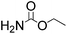
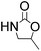
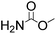
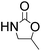
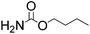
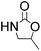
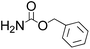
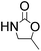
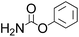
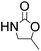
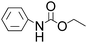
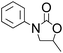
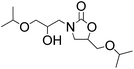
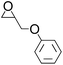
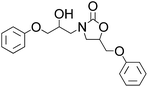
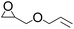Efficient synthesis of 2-oxazolidinones from epoxides and carbamates catalyzed by amine-functionalized ionic liquids†
Jianpeng Shang*a,
Zuopeng Lia,
Caina Sua,
Yong Guoa and
Youquan Dengb
aDepartment of Chemistry and Environmental Engineering, Shanxi Datong University, Datong, 037009, China. E-mail: nmsjp2006@126.com; Fax: +86 352 7624721
bCentre for Green Chemistry and Catalysis, Lanzhou Institute of Chemical Physics, Chinese Academy of Sciences, Lanzhou, 730000, China
First published on 18th August 2015
Abstract
A series of amine-functionalized ionic liquids were prepared and their catalytic performance was tested in the synthesis of 2-oxazolidinones from epoxides and carbamates. Under optimized reaction conditions, good to excellent yields of various 2-oxazolidinones were achieved with different epoxides and carbamates. Moreover, the amine-functionalized ionic liquid catalyst could be easily recovered and reused without significant loss in activity.
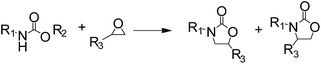
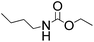
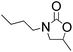
2-Oxazolidinones are important cyclic compounds in both fine chemicals and synthetic organic chemistry. They are widely used in the synthesis of pharmaceuticals, pesticides, cosmetics, and so on.1–3 Since the 2-oxazolidinones preparation generally uses highly toxic phosgene,4 and therefore involves environmental and safety problems, much effort has been made to explore green alternative methods for 2-oxazolidinones synthesis. Several non-phosgene routes for 2-oxazolidinones synthesis have been developed: (1) oxidative carbonylation of β-aminoalcohols with CO and O2, (2) carbonylation of β-aminoalcohols with organic carbonate, (3) cycloaddition of CO2 with β-aminoalcohols or aziridines. Although the oxidative carbonylation of β-aminoalcohols catalyzed by transition metals is an efficient way to produce 2-oxazolidinones,5–7 such a route is not eco-friendly due to the potential explosion hazards and poisonous CO. As an alternative, organic carbonates were used for the synthesis 2-oxazolidinones.8–10 It should be noted that the organic carbonates are mainly produced by phosgenation or oxidative carbonylation routes.11,12 Under these circumstances, the direct synthesis of 2-oxazolidinones from CO2 and β-aminoalcohols13–15 or aziridines16–19 is preferable from environmental and economical viewpoints, however, some stoichiometrically consumed dehydrating reagents and high pressure or high temperature are required in most of these works. Since epoxides are known to produce ring-opened addition products with nucleophilic reagents,20 another non-phosgene route for 2-oxazolidinones synthesis is the reaction between epoxides and carbamates, and then intramolecular cyclization to give the expected 2-oxazolidinones,21–23 Scheme 1. Several alkaline agents,23,24 such as triethylamine, have been reported as catalysts for the 2-oxazolidinones synthesis from epoxides and carbamates. However, the volatility, separation and recovery problems of those catalysts are still maintained and accordingly limit their practical application.
Ionic liquids (ILs), defined as organic salts with melting points below 100 °C, have attracted considerable attention as alternative reaction media and catalytic materials because of their peculiar physicochemical properties. Moreover, various functionalized ILs, incorporating different functional groups into the ILs structure, have been explored and used as solvents or catalysts in chemical processes.25–27 Herein, in order to avoid the use of volatile organic amines, a series of amine-functionalized ILs (Scheme 2) were prepared and their catalytic performances for 2-oxazolidinones synthesis from epoxides and carbamates were tested.
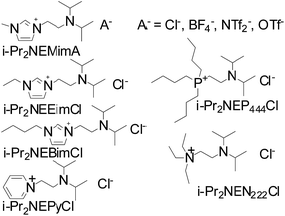 | ||
| Scheme 2 The structure and abbreviation of the amine-functionalized ionic liquids used in this study. | ||
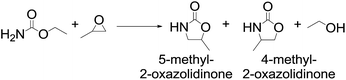
In our initial study, the reaction of propylene oxide (PO) and ethyl carbamate (EC) was chosen as the model reaction to explore the catalyst system, Table 1. Generally, the reaction was carried out at 140 °C for 24 h in the presence of 10 mol% catalyst. In the blank test (entry 1), the conversion of PO was only 12%, suggested that catalyst was essential for such reaction proceeded successfully. When the organic base triethylamine used as catalyst, the conversion of PO reached to 90%, entry 2. Owing to the volatility of organic amine and the high catalytic activity toward this reaction, some amine-functionalized ILs with different cations and anions were prepared and investigated in such reaction, entry 3–12. From the results, it could be found that both the cation and anion of the investigated ILs have strong impact on the catalytic activity, and the highest conversion of PO was achieved with i-Pr2NEMimCl as catalyst (entry 3). The effects of the cation structure on the catalytic performance were tested using pyridinium-, quaternary ammonium- and quaternary phosphonate-based amine-functionalized ILs as catalysts (entries 4–6), the results showed that the catalytic efficiency decreased in the order of i-Pr2NEMimCl > i-Pr2NEP444Cl > i-Pr2NEN222Cl > i-Pr2NEPyCl. The effects of imidazolium based amine-functionalized ILs with different anions (Cl−, BF4−, NTf2−, OTf−) on the reaction was also investigated. The halide anion Cl− gave excellent result, whereas, BF4−, NTf2−, OTf− were found to be inactive (entries 7–9). When the NaCl was used as catalyst (entry 10), the conversion of PO was only 12%, suggested that the single Cl− has no activity for this reaction and the high activity of the i-Pr2NEMimCl may derived from the combination effects of amine group and Cl−. Moreover, the alkyl length of the imidazolium cation has a weak effects on the reaction and the conversion of the PO decreased from 97% to 88% as alkyl chain length of the cation increased from C1 to C4 (entries 3, 11–12), probably duo to the alteration of the solubility of the ionic liquids in the reaction mixture. Furthermore, although the general solid base KF/Al2O3 was found to show some catalytic activity for this reaction (entry 13), the conversion of PO was lower than that of i-Pr2NEMimCl used as catalyst, which suggested that the ionic environment supplied by ILs may be helpful for such reaction.
| Entry | Catalyst (mol%) | T (°C) | t (h) | Conv. (%) | Sel.b (%) | Yieldc (%) |
|---|---|---|---|---|---|---|
| a Reaction conditions: 10 mmol PO; 15 mmol EC; 1 mmol catalysts; 120–140 °C; 8–24 h. Conversions and selectivities were determined by gas chromatography.b Selectivity of the 5-methyl-2-oxazolidinone.c Isolated yield based on the charged PO. | ||||||
| 1 | — | 140 | 24 | 12 | 98 | 11 |
| 2 | Et3N | 140 | 24 | 90 | 99 | 88 |
| 3 | i-Pr2NEMimCl | 140 | 24 | 97 | 99 | 96 |
| 4 | i-Pr2NEPyCl | 140 | 24 | 67 | 99 | 64 |
| 5 | i-Pr2NEN222Cl | 140 | 24 | 73 | 98 | 70 |
| 6 | i-Pr2NEP444Cl | 140 | 24 | 88 | 99 | 84 |
| 7 | i-Pr2NEMimBF4 | 140 | 24 | 0 | — | — |
| 8 | i-Pr2NEMimNTf2 | 140 | 24 | 0 | — | — |
| 9 | i-Pr2NEMimOTf | 140 | 24 | 0 | — | — |
| 10 | NaCl | 140 | 24 | 12 | 98 | 10 |
| 11 | i-Pr2NEEimCl | 140 | 24 | 88 | 98 | 85 |
| 12 | i-Pr2NEBimCl | 140 | 24 | 88 | 98 | 85 |
| 13 | KF/Al2O3 | 140 | 24 | 72 | 99 | 70 |
| 14 | i-Pr2NEMimCl | 120 | 24 | 57 | 98 | 54 |
| 15 | i-Pr2NEMimCl | 130 | 24 | 78 | 98 | 75 |
| 16 | i-Pr2NEMimCl | 140 | 8 | 80 | 99 | 78 |
| 17 | i-Pr2NEMimCl | 140 | 15 | 91 | 98 | 88 |
| 18 | i-Pr2NEMimCl | 140 | 20 | 95 | 98 | 92 |
Subsequently, the influence of the reaction temperature on the reaction was investigated under identical reaction condition. As is easily seen, reaction temperature has a great influence on the reaction outcome with variation of reaction temperature from 120 to 140 °C (entries 3, 14–15), the conversion of PO reached 97% as the reaction temperature was increased up to 140 °C. Additionally, the conversion of PO was increased with increasing the reaction time and reached 97% after 24 h (entries 3, 16–18).
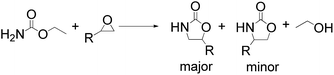
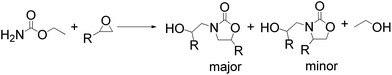
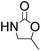
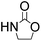
Notably, the 5-methyl-2-oxazolidinone was preferentially formed with high selectivity in all cases listed in Table 1. The major product corresponds to the nucleophilic ring opening of the PO at the less substitute site under basic conditions,28 followed by intramolecular cyclization to produce the corresponding 5-methyl-2-oxazolidinone. Moreover, the 4-methyl-2-oxazolidinone as byproduct was observed, which was formed duo to the nucleophilic ring opening of the PO at methyl substitute site.
The recycling performance of i-Pr2NEMimCl was also investigated in the reaction of EC and PO, Fig. 1. The results showed a slight decrease in the activity after each run, which could be ascribed to the fact that i-Pr2NEMimCl was recovered only 95% of the charged amount after each run by extracted with diethyl ether, suggesting that the i-Pr2NEMimCl catalyst could be reused without significantly loss in activity.
 | ||
| Fig. 1 Recycling test of i-Pr2NEMimCl. Reaction conditions: 10 mmol PO; 15 mmol EC; recovered i-Pr2NEMimCl; 140 °C; 24 h. | ||
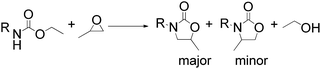
To demonstrate the utility and generality of this approach to formation 2-oxazolidinones, the reaction of EC with different epoxides were carried out under the optimized conditions, the results shown in Table 2. Excellent yields of the corresponding 2-oxazolidinones were obtained with terminal epoxides (entries 1–7), while the disubstituted epoxide, cyclohexene oxide (entry 8), gave lower activity towards the production of the corresponding 2-oxazolidinones, which might be due to the high hindrance of cyclohexene oxide. However, the selectivity of the 5-substituted-2-oxazolidinones for the styrene oxide (entry 7) was much lower than that of other epoxides, which might be ascribed to the conjugative effect derived from the benzene ring, which should favourably attack at the carbon atom at which phenyl substitute was connected to afford 4-substituted-2-oxazolidinones.29
| Entry | Epoxide | Major product | Conv. (%) | Sel.b (%) | Yieldc (%) |
|---|---|---|---|---|---|
| a Reaction conditions: 10 mmol epoxides; 15 mmol EC; 1 mmol catalysts; 140 °C; 24 h. Conversions and selectivities were determined by gas chromatography.b Selectivity of the 5-substituted-2-oxazolidinone.c Isolated yield based on the charged PO. | |||||
| 1 | 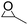 |
 |
97 | 99 | 96 |
| 2 |  |
 |
96 | 100 | 95 |
| 3 | 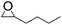 |
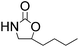 |
95 | 98 | 93 |
| 4 | 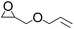 |
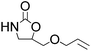 |
97 | 99 | 95 |
| 5 | 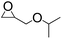 |
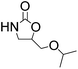 |
98 | 98 | 95 |
| 6 | 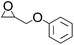 |
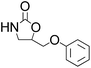 |
96 | 98 | 94 |
| 7 | 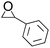 |
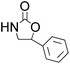 |
95 | 74 | 93 |
| 8 | 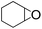 |
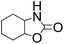 |
82 | 100 | 81 |
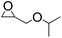
Moreover, the reaction of PO and different carbamates with various substituents on nitrogen or oxygen atom were also investigated, the results shown in Table 3. The carbamates with different alkyl or benzyl substituent on oxygen atom (entries 1–4) gave good to excellent yields of the corresponding 2-oxazolidinones, however, the much lower yield of the corresponding 2-oxazolidinones was obtained when the phenyl substituent on the oxygen atom (entry 5), which could be ascribed to the weaker leaving ability of the phenoxy than the alkoxy or benzyloxy during the intramolecular cyclization step. Additionally, the substituent on the nitrogen atom of carbamates gave lower yield of the corresponding 2-oxazolidinones, entries 6–7, which could be duo to the high hindrance of substituent on the nitrogen atom and unfavorable for the nucleophilic addition to the PO.
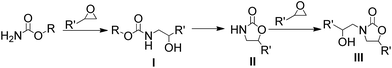
Based on the reaction mechanism reported in the previous literature,23 the 2-oxazolidinone was formed in two consecutive steps, Scheme 3. The first is the formation of compound I by nucleophilic addition between epoxide and carbamate, followed by the formation of the 2-oxazolidinone II in the second step by intramolecular cyclization.
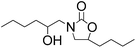
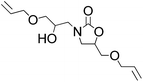
It is worth to note that during the reaction processes, we never detected the presence of the compound I. This fact indicated that the rate-limiting step for the conversion of epoxide into 2-oxazolidinone is the ring-opening step of epoxide. Thus, we supposed that if the epoxide is in excess, the further ring opening of epoxide with the formed 2-oxazolidinone II could be occurred and generate the hydroxyl functionalized 2-oxazolidinone III which also represent an important class of compounds have wide utility in pharmaceutical chemistry. So, we carried out the reaction under the optimized condition with the mole ratio of EC![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) epoxide equal to 1
epoxide equal to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3. The results are shown in Table 4. It can be seen that excellent yields of hydroxyl functionalized 2-oxazolidinones were obtained with various epoxides.
3. The results are shown in Table 4. It can be seen that excellent yields of hydroxyl functionalized 2-oxazolidinones were obtained with various epoxides.
Conclusions
In conclusion, an amine-functionalized ILs catalyst was developed for the synthesis of 2-oxazolidinones from epoxides and carbamates. Under the optimized reaction conditions, good to excellent yields of various 2-oxazolidinones were obtained with different epoxides and carbamates. Meanwhile, the hydroxyl functionalized 2-oxazolidinones were successfully synthesized when the epoxides were in excess.Acknowledgements
This work has been financially supported with Doctoral Scientific Research Foundation of Shanxi Datong University.Notes and references
- G. Zappia, E. Gacs-Baitz, G. Delle Monache, D. Misiti, L. Nevola and B. Botta, Curr. Org. Chem., 2007, 4, 81–135 CAS.
- X. Dou, L. He and Z. Yang, Synth. Commun., 2012, 42, 62–74 CrossRef CAS PubMed.
- T. A. Mukhtar and G. D. Wright, Chem. Rev., 2005, 105, 529–542 CrossRef CAS PubMed.
- S. Matsunaga, N. Kumagai, S. Harada and M. Shibasaki, J. Am. Chem. Soc., 2003, 125, 4712–4713 CrossRef CAS PubMed.
- B. Gabriele, G. Salerno, D. Brindisi, M. Costa and G. P. Chiusoli, Org. Lett., 2000, 2, 625–627 CrossRef CAS PubMed.
- J. Liu, X. Peng, J. Liu, S. Zheng, W. Sun and C. Xia, Tetrahedron Lett., 2007, 48, 929–932 CrossRef CAS PubMed.
- F. Li and C. Xia, J. Catal., 2004, 227, 542–546 CrossRef CAS PubMed.
- S. Pulla, V. Unnikrishnan, P. Ramidi, S. Z. Sullivan, A. Ghosh, J. L. Dallas and P. Munshi, J. Mol. Catal. A: Chem., 2011, 338, 33–43 CAS.
- B. Wang, S. Yang, L. Min, Y. Gu, Y. Zhang, X. Wu, L. Zhang, E. H. M. Elageed, S. Wu and G. Gao, Adv. Synth. Catal., 2014, 356, 3125–3134 CrossRef CAS PubMed.
- F. Li, H. Li, L. Wang, P. He and Y. Cao, Catal. Sci. Technol., 2015, 5, 1021–1034 CAS.
- B. Schäffner, F. Schäffner, S. P. Verevkin and A. Börner, Chem. Rev., 2010, 110, 4554–4581 CrossRef PubMed.
- S. A. Anderson and T. W. Root, J. Mol. Catal. A: Chem., 2004, 220, 247–255 CrossRef CAS PubMed.
- S. I. Fujita, H. Kanamaru, H. Senboku and M. Arai, Int. J. Mol. Sci., 2006, 7, 438–450 CrossRef CAS PubMed.
- K. I. Tominaga and Y. Sasaki, Synlett, 2002, 2, 307–309 CrossRef.
- R. Juárez, P. Concepción, A. Corma and H. García, Chem. Commun., 2010, 46, 4181–4183 RSC.
- A. W. Miller and S. T. Nguyen, Org. Lett., 2004, 6, 2301–2304 CrossRef CAS PubMed.
- M. T. Hancock and A. R. Pinhas, Tetrahedron Lett., 2003, 44, 5457–5460 CrossRef CAS.
- Y. Wu, L. He, Y. Du, J. Wang, C. Miao and W. Li, Tetrahedron, 2009, 65, 6204–6210 CrossRef CAS PubMed.
- R. A. Watile, D. B. Bagal, K. M. Deshmukh, K. P. Dhake and B. M. Bhanage, J. Mol. Catal. A: Chem., 2011, 351, 196–203 CrossRef CAS PubMed.
- S. Bonollo, F. Fringuelli, F. Pizzo and L. Vaccaro, Synlett, 2007, 17, 2683–2686 Search PubMed.
- L. I. Kas'yan, V. A. Pal'chikov and Ya. S. Bondarenko, Russ. J. Org. Chem., 2011, 47, 797–841 CrossRef.
- A. Cwik, A. Fuchs, Z. Hell, I. Bojtos, D. Halmai and P. Bombicz, Org. Biomol. Chem., 2005, 3, 967–969 CAS.
- G. Bartoli, M. Bosco, A. Carlone, M. Locatelli, P. Melchiorre and L. Sambri, Org. Lett., 2005, 7, 1983–1985 CrossRef CAS PubMed.
- Y. Iwakura and S. I. Izawa, J. Org. Chem., 1964, 29, 379–382 CrossRef CAS.
- R. D. Rogers and K. R. Seddon, Science, 2003, 302, 792–793 CrossRef PubMed.
- H. P. SteinrÜk and P. Wasserscheid, Catal. Lett., 2015, 145, 380–397 CrossRef.
- Y. Gu and G. Li, Adv. Synth. Catal., 2009, 351, 817–847 CrossRef CAS PubMed.
- S. Bonollo, F. Fringuelli, F. Pizzo and L. Vaccaro, Synlett, 2007, 17, 2683–2686 Search PubMed.
- S. Bonollo, D. Lanari and L. Vaccaro, Eur. J. Org. Chem., 2011, 14, 2587–2598 CrossRef PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra09838f |
| This journal is © The Royal Society of Chemistry 2015 |