Optical properties of armchair graphene nanoribbons with Stone–Wales defects and hydrogenation on the defects†
Min Wang*ab,
Yu Chen Wangab,
Hai Xing Zhaoab and
Si Xing Songab
aInstitute for Clean Energy & Advanced Materials, Faculty of Materials and Energy, Southwest University, Chongqing 400715, P. R. China. E-mail: minwang@swu.edu.cn
bChongqing Key Laboratory for Advanced Materials and Technologies of Clean Energies, Chongqing 400715, P. R. China
First published on 29th July 2015
Abstract
Armchair graphene nanoribbon (AGNR) is one of the most investigated semiconducting graphene materials. The controllable approach on AGNR is quite useful for future optical applications. To realize the aim, optical properties of three AGNRs with Stone–Wales (SW) defects and hydrogenation on the SW defects (SW-H) are theoretically investigated. W8, W9 and W10 AGNRs are chosen based on the width (W) index of n. SW defects enlarge the band gap of W8, and reduce the band gap of W9 and W10. The hydrogenations increase the band gaps of W8- and W9-SW, and decrease the one of W10-SW. The distributions of exciton wavefunctions located near one edge of W10-SW-H, revealed an obvious quantum confinement effect. In W10 serials, the exciton binding energy difference between SW and SW-H structures is only 0.08 eV, indicating tuneable optical applications with this small exciton binding energy switch. Due to the strong optical absorption and small exciton binding energy of W9-SW, it also possesses potential applications for luminescence and photovoltaic devices.
Introduction
Graphene has gained intensive investigations1–16 since its discovery. However, its zero band gap limits optical applications. For example, luminescence is hard to exist in pristine graphene. To improve the properties, various modifications are explored. Graphene nanoribbon (GNR), which is produced from graphene cracked through direct focused electron writing17 or plasma etching18 along one specific direction, can be turned into a semiconducting quasi one-dimensional structure. Moreover, GNRs can also be produced by unzipping carbon nanotubes through plasma etching or solution-based oxidation.19,20In particular, armchair GNRs (AGNRs) possess a unique “family effect”21 in the electronic properties including the band gaps, which are dependent on the ribbon widths and classified into three families: n = 3p − 1, 3p and 3p + 1, where p is a positive integer and n is the width index. Due to the special structures, AGNRs are expected to exhibit various remarkable electronic and optical properties and are potential for future carbon-based nanoelectronics.3,22–27
The defects widely exist in the samples. For example, Stone–Wales (SW) defect,28 composing of two pairs of five- and seven-membered rings and possesses an in-plane 90° rotation of two carbon atoms with respect to the midpoint of the bond, was observed in situ by high-resolution transmission electron microscope.29 Theoretical investigations demonstrate strong chemical reactivity upon the defects and adatoms onto defects are more preferable.30,31 Besides, hydrogenated progress is attracted special attentions since it can be controlled by patterned adsorption32 or be reversible by thermal annealing33,34 on graphene surface. Thus, the defects and hydrogenations could be considered as candidate approaches to control the optical properties. In this work, both SW defects and the hydrogenations are theoretically investigated in AGNRs to tune the optoelectronic properties.
Theoretical methods and models
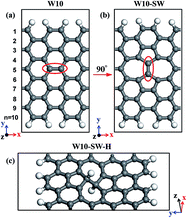
To obtain the optical properties, the first-principle calculation is used with a many-body perturbation approach, possessing a three-step procedure:35 (1) firstly, the fully relaxed system is studied within local density approximation (LDA) based on Quantum ESPRESSO code36 to obtain the ground-state electronic properties. Separable ultrasoft pseudopotentials and a plane-wave basis set are chosen for the calculations. The energy cutoff is set as 50 Ry, and 41 × 1 × 1 is used for k-point parameter. The force tolerance is chosen as 0.01 eV Å−1. (2) Secondly, since LDA is a good approximation for the quasiparticle wavefunctions, the quasiparticle corrections to LDA eigenvalues are used based on G0W0 approximation for the self-energy operator, where G and W are denoted as the single particle Green's function and the screened Coulomb interaction. Moreover, to treat the screening, the plasmon-pole approximation is considered.37 (3) Thirdly, the Bethe–Salpeter equation (BSE) is solved in the basis set of quasi-electron and quasi-hole states to gain the electron–hole interaction. The random-phase approximation38 is used to calculate the static screening in the direct term. All the GW and BS calculations are performed with Yambo code.39 Kinetic energy cut-off is set as 30 Ry. Five valence bands and five conduction bands are included for the calculation. A supercell of ten unit cells is used during the calculation. Due to the usage of the supercell method in the calculations, a rectangular-shape truncated Coulomb interaction is applied to eliminate the image effect between adjacent supercells to mimic isolated AGNRs.40 To be more convincing, we also do some benchmarks to graphene, and the data are close to the previous works (ESI,† Fig. S1).8,9 Additionally, as mentioned in previous work,41 the GW/BSE results are sensitive to the setup, thus we take W9-SW as an example to investigate the convergence of exciton binding energy based on different k-point grid (ESI,† Fig. S2). The results show that our data are reliable and our setup is also in agreement with previous work.41 The decimal place is chosen in accord with the previous computation work.40Previous theoretical reports demonstrated that graphene with defects has stronger chemical reactivity than the pristine one and moreover single adatom onto defect are more preferable than that adsorption on graphene.30,31,42 To consider two or more hydrogen atoms on the SW-defected graphene, it is also demonstrated that the defects decrease the chemisorption energy10 in comparison to the ideal graphene, and hydrogen atoms energetically prefer to stay on both sides.10,30 Herein the AGNRs with SW defects and the hydrogen atoms adsorbed on the SW defects are mainly investigated. The chosen concentration of SW defects are in agreement with previous computation works.43–45 The pristine AGNR, SW and SW-H AGNRs are shown in Fig. 1(a)–(c), respectively. The period direction is along x axis. The unit length along y axis and the height of a vacuum layer along z axis are set as 30 Å. AGNRs with SW defect and the one with hydrogen atoms adsorbed on SW defect are defined as W(n)-SW and W(n)-SW-H respectively, where n is the width index of AGNRs21 (marked in Fig. 1). In this study, we consider three series of AGNRs (n = 8, 9 and 10), which cover the distinct three families (n = 3p − 1, 3p and 3p + 1) of pristine AGNRs according to previous works.40,46 The dangling σ bonds at the AGNRs' edges are passivated by hydrogen atoms.
Results and discussion
The analysis of the variation of formation energies and band gaps
The related formation energies (Eformation) for W(n)-SW and W(n)-SW-H, listed in Table 1, are defined as EW(n)-SW − EW(n) and EW(n)-SW-H − EW(n) − 2EH, respectively. The formation energy for W(n)-SW decreases with the increase of the width of ribbons, while the formation energy for W(n)-SW-H has the reverse trend.| n | LDA (eV) | GW (eV) | BS (eV) | Eb (eV) | Eformation (eV per atom) |
|---|---|---|---|---|---|
| W8 | 0.26 (0.28) | 1.10 (1.00) | 0.48 (0.42) | 0.62 (0.58) | — |
| W8-SW | 0.71 | 2.29 | 1.07 | 1.22 | 0.1275 |
| W8-SW-H | 1.11 | 3.17 | 1.70 | 1.47 | −0.0398 |
| W9 | 0.81 (0.78) | 2.25 (2.10) | 1.10 (0.99) | 1.15 (1.11) | — |
| W9-SW | 0.16 | 0.81 | 0.14 | 0.67 | 0.1270 |
| W9-SW-H | 0.58 | 2.14 | 1.06 | 1.08 | −0.0252 |
| W10 | 1.10 (1.16) | 2.80 (2.82) | 1.50 (1.51) | 1.30 (1.31) | — |
| W10-SW | 0.86 | 2.32 | 1.17 | 1.15 | 0.1168 |
| W10-SW-H | 0.64 | 2.01 | 0.94 | 1.07 | −0.0233 |
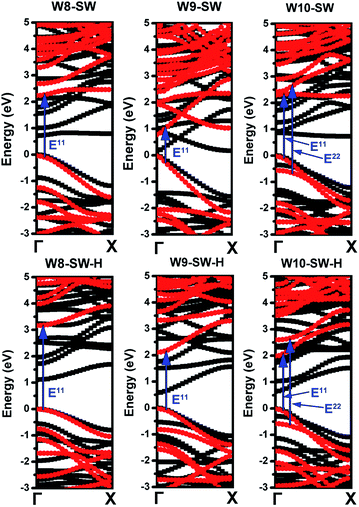
Table 1 also summarizes LDA band gaps and GW band gaps for W(n), W(n)-SW and W(n)-SW-H structures. It is noted that electronic and optical properties of pure AGNRs are detailed discussed in previous works,40,46 which are quite similar to our calculations, so we only summarizes related data in Table 1. Both LDA and GW band structures of SW and SW-H are plotted in Fig. 2. Each band line comes from Γ (0, 0, 0) to X (π/a, 0, 0), where a is the unit length along the ribbon axis. It is obvious that band gap corrections in GW band structures appear and also possess the main characteristics of LDA band structures. All the structures have the direct band gaps. In the pristine AGNRs, the GW band gaps of W8, W9 and W10 are 1.10, 2.25, and 2.80 eV, respectively, which are in agreement with previous results.40
Comparing with pristine and SW AGNRs, SW defects enlarge the band gap of W8, and reduce the band gap of W9 and W10. During the hydrogenation procedures, it was reported47 that the induced sp3 bonds by the surface hydrogen adsorption break sp2 network, raising the carbon–carbon bond order and there is a transition from small band gap to large band gap, called metal–insulator transitions. But the GW band gap of W10-SW-H is 2.32 eV, which is smaller than that of W10-SW (2.01 eV), revealing that the hydrogen adsorption can also decrease the band gap, though for W8 and W9 series, the hydrogen adsorptions enlarge the band gaps strongly with increases of 0.9 eV and 1.3 eV respectively.
To investigate the origination of the band gap difference, the bonding characteristics are first analyzed. There are complex carbon–carbon bond hybridizations in SW and SW-H AGNRs. The rotated carbon–carbon bond to form SW defect, marked in red circle in Fig. 1(a) and (b), is below sp2 hybridization and its bond length is only 1.32 Å. Other carbon atoms of 5–7 rings belong to the mixture of sp2 and sp3 hybridizations, and the bond lengths are between 1.45 Å and 1.50 Å. Moreover, in SW-H AGNRs, the hybridizations on the rotated carbon atoms (Fig. 1(c)) transform from sp2 to sp3, and the carbon–carbon bond length increases from 1.32 Å to 1.50 Å. The variations of sp2 and sp3 hybridizations and the bond lengths lead to different electronic properties including the band gap difference.
As shown in Table 1 and Fig. 2, the quasi-particle GW energy corrections are in the order of 1 eV. Due to the complicated band and energy dependence on GW correction, the so-called “scissors operator”, in which the self-energy is approximated by a rigid shift of energy bands, cannot be applied legitimately.48,49 GW corrections of W10 series are larger than those of W9 series. Additionally, GW corrections with bigger LDA band gaps tend to be larger.
The analysis of optical transition
The optical transitions are computed with the light polarization vector along the ribbon axis by solving Bethe–Salpeter (BS) equation numerically.35 The optical spectra are shown in Fig. 3, and the first peak positions of the transition energies (BS energies) are listed in Table 1. | ||
| Fig. 3 Optical absorption spectra of SW and SW-H AGNRs: (a) W8-SW and W8-SW-H, (b) W9-SW and W9-SW-H, (c) W10-SW and W10-SW-H with (w) or without (w/o) electron–hole interaction (e–h). | ||
In the presence of electron–hole (e–h) interaction, the optical positions are quite different from the one without e–h interactions (see W8-SW in Fig. 3(a)). Optical transitions give rise to the interband transitions and bright excitons, which are labelled as Enm (blue arrows in Fig. 2), which is denoted as the transition from the nth valence band to the mth conduction band.
The absolute values of BS energies are about half of GW band gaps, revealing a clear manifestation of strong binding of the electron and hole in SW AGNRs. The exciton binding energy is defined as the difference between GW band gaps and BS energies. It is well known that the screening in one or quasi-one dimensional material is an order of magnitude weaker than that in bulk materials, thus the exciton binding strength is expected to be higher in AGNRs. Most of the exciton binding energies of SW and SW-H AGNRs are larger than 1 eV. It is noted that GW band gap of W9-SW is about 0.81 eV, resulting in a significantly lighter effective mass (smaller kinetic energy) and fairly stronger screening to reduce the Coulomb interaction between electron and hole.8 Therefore, the exciton binding energy is the weakest in the SW series, which is only 0.67 eV.
As discussed above, the adsorption hydrogen atoms can tune the electronic and optical properties. In W8- and W9-SW-H structures, the hydrogenation procedures raise the band gap, the transition energies and corresponding exciton binding energies, and reduce the optical adsorption intensities. On the other hand, comparing with W10-SW and W10-SW-H, the hydrogenation procedure has the opposite effects. Additionally, the strong optical absorption intensity and small exciton binding energy of W9-SW reveal the potential optical applications including luminescence and photovoltaics.
In order to get more clear on the differences of the electron structures before and after the hydrogen adsorptions, valence band maximum (VBM) and conduction band minimum (CBM) are plotted in Fig. 4. CBMs and VBMs in W9-SW and W9-SW-H AGNRs keep the same symmetrical distribution, though the hydrogen atoms modify the connected carbon atoms' hybridization from sp2 into sp3 type and increase related carbon–carbon bond length. In W8 and W10 series, CBM of W8-SW and VBM of W10-SW are mostly confined near one edge, and after the hydrogen adsorption, the similar confinement are more obvious in both CBMs and VBMs of W8-SW-H and W10-SW-H, especially the latter structure. Moreover, this confinement is very similar to that in the hydrogenation on the pristine AGNRs.48–50
 | ||
| Fig. 4 VBMs and CBMs for W(n)-SW and W(n)-SW-H, where n is 8, 9 and 10. The pink arrows point out the positions of hydrogenations on SW defects. | ||
The analysis of exciton wavefunctions
To get an insight into the effects of electron–hole interaction, the exciton wavefunctions with the lowest exciton peaks in SW and SW-H series are plotted in Fig. 5 with one fixed hole position in the middle of a π bond (marked as black spots). The general patterns of wavefunction distribution are quite different with and without hydrogenation. For the asymmetric W8- and W10-SW-H, the hydrogenation make the exciton wavefunction distribute towards the edges, while due to the symmetry in W9-SW-H, the exciton distribution is more symmetric than those in W8- and W10-SW-H. Moreover, these features in spatial distribution can be understood by VBMs and CBMs (Fig. 4). When W8- and W10-SW structures adsorb hydrogen atoms, they do not possess the mirror planes and both VBMs and CBMs prefer to localized at the same edge, especially the latter structure. Consequently, the exciton wavefunctions are expected to locate near the same edge. Hence, there are obvious quantum confinement effects on the excitons in W10-SW-H structures, that is, most of the exciton wavefunction locates near one edge. Moreover, similar confinement phenomenon has also reported in other work.48 Additionally, since the hydrogen adsorption on graphene surface can be reversible by thermal annealing,33,34 a small exciton binding energy difference of 0.08 eV between W10-SW and W10-SW-H indicates that the W10-SW is a good candidate for nanoscale optical switch. It is noticeable that the edge excitons reported in the previous work40 do not appear in SW and SW-H systems.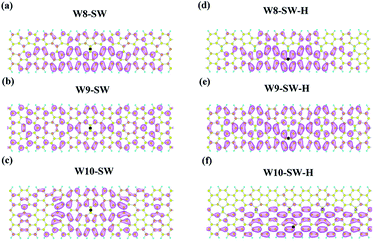 | ||
| Fig. 5 The lowest exciton wavefunctions for (a–c) (n)-SW and (d–f) W(n)-SW-H, where n is 8, 9 and 10. | ||
Fig. 6 displays the lowest exciton wavefunctions of W8-, W9- and W10-SW (and -SW-H) projected on x-axis direction, clearly revealing that the hydrogen adsorptions change one dimensional exciton wavefunction distribution. Due to the small exciton binding energy of W9-SW, it has a large distribution of exicton wavefunction, which agrees well with the previous studies about the relationship between the binding energy and the distribution of exciton wavefunction.40,51,52
 | ||
| Fig. 6 The lowest exciton wavefunctions projected on x-axis direction for (a–c) W(n)-SW and (d–f) W(n)-SW-H, where n is 8, 9 and 10. | ||
It is known that the binding energy of a Frenkel exciton53 can be estimated as EFrenkelb ∼ (2μ2)/(4πε0εrr3), where μ is the dipole moment, ε0 is the vacuum permittivity, εr ∼ 2.5 is the static dielectric constant of AGNR, and r ∼ 1.4 Å is the interatomic distance.48 Based on the calculated Eb in Table 1, the dipole moment of μ is estimated as 1 × 10−29 C m, which is an order larger than the typical one in a Frenkel exciton.54 Therefore, the exciton in this work is not a Frenkel one. Additionally, due to the binding energy (∼1 eV) and radius (∼10 Å) of exciton, it may be a charge-transfer exciton.50 The distribution of exciton wavefunction along x-axis (ribbon length) is determined by the Coulomb interaction with quasi-one-dimensional character, which are similar to carbon nanotubes55 and pure AGNRs.40
It is noted that though SW defect needs energy,45 but it occurs in graphene.56 Previous computation works43–45 have also discussed the electronic properties in the graphene or graphene ribbon with different concentrations of SW. The configurations of a larger supercell with a lower SW concentration may provide different properties due to the weaker interactions between the defects. We uncover the configurations with low concentrations presently, but it is necessary for us to continue the work in the future.
Conclusions
In summary, the optic and electronic properties of AGNRs with SW defects and hydrogen adsorption on SW defects are investigated. SW defects enlarge the band gap of W8, and reduce the band gap of W9 and W10. The hydrogenations increase the band gaps of W8- and W9-SW, and decrease the band gap of W10-SW. The variation of band gaps is analyzed in terms of bonding characteristics. There are obvious quantum confinement effects on the excitons in W10-SW-H structures, that is, the distribution of exciton wavefunction locates near one edge. Additionally, the exciton binding energy difference between W10-SW and W10-SW-H structures is only 0.08 eV, indicating tuneable optical applications with this small exciton binding energy switch. Due to the strong optical absorption and small exciton binding energy of W9-SW, it possesses potential optical applications for luminescence and photovoltaic devices.Acknowledgements
This work was financially supported by the National Natural Science Foundation of China (Grant No. 21203154 and 21375108). Computation resource is supported by Faculty of Materials and Energy (Southwest University).Notes and references
- K. S. Novoselov, A. K. Geim, S. V. Morozov, D. Jiang, Y. Zhang, S. V. Dubonos, I. V. Grigorieva and A. A. Firsov, Science, 2004, 306, 666–669 CrossRef CAS PubMed.
- K. S. Novoselov, A. K. Geim, S. V. Morozov, D. Jiang, M. I. Katsnelson, I. V. Grigorieva, S. V. Dubonos and A. A. Firsov, Nature, 2005, 438, 197–200 CrossRef CAS PubMed.
- Y. B. Zhang, Y. W. Tan, H. L. Stormer and P. Kim, Nature, 2005, 438, 201–204 CrossRef CAS PubMed.
- W. X. Zhang, C. He, T. Li and S. B. Gong, RSC Adv., 2015, 5, 33407–33413 RSC.
- B. Liu, F. Meng, C. D. Reddy, J. A. Baimova, N. Srikanth, S. V. Dmitriev and K. Zhou, RSC Adv., 2015, 5, 29193–29200 RSC.
- X. An and J. C. Yu, RSC Adv., 2011, 1, 1426–1434 RSC.
- S. Bai and X. Shen, RSC Adv., 2012, 2, 64–98 RSC.
- L. Yang, J. Deslippe, C. H. Park, M. L. Cohen and S. G. Louie, Phys. Rev. Lett., 2009, 103, 186802 CrossRef.
- P. E. Trevisanutto, M. Holzmann, M. Côté and V. Olevano, Phys. Rev. B: Condens. Matter Mater. Phys., 2010, 81, 121405 CrossRef.
- D. W. Boukhvalov and M. I. Katsnelson, Nano Lett., 2008, 8, 4373–4379 CrossRef CAS.
- M. Yang, A. Nurbawono, C. Zhang, Y. P. Feng and Ariando, Appl. Phys. Lett., 2010, 96, 193115 CrossRef PubMed.
- M. Yang, A. Nurbawono, C. Zhang, R. Wu, Y. Feng and Ariando, AIP Adv., 2011, 1, 032109 CrossRef PubMed.
- F. Karlický and M. Otyepka, J. Chem. Theory Comput., 2013, 9, 4155–4164 CrossRef.
- H.-X. Zhao, Y.-C. Wang, L.-Y. Zhang and M. Wang, New J. Chem., 2015, 39, 98–101 RSC.
- M. Wang and C. M. Li, Phys. Chem. Chem. Phys., 2013, 15, 3786–3792 RSC.
- D. Lee, J. Seo, X. Zhu, J. M. Cole and H. Su, Appl. Phys. Lett., 2015, 106, 172402 CrossRef PubMed.
- M. Remeika and A. Bezryadin, Nanotechnology, 2005, 16, 1172–1176 CrossRef CAS.
- M. Y. Han, B. Ozyilmaz, Y. B. Zhang and P. Kim, Phys. Rev. Lett., 2007, 98, 206805 CrossRef.
- D. V. Kosynkin, A. L. Higginbotham, A. Sinitskii, J. R. Lomeda, A. Dimiev, B. K. Price and J. M. Tour, Nature, 2009, 458, 872–U875 CrossRef CAS PubMed.
- L. Y. Jiao, L. Zhang, X. R. Wang, G. Diankov and H. J. Dai, Nature, 2009, 458, 877–880 CrossRef CAS PubMed.
- Y. W. Son, M. L. Cohen and S. G. Louie, Phys. Rev. Lett., 2006, 97, 216803 CrossRef.
- Y. W. Son, M. L. Cohen and S. G. Louie, Nature, 2006, 444, 347–349 CrossRef CAS PubMed.
- K. Wakabayashi, Phys. Rev. B: Condens. Matter Mater. Phys., 2001, 64, 125428 CrossRef.
- V. Barone, O. Hod and G. E. Scuseria, Nano Lett., 2006, 6, 2748–2754 CrossRef CAS PubMed.
- D. A. Areshkin, D. Gunlycke and C. T. White, Nano Lett., 2007, 7, 204–210 CrossRef CAS PubMed.
- M. Wang and C. M. Li, Nanoscale, 2012, 4, 1044–1050 RSC.
- M. Wang and C. M. Li, Nanoscale, 2011, 3, 2324–2328 RSC.
- A. J. Stone and D. J. Wales, Chem. Phys. Lett., 1986, 128, 501–503 CrossRef CAS.
- A. Hashimoto, K. Suenaga, A. Gloter, K. Urita and S. Iijima, Nature, 2004, 430, 870–873 CrossRef CAS PubMed.
- P. A. Denis and F. Iribarne, J. Phys. Chem. C, 2013, 117, 19048–19055 CAS.
- M. Ijas, P. Havu and A. Harju, Phys. Rev. B: Condens. Matter Mater. Phys., 2013, 87, 205430 CrossRef.
- R. Balog, B. Jorgensen, L. Nilsson, M. Andersen, E. Rienks, M. Bianchi, M. Fanetti, E. Laegsgaard, A. Baraldi, S. Lizzit, Z. Sljivancanin, F. Besenbacher, B. Hammer, T. G. Pedersen, P. Hofmann and L. Hornekaer, Nat. Mater., 2010, 9, 315–319 CrossRef CAS PubMed.
- D. C. Elias, R. R. Nair, T. M. G. Mohiuddin, S. V. Morozov, P. Blake, M. P. Halsall, A. C. Ferrari, D. W. Boukhvalov, M. I. Katsnelson, A. K. Geim and K. S. Novoselov, Science, 2009, 323, 610–613 CrossRef CAS PubMed.
- S. Ryu, M. Y. Han, J. Maultzsch, T. F. Heinz, P. Kim, M. L. Steigerwald and L. E. Brus, Nano Lett., 2008, 8, 4597–4602 CrossRef CAS PubMed.
- G. Onida, L. Reining and A. Rubio, Rev. Mod. Phys., 2002, 74, 601 CrossRef CAS.
- P. Giannozzi, S. Baroni, N. Bonini, M. Calandra, R. Car, C. Cavazzoni, D. Ceresoli, G. L. Chiarotti, M. Cococcioni, I. Dabo, A. Dal Corso, S. de Gironcoli, S. Fabris, G. Fratesi, R. Gebauer, U. Gerstmann, C. Gougoussis, A. Kokalj, M. Lazzeri, L. Martin-Samos, N. Marzari, F. Mauri, R. Mazzarello, S. Paolini, A. Pasquarello, L. Paulatto, C. Sbraccia, S. Scandolo, G. Sclauzero, A. P. Seitsonen, A. Smogunov, P. Umari and R. M. Wentzcovitch, J. Phys.: Condens. Matter, 2009, 21, 395502 CrossRef PubMed.
- R. W. Godby and R. J. Needs, Phys. Rev. Lett., 1989, 62, 1169 CrossRef.
- M. Rohlfing and S. G. Louie, Phys. Rev. B: Condens. Matter Mater. Phys., 2000, 62, 4927 CrossRef CAS.
- A. Marini, C. Hogan, M. Gruning and D. Varsano, Comput. Phys. Commun., 2009, 180, 1392–1403 CrossRef CAS PubMed.
- D. Prezzi, D. Varsano, A. Ruini, A. Marini and E. Molinari, Phys. Rev. B: Condens. Matter Mater. Phys., 2008, 77, 041404 CrossRef.
- F. Karlický and M. Otyepka, Ann. Phys., 2014, 526, 408–414 CrossRef PubMed.
- S. Letardi, M. Celino, F. Cleri and V. Rosato, Surf. Sci., 2002, 496, 33–38 CrossRef CAS.
- S. Bhowmick and U. V. Waghmare, Phys. Rev. B: Condens. Matter Mater. Phys., 2010, 81, 155416 CrossRef.
- P. Koskinen, S. Malola and H. Häkkinen, Phys. Rev. Lett., 2008, 101, 115502 CrossRef.
- M. T. Lusk and L. D. Carr, Phys. Rev. Lett., 2008, 100, 175503 CrossRef.
- L. Yang, M. L. Cohen and S. G. Louie, Nano Lett., 2007, 7, 3112–3115 CrossRef CAS PubMed.
- L. Hornekaer, E. Rauls, W. Xu, Z. Sljivancanin, R. Otero, I. Stensgaard, E. Laegsgaard, B. Hammer and F. Besenbacher, Phys. Rev. Lett., 2006, 97, 186102 CrossRef CAS.
- X. Zhu and H. Su, J. Phys. Chem. C, 2010, 114, 17257–17262 CAS.
- X. Zhu and H. Su, J. Phys. Chem. A, 2011, 115, 11998–12003 CrossRef CAS PubMed.
- X. Zhu and H. Su, ACS Nano, 2014, 8, 1284–1289 CrossRef CAS PubMed.
- F. Wang, D. J. Cho, B. Kessler, J. Deslippe, P. J. Schuck, S. G. Louie, A. Zettl, T. F. Heinz and Y. R. Shen, Phys. Rev. Lett., 2007, 99, 227401 CrossRef.
- L. Yang, C.-H. Park, Y.-W. Son, M. L. Cohen and S. G. Louie, Phys. Rev. Lett., 2007, 99, 186801 CrossRef.
- S. Nakajima, Y. Toyozawa and R. Abe, The Physics of Elementary Excitations, Springer-Verlag, Berlin, Germany, 1980 Search PubMed.
- M. Pope and C. E. Swenberg, Electronic Processes in Organic Crystals and Polymers, Oxford University Press, Oxford, UK, 1999 Search PubMed.
- C. D. Spataru, S. Ismail-Beigi, L. X. Benedict and S. G. Louie, Phys. Rev. Lett., 2004, 92, 077402 CrossRef.
- J. C. Meyer, C. Kisielowski, R. Erni, M. D. Rossell, M. F. Crommie and A. Zettl, Nano Lett., 2008, 8, 3582–3586 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Optical spectrum of graphene, and the relation of exciton binding energy and k-point grid in W9-SW. See DOI: 10.1039/c5ra08836d |
| This journal is © The Royal Society of Chemistry 2015 |