Pyridine-2,6-dicarboxylic acid for the sensitization of europium(III) luminescence with very long lifetimes
Chaoyou Tao,
Kai Du,
Qiang Yin,
Jiayi Zhu,
Hongwei Yan,
Fanghua Zhu and
Lin Zhang*
Research Center of Laser Fusion, China Academy of Engineering Physics, Mianyang, 621900, China. E-mail: zhlmy@sina.com
First published on 22nd June 2015
Abstract
Convenient and mild syntheses of two europium(III) complexes, Na[Eu(dipic)2·3H2O]·4H2O (dipic = dipicolinate anions) and Na[Eu(dipic)2·phen]·H2O (phen = 1,10-phenanthroline), were reported. These two europium(III) complexes were confirmed by 1H NMR, FT-IR, UV-Vis spectroscopy, and elemental and lifetime analysis. Interestingly, the lifetimes were 2.31 ms and 2.38 ms for Na[Eu(dipic)2·3H2O]·4H2O in the solid state and in ethanol solution, respectively, though high-frequency vibrations were present in the first coordination sphere. Furthermore, the lifetime for Na[Eu(dipic)2·phen]·H2O in ethanol solution is 3.23 ms, which is the longest lifetime reported to date. The Na[Eu(dipic)2·phen]·H2O complexes exhibited a quantum yield of 36.1% in ethanol solution, which was longer than that of Na[Eu(dipic)2·3H2O]·4H2O. The value of the stimulated emission cross-section (σp) for Na[Eu(dipic)2·phen]·H2O in ethanol was very close to the values of the Nd-glass laser for practical use, which showed that it had potential application as a high-powered liquid laser medium.
1. Introduction
The unique luminescence properties of lanthanide(III) ions, such as sharp emission, large Stokes shift, insensitivity to oxygen and particularly their long excited state lifetimes, ranging from microseconds (Yb, Nd) to milliseconds (Eu, Tb), triggered the development of optical imaging,1 time-resolved bio-labelling probes,2 luminescent probes,3 OLED,4 and lasing systems.5 The emission originates from 4f–4f intra-atomic electronic and magnetic dipole transitions that are parity-forbidden. As such, the absorption coefficients of the optical transitions for these ions are, however, extremely low, which limits their practical application considerably. This drawback can be overcome by the use of highly absorbent chelating ligands, which serve as efficient sensitizers. The luminescent ligands act as antenna chromophores analogous to the light harvesting center in photosynthetic systems. Lanthanide(III) ions have a large radius and a high affinity for hard donor centers and ligands with oxygen or hybrid oxygen–nitrogen atoms, particularly multi-carboxylate ligands. Lanthanide(III) complexes with organic ligands containing carboxylic groups are of enduring interest due to their usually high thermodynamic stability and peculiar magnetic and luminescent properties. Pyridine-2,6-dicarboxylic acid (H2dipic), which has a rigid 120° angle between the central pyridine ring and the two carboxylate groups, is one of the suitable polydentate ligands that has attracted considerable attention in coordination chemistry. The H2dipic group could potentially provide various coordination motifs to form both discrete and consecutive metal complexes under appropriate synthesis conditions6 and to efficiently sensitize the visible emitter, europium(III), leading to good luminescence quantum yields in solution and in the solid state.7,8 The tridentate dipicolinate anions (dipic2−) formed various anionic homoleptic complexes [Ln(dipic)3]3−, which completely satisfied the eight and nine coordination requirements of lanthanide(III) and showed a reasonable stability with respect to ligand dissociation, even in the presence of polar solvents. The [Eu(dipic)3]3− chelates displayed intense luminescence due to sensitization through the dipic2− triplet state9 with an efficiency of 61% for the tris complex in solution.7 These chelates have even been proposed as secondary standards for quantum yield determination.7 Moreover, several reports dealt with unsaturated lanthanide(III) coordination polymers, which were synthesized by the reaction between H2dipic and lanthanum(III) nitrate under hydrothermal conditions.10–14 However, these reports concentrated on the structures of coordination polymers rather than on their photophysical properties. Focusing on the photophysical properties of europium(III) complexes will be of interest because of their excellent stability, unique red emission and other notable properties.In this study, two novel europium(III) complexes, Na[Eu(dipic)2·3(H2O)]·4H2O and Na[Eu(dipic)2·phen]·H2O, have been investigated systematically. Their synthesis, structure and photophysical properties were reported. The lifetimes were 2.31 ms and 2.38 ms for Na[Eu(dipic)2·3H2O]·4H2O in the solid state and in ethanol solution, respectively, though high-frequency vibrations were present in the first coordination sphere. Interestingly, the lifetime for Na[Eu(dipic)2·phen]·H2O in ethanol solution is 3.23 ms, which is the longest lifetime reported to date. Na[Eu(dipic)2·3H2O]·4H2O exhibited a quantum yield of 36.1% in ethanol solution. Particular attention was focused on the stimulated emission cross-section (σp) values of these complexes, which were very close to the values of a Nd-glass laser.
2. Experimental
2.1. Materials and synthesis
Europium(III) chloride (99.99%, TCI), pyridine-2,6-dicarboxylic acid (99%, TCI), 1,10-phenanthroline·H2O (99%, TCI) and other chemicals (analytically pure) were used without further purification.2.2. Synthesis of Na[Eu(dipic)2·3(H2O)]·4H2O
H2dipic (0.334 g, 2 mmol) was added to 20 mL ethanol and its pH value was adjusted to 7 by adding sodium hydroxide (1 M aqueous solution, 4 mL) while stirring. Then, EuCl3·6H2O (0.366 g, 1 mmol) was dissolved in another 20 mL ethanol. The precipitate was produced by adding EuCl3·6H2O solution dropwise. Then, the precipitate was filtered and washed with ethanol. Finally, the precipitate was dried and stored in a silica-gel drier. 1H NMR (300 MHz, DMSO-d6, TMS, ppm): 5.36 (s, 2H), 4.77 (s, 4H). Elemental analysis: calculated for Eu1Na1C14H20O15N2: C, 27.07; H, 3.01; N, 5.17%. Found: C, 26.64; H, 3.19; N, 4.84%. IR (KBr, cm−1): 1391, 1625 (CO); 482 (OEu); 416 (NEu).2.3. Synthesis of Na[Eu(dipic)2·phen]·H2O
A solution of EuCl3·6H2O (0.366 g, 1 mmol) in deionized water (5 mL) was added to a solution of H2dipic (0.334 g, 2 mmol) in ethanol (10 mL). Then, a solution of phen (0.180 g, 1 mmol) in ethanol (10 mL) was added to the abovementioned mixture. The reaction mixture was stirred for 24 h at 60 °C, and then the solution was filtered to remove any insoluble material. The solution was concentrated and allowed to stand. Precipitates were filtered and dried. 1H NMR (300 MHz, DMSO-d6, TMS, ppm): 9.10 (m, 2H), 8.50 (d, 2H), 8.00 (s, 2H), 7.78 (m, 2H), 5.26 (t, 2H), 4.66 (d, 4H). Elemental analysis: calculated for Eu1Na1C26H16O9N4: C, 44.40; H, 2.29; N, 7.97%. Found: C, 44.71; H, 2.13; N, 7.82%. IR (KBr, cm−1): 1625 (CO); 502 (OEu); 416 (NEu).2.4. Characterization
1H NMR spectra were recorded in DMSO-d6 at room temperature using a Bruker Avance 300 spectrometer. Elemental analysis data were obtained from a Vario EL elemental analyzer. Absorption spectra (solution) were recorded using a 6800 double-beam Jenway spectrophotometer. FT-IR spectra were obtained using a Nicolet 6700 Fourier transform infrared spectrometer. The photoluminescence (PL) emission, excitation spectra and decay curves were measured using a Fluorolog-3-Tau fluorescence spectrophotometer equipped with a 150 W xenon lamp as the excitation source.3. Results and discussion
3.1. Synthesis of europium(III) complexes
Synthetic routes for the europium(III) complexes are outlined in Scheme 1. The synthesis of Na[Eu(dipic)2·3H2O]·4H2O was carried out quickly and conveniently by simply mixing a solution of H2dipic in ethanol, after pH adjustment, with a solution of EuCl3·6H2O in ethanol. Then, white precipitates were quickly obtained. For Na[Eu(dipic)2·phen]·H2O complexes, an aqueous solution of EuCl3·6H2O was added to a solution of H2dipic in ethanol. Then, phen in ethanol solution was added subsequently. The reaction mixture was stirred for 24 h at 60 °C. Precipitates emerged when the solution was concentrated and allowed to stand.3.2. 1H NMR spectra analysis
The 1H NMR spectra of the Na[Eu(dipic)2·3H2O]·4H2O and Na[Eu(dipic)2·phen]·H2O complexes were recorded in DMSO-d6 (Fig. 1). Here, we chose the europium(III) complex of Na[Eu(dipic)2·phen]·H2O as an example to investigate the chemical shift in the 1H NMR spectrum. The 1H NMR spectrum of Na[Eu(dipic)2·phen]·H2O exhibited a doublet at 4.66 ppm, which integrated four protons and was assigned to the protons at the c position. The peak at 5.26 ppm integrated two protons and was attributed to the protons at the d position. The h, g, e, and f position protons of phen were observed at 9.10, 8.50, 8.00, and 7.78 ppm, respectively, and each of these peaks integrated 2 protons.3.3. UV-Vis absorption spectroscopy
The UV-Vis absorption spectra of ligands and europium(III) complexes were recorded in ethanol solution and are depicted in Fig. 2. All chromophores exhibited broad, intense, structureless, high-energy absorption transitions in the UV part of the spectra with λmax between 210 and 300 nm. A careful inspection of the absorption spectra of the ligands revealed the presence of a peak situated between 220 and 270 nm that coincides with the π–π* absorption observed for the corresponding complexes, mainly involving orbitals located on the pyridine ring and carbonyl functions. Furthermore, the absorption maximal peaks of europium(III) complexes underwent slight red shifts, which come from the formation of larger conjugated rings when the ligands coordinated to the europium(III) ions.15 | ||
| Fig. 2 UV-Vis spectra of ligands and europium(III) complexes in ethanol solution (1 × 10−5 mol L−1). | ||
3.4. FT-IR spectroscopy
The FT-IR spectra for the ligands H2dipic and phen and the corresponding europium(III) complexes are shown in Fig. 3. The spectra of the europium(III) complexes were not similar with that of the ligands. | ||
| Fig. 3 Infrared spectra of the ligands: phen, H2dipic and complexes: Na[Eu(dipic)2·3(H2O)·4H2O, Na[EU(dipic)2·phen]·H2O. | ||
For the complex Na[Eu(dipic)2·3H2O]·4H2O, all the bonds involving O–H motions of the carboxylate ground state disappeared. For instance, the characteristic carboxyl vibration in the free H2dipic ligand was found at 1701 cm−1 as a strong and broad vibration (νC![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), and the peaks at 1326 and 1289 cm−1 were attributed to the νC–O stretching vibration, which transformed into the asymmetric νsCOO− at 1625 cm−1 and the symmetric νasCOO− at 1391 cm−1 of Na[Eu(dipic)2·3H2O]·4H2O, respectively, indicating that all the carboxylic groups took part in coordination. Moreover, two new absorption peaks were observed at about 482 cm−1 and 416 cm−1, which were attributed to the stretching of O → Eu and N → Eu, respectively. The FT-IR spectrum of Na[Eu(dipic)2·phen]·H2O was similar to that of Na[Eu(dipic)2·3H2O]·4H2O, except for the presence of the characteristic phen vibrations. The FT-IR results further confirmed the conformation of the europium(III) complexes.
O), and the peaks at 1326 and 1289 cm−1 were attributed to the νC–O stretching vibration, which transformed into the asymmetric νsCOO− at 1625 cm−1 and the symmetric νasCOO− at 1391 cm−1 of Na[Eu(dipic)2·3H2O]·4H2O, respectively, indicating that all the carboxylic groups took part in coordination. Moreover, two new absorption peaks were observed at about 482 cm−1 and 416 cm−1, which were attributed to the stretching of O → Eu and N → Eu, respectively. The FT-IR spectrum of Na[Eu(dipic)2·phen]·H2O was similar to that of Na[Eu(dipic)2·3H2O]·4H2O, except for the presence of the characteristic phen vibrations. The FT-IR results further confirmed the conformation of the europium(III) complexes.
3.5. Excitation and emission spectroscopy
To obtain further information about the interaction between the ligands and the europium(III) ion, excitation spectra were obtained. Fig. 4 shows the excitation spectrum for each of the complexes in the solid state and in ethanol solution obtained at 615 nm (5D0 → 7F2 europium(III) emission). For all the complexes in the solid state, intense peaks were observed at about 260 nm. These can be assigned to the excitation of the π–π* transition centered on the H2dipic ligand, followed by an energy-transfer process to the europium(III) ion.14 The excitation spectra showed that europium(III) emission was present for excitation at short wavelengths in bands that were attributed to ligand absorption. The observed multiple bands centered at 395 nm are attributed to the 7F0 → 5L6, 5G2, 5L7 and 5G3 transitions of europium(III).16 The maximal peaks were 283 nm and 275 nm for Na[Eu(dipic)2·3H2O]·4H2O and Na[Eu(dipic)2·phen]·H2O in ethanol solution, respectively. Fig. 4b shows that some of the excitation peaks from europium(III) were weakened or even disappeared in ethanol solution, which may be attributed to the fluorescence quenching caused by the solvent. A similar phenomenon can be found elsewhere.8 The excitation spectra for europium(III) complexes in ethanol solution indicated that most of the excitation energy was absorbed by the ligand and then transferred to the central europium(III) ion, emitting characteristic luminescence. The emission spectrum for each complex is shown in Fig. 4. Each of these was obtained by excitation using the optimum excitation wavelength. For each complex, the lines were distributed mainly in the 550–750 nm range and were associated with the 4f → 4f transitions of the 5D0 excited state to the low-lying 7FJ (J = 0, 1, 2, 3 and 4) levels of europium(III) ions. No emission peaks from the ligands were observed under this excitation, confirming that the energy transfer from the ligands to the europium(III) ion center was quite efficient in all the europium(III) complexes. From the emission spectra, the transitions of 5D0 → 7F0 (forbidden in the inversion center) and 5D0 → 7F3 (magnetic and electric dipole transitions) are very weak, while those of 5D0 → 7F1 (magnetic dipole transition), 5D0 → 7F2 and 5D0 → 7F4 (electric dipole transition) are strong. A prominent feature noted in these spectra is the hypersensitive 5D0 → 7F2 transition at 615 nm, which is the most intense, pointing to a highly polarizable chemical environment around the europium(III) center. It is known that the nature of the magnetic dipole transition (5D0 → 7F1) is independent of the coordination environment surrounding the metal ion. When the interactions of the lanthanide(III) complex with its local environment were stronger, the complex became more unsymmetrical and the intensity of the electric dipole transitions became more intense. We can therefore use the 5D0 → 7F1 transition at 590 nm emission to assess the effect of the ligands on the emissions at other wavelengths. The emission line strength of the 615 nm (5D0 → 7F2) transition was about 3 times that of the invariant 5D0 → 7F1 transition for all complexes in the solid state as well as in ethanol solution, which indicated the significantly low symmetry of the ligand field. The calculated results are shown in Table 1.| Complex | τobs [ms] | Δλeff [nm] | Irel | Ar [s−1] | σp × 10−20 [cm2] | τ [ms] | ΦLN [%] | Φ [%] | |
|---|---|---|---|---|---|---|---|---|---|
| a FWHM of the 5D0 → 7F2 (615 nm) transition (Δλeff).Emission lifetimes (τobs) of the europium(III) complexes were measured by the excitation at 355 nm. Quantum yields (Φ) of the europium(III) complexes were measured with excitation at optimal excitation wavelengths (5D0 → 7F2) in ethanol solution (1 × 10−5 M). Einstein coefficient (Ar) was determined using eqn (3). Stimulated emission cross-sections (σp) values were determined using eqn (4). | |||||||||
| Na[Eu(dipic)2·3H2O]·4H2O | Solid | 2.31 | 2.21 | 2.56 | 202 | 0.77 | 4.95 | 46.7 | — |
| Ethanol | 2.38 | 2.50 | 3.33 | 177 | 0.72 | 5.65 | 42.1 | 14.8 | |
| Na[Eu(dipic)2·phen]·H2O | Solid | 2.08 | 3.18 | 3.21 | 252 | 0.67 | 3.97 | 52.4 | — |
| Ethanol | 3.23 | 2.61 | 2.75 | 190 | 0.75 | 5.26 | 61.4 | 36.1 | |
The emission spectrum for each complex was recorded in solid state and in ethanol solution by excitation using the europium(III) 5L6 ← 7F0 395 nm transition to allow comparison with emission spectra under ligand excitation (see Fig. 5) and between the complexes. For each complex, three main emission bands were also observed for peak at about 590 nm (5D0 → 7F1), 615 nm (5D0 → 7F2), and 690 nm (5D0 → 7F2). No emission was observed from the ligands in this study or from transitions within the europium(III) ion. The excitation of the complexes at the emission spectra peaks observed in Fig. 5 had no effect on the shape of the emission spectra.
 | ||
| Fig. 5 Emission spectra of europium(III) complexes in solid state and in ethanol solution under direct f–f excitation (5L6 ← 7F0, 395 nm). | ||
3.6. Emission lifetime and quantum yields (Φ)
To better understand the luminescent properties of the europium(III) complexes in the solid state and in ethanol solution, the room temperature luminescence decay curves of the 5D0 excited state were measured by monitoring the most intense emission lines (5D0 → 7F2) of the europium(III) ion center at 615 nm under excitation with a 355 nm xenon lamp. As shown in Fig. 6, the luminescence decays of the excited states were best fitted to the monoexponential decay for all of the complexes. This demonstrated the presence of only one emissive europium(III) ion center in the solid state as well as in ethanol solution, which agreed with the analysis of the luminescence spectra of the europium(III) complexes. From the monoexponential decays, the luminescent lifetime values (τobs) of Na[Eu(dipic)2·phen]·H2O and Na[Eu(dipic)2·3H2O]·4H2O could be calculated as 2.31 ms and 2.38 ms in the solid state and 2.08 ms and 3.23 ms in ethanol solution, respectively. The location of the ligand states at high energy prevents both a mixing of these states with the 4f states and a back-transfer from the excited europium(III) ion to the ligand.17 Therefore, the lifetimes measured for the europium(III) (5D0) excited levels in Na[Eu(dipic)2·phen]·H2O and Na[Eu(dipic)2·3H2O]·4H2O were even higher than the values found for the complexes Na3[Eu(dipic)3]·nH2O (τobs = 1.3 ms)18 and [N(C2H5)4]3[Eu(dipic)3]·nH2O (τobs = 2.02 ms).19 This may reflect differences in the electronic structure and/or different coupling between the electronic structures of the europium(III) ions and ligands compared with other europium(III) complexes mentioned in ref. 18 and 19. | ||
| Fig. 6 The room temperature luminescence decay curves of the europium(III) complexes: (a) in the solid state, (b) in ethanol solution (λem = 615 nm, λex = 355 nm). | ||
Furthermore, the lifetime for Na[Eu(dipic)2·phen]·H2O in ethanol solution is 3.23 ms, which to the best of our knowledge is the longest lifetime reported to date. The relatively long lifetime for Na[Eu(dipic)2·phen]·H2O in ethanol solution was diagnostic of the absence of high-frequency oscillators in the first coordination sphere. Although there were high-energy C–H and O–H vibrations in the complexes, these were not in the first coordination sphere and thus were less effective in causing radiationless deactivation.
The effect of the coordination water on the europium(III) emission lifetimes can be clearly observed (Table 1) with the lifetime increasing for all the complexes when the solid state europium(III) complexes were dissolved in ethanol. This arose from the reduction of nonradiative relaxation of the excited europium(III) ion through coupling with OH vibrational modes. When these two complexes were compared, the lifetime of the emission from Na[Eu(dipic)2·3H2O]·4H2O in ethanol solution was always less than that from Na[Eu(dipic)2·phen]·H2O. However, for these two complexes in the solid state, the lifetime of Na[Eu(dipic)2·3H2O]·4H2O was longer than that of Na[Eu(dipic)2·phen]·H2O, regardless of the unsaturated coordination number in the former. The mechanism was an open question.
The quantum yields for the europium(III) emission of Na[Eu(dipic)2·3H2O]·4H2O and Na[Eu(dipic)2·phen]·H2O in ethanol solution were determined using quinine sulfate (dissolved in 0.5 M H2SO4 with a concentration of 10−6 M, assuming ΦPL of 0.55) as a standard.20 The luminescent efficiency was calculated according to the following formula:
 | (1) |
The quantum yield of Na[Eu(dipic)2·phen]·H2O (36.1%) was significantly higher than that of Na[Eu(dipic)2·3H2O]·4H2O (14.8%). Both of their quantum yields were slightly higher than that of Cs3[Eu(dipic)3] (13.5%), which was reported as a secondary standard for quantum yield determination.7 According to energy gap theory, the radiationless transitions are promoted by ligands and solvents with high-frequency vibrational modes. The increase in quantum yield is due to the suppression of radiationless transitions caused by vibrational relaxation. However, this comparatively low quantum yield of Na[Eu(dipic)2·3H2O]·4H2O was not particularly surprising, given the presence of high-frequency vibrations, which is an effective europium(III) excited-state quencher. Although there were high-energy O–H vibrations in the complex Na[Eu(dipic)2·phen]·H2O, these were not in the first coordination sphere and thus were less effective in causing radiationless deactivation. The exclusion of water from the inner sphere of the Na[Eu(dipic)2·3H2O]·4H2O complex minimized the nonradiative quenching of the europium(III) emission, resulting in a quantum yield that was higher than that of Na[Eu(dipic)2·3H2O]·4H2O.
3.7. Intrinsic quantum yield (ΦLN) and radiative lifetime (τ)
Assuming that non-radiative and radiative processes were essentially involved in the depopulation of the 5D0 state, the intrinsic quantum yield ΦLN of the 5D0 emission level in europium(III) complexes at room temperature was obtained based on the luminescence data (emission spectra and emission decay curves).The intrinsic quantum yield of the luminescence step, expressing how well the radiative processes (characterized by the rate constant Ar) compete with non-radiative processes (characterized by the rate constant Anr),21 can be obtained from the experimental spectroscopic data (emission spectra and emission decay curves).22
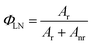 | (2) |
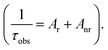
The radiative lifetime (τ), which can be calculated from the simple treatment of its corrected emission spectrum according to eqn (3), assuming that the energy of the 5D0 → 7F1 transition and its oscillator strength are constant,23 is directly related to the radiative and non-radiative decay rates of the lanthanide ion.
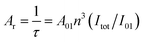 | (3) |
3.8. The stimulated emission cross-section (σp)
To estimate the capability of these europium(III) complexes as a laser medium, σp values were determined. The σp value is one of the most important factors for laser amplification. With the corresponding emission spectrum, for a Lorentz line, σp can be related to the radiative transition rate by
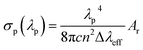 | (4) |
In general, the transition 5D0 → 7F2 of the europium(III) ion is interesting with respect to its use in laser systems. In this work, σp values of the 5D0 → 7F2 fluorescence transition of the europium(III) ion were of the same order as those shown by glasses used in solid state laser applications.24 When factors including the lifetime, the quantum yield and the stimulated emission cross-section were taken into consideration, Na[Eu(dipic)2·phen]·H2O was found to be more promising than Na[Eu(dipic)2·3H2O]·4H2O for optical applications such as high-powered laser media. In accordance with the above discussion, the relative emission intensities of the 5D0 → 7F2 to 5D0 → 7F1 transition (Irel), the FWHM of the 5D0 → 7F2 transition (Δλeff), emission lifetimes (τobs), quantum yields (Φ), radiative rates (Ar), stimulated emission cross-section (σp), radiative lifetime (τ), and intrinsic quantum yields (ΦLN) of the europium(III) complexes are summarized in Table 1.
4. Conclusion
Two new europium(III) complexes, Na[Eu(dipic)2·phen]·H2O and Na[Eu(dipic)2·3H2O]·4H2O were synthesized under mild conditions and characterized by various techniques. The photophysical properties in the solid state and in ethanol revealed the presence of a single luminescent site in all europium(III) complexes and efficient ligand-to-metal energy transfer. Europium(III) complexes showed relatively high quantum yields and long lifetimes. The lifetime for Na[Eu(dipic)2·phen]·H2O in ethanol solution is 3.23 ms, which is the longest lifetime reported to date. The stimulated emission cross-section was also determined and was very close to the values of a Nd-glass laser. Therefore, the luminescent europium(III) complex Na[Eu(dipic)2·phen]·H2O showed more potential application as a high-powered liquid laser medium.Acknowledgements
We gratefully acknowledge the financial support by the Foundation of China Academy of Engineering Physics (no. 2012A0302015, 2012B030250, 2013B0302051).References
- S. Bera, M. Ghosh, M. Pal, N. Das, S. Saha, S. Duttab and S. Jana, RSC Adv., 2014, 4, 37479–37490 RSC.
- Z. Liang, C. Chan, Y. Liu, W. Wong, C. Lee, G. Law and K. Wong, RSC Adv., 2015, 5, 13347–13356 RSC.
- R. Manigandan, K. Giribabu, R. Suresh, S. Munusamy, S. Kumar, S. Muthamizh, T. Dhanasekaran, A. Padmanaban and V. Narayanan, RSC Adv., 2015, 5, 7515–7521 RSC.
- K. Machado, S. Mukhopadhyay, R. Videira, J. Mishra, S. Mobin and G. Mishra, RSC Adv., 2015, 5, 35675–35682 RSC.
- Y. Hasegawa, H. Kawai, K. Nakamura, N. Yasuda and Y. Wada, J. Alloys Compd., 2006, 408–412, 771–775 Search PubMed.
- H. Gao, L. Yi, B. Ding, H. Wang, P. Cheng, D. Liao and S. Yan, Inorg. Chem., 2006, 45, 481–483 CrossRef CAS PubMed.
- A. Chauvin, F. Gumy, D. Imbert and J. Bünzli, Spectrosc. Lett., 2004, 37, 517–532 CrossRef CAS.
- A. Aebischer, F. Gumy and J. Bünzli, Phys. Chem. Chem. Phys., 2009, 11, 1346–1353 RSC.
- F. Prendergast, J. Lu and P. Callahan, J. Biol. Chem., 1983, 258, 4075–4078 CAS.
- L. Yang, S. Song, C. Shao, W. Zhang, H. Zhang, Z. Bu and T. Ren, Synth. Met., 2011, 161, 1500–1508 CrossRef CAS PubMed.
- L. Yang, L. Wu, H. Zhang, S. Song, L. Liu and M. Li, Dyes Pigm., 2013, 99, 257–267 CrossRef CAS PubMed.
- L. Yang, S. Song, C. Shao, W. Zhang, H. Zhang, Z. Bu and T. Ren, Synth. Met., 2011, 161, 925–930 CrossRef CAS PubMed.
- L. Yang, S. Song, H. Zhang, W. Zhang, L. Wu, Z. Bu and T. Ren, Synth. Met., 2012, 162, 261–267 CrossRef CAS PubMed.
- T. Zhu, K. Ikarashi, T. Ishigaki, K. Uematsu, K. Toda, H. Okawa and M. Sato, Inorg. Chim. Acta, 2009, 362, 3407–3414 CrossRef CAS PubMed.
- N. Shavaleev, S. Eliseeva, R. Scopelliti and J. Bünzli, Inorg. Chem., 2010, 49, 3927–3936 CrossRef CAS PubMed.
- W. Carnall, P. Fields and K. Rajnak, J. Chem. Phys., 1968, 49, 4412 CrossRef CAS PubMed.
- S. Tanase, P. Gallego, R. Gelder and W. Fu, Inorg. Chim. Acta, 2007, 360, 102–108 CrossRef CAS PubMed.
- W. Horrocks and D. Sudnick, J. Am. Chem. Soc., 1979, 101, 334–340 CrossRef CAS.
- P. Brayshaw, J. Buenzli, P. Froidevaux, J. Harrowfield, Y. Kim and A. Sobolev, Inorg. Chem., 1995, 34, 2068–2076 CrossRef CAS.
- C. Yang, J. Luo, J. Ma, M. Lu, L. Liang and B. Tong, Dyes Pigm., 2011, 92, 696–704 CrossRef PubMed.
- M. Werts, R. Jukes and J. Verhoeven, Phys. Chem. Chem. Phys., 2002, 4, 1542–1548 RSC.
- A. Balamurugan, M. Reddy and M. Jayakannan, J. Phys. Chem. B, 2009, 113, 14128–14138 CrossRef CAS PubMed.
- H. Roesky and M. Andruh, Coord. Chem. Rev., 2003, 236, 91–119 CrossRef CAS.
- W. Koechner, Solid-state laser engineering, Springer-Verlag, New York, 5th edn, 2005, ch. 2, pp. 53–54 Search PubMed.
| This journal is © The Royal Society of Chemistry 2015 |



