Temperature-responsive proton-conductive liquid crystals formed by the self-assembly of zwitterionic ionic liquids†
Xinpei Gaoa,
Fei Lua,
Bin Dongb,
Tao Zhoua,
Yizhi Liua and
Liqiang Zheng*a
aKey laboratory of Colloid and Interface Chemistry, Shandong University, Ministry of Education, Jinan, 250100, P. R. China. E-mail: lqzheng@sdu.edu.cn; Fax: +86-531-88564750; Tel: +86-531-88361528
bDalian Institute of Chemical Physics, Chinese Academy of Sciences, Dalian, 116023, P. R. China
First published on 20th July 2015
Abstract
In the present study, we synthesized a series of ionic liquids by mixing amphiphilic imidazolium-type zwitterions with sulfonic acids containing different substituent groups. Nanostructured proton conductors having hexagonal and cubic structures were constructed by the self-assembly of these zwitterionic ionic liquids. These nanostructured proton conductors exhibited an assembled-structure dependent proton conduction behavior. The introduction of highly ordered liquid crystal structures efficiently improved ionic conductivity, suggesting the induction of proton conduction through a hopping mechanism. Temperature-responsive ionic conductivity behavior based on phase transition within the self-assembled liquid crystal structures was also observed.
Introduction
Proton conducting gel electrolytes, as appropriate alternatives to liquid and solid electrolytes, are very important components of clean energy devices.1–4 Current studies are mainly about polymer gel electrolytes and improving their ionic conductivity behaviors.5–7 Introduction of liquid-crystalline properties provides a new perspective for the development of advanced functional gel electrolytes.8 Ionic liquid crystals (ILCs), which combined the properties of liquid crystals (LCs) and ionic liquids (ILs), have attracted a great deal of attention due to their unique ability to construct nanostructured and well-organized ion-conductive channels.9,10 The proper use of ILCs has been found to be a versatile strategy for the development of new electrolyte materials forming efficient 1D,11 2D,12 and 3D13 ion-conductive pathways. More significantly, due to the additive effects of the LCs intrinsic anisotropy, several stimuli responsive ion-conductive liquid-crystalline materials such as magnetic-field-responsive,14 electric-field-responsive,15 and photo-responsive16 have been developed.ILs are now not only considered as important alternative solvents, but as materials with unique and tunable properties which can be easily adjusted by suitable selection of cations and anions for a specific need.17 As a result of their unique combination of properties such as low volatility, nonflammability, high thermal stability, and high ionic conductivity,18 ILs have been widely studied as designable electrolyte materials for energy devices such as fuel cells,19 dye-sensitized solar cells,20 thermoelectrochemical cells,21 and lithium ion batteries.22 However, ILs also have drawbacks as candidates for new generation electrolyte materials. On the one hand, migration of ILs component ions along with the potential gradient inevitably reduces the transport number of the target ion.23 On the other hand, the lack of selectivity in ion transportation also remains as a serious problem in devices where target ion conduction is required.24 Taking all these factors into account, it is very necessary to design a new type of ILs for better ion transportation system.
To overcome these drawbacks, zwitterions, in which a pair of cation and anion is covalently tethered, have been proposed as a medium for selective target ion transport.25–27 Due to their intramolecularly electroneutral characteristic, the zwitterions are expected not to migrate along with the potential gradient. Therefore the applied voltage can be used to operate the target ions.23 These compounds were first pioneered by Ohno and co-workers, which were initially considered as solvents or ion dissociators.25 Although zwitterions were generally farinose solids with high isotropization temperatures, a new type of IL derivatives, zwitterionic ILs, were obtained upon equimolar addition of ionic salts. The high dipole moment of the zwitterions explains their ability to solvate various ionic salts.28 Development of zwitterionic ILs-based liquid-crystalline materials through self-assembly enables the formation of ion-conductive channels where target ions can efficiently transport in desired directions.29
In the present work, we synthesized a series of amphiphilic imidazolium-based zwitterions. A series of zwitterionic ILs were obtained by mixing equimolar zwitterions with sulfonic acids containing different substituent groups. Nanostructured proton conductors having hexagonal and cubic structures were constructed by the self-assembly of these zwitterionic ILs. The microstructures and phase behaviors of the obtained ILCs were studied by polarized optical microscope (POM) and small-angle X-ray scatting (SAXS). The ionic conductivities of ILCs were investigated by alternating current impedance method. All these nanostructured proton conductors exhibited an assembled-structure dependent proton conduction behavior. Temperature-responsive ionic conductivity behavior based on phase transitions within the self-assembled liquid crystal structures was also confirmed.
Experimental section
Materials
Methanesulfonic acid (CH3SO3H, 99%), trifluoromethanesulfonic acid (CF3SO3H, 99%), benzenesulfonic acid (C6H5SO3H, 95%), 1-bromododecane (98%), 1-bromotetradecane (98%), 1-bromohexadecane (98%), 1,3-propanesultone (99%), and imidazole (99%) were purchased from J&K Scientific Ltd. Sodium hydroxide, acetone and tetrahydrofuran were purchased from Shanghai Chemical Co. All the above materials were used without further purification. Deionized water was used through the experiment.C12IPS: δ = 9.177 (1H, s), 7.800 (1H, t, J = 1.8 Hz), 7.780 (1H, t, J = 1.8 Hz), 4.298 (t, 2H, J = 7.2 Hz), 4.143 (2H, t, J = 7.2 Hz), 2.392 (2H, t, J = 7.2 Hz), 2.088 (2H, quint, J = 7.2 Hz), 1.804 (2H, m), 1.239 (18H, m), 0.85 (t, 3H, J = 6.6 Hz).
C14IPS: δ = 9.805 (1H, s), 7.307 (1H, t, J = 1.8 Hz), 7.151 (1H, t, J = 1.8 Hz), 4.609 (t, 2H, J = 6.9 Hz), 4.258 (2H, t, J = 7.5 Hz), 2.876 (2H, t, J = 6.9 Hz), 2.418 (2H, quint, J = 6.9 Hz), 1.891 (2H, m), 1.323 (22H, m), 0.878 (t, 3H, J = 6.6 Hz).
C16IPS: δ = 9.804 (1H, s), 7.334 (1H, t, J = 1.8 Hz), 7.152 (1H, t, J = 1.8 Hz), 4.604 (t, 2H, J = 6.9 Hz), 4.260 (2H, t, J = 7.5 Hz), 2.876 (2H, t, J = 6.9 Hz), 2.419 (2H, quint, J = 6.9 Hz), 1.888 (2H, m), 1.250 (26H, m), 0.878 (t, 3H, J = 6.6 Hz).
Results and discussion
The aim of the present study is to construct nanostructured proton conductors with well-defined proton nanochannels by self-assembly of zwitterionic ILs that contain a mobile proton. The advantage of using zwitterionic ILs rather than conventional ILs lies in the feasibility to select counterions and tune self-assembled structures by changing the species of acids. A large amount of researches have proved that the aggregation behavior of ILs can be consciously adjusted by variation of alkyl chain lengths in imidazolium cations and the anionic counterion species.32–34 Benefiting from avoiding the time-consuming ion-exchanges process, this strategy becomes more convenient for zwitterionic ILs systems. Sulfonic acids containing different substituent groups (CH3SO3H, C6H5SO3H, and CF3SO3H) shown in Fig. 1a–c were chosen as proton donors. The chemical structure of the zwitterions designed in the present study is shown in Fig. 1d. The amphiphilic zwitterion 3-(1-alkyl-3-imidazolio)propanesulfonate (CnIPS, n is the number of carbon atoms in the alkyl chain) consists of two incompatible parts: a hydrophobic alkyl chain part and a hydrophilic imidazolium zwitterionic part. These zwitterions were obtained as white farinose solids with high melting points. A series of hydrophilic zwitterionic ILs were obtained by evaporation of methanol solutions of equimolar CnIPS and sulfonic acids. The decrease in the melting point of the zwitterions with the addition of sulfonic acids is explained by the hard–soft acid–base principle. The preferential interactions between the soft imidazolium cation and the soft CH3SO3−, C6H5SO3−, and CF3SO3− anions play the key role in the formation of ILs [CnIPS][R–SO3H]. The melting points of the synthesized zwitterionic ILs were characterized by differential scanning calorimetry (DSC) measurements and the Tm values are listed in Table 1. The IL [C12IPS][CH3SO3H] exhibits the lowest Tm value, and the values of Tm appear to rise steadily with increasing the alkyl chain length. The same varying tendency is observed with increasing anionic radii of sulfonic acids. However, the melting temperatures of ILs are generally decreased with increasing anion radius. It is because the larger anion radius induces weaker electrostatic interaction with imidazolium cation.35 Among the ILs [C12IPS][R–SO3H], [C12IPS][CF3SO3H] has a much higher Tm than [C12IPS][CH3SO3H]. This phenomenon could be attributed to strong hydrogen bonds for the sake of F atoms.35 Due to the additional π–π stacking interactions between phenyl ring and imidazolium cation,36 the melting point of [C12IPS][C6H5SO3H] is also comparatively higher than [C12IPS][CH3SO3H].| [C12IPS][CF3SO3H] | [C12IPS][C6H6SO3H] | [C12IPS][CH3SO3H] | [C14IPS][CH3SO3H] | [C16IPS][CH3SO3H] | |
|---|---|---|---|---|---|
| Tm/°C | 84 | 71 | 65 | 77 | 85 |
It is generally accepted that nanosegregation at a molecular scale, leads to the formation of a variety of sophisticated topological structures in ILCs.8 To promote the nanosegregation between the hydrophobic alkyl chain part and hydrophilic part of amphiphilic zwitterions, the ILs [CnIPS][R–SO3H] underwent further addition of water. The phase transition behavior of ILs [CnIPS][R–SO3H] aqueous mixtures is summarized in Fig. 2 using a bar graph. The characterization of these LC phases was determined by combination of POM and SAXS measurements. For all the systems, single-phase regions consists of the isotropic solution phase (L1) and anisotropic hexagonal phase (H1). It is noteworthy that isotropic cubic phase (Pm3n) exists in the [C12IPS][CH3SO3H] and [C14IPS][CH3SO3H] systems but disappears when the alkyl chain length is elongated to 16. Interestingly, the isotropic cubic phase also disappears with increasing anionic radii of sulfonic acids in [C12IPS][R–SO3H] systems. It is not difficult to find that the phase transition behavior of ILs [CnIPS][R–SO3H] is consistent with the Tm variation tendency. That is because both Tm and phase transition behavior of ILs are essentially governed by electrostatic interactions between anions and cations. The hydrophilic headgroup area of the amphiphilic zwitterions is determined by the effective occupied area of anion and cation, which ultimately affects the critical packing parameter. This explains why the IL [C12IPS][CH3SO3H] with lower Tm has a more diverse LC phase behavior. Besides the nanosegregation between hydrophilic and hydrophobic moieties of the mixtures, the formation of hydrogen bonding networks through the water molecules and sulfonic acid groups also plays a key role in the formation of LCs,29 which determines that the [C12IPS][CF3SO3H] system possess a relatively wide LC phase region from 30 wt% to 80 wt%.
 | ||
| Fig. 2 Phase diagram of aqueous mixture of [CnIPS][R–SO3H] at 25 °C. L1, isotropic solutions; Pm3n, cubic liquid crystalline phase; H1, hexagonal liquid crystalline phase. | ||
Fig. 3a shows the SAXS patterns of the [C12IPS][CH3SO3H] aqueous mixture system with increasing [C12IPS][CH3SO3H] content collected at 25 °C. As shown in Fig. 2, mixtures containing up to 50 wt% [C12IPS][CH3SO3H] is isotropic solution phase (L1), and further increase of [C12IPS][CH3SO3H] content turns the mixtures into LCs, and an isotropic cubic phase is found when the content is between 55 and 75 wt%. The SAXS pattern for 60 wt% sample collected at 25 °C shows three intensive reflection peaks (marked with red arrows) with relative ratios of

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif)
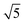
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif)
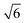 , which can be indexed as the (200), (210) and (211) reflections of a cubic structure with Pm3n symmetry. This space group37,38 allows the Bragg reflections hkl = 110, 200, 210, 211, 220, …, which give the d ratios at the peaks follow the order
, which can be indexed as the (200), (210) and (211) reflections of a cubic structure with Pm3n symmetry. This space group37,38 allows the Bragg reflections hkl = 110, 200, 210, 211, 220, …, which give the d ratios at the peaks follow the order 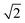 ,
, 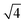 ,
,  ,
, 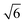 ,
,  , …, of which the second and third reflections are the most intense corresponding to hkl = 200 and 210, respectively, while the first reflection corresponding to hkl = 110 is usually weak and sometimes cannot be observed.39 As the content of [C12IPS][CH3SO3H] increased to 80 wt%, a phase transition from the Pm3n cubic phase to the H1 phase occurres. The SAXS pattern of 80 wt% sample shows three reflection peaks (marked with blue arrows) with relative ratios of 1
, …, of which the second and third reflections are the most intense corresponding to hkl = 200 and 210, respectively, while the first reflection corresponding to hkl = 110 is usually weak and sometimes cannot be observed.39 As the content of [C12IPS][CH3SO3H] increased to 80 wt%, a phase transition from the Pm3n cubic phase to the H1 phase occurres. The SAXS pattern of 80 wt% sample shows three reflection peaks (marked with blue arrows) with relative ratios of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif)

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, which are assigned to be the (100), (110), and (200) reflections of the hexagonal columnar structure. The phase behavior of [C12IPS][CH3SO3H] aqueous mixture system depends on not only the water content, but also the temperature. Fig. 3b shows the SAXS patterns of 70 wt% sample collected as temperature varying from 5 to 85 °C. The SAXS patterns of 70 wt% sample give reflections of the cubic structure with Pm3n symmetry below 45 °C. The scattering peak assigned to the (200), (210) reflections of Pm3n cubic structure gradually disappeared as the temperature rises. Eventually, only reflections of hexagonal columnar structure were observed when the temperature was above 65 °C. The phase transition from isotropic Pm3n to anisotropic H1 on heating and phase transition from H1 to Pm3n on cooling were also visually observed through POM (Fig. 4). The POM image of 70 wt% sample showed no birefringence at 25 °C, and the dark image indicates the existence of an optically isotropic cubic LC phase. Bright and colorful birefringence corresponds to H1 phase gradually appeared as the temperature rose and faded away as the temperature dropped. This observation significantly indicates the reversible transition between Pm3n isotropic cubic phase and anisotropic H1 hexagonal phase. The same protocols were used to assess other [CnIPS][R–SO3H] systems and the results were shown in ESI.† The relevant structural parameters of the different types of LC phases were deduced from the SAXS pattern and the values are listed in Table S1 in ESI.†
2, which are assigned to be the (100), (110), and (200) reflections of the hexagonal columnar structure. The phase behavior of [C12IPS][CH3SO3H] aqueous mixture system depends on not only the water content, but also the temperature. Fig. 3b shows the SAXS patterns of 70 wt% sample collected as temperature varying from 5 to 85 °C. The SAXS patterns of 70 wt% sample give reflections of the cubic structure with Pm3n symmetry below 45 °C. The scattering peak assigned to the (200), (210) reflections of Pm3n cubic structure gradually disappeared as the temperature rises. Eventually, only reflections of hexagonal columnar structure were observed when the temperature was above 65 °C. The phase transition from isotropic Pm3n to anisotropic H1 on heating and phase transition from H1 to Pm3n on cooling were also visually observed through POM (Fig. 4). The POM image of 70 wt% sample showed no birefringence at 25 °C, and the dark image indicates the existence of an optically isotropic cubic LC phase. Bright and colorful birefringence corresponds to H1 phase gradually appeared as the temperature rose and faded away as the temperature dropped. This observation significantly indicates the reversible transition between Pm3n isotropic cubic phase and anisotropic H1 hexagonal phase. The same protocols were used to assess other [CnIPS][R–SO3H] systems and the results were shown in ESI.† The relevant structural parameters of the different types of LC phases were deduced from the SAXS pattern and the values are listed in Table S1 in ESI.†
 | ||
| Fig. 4 Polarizing optical microscopy images recording the 70 wt% [C12IPS][CH3SO3H] phase transition behavior. Phase transition from Pm3n to H1 on heating and H1 to Pm3n on cooling. | ||
The phase behavior of the ILs [CnIPS][R–SO3H] aqueous mixtures was taken as a guideline for the study of ionic conduction behavior. The study of well-defined nanostructure in the LCs could help us have a better understanding of the ionic conduction behavior. To evaluate these ILs [CnIPS][R–SO3H] aqueous mixtures as proton conductive materials, we performed ionic conductivity measurements by the alternating current impedance method. Fig. 5a shows the Arrhenius plots of ionic conductivity of [C12IPS][CH3SO3H] aqueous mixture in different LC phases during the heating process: 60 wt% sample in Pm3n phase (black circles) and 80 wt% sample in H1 phase (red circles). The ionic conductivities of 70 wt% sample on heating (black squares) are shown in Fig. 5b. It is of interest to note that, although the ionic conductivity of [C12IPS][CH3SO3H] aqueous mixture simply increases with the rise in temperature, all these nanostructured LCs exhibit an assembled-structure dependent proton conduction behavior. The ionic conductivity of the sample with 80 wt% [C12IPS][CH3SO3H] in H1 phase is 1.53 × 10−4 S cm−1 at 20° C and the conductivity increases to 5.57 × 10−3 S cm−1 upon heating. The highest ionic conductivity observed at 80 °C is about 36 times higher than that observed at 20 °C. But at the same temperature range from 20 to 80 °C, the ionic conductivity of the sample with 60 wt% [C12IPS][CH3SO3H] in Pm3n phase slightly increased from 3.06 × 10−2 to 4.68 × 10−2 S cm−1, which is virtually free from the influence of temperature. The activation energies of [C12IPS][CH3SO3H] aqueous mixture in different LC phases were estimated from the Arrhenius plots of the conductivity to better illustrate the proton conduction mechanism. The activation energy of the sample in H1 phase is estimated to be 22.35 kJ mol−1 while the activation energy of the sample in Pm3n cubic phase is estimated to be 2.69 kJ mol−1, which is about 10 times lower than that of the sample in H1 phase. The decrease in activation energies can be attributed to the different conduction mechanism in each material. We conclude that the proton conduction within the hexagonal columnar phase is mainly dominated by the diffusion controlled transport (vehicle) mechanism.10,29 As a result of their ordered assemblies at the molecular level, the sample in H1 phase can form low-dimensional hydrophilic channels between the columnar structures. The proton transporting inside these hydrophilic channels should be induced by the hopping conduction via hydrogen-bonded water networks as proton conductive pathways.8,40 But these hydrophilic channels between hexagonal columnar structure are not continuous on macroscopic scale. The proton transporting between these hydrophilic channels is still controlled by the vehicle mechanism, which ultimately acts as the rate-controlling step. While in the Pm3n cubic phase, 3D continuous hydrophilic channels can provide macroscopically continuous proton conductive pathways as a result of their 3D periodical LC structure. We infer that the drastic enhancement of ionic conductivity and the decrease in activation energy within the Pm3n cubic phase can be attributed to the change of conduction mechanism from vehicular to hopping (Grotthuss) mechanism.8,10 Another remarkable result was observed in the Arrhenius plots of ionic conductivity of 70 wt% sample on heating process (black squares) as shown in Fig. 5b. At the temperature range from 20 to 40 °C, the ionic conductivity and variation tendency of 70 wt% sample is quite similar to that of 60 wt% sample in Pm3n phase. While the ionic conductivity decreases suddenly as the temperature around the phase transition temperature from Pm3n cubic phase to hexagonal columnar phase following the disruption of the 3D continuous hydrophilic channel structures, which further validated our speculation about the assembled-structure dependent proton conduction behavior.
Conclusion
In summary, we have synthesized a series of ionic liquids by mixing amphiphilic imidazolium-type zwitterions with sulfonic acids containing different substituent groups. Benefiting from the using of zwitterionic ILs, it is very convenient to select target ions and tune self-assembled structures by changing the structure of counterions. Based on the aggregation behaviors of these amphiphilic zwitterionic ILs, nanostructured proton conductors having hexagonal and cubic structures were constructed. These nanostructured proton conductors all exhibited an assembled-structure dependent proton conduction behavior. The introduction of highly ordered liquid crystalline structures efficiently improved ionic conductivity, suggesting the induction of proton conduction through a hopping mechanism. Temperature-responsive ionic conductivity behavior based on phase transition within the self-assembled liquid crystalline structures was also observed. We believe that the design of materials based on the principle of self-organization of zwitterionic ILs has clear potential to create a new generation of functional ion transportation system.Acknowledgements
The authors are grateful to the National Basic Research Program (2013CB834505), the Shandong Provincial Natural Science Foundation, China (ZR2012BZ001) and Specialized Research Fund for the Doctoral Program of Higher Education of China (no. 20120131130003).References
- M. Armand, F. Endres, D. R. MacFarlane, H. Ohno and B. Scrosati, Nat. Mater., 2009, 8, 621–629 CrossRef CAS PubMed.
- E. Tunkara, C. Albayrak, E. O. Polat, C. Kocabas and O. Dag, ACS Nano, 2014, 8, 11007–11012 CrossRef CAS PubMed.
- L. E. Shmukler, N. V. Thuc, Y. A. Fadeeva and L. P. Safonova, J. Appl. Polym. Sci., 2014, 131, 40674 CrossRef PubMed.
- Q. Chen, X. Li, X. Zang, Y. Cao, Y. He, P. Li, K. Wang, J. Wei, D. Wu and H. Zhu, RSC Adv., 2014, 4, 36253–36256 RSC.
- S. Y. Shen, R. X. Dong, P. T. Shih, V. Ramamurthy, J. J. Lin and K. C. Ho, ACS Appl. Mater. Interfaces, 2014, 6, 18489–18496 CAS.
- S. T. Senthilkumar, R. Kalai Selvan, N. Ponpandian and J. S. Melo, RSC Adv., 2012, 2, 8937–8940 RSC.
- H. Gao and K. Lian, RSC Adv., 2014, 4, 33091–33113 RSC.
- K. Ichikawa, T. Kato and H. Ohno, J. Am. Chem. Soc., 2012, 134, 11354–11357 CrossRef PubMed.
- K. Binnemans, Chem. Rev., 2005, 105, 4148–4204 CrossRef CAS PubMed.
- X. Gao, F. Lu, L. Shi, H. Jia, H. Gao and L. Zheng, ACS Appl. Mater. Interfaces, 2013, 5, 13312–13317 CAS.
- M. Yoshio, T. Kagata, K. Hoshino, T. Mukai, H. Ohno and T. Kato, J. Am. Chem. Soc., 2006, 128, 5570–5577 CrossRef CAS PubMed.
- J. Sakuda, E. Hosono, M. Yoshio, T. Ichikawa, T. Matsumoto, H. Ohno, H. Zhou and T. Kato, Adv. Funct. Mater., 2015, 25, 1206–1212 CrossRef CAS PubMed.
- R. L. Kerr, S. A. Miller, R. K. Shoemaker, B. J. Elliott and D. L. Gin, J. Am. Chem. Soc., 2009, 131, 15972–15973 CrossRef CAS PubMed.
- X. Feng, M. E. Tousley, M. G. Cowan, B. R. Wiesenauer, S. Nejati, Y. Choo, R. D. Noble, M. Elimelech, D. L. Gin and C. O. Osuji, ACS Nano, 2014, 8, 11977–11986 CrossRef CAS PubMed.
- H. Shimura, M. Yoshio, A. Hamasaki, T. Mukai, H. Ohno and T. Kato, Adv. Mater., 2009, 21, 1591–1594 CrossRef CAS PubMed.
- B. Soberats, E. Uchida, M. Yoshio, J. Kagimoto, H. Ohno and T. Kato, J. Am. Chem. Soc., 2014, 136, 9552–9555 CrossRef CAS PubMed.
- R. D. Rogers and K. R. Seddon, Science, 2003, 302, 792–793 CrossRef PubMed.
- M. Smiglak, J. M. Pringle, X. Lu, L. Han, S. Zhang, H. Gao, D. R. MacFarlane and R. D. Rogers, Chem. Commun., 2014, 50, 9228–9250 RSC.
- S. Y. Lee, A. Ogawa, M. Kanno, H. Nakamoto, T. Yasuda and M. Watanabe, J. Am. Chem. Soc., 2010, 132, 9764–9773 CrossRef CAS PubMed.
- Y. Bai, Y. Cao, J. Zhang, M. Wang, R. Li, P. Wang, S. M. Zakeeruddin and M. Gratzel, Nat. Mater., 2008, 7, 626–630 CrossRef CAS PubMed.
- T. J. Abraham, D. R. MacFarlane and J. M. Pringle, Energy Environ. Sci., 2013, 6, 2639–2645 CAS.
- H. Yoon, G. H. Lane, Y. Shekibi, P. C. Howlett, M. Forsyth and A. S. Best, Energy Environ. Sci., 2013, 6, 979–986 CAS.
- M. Y. Fujita, N. Byrne, M. Forsyth, D. R. MacFarlane and H. Ohno, J. Phys. Chem. B, 2010, 114, 16373–16380 CrossRef PubMed.
- H. Ohno, M. Yoshizawa and W. Ogihara, Electrochim. Acta, 2003, 48, 2079–2083 CrossRef CAS.
- M. Yoshizawa, M. Hirao, K. I. Akita and H. Ohno, J. Mater. Chem., 2001, 11, 1057–1062 RSC.
- C. Tiyapiboonchaiya, J. M. Pringle, J. Sun, N. Byrne, P. C. Howlett, D. R. MacFarlane and M. Forsyth, Nat. Mater., 2004, 3, 29–32 CrossRef CAS PubMed.
- N. Byrne, P. C. Howlett, D. R. MacFarlane and M. Forsyth, Adv. Mater., 2005, 17, 2497–2501 CrossRef CAS PubMed.
- H. Park, H. S. Kim and Y. M. Jung, J. Phys. Chem. B, 2011, 115, 1743–1750 CrossRef CAS PubMed.
- S. Ueda, J. Kagimoto, T. Ichikawa, T. Kato and H. Ohno, Adv. Mater., 2011, 23, 3071–3074 CrossRef CAS PubMed.
- U. H. Choi, M. Lee, S. Wang, W. Liu, K. I. Winey, H. W. Gibson and R. H. Colby, Macromolecules, 2012, 45, 3974–3985 CrossRef CAS.
- L. M. Gan, P. Y. Chow, Z. Liu, M. Han and C. H. Quek, Chem. Commun., 2005, 35, 4459–4461 RSC.
- L. Shi and L. Zheng, J. Phys. Chem. B, 2012, 116, 2162–2172 CrossRef CAS PubMed.
- H. Yan, Y. Long, K. Song, C. H. Tung and L. Zheng, Soft Matter, 2014, 10, 115–121 RSC.
- F. Lu, X. Gao, S. Xie, N. Sun and L. Zheng, Soft Matter, 2014, 10, 7819–7825 RSC.
- S. Zhang, N. Sun, X. He, X. Lu and X. Zhang, J. Phys. Chem. Ref. Data, 2006, 35, 1475–1517 CrossRef CAS PubMed.
- Y. Gu, L. Shi, X. Cheng, F. Lu and L. Zheng, Langmuir, 2013, 29, 6213–6220 CrossRef CAS PubMed.
- P. Sakya, J. M. Seddon, R. H. Templer, R. J. Mirkin and G. J. T. Tiddy, Langmuir, 1997, 13, 3706–3714 CrossRef CAS.
- D. V. Perroni and M. K. Mahanthappa, Soft Matter, 2013, 9, 7919–7922 RSC.
- X. Li and H. Kunieda, Langmuir, 2000, 16, 10092–10100 CrossRef CAS.
- K. D. Kreuer, S. J. Paddison, E. Spohr and M. Schuster, Chem. Rev., 2004, 104, 4637–4678 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available: SAXS patterns and POM images of [C12IPS][C6H5SO3H], [C12IPS][CF3SO3H], [C14IPS][CH3SO3H] and [C16IPS][CH3SO3H] systems. Structural parameters deduced from the SAXS patterns are listed in Table S1. See DOI: 10.1039/c5ra10830f |
| This journal is © The Royal Society of Chemistry 2015 |



