Co3O4 nanoparticles/cellulose nanowhiskers-derived amorphous carbon nanoneedles: sustainable materials for supercapacitors and oxygen reduction electrocatalysis†
R. Silvaa,
G. M. Pereiraa,
D. Voiryb,
M. Chhowallab and
T. Asefa*cd
aDepartamento de Química, Universidade Estadual de Maringá, Avenida Colombo 5790, CEP: 87020-900-Maringá, Paraná, Brazil
bDepartment of Materials Science and Engineering, Rutgers, The State University of New Jersey, 607 Taylor Road, Piscataway, New Jersey 08854, USA
cDepartment of Chemistry and Chemical Biology, Rutgers, The State University of New Jersey, 610 Taylor Road, Piscataway, New Jersey 08854, USA. E-mail: tasefa@rci.rutgers.edu
dDepartment of Chemical and Biochemical Engineering, Rutgers, The State University of New Jersey, 98 Brett Road, Piscataway, New Jersey 08854, USA
First published on 27th May 2015
Abstract
Hybrid nanostructured materials comprised of amorphous carbon nanoneedles (CNN)-supported Co3O4 nanoparticles (Co3O4-CNN) were synthesized. The synthesis involved layer-by-layer nanocasting of cellulose nanowhiskers with precursors of cobalt oxide and silica, followed by pyrolysis of the core–shell–shell composite materials and etching of the outer silica shells from the carbonized materials. Notably, cotton-derived cellulose nanowhiskers were used as the carbon precursors, and also as the hard templates for needle-shaped carbons, in the synthesis. The effectiveness of the core–shell–shell nanoreactors, possessing the silica shell-entrapped cellulose nanowhiskers and Co(II) ions, in generating organized carbon nanomaterials with metal oxide nanoparticles, or otherwise, as a function of the loading of Co(II) ions was evaluated. Details of the synthetic method and the different materials in terms of composition and morphology it results in as a function of the relative amount of metal ions have also been discussed. The materials showed promising supercapacitive properties and electrocatalytic activity for the oxygen reduction reaction (ORR). The materials' double layer capacitance and performance for ORR electrocatalysis as a function of their Co3O4 content and particles size have also been discussed. The results indicated that the electrochemical properties of these hybrid materials are strongly related to the morphology of their carbon nanostructures. The synthetic method demonstrated here can potentially serve as a facile route to produce other metal oxide/carbon nanomaterials, with different morphologies and similar or better properties, using other carbon precursors.
Introduction
Fuel cells allow the energy stored in chemical form in sustainable energy sources such as hydrogen to be recovered as electrical energy at/or near ambient temperature.1,2 However, in many conventional and commercially available fuel cells the cathode and anode electrodes contain electrocatalysts composed of expensive and less earth abundant noble metals such as platinum.3 And, the relative low earth abundance and high cost of such noble metals has long posed serious concerns on the feasibility of fuel cell technologies for large scale uses. Additionally, even with such expensive electrocatalysts, a great portion of the energy in fuel cells is lost due to the large overpotential needed to overcome the oxygen reduction reaction (ORR) at the cathodic half-reaction of fuel cells (as the ORR typically requires overpotentials as high as 500–600 mV to take place even with platinum-based catalysts).4 The severe activation loss associated with ORR, coupled with the intrinsic low kinetics of the reaction, is therefore often responsible for the limited current density as well as cell voltage given off by many conventional fuel cells.5,6Hence, breakthrough technological advances in fuel cells demand the development of sustainable, noble metal-free, alternative electrocatalysts to platinum, and those that can catalyze ORR with at least the same efficiency as,7 if not better than, platinum.8 It is only then that the large scale deployment of fuel cells based on energy sources such as hydrogen to meet the so-called “hydrogen economy” can be realized.9
To this end, intensive research in this area has recently led to various ORR electrocatalysts that contain inexpensive and earth-abundant early transition metals.10,11 In particular, cobalt and cobalt oxides supported on carbon materials, especially graphene, have emerged as viable alternatives to conventional Pt/C electrocatalysts.12–14 This is because supported cobalt or cobalt oxides can catalyze ORR effectively while cobalt meets the requirements of sustainable materials due to its relatively high earth-abundance, low cost (which is only a fraction of that of platinum), and low toxicity especially compared with many other heavy metals.15
On the other hand, owing to its conducive electronic properties for electrochemical processes, graphene has been widely considered as a support material for many metallic- and metal oxide-based nanoparticles for electrocatalysis.16,17 However, the potential use of graphene and its derivatives as a support material for electrocatalysts has some limitations too, with the most notable of which being the unavailability of synthetic methods that can produce pristine graphene on large scale for scalable production of such electrocatalysts. This also explains why despite reports of a number of graphene-based electrocatalysts, many of them in actuality contain either graphene oxide (GO) or reduced graphene oxide (rGO), and not pristine graphene.18,19 Unlike pristine graphene, which has no band gap, GO and rGO are indirect band gap materials with substantially different electronic properties.20 This is merely due to the presence of hydrogen and oxygen moieties (or impurities) on the latter. Nevertheless, besides substantially changing the carbon material's inherent properties, the presence of such ‘impurity’, even in tiny amount, on carbonaceous materials such as graphene affects the surface interactions and charge transfers between the carbon support materials and the supported nanoparticles, and thereby the overall materials' potential applications in areas such as (electro)catalysis and charge storage.21,22
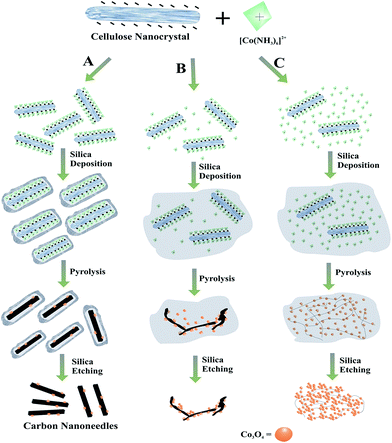
Therefore, other viable support materials, alternative to graphene but still allow cobalt-based electroactive moieties to transfer their charges easily and give enhanced electrocatalytic activity toward ORR, are highly desired. In view of this and the issues mentioned above, we had thought that the nanoreactor method we had previously used to make pure crystalline carbon nanoneedles23 could be exploited to produce carbon nanoneedle (CNN)-supported early transition metal oxide nanoparticles for electrocatalysis (i.e., unique types of carbon nanomaterials with high proportions of plane edges and good electrical conductivity decorated with electrocatalytic active metal oxides on their surfaces). Herein this synthetic idea is shown to work, with the report of the synthesis of carbon nanoneedles-supported cobalt oxide electrocatalysts, as illustrated in Fig. 1, that exhibit good electrocatalytic activity for ORR. Moreover, the ability of the materials to serve as high performance supercapacitors is also demonstrated.
Results and discussion
Materials synthesis and characterization
The first step in the synthesis of the Co3O4/CNN materials involved the successive encapsulation of nanostructured carbon precursors derived from a natural source with metal complexes, followed by thermally robust silica shells (or nanocages). Specifically, cellulose nanowhiskers prepared by acid hydrolysis from cotton were coated by hexaamminecobalt(II) complex ([Co(NH3)6]2+) and then by silica shells, both via electrostatic self-assembly, as schematically shown in Fig. 1. The resulting Co2+/cellulose@SiO2 core@shell nanomaterials were treated at high temperature under inert atmosphere (N2) in different stages as presented in Fig. S1.† This allowed the cellulose nanowhiskers trapped inside the nanocages to undergo carbonization and form carbon nanoneedles with shapes akin to the shapes of the nanocages. At the same time, the metal complexes sandwiched between the cellulose cores and the silica shells were thermally transformed into metal oxide nanoparticles. Upon removal of the silica shells, freestanding carbon nanoneedles decorated with Co3O4 nanoparticles (Co3O4/CNN) that can efficiently electrocatalyze ORR and effectively store charges were obtained. Although we used this type of synthetic method to produce crystalline carbon nanoneedles before,23 it is in this present work that the method is shown, for the first time, to be applicable for making amorphous carbon nanoneedles-supported early transition metal oxides in situ. Moreover, it is also for the first time that materials synthesized this way are shown to have high electrocatalytic activity for ORR as well as good charge storage capacity.Besides serving as precursor for the desired metal oxide catalytic sites (i.e., Co3O4 nanoparticles, in this case), the metal complexes ([Co(NH3)6]2+) anchored onto the cellulose nanowhiskers play two more important roles. First, they allow the formation of silica shells around the cellulose nanowhiskers, by enabling the spontaneous electrostatic deposition on the surfaces of the cellulose nanowhiskers of negatively charged silicate intermediates formed from the hydrolysis and condensation silica precursors under basic conditions. Second, they serve as catalyst, promoting the formation of graphitic carbon during the carbonization step (please note that metallic phases composed of Fe, Cu and Ni are well-known to promote graphitization of carbon during high temperature carbonization processes24).
Specifically, as depicted in Fig. 1, three different concentrations (i.e., relatively low, moderate or high) of [Co(NH3)6]2+ were used in the synthesis, which after carbonization led to three Co3O4/CNN@SiO2 materials, labeled as Co3O4/CNN@SiO2-A, -B and -C, respectively. After removal of the silica shells, three different Co3O4/CNN materials, denoted as Co3O4/CNN-A, Co3O4/CNN-B and Co3O4/CNN-C, respectively, with less, moderate and large relative amount Co3O4, respectively, were ultimately obtained, as discussed in more detail below.
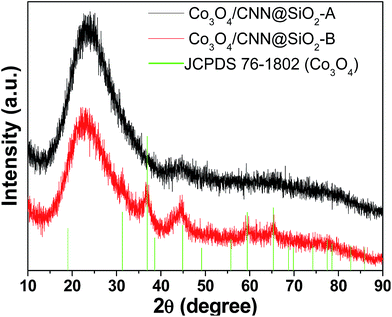
The structure and composition of the resulting Co3O4/CNN materials were then characterized by different analytical techniques. Fig. 2 displays the powder X-ray diffraction (XRD) patterns of Co3O4/CNN@SiO2-A and Co3O4/CNN@SiO2-B. In the XRD pattern of Co3O4/CNN@SiO2-A (which was synthesized with the lowest amount of [Co(NH3)6]2+ ions), no visible well-defined peaks corresponding to crystalline phases are observed. The result suggests that the Co3O4 on this material, whose presence is confirmed by other methods (vide infra), are either too small in size or in amount, or are inherently amorphous to diffract X-ray beams. On the other hand, the diffraction pattern of Co3O4/CNN@SiO2-B clearly displays peaks that correspond to Co3O4. Nevertheless, the diffraction peaks are still broad in the latter case, suggesting that the Co3O4 particles formed in Co3O4/CNN@SiO2-B are small in size.
The relative amount of Co3O4 in the Co3O4/CNN materials was determined by thermogravimetric analysis (TGA) under a flow of synthetic air (20% O2 in N2). The TGA results are presented in Fig. 3. From the residual weight obtained after the complete decomposition of the composite materials under synthetic air, the content of Co3O4 in each material was then determined. Accordingly, the relative amount of Co3O4 in Co3O4/CNN@SiO2-A, -B and -C were found to be 6.4, 57.5, and 81.4%, respectively. This also means that Co3O4/CNN-A has the least amount of Co3O4 (ca. 6%) by weight with respect to carbon, Co3O4/CNN-B contains comparable weight ratio of Co3O4 and carbon, and Co3O4/CNN-C has the highest relative amount of Co3O4 (ca. 81%) with respect to carbon. The result also clearly indicates the broadly varying composition or content of Co3O4 in the materials, as a result of a simple change in the relative amount of [Co(NH3)6]2+ complex used for the synthesis of the materials.
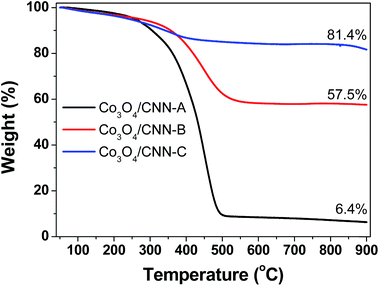 | ||
| Fig. 3 Thermogravimetric traces obtained for the different Co3O4/CNN materials at a heating rate of 10 °C min−1 under a constant flow of synthetic air (20% O2 in N2) at a rate of 20 mL min−1. | ||
The morphologies of the Co3O4/CNN materials were probed by TEM microscopy, and the results are displayed in Fig. 4. The TEM images generally suggest that the amount of [Co(NH3)6]2+ used in the synthesis also significantly affects the morphology of the final materials, besides their compositions or relative amounts of Co3O4 as discussed above. The TEM image of Co3O4/CNN@SiO2-A (Fig. 4a) shows the presence of needle-shaped nanostructures. Under higher magnification, the TEM image (Fig. 4b) further shows the presence of Co3O4 nanoparticles with ca. 8–10 nm in diameter.
 | ||
| Fig. 4 TEM images of Co3O4/CNN@SiO2-A (a and b), Co3O4/CNN@SiO2-B (c and d), and Co3O4/CNN@SiO2-C (e and f). | ||
On the other hand, no needle-shaped carbon nanostructures were observed in the TEM images of Co3O4/CNN@SiO2-B and Co3O4/CNN@SiO2-C (Fig. 4c–e). The TEM images of Co3O4/CNN@SiO2-B rather show the presence of large numbers of Co3O4 nanoparticles with an average size of ca. 10 nm distributed over highly interconnected carbon nanostructures (Fig. 4d). On the other hand, despite the TEM images of Co3O4/CNN@SiO2-C (Fig. 4e and f) also show small nanoparticles (10–20 nm), the nanoparticles are highly interconnected or aggregated, reaching sizes up to few micrometers. This is most likely to do with the relatively high amount of [Co(NH3)6]2+ ions used for the synthesis of this particular material, compared with those used to make the other two materials. In fact, this material had 81.4% Co3O4 by weight, or conversely relatively little carbon.
Supercapacitive properties of Co3O4/CNN materials
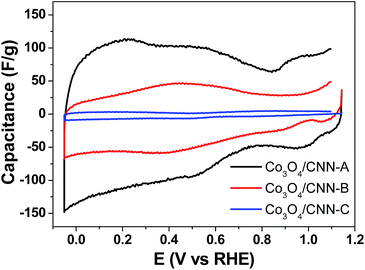
Next, the specific capacitance of the Co3O4/CNN materials was measured with cyclic voltammetry (Fig. 5). Using the results, the relationship between the specific capacitance and the structure/morphology of the materials was also elucidated. All the electrochemical measurements were carried out using a saturated calomel electrode (SCE) as reference, which was converted to reversible hydrogen electrode (RHE) as described in ESI and Fig. S2.†As the Co3O4/CNN materials obtained by using different concentrations of [Co(NH3)6]2+ were found to possess different amounts of Co3O4 and have distinct structures, they were expected to exhibit different charge storage capacity as well. Moreover, as the electrochemical capacitance of carbonaceous materials like Co3O4/CNN often stems from the electrical double-layers forming on the surfaces of the materials, the capacitance (C) of the materials can be directly correlated to the electrochemical specific surface areas of the materials, with eqn (1):25
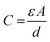 | (1) |
In Fig. 5, the graphs of electrochemical capacitance versus potential for the three materials are presented. The values of specific capacitance in the potential range of −0.05 to 1.15 V vs. RHE were found to be 90.8, 33.6, and 3.1 F g−1 for Co3O4/CNN-A, Co3O4/CNN-B, and Co3O4/CNN-C, respectively. The result clearly shows that the three materials have significantly different capacitance. The result also shows a very clear inverse relationship between the capacitance of the materials and the amount of Co3O4 they contain. The lowest capacitance was found for Co3O4/CNN-C, which has a large density of highly aggregated clusters of Co3O4 nanoparticles with 10–20 nm in size, whereas the highest specific capacitance was obtained for Co3O4/CNN-A, which has needle-shaped nanostructured carbon with relatively small amount of Co3O4 with ca. 8–10 nm in size. Although needle-like structures are also verified in sample B by TEM, the specific capacitance of Co3O4/CNN-B, which is composed of 57.5% of Co3O4, is 3-fold lower than that of Co3O4/CNN-A, which has only 6.4% of Co3O4. Consequently, it can be fairly concluded that Co3O4/CNN-A' relatively high amount of carbon and needle-shaped nanostructures, which can provide the material with large electrochemical active surface area, are responsible for Co3O4/CNN-A's high double layer capacitance.
Electrocatalytic properties of Co3O4/CNN materials for ORR
The electrocatalytic properties of all the Co3O4/CNN materials toward ORR were then tested, and by using the results the interplay between the composition/structural features and the electrocatalytic activities of the materials was probed. To quickly check if the Co3O4/CNN materials can catalyze ORR, cyclic voltammogram (CV) over Co3O4/CNN-A in O2-saturated, 0.1 M KOH solution was first performed (Fig. 6). As a negative control, the cyclic voltammogram (CV) of the same material in N2-saturated KOH solution (0.1 M) was also conducted, and the result was included (Fig. 6). When comparing the two curves, a strong catalytic reduction peak over Co3O4/CNN-A (with an onset potential of 0.81 V vs. RHE) is seen in the curve obtained under the O2-saturated solution. Conversely, no such reduction peak is observed for the one obtained in the N2-saturated solution. These results clearly indicate that Co3O4/CNN-A effectively catalyzes the ORR.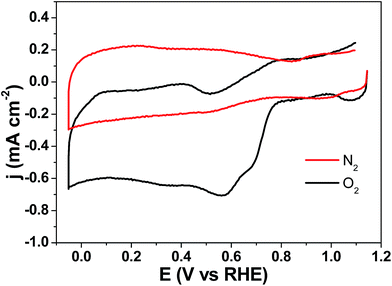 | ||
| Fig. 6 Cyclic voltammograms over Co3O4/CNN-A in O2- and N2-saturated KOH (0.1 M) solutions at scan rate of 20 mV s−1. | ||
In Fig. 7 the CVs of all the three Co3O4/CNN materials obtained in O2-saturated KOH solution (0.1 M) are shown together. From the graphs, all the three materials, Co3O4/CNN-A, -B, and -C can be seen to electrocatalyze ORR, with onset potentials of 0.81 V vs. RHE, 0.82 V vs. RHE and 0.68 V vs. RHE, respectively. So, among the three materials, Co3O4/CNN-C can be said to have the highest overpotential or the lowest electrocatalytic activity toward ORR, despite its highest amount of Co3O4. On the other hand, Co3O4/CNN-A and Co3O4/CNN-B show lower overpotentials and better electrocatalytic activity toward ORR. It is also interestingly to note that the catalytic activity of Co3O4/CNN-A is similar to that of Co3O4/CNN-B, despite the former contains approximately 10-fold lower amount of Co3O4 than the latter. Moreover, when the activities of the materials towards ORR are compared based on current density, it can be seen Co3O4/CNN-A is once again a better electrocatalyst than either Co3O4/CNN-B or Co3O4/CNN-C. So, these results indicate that the difference in electrocatalytic activity between these three materials can be attributed to their carbon nanostructures and the relative amount of carbon materials they contain. It is worth reiterating here again that the former is composed of a large amount of, needle-shaped carbon nanostructures while the latter two possess relatively less amount of carbon with disorganized nanostructures. Similarly, Co3O4/CNN-B contains relatively larger amount of carbon materials compared with Co3O4/CNN-C. These said, it is worth adding here that CNN material made without Co3O4 or by using a small amount FeCl3, which was added only to promote the formation of needle-shaped carbons for the synthesis of CNN, did show significantly lower activity than Co3O4/CNN-A, with onset potential of 0.67 V vs. RHE (Fig. S3†). So, overall, besides the needle-shaped carbon nanostructures, the presence of some optimal amount of Co species (and Co3O4 nanoparticles) directly or indirectly assists the material with its electrocatalytic activity toward ORR. This is most likely why Co3O4/CNN-A showed the best electrocatalytic activity for ORR compared with the materials we have investigated here. It is worth adding here that the electrocatalytic activity of Co3O4/CNN-A is also actually comparable to that of some of the most efficient graphene and carbon nanotube-supported Co3O4 materials reported previously, albeit it is lower than that of Pt/C (20%) (Fig. 7).26
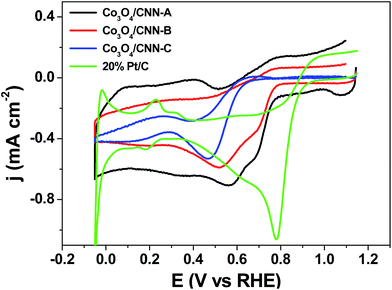 | ||
| Fig. 7 Cyclic voltammograms of Co3O4/CNN-A, Co3O4/CNN-B, and Co3O4/CNN-C in O2 saturated KOH solutions (0.1 M) at scan rate of 20 mV s−1. | ||
The stability of Co3O4/CNN-A and Co3O4/CNN-B during ORR were tested with chronoamperometry for a period of 14 h at constant applied potential of 0.73 V vs. RHE (Fig. 8). In the chronoamperometric curve of Co3O4/CNN-A, the current density is seen to decrease slightly over 9 h of direct use, but remains stable afterwards. At the end of the experiment, while the current density of Co3O4/CNN-A reaches ca. 68.8% of the initial value, that of Co3O4/CNN-B decreases continuously and reaches as low as 36% of the initial value after 14 h of direct use. So, the results show that compared with Co3O4/CNN-B, Co3O4/CNN-A not only gives higher current density but also retains the higher current density over longer time period. This huge difference between Co3O4/CNN-A and Co3O4/CNN-B and Co3O4/CNN-A's more stability as electrocatalyst in ORR further highlights the benefits of the carbon nanoneedle structures and the optimal density of Co3O4 in it for electrocatalysis.
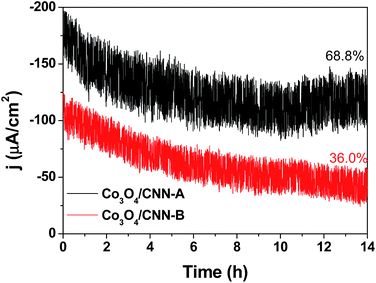 | ||
| Fig. 8 Chronoamperometric curves ORR over Co3O4/CNN-A and Co3O4/CNN-B in O2-saturated KOH solution (0.1 M) at constant potential of 0.73 V vs. RHE. | ||
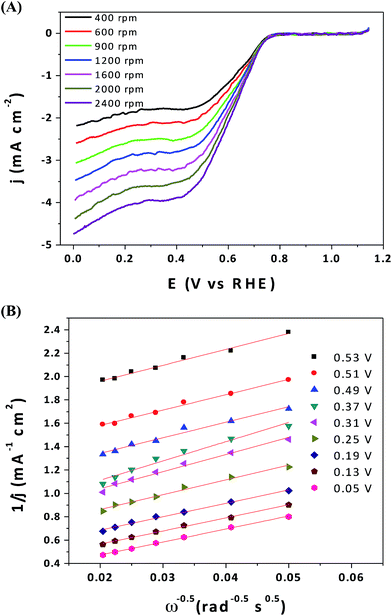
Finally, polarization curves were obtained for the ORR over Co3O4/CNN-A electrocatalyst at different spin rates on a rotating disk electrode (RDE), and the results are presented in Fig. 9A. The corresponding Koutecky–Levich plots obtained from the polarization curves (Fig. 9B) indicate that the average number of electron transferred in the range of 0.07 to 0.56 V vs. RHE is 2.22. This suggests the high selectivity of Co3O4/CNN-A electrocatalysts for 2-electron process in ORR, which favors the formation of hydrogen peroxide from the reduction of O2.8
Conclusions
The synthesis of carbon nanoneedles-supported Co3O4 nanoparticles with different amount of Co3O4 nanoparticles using cotton-derived cellulose nanowhiskers as precursors has been described. The materials have been shown to have high supercapacitive charge storage properties as well as efficient catalytic activities for ORR. Specifically, three different materials containing amorphous carbon nanoneedles with relatively low (6.4%), moderate (57.5%), and high (81.4%) amount of Co3O4, denoted as Co3O4/CNN-A, Co3O4/CNN-B, and Co3O4/CNN-C, respectively, have been synthesized and characterized by various analytical methods. While Co3O4/CNN-B and Co3O4/CNN-C showed disorganized structure, Co3O4/CNN-A showed well-defined carbon nanoneedles decorated with Co3O4 nanoparticles, as characterized by TEM and XRD. Their XRD patterns, on the other hand, have indicated that the carbon phases in the materials are amorphous. Among the samples investigated here, Co3O4/CNN-A has afforded a high specific capacitance, which is attributed to its nanoneedle morphology, and most importantly, its high electrochemical active surface area. The results have also clearly revealed the trends between the electrochemical capacitance and the structures of the materials or the size of Co3O4 nanoparticles they contain. Co3O4/CNN-A has also exhibited the highest catalytic activity for ORR, in terms of overpotential, current density and stability. The electrocatalytic activity of these amorphous carbon nanoneedles-supported Co3O4 nanoparticles, especially of Co3O4/CNN, has been found to be comparable with some of the most efficient graphene and carbon nanotube-supported Co3O4 nanomaterials reported previously.26 The results suggested that the structures of the underlying carbon needle support materials and the presence of optimal amount of Co3O4 nanoparticles play outstanding roles in the Co3O4/CNN-A's catalytic activities toward ORR.Experimental
Synthesis of cellulose nanowhiskers
To synthesize cellulose nanowhiskers, cotton (6 g) was taken and soaked in aqueous NaOH (1 M) solution (300 mL) for 1 h. The cotton was then removed from the solution and washed with copious amount of distilled water and dried in oven at 60 °C for 6 h. One gram of the washed cotton was taken and hydrolyzed in sulfuric acid solution (65%) (40 mL) at 72 °C for 30 min under magnetic stirring (800 rpm). The resulting solution was centrifuged for 5 min. The supernatant was decanted and the precipitate was recovered and re-dispersed in water. Centrifugation of the solution, followed by decantation of the supernatant and re-dispersion of the precipitate in fresh water, were repeated until the liquid phase became opaque. At this point, the liquid phase and the solid were sonicated for 6 h. The dispersion was centrifuged again at 5000 rpm for 5 min. The liquid phase was collected and the precipitate was discarded. The liquid phase was then centrifuged for 15 minutes at 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm, resulting in the cellulose nanowhiskers.
000 rpm, resulting in the cellulose nanowhiskers.
Syntheses of amorphous carbon nanoneedles supported Co3O4 nanoparticles
The cellulose nanowhiskers obtained above were dispersed in 12 mL of water and 300 mL of absolute ethanol. Three different samples were prepared by adding different amounts of Co(NO3)2 into the above mixture. For sample A, 25 mg; for sample B, 75 mg; and for sample C, and 150 mg of Co(NO3)2 were stirred with the mixture for 30 min. After further addition of 5 mL of ammonia solution (25%), the dispersion was stirred for another 30 min. While the dispersion was kept stirring, 0.5 mL of tetraethyl orthosilicate was added into it. After stirring the dispersion for 12 h, the solid product was recovered by centrifugation and washed with ethanol and re-centrifuged. The precipitate was recovered, dried, and crushed in a mortar and pestle. The resulting powdered material (presumably containing Co3O4/CNN@SiO2 core–shell nanoparticles) was thermally treated at different thermal stages as presented in Fig. S1.† The silica shells were etched off of the particles by stirring the material in 1 M aqueous NaOH solution for 2 h at 80 °C. The resulting solid (powdered) material was washed three times with water, via centrifugation at 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 30 minutes, followed by decantation. It was then dried under ambient conditions, giving the desired Co3O4/CNN samples.
000 rpm for 30 minutes, followed by decantation. It was then dried under ambient conditions, giving the desired Co3O4/CNN samples.
Acknowledgements
T.A. gratefully acknowledges the financial assistance provided to his group by the U.S. National Science Foundation (grants CBET-1134289, NSF-ACIF, and NSF Special Creativity grant in 2011).Notes and references
- V. Mazumder, Y. Lee and S. Sun, Adv. Funct. Mater., 2010, 20, 1224 CrossRef CAS PubMed.
- R. Silva and T. Asefa, Adv. Mater., 2012, 24, 1878 CrossRef CAS PubMed.
- Y. Zhao, L. Z. Fan, H. Z. Zhong, Y. F. Li and S. H. Yang, Adv. Funct. Mater., 2007, 17, 1537 CrossRef CAS PubMed.
- O. E. Herrera, D. P. Wilkinson and W. Merida, J. Power Sources, 2012, 198, 132 CrossRef CAS PubMed.
- J. X. Wang, J. L. Zhang and R. R. Adzic, J. Phys. Chem. A, 2007, 111, 12702 CrossRef CAS PubMed.
- J. K. Nørskov, J. Rossmeisl, A. Logadottir, L. Lindqvist, J. R. Kitchin, T. Bligaard and H. Jonsson, J. Phys. Chem. B, 2004, 108, 17886 CrossRef.
- Y. Q. Dong, H. C. Pang, H. B. Yang, J. Jiang, Y. W. Chi and T. Yu, RSC Adv., 2014, 4, 32791 RSC.
- R. Silva, D. Voiry, M. Chhowalla and T. Asefa, J. Am. Chem. Soc., 2013, 135, 7823 CrossRef CAS PubMed.
- X. Zou, X. Huang, A. Goswami, R. Silva, B. R. Sathe, E. Mikmekova and T. Asefa, Angew. Chem., Int. Ed., 2014, 53, 4372 CrossRef CAS PubMed.
- K. P. Gong, F. Du, Z. H. Xia, M. Durstock and L. M. Dai, Science, 2009, 323, 760 CrossRef CAS PubMed.
- P. M. Hallam, B. L. Riehl, B. D. Riehl and C. E. Bank, RSC Adv., 2011, 1, 93 RSC.
- S. Guo, S. Zhang, L. Wu and S. Sun, Angew. Chem., Int. Ed., 2012, 51, 11770 CrossRef CAS PubMed.
- Y. Liang, Y. Li, H. Wang, J. Zhou, J. Wang, T. Regier and H. Dai, Nat. Mater., 2011, 10, 780 CrossRef CAS PubMed.
- Y. M. Tan, C. F. Xu, G. X. Chen, X. L. Fang, N. F. Zheng and Q. J. Xie, Adv. Funct. Mater., 2012, 22, 4584 CrossRef CAS PubMed.
- Y. Liang, H. Wang, P. Diao, W. Chang, G. Hong, Y. Li, M. Gong, L. Xie, J. Zhou, J. Wang, T. Z. Regier, F. Wei and H. Dai, J. Am. Chem. Soc., 2012, 26, 15849 CrossRef PubMed.
- F. Zhang, F. M. Yasin, X. J. Chen, J. X. Mo, C. L. Raston, L. Colin and H. B. Zhang, RSC Adv., 2013, 3, 25166 RSC.
- A. H. Castro Neto, F. Guinea, N. M. R. Peres, K. S. Novoselov and A. K. Geim, Rev. Mod. Phys., 2009, 81, 109 CrossRef CAS.
- X.-H. Guan, L. Yang, X. Guan and G.-S. Wang, RSC Adv., 2015, 5, 36185 RSC.
- D. R. Dreyer, S. Park, C. W. Bielawski and R. S. Ruoff, Chem. Soc. Rev., 2010, 39, 228 RSC.
- G. Eda, G. Fanchini and M. Chhowalla, Nat. Nanotechnol., 2008, 3, 270 CrossRef CAS PubMed.
- S. Kundu, T. C. Nagaiah, X. X. Chen, W. Xia, M. Bron, W. Schuhmann and M. Muhler, Carbon, 2012, 50, 4534 CrossRef CAS PubMed.
- L. Li, Z. D. Wei, Y. Zhang, X. Q. Qi, M. R. Xia, J. Zhang, Z. G. Shao and C. X. Sun, Sci. China, Ser. B: Chem., 2009, 52, 571 CrossRef CAS PubMed.
- R. Silva, J. Al-Sharab and T. Asefa, Angew. Chem., Int. Ed., 2012, 51, 7171 CrossRef CAS PubMed.
- F. J. Maldonado-Hodar, C. Moreno-Castilla, J. Rivera-Utrilla, Y. Hanzawa and Y. Yamada, Langmuir, 2000, 16, 4367 CrossRef CAS.
- P. Simon and A. Burke, Electrochem. Soc. Interface, 2008, 17, 38 CAS.
- S. Guo, S. Zhang, L. Wu and S. Sun, Angew. Chem., Int. Ed., 2012, 51, 11770 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra08037a |
| This journal is © The Royal Society of Chemistry 2015 |