Removal of dyes and heavy metal ions from water by magnetic hydrogel beads based on poly(vinyl alcohol)/carboxymethyl starch-g-poly(vinyl imidazole)
Abstract
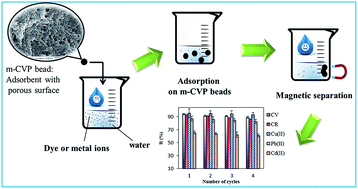
Novel magnetic nanocomposite hydrogel (m-CVP) beads were prepared by instantaneous gelation of carboxymethyl starch-g-polyvinyl imidazole (CMS-g-PVI), poly(vinyl alcohol) (PVA), and Fe3O4 mixture in boric acid solution followed by crosslinking by glutaraldehyde (GA). Highly porous m-CVP beads with magnetic sensitivity were fully characterized and used as eco-friendly adsorbent for the removal of crystal violet (CV) and congo red (CR) dye and Pb(II), Cu(II) and Cd(II) from water. Batch adsorption results showed that the sorbent had high affinity to the heavy metals and dyes in water. The adsorption kinetics results showed that the adsorption rates could be described by pseudo-second-order kinetics, indicating chemical sorption as the rate determining step. Sorption of Pb(II), Cu(II), Cd(II), CR and CV to m-CVP beads agreed well with the Langmuir adsorption model at different ionic strengths, with maximum adsorption capacities of 65.00, 83.60, 53.20, 83.66 and 91.58 mg g−1, respectively. Positive values of ΔH0 showed that the adsorption was chemisorption, and the negative values of ΔG0 indicated the spontaneity of the pollutant adsorption. Desorption and reusability of m-CVP beads was also investigated for tested heavy metal ions and dyes based on sequential adsorption–desorption cycles. All the studied results indicated that m-CVP beads were an efficient, low cost, and reusable adsorbent for the removal of dye and heavy metal ions from aqueous solutions.

 Please wait while we load your content...
Please wait while we load your content...