation of a polyion chain. The influences of (a) the molecular weight and the concentration of the polyelectrolyte, (b) the added electrolyte concentration, and (c) the temperature on polyion–counterion interactions were investigated. The extent of counterion condensation was found to be greatly affected by the concentration and molecular weight of the polyelectrolyte, the concentration of the added electrolyte, and the temperature. The polyion equivalent conductivity in conjunction with the derived coefficient of friction between the monomer units and the solvent provided important information concerning the relative importance of the size and charge of the polyions. The overall results have been elucidated taking the medium dielectric constant, the hydration behaviour of the counterions, and the coiling behavior of the polyion chains into account. The present study provides new insight as to how the polyelectrolyte molecular weight influences the counterion condensation behaviour in a polyelectrolyte solution in the presence of an added electrolyte.
Introduction
Polyelectrolytes are polymers whose repeating units bear an electrolyte group which dissociates in aqueous solutions to yield a macroion or polyion and oppositely charged counterions.1,2 The Coulombic attraction leads to a spatial correlation of the counterions with the macroion. This correlation makes the aqueous solutions of polyelectrolytes behave entirely differently from the neutral (uncharged) polymeric species, thus influencing a variety of their transport and colligative properties. The unique properties of polyelectrolytes render them a system of fundamental interest in the physics of soft-condensed matter including biomacromolecules.1–9 They also find a variety of technological applications. In many applications as well as in biological systems both polyelectrolytes and ions derived from electrolytes coexist, where polyelectrolyte–salt interactions play a pivotal role. A quantitative elucidation of these interactions requires a thorough understanding of the condensation of counterions onto the polyion chains which would help unravel the effects of solution conditions on biological and industrial systems involving polyelectrolytes. Despite decades of research, the phenomenon of counterion condensation in polyelectrolyte solutions is not fully understood owing primarily to the intricate coupling of various interactions in these systems.5–17 The ionic interactions between the polyion and the counterions characterizing the polyelectrolyte behavior can be conveniently investigated by measuring the electrical conductivity because it takes into account the movement of any charged entity present in aqueous solutions (polyion, counterions, and ions derived from the added salt) when an external electric field is applied.Electrical conductivities of polyelectrolyte solutions were first considered by Manning quantitatively on the basis of counterion condensation model.18–20 Within this model, the polyion is represented as an infinitely long charged line, small ions (counterions) are assumed to form an ionic atmosphere whose density depends on the frame of the polyion and their interactions with the charged polyions are purely coulombic, so that the screening effect extends over the Debye length. The uncondensed mobile ions are treated in the Debye–Hückel approximation. The solvent is assumed to be a dielectric continuum characterized by a spatially uniform dielectric constant ε. Interactions among the polyions are neglected, the theory being addressed to highly diluted solutions. The equations derived on the basis of these assumptions can, therefore, be considered as the limiting laws.
The equivalent conductivity (Λ) of a polyelectrolyte dissolved in solution is given by20
| Λ = f(λ0c + λp) | (1) |
In the frame of the Manning counterion condensation model, a polyelectrolyte is characterized by the charge parameter (ξ) which is defined through the ratio of the Bjerrum length (λB) to the distance between two adjacent charged groups on the polyion (b):
 | (2) |
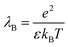 | (3) |
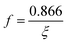 | (4) |
Manning20 derived the following expression for the equivalent conductivity of a polyion along with counterions each with a charge of zc taking into account the electrophoretic and relaxation effects
 | (5) |
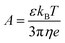 | (6) |
 | (7) |
Therefore, the equivalent conductivity of a polyelectrolyte solution (Λ) can, according to Manning,20 be expressed as
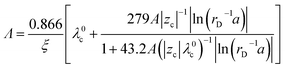 | (8) |
The specific conductivity of the polyelectrolyte solution in presence of an added electrolyte (σ) will be given by33,34
 | (9) |
It should be pointed out that the Manning theory is, in principle, applicable to polyelectrolyte solutions as the polyelectrolyte concentration approaches zero characterizing the absence of polyion–polyion interactions where polyions can be modeled as a rigid linear array of point charges. Hence, the quantitative conclusions drawn from this theory cannot be compared with the experimental data measured at finite concentrations especially in the semidilute regime where the interpolyionic interactions cannot be neglected. Flexible polyelectrolytes cannot also be treated with this model since the conformation of the polyion chains in these cases varies with their concentration as opposed to the underlying assumption of this model.21–26
Later, Colby et al.27 developed a simple model of AC conductivity for salt-free semidilute polyelectrolyte solutions using the de Gennes scaling picture for the configuration of a polyelectrolyte chain in semidilute solution according to Dobrynin et al.28 This model considered the concentration dependence of polyion chain flexibility and the mutual interactions among the polyion chains which were not taken into account in the Manning counterion condensation theory. This approach has been found to be successful to describe the conductivity of semidilute polyelectrolyte solutions.27,29–32
In a semidilute polyelectrolyte solution, Colby et al.,27 considered a polyion chain as an assembly of randomly walking Nξ correlation blobs each with a size of ξ0. Each correlation blob contains g monomers and bears an electric charge of qξ = |zc|efg. The entire chain with a contour length of Nξξ0, carries a charge of |Zc|efgNξ. The strong electrostatic interactions within the correlation blobs cause the chain to attain a completely extended conformation of ge electrostatic blobs each of size D.
Within this model, Colby et al.,27 arrived at the following expression of the equivalent conductivity of a salt-free solution of a polyion (λp)
 | (10) |
The equivalent conductivity of a the polyelectrolyte solution is, therefore, given by
 | (11) |
The specific conductivity of a polyelectrolyte solution in presence of an added electrolyte will be given by
 | (12) |
The above equation has only one adjustable parameter, namely the fraction of free counterions. This parameter can be ascertained by least-squares fit of the measured specific conductivity of the polyelectrolyte-salt solution (σ) to eqn (12) in conjunction with the experimental specific conductance (σs) of the polyelectrolyte-free salt solution. The value of f appearing in the above equation takes care of the modified polyion–counterion interactions upon addition of the salt to the polyelectrolyte solution.
This equation has been successfully employed to describe quantitatively the electrical conductivities of polyelectrolyte-salt solutions,33–35 and hence would help unravel the interactions characterizing the polyelectrolyte-salt solutions, in general, and the biopolymer-salt solutions (e.g., protein–salt, DNA–salt solutions etc.), in particular.
Thus, it is intended in this study to investigate the counterion condensation behavior of aqueous sodium polystyrenesulfonate (NaPSS) solutions in presence of an added electrolyte considering a multitude of parameters including polymer molecular weight, polyelectrolyte concentration, added salt concentration and the temperature. The present study reported for the first time the effect of polymer molecular weight on the counterion condensation phenomena in polyelectrolyte-salt solutions and clearly demonstrated the significance of the polyion chain conformation on the polyion mobility.
Experimental section
Materials
Two NaPSS samples with average molecular weights of 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 and 1
000 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000
000![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 each with a degree of substitution of one employed in this investigation was purchased from Aldrich Chemical Company, Inc. The chemical structure of the NaPSS sample used in this study is depicted in Scheme 1. The average molecular weights of the samples were estimated from the intrinsic viscosity value determined in presence of 0.05 mol L−1 NaCl aqueous solution at 298.15 K using the Mark-Houwink equation with the constants obtained from the literature.36 The molecular weights reported by the manufacturer agreed well with those determined in the present study. The Fourier Transform Infrared Spectra (FT-IR) of both polymer samples are shown Fig. 1 with characteristic peaks indicated by * symbol. The characteristic peaks were observed at about 1182, 1128, 1040, 1010 and 669 cm−1. These characteristic peaks agree very well with those reported earlier for NaPSS samples.37,38 The SO−3 group antisymmetric and symmetric vibrational adsorption peaks can be assigned to the peaks at 1182 and 1038 cm−1, respectively.38 Peaks at 1130 and 1011 cm−1 can be assigned to the in-plane skeleton vibration of benzene ring and in-plane bending vibration of benzene ring.38 Incomplete sulfonation of NaPSS would show peaks at around 699 and 758 cm−1; absence of such peaks in the FT-IR spectra confirms the manufacturer's report that the studied NaPSS samples are fully sulfonated.
000 each with a degree of substitution of one employed in this investigation was purchased from Aldrich Chemical Company, Inc. The chemical structure of the NaPSS sample used in this study is depicted in Scheme 1. The average molecular weights of the samples were estimated from the intrinsic viscosity value determined in presence of 0.05 mol L−1 NaCl aqueous solution at 298.15 K using the Mark-Houwink equation with the constants obtained from the literature.36 The molecular weights reported by the manufacturer agreed well with those determined in the present study. The Fourier Transform Infrared Spectra (FT-IR) of both polymer samples are shown Fig. 1 with characteristic peaks indicated by * symbol. The characteristic peaks were observed at about 1182, 1128, 1040, 1010 and 669 cm−1. These characteristic peaks agree very well with those reported earlier for NaPSS samples.37,38 The SO−3 group antisymmetric and symmetric vibrational adsorption peaks can be assigned to the peaks at 1182 and 1038 cm−1, respectively.38 Peaks at 1130 and 1011 cm−1 can be assigned to the in-plane skeleton vibration of benzene ring and in-plane bending vibration of benzene ring.38 Incomplete sulfonation of NaPSS would show peaks at around 699 and 758 cm−1; absence of such peaks in the FT-IR spectra confirms the manufacturer's report that the studied NaPSS samples are fully sulfonated.
 | ||
Fig. 1 FT-IR spectra NaPSS samples: molecular weight 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 (top spectrum) and 1 000 (top spectrum) and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 (bottom spectrum). 000 (bottom spectrum). | ||
NaCl was purchased from Fluka (puriss grade) and was dried in vacuo for a prolonged period immediately prior to use.
Triply distilled water having a specific conductivity of ca. 10−6 S cm−1 at 308.15 K was employed for the preparation of the solutions.
Conductivity measurements
An Orion (Thermo Fisher, USA) conductivity meter was used to measure the specific conductivities taking solutions in a wide mouth test tube fitted with a dip-type cell (cell constant = 1 cm−1). The uncertainty of measurements was 0.01%. The conductivity measurements were performed in a water thermostat maintained within ±0.01 K of the experimental temperature. Calibration of the cell was done with potassium chloride (aq.) solutions by using the method of Lind et al.39 The specific conductivities of the solutions (σ) were always solvent corrected. Each experiment was performed in 3–5 replicates and their average values were used for calculation.Viscosity measurements
A suspended level Ubbelohde viscosimeter was used for measuring the viscosities of the experimental solutions The viscosimeter was kept vertically in a water thermostat maintained within ±0.01 K of the desired temperature. The absolute viscosity (η) can then be obtained from:
 | (13) |
Fourier transform-infrared (FTIR) spectral measurements
FT-IR spectra of NaPSS samples were acquired by attenuated total reflectance (ATR) technique using a Jasco FTIR-4200 spectrometer with a frequency resolution of 2 cm−1.Results and discussion
Solvent properties and the limiting equivalent conductivity of Na+ ion
Table 1 reports the measured coefficients of viscosity (η) and the literature43 dielectric constants (ε) of water at the experimental temperatures. The values of the equivalent conductivities of the counterion (Na+) at infinite dilution in water (λ0c) were taken from the literature43 and are also included in this table.| Temperature (K) | η (P) | ε | λ0c (S cm2 eq.−1) |
|---|---|---|---|
| 298.15 | 0.008903 | 78.30 | 50.10 |
| 308.15 | 0.007194 | 74.82 | 61.54 |
| 318.15 | 0.005963 | 71.51 | 73.73 |
Experimental specific conductivities
A representative plot (Fig. 2) shows the variation of the experimental specific conductivities (σ) of aqueous solutions of NaPSS as a function of the square root of the moles of monomers of the polyelectrolyte sample per cm3 (cp) with varying concentrations of the added salt NaCl (0.0001, 0.0010, and 0.0100 mol L−1) for two different molecular weights of the polyelectrolyte, namely 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 and 1
000 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000
000![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 at 298.15 K. The corresponding figures at 308.15 and 318.15 K are available as Supplementary material (Fig. 1S and 2S†). The specific conductivities are, in all cases, found to be increased with polyelectrolyte concentration. Addition of salt caused an increase in the specific conductivities of the polyelectrolyte solutions as expected.
000 at 298.15 K. The corresponding figures at 308.15 and 318.15 K are available as Supplementary material (Fig. 1S and 2S†). The specific conductivities are, in all cases, found to be increased with polyelectrolyte concentration. Addition of salt caused an increase in the specific conductivities of the polyelectrolyte solutions as expected.
Fraction of free counterions
The present specific conductivity data have been analyzed on the basis of eqn (9). Two key size parameters, namely the sizes of the electrostatic blob (D) and that of the correlation blob (ξ0) appearing in eqn (9) depend upon the quality of the solvent. For poor solvents, these are given by27| D = b(ξf′2)−1/3 | (14) |
| ξ0 = (c′b)−1/2(ξf′2)−1/3 | (15) |
| D = b(ξf′2)−3/7. | (16) |
| ξ0 = (c′b)−1/2(ξf′2)−1/7 | (17) |
As pointed out by Colby et al.,27 we used the poor solvent correlations for the analysis of the present experimental data on aqueous NaPSS solutions.
The scaling theory approach, with only one adjustable parameter f over the entire concentration range provided a very good description of the experimental specific conductivity vs. concentration data for polyelectrolyte solutions in presence of an added salt due mainly to the fact that this approach takes into account the concentration-dependent conformational change of the polyions, and the interactions between them in these solutions. A quantitative description of the experimental results could, however, not be achieved in some cases.33 It can thus be inferred that the assumption of the independence of the effective charge on a polyion chain of the polymer concentration is no longer valid for the system under investigation. Keeping this in view, in the present investigation, we have calculated the fractions of free counterions (f) independently at each polyelectrolyte concentration with eqn (9), (14) and (15). The values of f are found to vary significantly with NaPSS concentration. This observation suggests that the effective charge on the polystyrenesulfonate chains is dependent on NaPSS concentration.
The results obtained will now be discussed to extract useful information concerning the influence of added salt on the counterion condensation behavior in polyelectrolyte solutions with particular reference to the effect of polyelectrolyte molecular weight which has not been investigated earlier.
The fractions of free counterions are found to increase as the polyelectrolyte concentration is increased for all the NaPSS–NaCl systems under investigation (Fig. 3 and 4). Higher degree of dissociation of counterions with increasing polyelectrolyte concentration might be attributed to a variation of the dielectric constant with polyelectrolyte concentration. Owing to the polarizability of the polyelectrolyte, the dielectric constant of the medium is known to increase as the polyelectrolyte concentration is increased.44 Although the polyelectrolyte concentration-dependent increase in the effective dielectric constant of the medium has not yet been completely understood,45 the experimental evidence is now well established.44,46 For NaPSS in particular, the frequency-dependent dielectric constant of the medium has been shown to increase with NaPSS concentration.47 The increase in dielectric constant value will cause a reduction in the Bjerrum length. Now, since the Bjerrum length sets the length scale for the separation between the dissociated sites on the polyion chain,18,44,48 there will be lower level of counterion condensation with the increase in the dielectric constant of the medium. Obviously, this effect becomes more important at higher polyelectrolyte concentrations. Further theoretical work on the polarizability of polyelectrolytes is, however, still awaited for a detailed quantitative description of the conductivity of polyelectrolyte solutions at high concentration.
 | ||
Fig. 4 (a) Polyion equivalent conductivity in aqueous solution of NaPSS (molecular weight = 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000) in presence of NaCl at different temperatures. For the significance of symbols, please refer to Fig. 3(a). (b) Polyion equivalent conductivity in aqueous solution of NaPSS (molecular weight = 1 000) in presence of NaCl at different temperatures. For the significance of symbols, please refer to Fig. 3(a). (b) Polyion equivalent conductivity in aqueous solution of NaPSS (molecular weight = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000) in presence of NaCl at different temperatures. For the significance of symbols, please refer to Fig. 3(a). 000) in presence of NaCl at different temperatures. For the significance of symbols, please refer to Fig. 3(a). | ||
Fig. 3(a) and (b) reveal a decrease in the fraction of free counterions as the temperature is increased over the entire polyelectrolyte concentration region for any given NaPSS–NaCl system. This observation may be ascribed to a variation in hydration and, hence, a concomitant alteration in the condensation behavior of the counterions as the temperature changes. Elevation of temperature causes a gradual dehydration of the counterions as well as of the dissociated sites on the polyion chain; this would result in more condensation of counterions on the polyion as there will be greater electrostatic attraction between the dehydrated ionic species with higher surface charge densities as the temperature is raised. This is amply demonstrated by the lower degree of counterion condensation at higher temperatures. Dehydration of the sodium ions at higher temperatures has already been inferred in an earlier investigation where the mobility of sodium ions was found to increase appreciably with temperature.49 Similar behavior was also observed with sodium polystyrenesulfonate29 and sodium carboxymethylcellulose30 in mixed-solvent media earlier.
The higher molecular weight NaPSS sample manifests a higher degree of counterion condensation than its lower molecular weight counterpart over the investigated polyelectrolyte concentration range at any given temperature and in presence of a given amount of the added salt (cf. Fig. 3(a) and (b)). The charge density parameter has nothing to do with the observed difference since it is the same for both the polymer samples. This may, however, be interpreted in terms of the confinement of more counterions in the more coiled structure of the higher molecular weight polymer sample in comparison with its lower molecular weight fraction. There has, so far, been only one report on the polyelectrolyte molecular weight dependence of the fraction of free counterions in salt-free polyelectrolyte solutions.15 Influence of polyelectrolyte molecular weight on the counterion condensation behavior in salt-containing polyelectrolyte solutions, however, has not been yet investigated. The present study, therefore, reports for the first time the polyelectrolyte molecular weight dependence of the counterion condensation phenomena in polyelectrolyte-salt solutions.
Polyion equivalent conductivity
An inspection of the effects of polyelectrolyte concentration, added salt concentration, the experimental temperature and the polyelectrolyte molecular weight on the equivalent conductivity (λp) of the polyion (cf. Fig. 4(a) and (b)) helped provide an important insight as to the relative role played by the effective charge and size of the monomer units in determining the polyion equivalent conductivities.The polyion equivalent conductivity is found to decrease as the concentration of the added salt increases and also as the molecular weight of the polyelectrolyte increases. As discussed earlier, there would be more counterion condensation and hence less effective charge on the monomer units either with the increase in the concentration of the added salt or with the increase in the molecular weight of the polymer sample. It, thus, appears that a decrease in the charge of the monomer units alone might have caused the reduction of the polyion equivalent conductivity. On this basis, the dependence of the polyion equivalent conductivity on the polyelectrolyte concentration or on the experimental temperature seems to be anomalous, where with the increase in the polyelectrolyte concentration, although the effective monomer charge increases (please see above), there is a reduction in polyion equivalent conductivity, and with the increase in the experimental temperature, although the monomer charge decreases (please see above), there is an increase in polyion equivalent conductivity. These observations can be interpreted if both the effective monomer size and effective monomer charge are simultaneously taken into consideration. It may be noted that while increasing effective monomer size should cause an increase in the polyion equivalent conductivity, an increase in the monomer size should result in a reduction in it, and it is the subtle balance between these opposing effects which would decide the observed mobility in the present NaPSS–NaCl aqueous solutions. The results obtained thus indicated clearly that the charge effect predominates over the size effect when either the added salt concentration or the polyelectrolyte molecular weight is varied, whereas the size effect overrides the charge effect when either polyelectrolyte concentration or temperature is varied.
Coefficient of friction between the polyion and the solvent (fpw)
Considering that the charge of the polyion is partly screened by the atmospherically bound counterions, the force of the external electric field per monomer unit is supposed to be reduced to the fraction f of its value in absence of counterions.50 According to this assumption, the hydrodynamic friction coefficient per mole of monomer units, fpw, can be obtained from the relation51
 | (18) |
Fig. 5(a) and (b) show the effects of the molecular weight of the polyelectrolyte, added salt concentration and temperature on the fpw vs.  profiles.
profiles.
 | ||
Fig. 5 (a) Coefficient of friction between polystyrenesulfonate ion and the solvent in aqueous solution of NaPSS (molecular weight = 70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000) in presence of NaCl at different temperatures. For the significance of symbols, please refer to Fig. 3(a). (b) Coefficient of friction between polystyrenesulfonate ion and the solvent in aqueous solution of NaPSS (molecular weight = 1 000) in presence of NaCl at different temperatures. For the significance of symbols, please refer to Fig. 3(a). (b) Coefficient of friction between polystyrenesulfonate ion and the solvent in aqueous solution of NaPSS (molecular weight = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000) in presence of NaCl at different temperatures. For the significance of symbols, please refer to Fig. 3(a). 000) in presence of NaCl at different temperatures. For the significance of symbols, please refer to Fig. 3(a). | ||
A delicate balance of three important factors, namely the effective monomer size, the effective monomer charge, and the presence of other species if any, in the aqueous environment governs the value of the coefficient of friction between the polyion and the solvent molecules. These factors will be taken into account in order to interpret the present observations.
A significant increase in the coefficient of friction value of the polystyrenesulfonate ion as a function of the polyelectrolyte concentration was observed in presence of a given amount of the added salt in a solution of a polyelectrolyte with a particular molecular weight and at a particular temperature. Both the monomer charge and the monomer size have been shown to increase as the concentration of polyelectrolyte increases. Both these factors would, of course, cause a greater frictional resistance between the monomers and the solvent molecules in their vicinity at higher polyelectrolyte concentrations.
The monomer units are found to experience lower friction the higher the concentration of the added salt (NaCl) over the entire polyelectrolyte concentration range. Addition of NaCl to a polyelectrolyte solution of a particular concentration leads to a reduction both in charge and the size of the monomer units, and this would result in a consequent decrease in the friction of the monomer units with the solvent molecules.
An elevation of temperature is found to reduce the frictional resistance of the monomers with the solvent molecules in a specified polyelectrolyte solution provided the polymer molecular weight remains the same. Increasing temperature induces more counterion condensation thus causing polyion chain contraction, i.e., there is a reduction both in monomer charge and monomer size upon temperature elevation and hence gives rise to weaker frictional resistance. This supports the phenomenon of gradual dehydration of the monomer units as inferred earlier.
A significant decrease in the friction coefficient with increasing molecular weight of the polymer over the entire polyelectrolyte concentration range in presence of a given amount of the added salt at a given temperature was also observed. Here, the fraction of free counterions and hence the charge on the monomer units decrease with increasing molecular weight. This would result in a reduction in the effective monomer size. Hence the coefficient of friction would also decrease with increasing polyelectrolyte molecular weight.
Conclusions
The interaction between a polyelectrolyte sodium polystyrenesulfonate and sodium chloride in aqueous milieu has been investigated using electrical conductivity data coupled with a recently developed model using the scaling description for the conformation of a polyion chain. Higher level of counterion condensation takes place with decreasing polyelectrolyte concentration, increasing temperature, increasing polyelectrolyte molecular weight and increasing added salt concentration. Increasing polyelectrolyte concentration caused an increase in the dielectric constant of the medium with a concomitant reduction in counterion condensation. Higher temperatures led to a dehydration of counterions and hence to a higher level of counterion condensation. The variations of polyion equivalent conductivity with polyelectrolyte concentration, concentration of the added salt, experimental temperature, and the polyelectrolyte molecular weight have been interpreted considering simultaneously the roles of the effective monomer size and effective monomer charge. In the present system, the variation of polyion equivalent conductivity with the added salt concentration or with the polyelectrolyte molecular weight is governed by the charge effect, while that with the polyelectrolyte concentration or with the temperature is governed by the size effect. The values of the coefficient of friction of the monomer with the solvent molecules are governed both by the monomer charge and the monomer size. It has been demonstrated that the coefficients of friction increase with increasing polyelectrolyte concentration, whereas these decrease either with an increase in the temperature, or an increase in the concentration of the added salt, or with the increase in the polyelectrolyte molecular weight. The present study provided new insight into the effects of polyelectrolyte molecular weight on the fractions of free counterions, the polyion mobility and the monomer-solvent friction coefficients in polyelectrolyte-salt solutions, which was not investigated earlier.Acknowledgements
This work is supported by the Science & Engineering Research Board/Department of Science and Technology, New Delhi, Government of India [SR/S1/PC-67/2010]. The authors gratefully acknowledge the partial financial support by the Presidency University through Faculty Research & Professional Development Fund (FRPDF) Scheme for 2014–15.References
- H. Dautzenberg, W. Jager, J. Koetz, B. Philipp, C. Seidel and D. Stscherbina. Polyelectrolytes: formation, characterization and application, Hanser Publishers, Munich, 1994, ch. 5 Search PubMed.
- C. Wandrey and D. Hunkeler, Handbook of polyelectrolytes and their applications, ed. Tripathy S. K., Kumar J. and Nalwa H. S., American Scientific, Stevenson Ranch, 2002, vol. 2, p. 147 Search PubMed.
- S. Förster and M. Schmidt, Polyelectrolytes in solution, Adv. Polym. Sci., 1995, 120, 51–133 CrossRef.
- A. Bhattacharya, Radiation and Industrial Polymers, Prog. Polym. Sci., 2000, 25, 371–401 CrossRef CAS.
- G. S. Manning, Counterion condensation theory of attraction between like charges in the absence of multivalent counterions, Eur. Biophys. J., 2011, 34, 11132–11136 Search PubMed.
- Q. Liao, A. Dobrynin and M. Rubinstein, Molecular Dynamics Simulations of Polyelectrolyte Solutions: Osmotic Coefficient and Counterion Condensation, Macromolecules, 2003, 36, 3399–3410 CrossRef.
- G. S. Manning, Excess counterion condensation on polyelectrolyte kinks and branch points and the interaction of skewed charged lines, Soft Matter, 2014, 10, 3738–3747 RSC.
- W. Essafi, F. Lafuma, D. Baigl and C. E. Williams, Anomalous counterion condensation in salt-free hydrophobic polyelectrolyte solutions: Osmotic pressure measurements, Europhys. Lett., 2005, 71, 10175–10182 CrossRef.
- H. Takano, I. Baba, D. Kubota and S. Miyashita, Counterion condensation and self-condensation of single polyelectrolytes, J. Phys. Soc. Jpn., 2010, 79, 013801–013804 CrossRef.
- B. O'Shaughnessy and Q. Yang, Manning–Oosawa counterion condensation, Phys. Rev. Lett., 2005, 94, 048302 CrossRef.
- G. Lamm and G. R. Pack, Counterion condensation and shape within Poisson–Boltzmann theory, Biopolymers, 2010, 93, 619–639 CrossRef CAS PubMed.
- H.-P. Hsu and E. Lee, Counterion condensation of a polyelectrolyte, Electrochem. Commun., 2012, 15, 59–62 CrossRef CAS PubMed.
- T. Vuletić, S. Dolanski-Babić, D. Grgičin, D. Aumiler, J. Rädler, F. Livilant and S. Tomić, Manning free counterion fraction for rodlike polyion: Aqueous solutions of short DNA fragments in presence of very low added salt, Phys. Rev. E: Stat., Nonlinear, Soft Matter Phys., 2011, 83, 04180301–04180308 CrossRef.
- F. Bordi, F. Cametti and T. Gili, Electrical conductivity of polyelectrolyte solutions in the presence of added salt: The role of solvent quality factor in light of a scaling approach, Phys. Rev. E: Stat., Nonlinear, Soft Matter Phys., 2003, 68, 011805 CrossRef CAS.
- F. Bordi, H. Colby, F. Cametti, L. de Lorenzo and T. Gili, Electrical conductivity of polyelectrolyte solutions in the semidilute regime: The role of counterion condensation, J. Phys. Chem., 2002, 106, 6887–6893 CrossRef CAS.
- F. Bordi, H. Colby, F. Cametti, L. de Lorenzo and T. Gili, Electrical conductivity of dilute and semidilute aqueous polyelectrolyte solutions. A scaling theory approach, J. Phys. Chem., 1999, 103, 5092–5097 CrossRef CAS.
- F. Bordi, C. Cametti and R. H. Colby, Dielectric spectroscopy and conductivity of polyelectrolyte solutions, J. Phys.: Condens. Matter, 2004, 16, R1423–R1463 CrossRef CAS.
- G. S. Manning, Limiting laws and counterion condensation polyelectrolyte solutions. I. Colligative properties, J. Chem. Phys., 1969, 51, 924–933 CrossRef CAS PubMed.
- G. S. Manning, Limiting laws and counterion condensation polyelectrolyte solutions. II. Self-diffusion of the small ions, J. Chem. Phys., 1969, 51, 934–938 CrossRef PubMed.
- G. S. Manning, A limiting law for the conductance of the rod model of a salt-free polyelectrolyte solution, J. Phys. Chem., 1975, 79, 262–265 CrossRef CAS.
- J. C. T. Kwak and R. C. Hayes, Electrical conductivity of aqueous solutions of salts of polystyrenesulfonic acid with univalent and divalent counterions, J. Phys. Chem., 1975, 79, 265–269 CrossRef CAS.
- J. C. T. Kwak and A. J. Johnston, The equivalent conductivity of aqueous solutions of salts of carboxymethylcellulose: A test of Manning’s limiting law, Can. J. Chem., 1975, 53, 792–796 CrossRef CAS PubMed.
- P. Beyer and E. Nordmeier, Some phenomena of counterion condensation on dextran sulfate, Eur. Polym. J., 1995, 31, 1031–1036 CrossRef CAS.
- H. Abramovic and C. Klofutar, Conductivity of some tetra-n-alkylammonium salts of pectic acid, Eur. Polym. J., 1997, 33, 1295–1300 CrossRef CAS.
- H. E. Rios, Electrical conductivity properties of poly[(maleic acid)-co-(vinyl pyrrolidone) salts in aqueous solutions, Polym. Int., 2001, 50, 885–890 CrossRef CAS PubMed.
- P. Nandi and B. Das, Effects of concentration, relative permittivity, and temperature on the solution behavior of sodium carboxymethylcellulose as probed by electrical conductivity, J. Phys. Chem. B, 2005, 109, 3238–3242 CrossRef CAS PubMed.
- R. H. Colby, D. C. Boris, W. E. Krause and J. S. Tan, Polyelectrolyte conductivity, J. Polym. Sci., Part B: Polym. Phys., 1997, 35, 2951–2960 CrossRef CAS.
- A. V. Dobrynin, R. H. Colby and M. Rubinstein, Scaling theory of polyelectrolyte solutions, Macromolecules, 1995, 28, 1859–1871 CrossRef CAS.
- D. Ghosh, A. Bhattarai and B. Das, Electrical conductivity of sodium polystyrenesulfonate in acetonitrile–water mixed solvent media: Experiment and data analysis using the Manning counterion condensation model and the scaling theory approach, Colloid Polym. Sci., 2009, 287, 1005–1011 CAS.
- A. Chatterjee, B. Das and C. Das, Polyion–counterion interaction behavior for sodium carboxymethylcellulose in methanol–water mixed solvent media, Carbohydr. Polym., 2012, 87, 1144–1152 CrossRef CAS PubMed.
- R. Sharma, C. Das, S. Dahal and B. Das, Polyion–counterion interactions in sodium carboxymethylcellulose–ethylene glycol–water ternary solutions, Carbohydr. Polym., 2013, 92, 1546–1554 CrossRef CAS PubMed.
- C. Das and B. Das, Transport, and thermodynamic investigations on sodium polystyrenesulfonate in ethylene glycol-water media, J. Chem. Thermodyn., 2014, 68, 293–302 CrossRef CAS PubMed.
- R. De and B. Das, Electrical conductances of sodium polystyrenesulphonate in 2-ethoxyethanol (1) + water (2) mixed solvent media in presence of sodium chloride at (308.15, 313.15, 318.15, and 323.15), Korean J. Chem. Eng., 2010, 55, 2108–2115 CAS.
- P. Nandi and B. Das, Electrical conductances of sodium carboxymethylcellulose in acetonitrile (1) + water (2) mixed solvent media in the presence of sodium chloride at 308.15 K, J. Chem. Eng. Data, 2011, 56, 2870–2876 CrossRef CAS.
- B. Das and A. Chatterjee, Salt-induced counterion condensation and related phenomena in sodium carboxymethylcellulose-sodium halide-methanol-water quaternary systems, Soft Matter, 2015, 11, 4133–4140 RSC.
- A. Takahashi, T. Kato and M. Nagasawa, The osmotic pressure of polyelectrolyte in neutral salt solutions, J. Phys. Chem., 1967, 71, 2001–2010 CrossRef.
- J. C. Yang, M. J. Jablonsky and J. W. Mays, NMR and FT-IR studies of sulfonated styrene-based homopolymers and copolymers, Polymer, 2002, 43, 5125–5132 CrossRef CAS.
- J. Xu, X. Cui, H. Wang, J. Li and S. Dong, Preparation of ultrafine poly(sodium 4-styrenesulfonate) fibres via electrospinning, Bull. Mater. Sci., 2011, 34, 531–533 CrossRef CAS.
- J. E. Lind Jr, J. J. Zwolenik and R. M. Fuoss, Calibration of conductance cells at 25° with aqueous solutions of potassium chloride, J. Am. Chem. Soc., 1959, 81, 1557–1559 CrossRef.
- B. Das and D. K. Hazra, Apparent and partial molal volumes of selected symmetrical tetraalkylammonium bromides in 2-methoxy-1-ethanol at 25 °C, J. Chem. Eng. Data, 1991, 36, 403–405 CrossRef CAS.
- B. Das and N. Saha, Electrical conductances of some symmetrical tetraalkylammonium salts in methanol, acetonitrile, and methanol(1) + acetonitrile(2) Mixtures at 298.15 K, J. Chem. Eng. Data, 2000, 45, 2–5 CrossRef CAS.
- B. Das, N. Saha and D. K. Hazra, Ionic association and conductances of some symmetrical tetraalkylammonium salts in methanol, acetonitrile, and methanol(1) + acetonitrile(2) mixtures at 298.15 K, J. Chem. Eng. Data, 2000, 45, 353–357 CrossRef CAS.
- Electrolyte solutions, ed. Robinson R. A. and Stokes R. H., Butterworths, London, 1959 Search PubMed.
- F. Oosawa, Polyelectrolytes, Marcel Dekker, New York, 1993 Search PubMed.
- U. Mohanty and Y. Zhao, Polarization of Counterions in Polyelectrolytes, Biopolymers, 1996, 38, 377–388 CrossRef CAS.
- R. L. Cleland, Electrophoretic mobility of wormlike chains. 2. Theory, Macromolecules, 1991, 24, 4391–4402 CrossRef CAS.
- F. V. D. Touw and M. Mandel, Dielectric increment and dielectric dispersion of solutions containing simple charged linear macromolecules. II. Experimental results with synthetic polyelectrolytes, Biophys. Chem., 1974, 2, 231–241 CrossRef.
- G. S. Manning, The critical onset of counterion condensation: A survey of its experimental and theoretical basis, Ber. Bunsenges. Phys. Chem., 1996, 100, 909–922 CrossRef CAS PubMed.
- A. Chatterjee and B. Das, Electrical conductances of tetrabutylammonium bromide, sodium tetraphenylborate and sodium bromide in methanol (1) + water (2) at (298.15, 308.15, and 318.15), Korean J. Chem. Eng., 2006, 51, 1352–1355 CAS.
- A. Katchalsky, Polyelectrolytes, Pure Appl. Chem., 1971, 26, 327–373 CrossRef CAS.
- I. A. Kuznestov, O. B. Stanislavskii and I. S. Kudryavtseva, Equivalent electrical conductivity of salt-free solutions of composite tRNA from E. coli. Structural charge density parameter of the “stretched” conformation, Russ. J. Phys. Chem., 1990, 64, 1091–1094 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra10933g |
| This journal is © The Royal Society of Chemistry 2015 |



