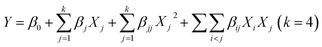Optimization of ultrasound-assisted extraction (UAE) of phenolic compounds from Crataegus pinnatifida leaves and evaluation of antioxidant activities of extracts
Meng Luoabd,
Jiao-Yang Hua,
Zhuo-Yue Songa,
Jiao Jiaoac,
Fan-Song Mu*ab,
Xin Ruana,
Qing-Yan Gaia,
Qi Qiaoa,
Yuan-Gang Zua and
Yu-Jie Fuab
aKey Laboratory of Forest Plant Ecology, Ministry of Education, Northeast Forestry University, Harbin 150040, PR China. E-mail: fansong8867@nefu.edu.cn; Fax: +86 451 82102082; Tel: +86 451 82191517
bCollaborative Innovation Center for Development and Utilization of Forest Resources, Harbin 150040, PR China
cState Key Laboratory of Tree Genetics and Breeding, Northeast Forestry University, Harbin 150040, PR China
dSchool of Integrative Plant Science, Cornell University, Ithaca, NY 14853, USA
First published on 27th July 2015
Abstract
In this study, an accurate method using high pressure liquid chromatography (HPLC) has been established for the simultaneous determination of chlorogenic acid (CA), vitexin-4′′-o-glucoside (VG), vitexin-2′′-o-rhamnoside (VR), orientoside (ORT), rutin (RT), vitexin (VIT) and hyperoside (HYP) from Crataegus pinnatifida leaves. The Box–Behnken design (BBD) combined with response surface methodology (RSM) was used to optimize the ultrasound-assisted extraction (UAE) method. Under optimized conditions (extraction temperature 41 °C, extraction time 31 min, ratio of liquid/solid 15 and ethanol concentration 39%), the yields of CA, VG, VR, ORT, RT, VIT and HYP were 0.46, 0.38, 4.37, 0.033, 0.036, 0.30 and 1.19, mg g−1, respectively. Additionally, antioxidant activities of UAE extracts in DPPH radical scavenging (DPPH), ABTS radical scavenging (ABTS) and reducing power tests were determined as 0.69 mg mL−1 (IC50 value), 0.86 mM Trolox and 0.24 mg mL−1 (IC50 value), respectively. Compared with conventional maceration extraction (ME) and heat-refluxing extraction (HRE) methods, the UAE method showed remarkable advantages in terms of higher extraction yields and better antioxidant activities. Overall, the present results showed that UAE was an efficient and alternative method for the extraction of phenolic compounds from C. pinnatifida leaves and its extracts were potential antioxidant ingredients or additives for the food industry with potential preservation properties.
1. Introduction
Crataegus pinnatifida Bge. Var. Major N. E. Br. (C. pinnatifida) (Rosaceae) is frequently utilized in the food industry because of its beneficial health effects and low toxicity.1 Northeast China is the leading production area of C. pinnatifida. Recent studies have shown that C. pinnatifida fruits and leaves possess various biological activities such as antioxidant, anti-inflammatory, vaso-relaxing and hypolipidemic effects.2–5 Many active compounds have been isolated from C. pinnatifida leaves, such as polyphenols, triterpenes, proanthocyanidins, and organic acids and so on.6–8 Among them, phenolic compounds including CA, VG, VR, ORT, RT, VIT and HYP9,10 are responsible for the treatment of cardiovascular, cerebrovascular, hypertension, antioxidative and hyperlipoidemia diseases.11–14 Therefore, it is necessary to develop a more effective and reliable extraction method to extract these health-promoting phenolic compounds from C. pinnatifida leaves for the pharmaceutical and healthy food industry.Several methods such as HPLC-UV and HPLC-MS/MS have been established to analysis phenolic compounds from C. pinnatifida leaves in previous studies.15–18 Conventional extraction methods such as ME and HRE which were associated with disadvantages of high consumption of organic solvent and energy along with long extraction time are always used to extract active compounds from plant materials.19,20 UAE is an environmental friendly method that can decrease solvent consumption, shorten extraction time, enhance extraction yield and improve extract quality.21,22 UAE has been widely applied in the fields of phytochemistry, biochemistry, physics and metallurgy, especially used for the extraction of active ingredients from medicinal plants.23–26
In the present study, UAE was used for the simultaneous extraction of seven phenolic compounds including CA, VG, VR, ORT, RT, VIT and HYP (Fig. 1) from C. pinnatifida leaves. Moreover, a sensitive and accurate HPLC method was successfully established for simultaneous determination of CA, VG, VR, ORT, VIT, RT and HYP in C. pinnatifida leaves. Furthermore, antioxidant activities of C. pinnatifida leaf extracts were comprehensively evaluated by several in vitro antioxidant methods, which can provide a basic research of antioxidant benefits of C. pinnatifida leaves. To our best knowledge, there is seldom report on the extraction, antioxidant evaluation and simultaneous quantification of seven main phenolic compounds from C. pinnatifida leaves.
2. Materials and methods
2.1. Materials and chemicals
C. pinnatifida leaves was obtained from Northeast Forestry University (Harbin, China). Voucher specimens were deposited in the herbarium of Key Laboratory of Forest Plant Ecology, Ministry of Education, Northeast Forestry University. C. pinnatifida leaves were dried in air, powered by a disintegrator, passed through a sieve (20–60 mesh) and stored at 4 °C until used. Standard compounds including CA, VG, VR, ORT, RT, VIT and HYP were bought from Ronghe Pharmaceutical Technology Company (Shanghai, China). DPPH and ABTS kits were obtained from Sigma-Aldrich Co. (Steinheim, Germany). Other reagents of either analytical or optical grades were obtained from Beijing Chemical Reagents Co. (Beijing, China). Water for HPLC was purified using a Milli-Q Water Purification system (Millipore, MA, USA).2.2. HPLC condition
HPLC analyses were conducted on a Waters HPLC system (Waters Co., Miford, MA) which consisted of a 1525 pump, a 2996 UV-vis photodiode array detector and a 7725 injection valve with a 20 μL loop. All samples were separated on a Waters C18 column (5 μm, 250 mm × 4.6 mm, i.d.) at 30 °C. The mobile phase was THF–acetonitrile–water–phosphoric acid (175![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 31
31![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 794
794![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.385, v/v/v/v). The flow rate was 1.0 mL min−1, and the injection volume was 20 μL. All target compounds were monitored at the wavelength of 350 nm. The chromatographic peaks of analytes were confirmed by comparing their retention times and UV spectra with those of the reference compounds. LC chromatograms of standards and UAE extracts of C. pinnatifida leaves are shown in Fig. 2. Contents of target compounds were calculated by corresponding calibration curves based on the dry weight of C. pinnatifida leaves.
0.385, v/v/v/v). The flow rate was 1.0 mL min−1, and the injection volume was 20 μL. All target compounds were monitored at the wavelength of 350 nm. The chromatographic peaks of analytes were confirmed by comparing their retention times and UV spectra with those of the reference compounds. LC chromatograms of standards and UAE extracts of C. pinnatifida leaves are shown in Fig. 2. Contents of target compounds were calculated by corresponding calibration curves based on the dry weight of C. pinnatifida leaves.
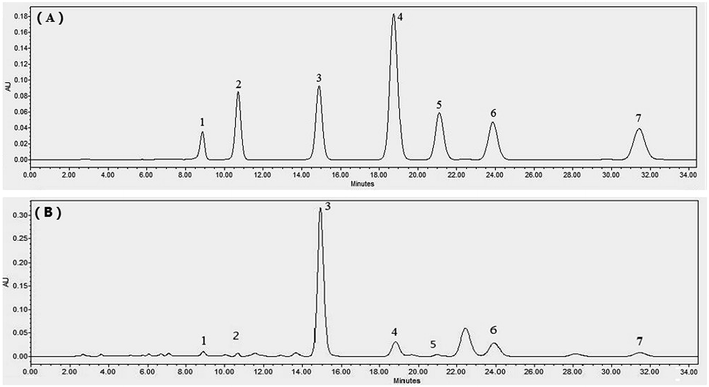 | ||
| Fig. 2 LC chromatograms of standards (A) and UAE extracts of C. pinnatifida leaves (B). 1. CA, 2. VG, 3. VR, 4. ORT, 5. RT, 6. VIT, 7. HYP. | ||
2.3. Method validation
Method validation was performed according to guidelines set by International Conference on Harmonisation (ICH (Q2), 2005) and International Union of Pure and Applied Chemistry (IUPAC, 2002). The method was validated in terms of linearity, sensitivity, selectivity, accuracy and precision.2.4. Extraction procedures
For UAE, 10 g of C. pinnatifida leave powders were mixed with 40% ethanol solution (150 mL) and extracted in an ultrasonic bath (KQ-250DB, Kunshan Ultrasonic Instrument Co., Ltd, China) at 40 °C for 30 min. After the extraction, the obtained solution was then centrifuged and filtered through a 0.22 μm nylon membrane for HPLC analysis. For ME, 10 g of C. pinnatifida leave powders were put into a conical flask with 40% ethanol solution (150 mL). The conical flask was then placed at room temperature for 12 h. The subsequent process was the same as UAE. For HRE, 10 g of C. pinnatifida leave powders were put into a round-bottomed flask with 40% ethanol solution (150 mL). The extraction was performed at 80 °C for 1 h. The subsequent process was the same as UAE.2.5. Experimental design
On the basis of single factor experimental results, a 24 factorial portion BBD combined with RSM was used to survey effects of the major factors including ratio of liquid/solid, extraction temperature (°C), ethanol concentration (%) and extraction time (min) on the total yields of CA, VG, VR, ORT, RT, VIT and HYP. A total of 29 randomized experiments including 24 factorial and 5 zero-point tests were performed. The regression analysis was carried out to evaluate the response function as a quadratic polynomial:where, Y is the predicted response; β0, βj, βjj and βij are the regression coefficients for intercept, linearity, square and interaction, respectively; Xi and Xj are the independent coded variables; and k represents the number of variables. The actual and coded levels of the independent variables used in the experimental design are summarized in Table 1.
| Run | Factors | Total yield (mg g−1) | |||
|---|---|---|---|---|---|
| X1 (RLS a) | X2 (Tb, °C) | X3 (ECc, %) | X4 (td, min) | ||
| a RLS is expressed as the ratio of liquid/solid (mL g−1).b T is expressed as the temperature (°C).c EC is expressed as the concentration of ethanol (%).d t is expressed as the time (min). | |||||
| 1 | −1 (10) | −1 (30) | 0 (40) | 0 (25) | 4.816 |
| 2 | 1 (20) | −1 (30) | 0 (40) | 0 (25) | 5.062 |
| 3 | −1 (10) | 1 (50) | 0 (40) | 0 (25) | 5.065 |
| 4 | 1 (20) | 1 (50) | 0 (40) | 0 (25) | 5.117 |
| 5 | 0 (15) | 0 (40) | −1 (30) | −1 (15) | 5.279 |
| 6 | 0 (15) | 0 (40) | 1 (50) | −1 (15) | 5.154 |
| 7 | 0 (15) | 0 (40) | −1 (30) | 1 (35) | 6.371 |
| 8 | 0 (15) | 0 (40) | 1 (50) | 1 (35) | 6.289 |
| 9 | −1 (10) | 0 (40) | 0 (40) | −1 (15) | 4.606 |
| 10 | 1 (20) | 0 (40) | 0 (40) | −1 (15) | 4.987 |
| 11 | −1 (10) | 0 (40) | 0 (40) | 1 (35) | 5.857 |
| 12 | 1 (20) | 0 (40) | 0 (40) | 1 (35) | 5.957 |
| 13 | 0 (15) | −1 (30) | −1 (30) | 0 (25) | 5.490 |
| 14 | 0 (15) | 1 (50) | −1 (30) | 0 (25) | 5.561 |
| 15 | 0 (15) | −1 (30) | 1 (50) | 0 (25) | 5.369 |
| 16 | −1 (10) | 0 (40) | −1 (30) | 0 (25) | 5.478 |
| 17 | 0 (15) | 1 (50) | 0 (40) | −1 (15) | 5.227 |
| 18 | 0 (15) | 1 (50) | 0 (40) | 1 (35) | 5.821 |
| 19 | 0 (15) | 0 (40) | 0 (40) | 0 (25) | 6.121 |
| 20 | 0 (15) | 0 (40) | 0 (40) | 0 (25) | 6.681 |
| 21 | 0 (15) | 0 (40) | 0 (40) | 0 (25) | 6.597 |
| 22 | 0 (15) | 0 (40) | 0 (40) | 0 (25) | 6.385 |
| 23 | −1 (10) | 0 (40) | 1 (50) | 0 (25) | 5.316 |
| 24 | 1 (20) | 0 (40) | −1 (30) | 0 (25) | 5.789 |
| 25 | 1 (20) | 0 (40) | 1 (50) | 0 (25) | 5.606 |
| 26 | 0 (15) | 1 (50) | 1 (50) | 0 (25) | 5.361 |
| 27 | 0 (15) | 0 (40) | 0 (40) | 0 (25) | 6.635 |
| 28 | 0 (15) | −1 (30) | 0 (40) | −1 (15) | 5.255 |
| 29 | 0 (15) | −1 (30) | 0 (40) | 1 (35) | 5.616 |
2.6. Antioxidant activities
Scavenging activities of C. pinnatifida leaf extracts towards DPPH, ABTS radicals scavenging and the reducing power assay were determined according to the previous reports.27 Antioxidant activities of samples in DPPH radicals scavenging and reducing power tests were reported as IC50 values. Antioxidant activities of samples in ABTS radicals scavenging test were expressed as mmol g−1 Trolox equivalent antioxidant capacity (TEAC).2.7. Statistical analysis
All experiments were conducted in triplicate. The experiment data were analyzed statistically with Design-Expert 8.0.6 (State-Ease, Inc., Minneapolis MN). Analysis of variance (ANOVA) was performed for calculations and modeling of optimal conditions for UAE. Values of P < 0.05 were regarded as significant.3. Results
3.1. Establishment of HPLC method
Selecting a proper HPLC method is of great importance to ensure the seven constituents detected precisely and achieve the goal of minimizing the interfere. HPLC C18 column without any detrimental effect on performance and without interfering peaks when analysing complex matrices. After a review of the literature, we tried gradient elution using (A) acetonitrile and (B) water (containing 0.1% formic acid) and the isocratic mobile phase, consisting of tetrahydrofuran/acetonitrile/0.05% phosphoric acid solution (20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3
3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7 v/v/v).28,29 After trial and error, the HPLC C18 column using THF–acetonitrile–water–phosphoric acid (175
7 v/v/v).28,29 After trial and error, the HPLC C18 column using THF–acetonitrile–water–phosphoric acid (175![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 31
31![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 794
794![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.385, v/v/v/v) system was effective for the separation of target compounds in C. pinnatifida leaves. Phosphoric acid (0.0385%) was used to improve the chromatographic behavior and to reduce the peak tailing. Moreover, 350 nm was chosen as the optimal detection wavelength for the simultaneous determination of seven phenolic compounds. Under this system, CA, VG, VR, ORT, RT, VIT and HYP had the retention times of 8.82, 10.69, 14.88, 18.59, 21.16, 23.89 and 31.55 min, respectively (Fig. 2).
0.385, v/v/v/v) system was effective for the separation of target compounds in C. pinnatifida leaves. Phosphoric acid (0.0385%) was used to improve the chromatographic behavior and to reduce the peak tailing. Moreover, 350 nm was chosen as the optimal detection wavelength for the simultaneous determination of seven phenolic compounds. Under this system, CA, VG, VR, ORT, RT, VIT and HYP had the retention times of 8.82, 10.69, 14.88, 18.59, 21.16, 23.89 and 31.55 min, respectively (Fig. 2).
The developed HPLC method was validated to verify that its performance was compatible with the required performance for the routine analysis of phenolic compounds. The results of the linearity, limits of quantification and detection, repeatability, precision, stability and recovery test of seven phenolic compounds are summarized in Table 2. Coefficients of correlation (R2 > 0.999) were exhibited for all the compounds within the range of tested concentrations. Table 2 summarizes the limit of detection (LOD) (S/N = 3) and the limit of quantification (LOQ) (S/N = 10) for the analytes were less than 4.34 and 19.3 μg mL−1, respectively. It indicates that the analytical method has excellent sensitivity.
| Compound | R2a | Linear range (μg mL−1) | LODb (μg mL−1) | LOQc (μg mL−1) | Precision (% RSDd) | Repeatability (% RSD) | Stability (% RSD) | Recoveryg (%) | |||
|---|---|---|---|---|---|---|---|---|---|---|---|
| RTe | PAf | RT | PA | RT | PA | ||||||
| a Coefficients of correlation.b Limit of detection, the lowest analyte concentration that produces a response detectable above the noise level of the system.c Limit of quantification, the lowest level of analyte that can be accurately and precisely measured.d Relative standard deviation, expressed as %.e Retention time.f Peak area.g Average of recoveries at two spike levels (high and low) (n = 6). | |||||||||||
| CA | 0.9999 | 19.0–304 | 4.34 | 19.0 | 0.41 | 0.54 | 0.47 | 0.62 | 0.61 | 0.82 | 99.75 |
| VG | 0.9996 | 15.6–250 | 3.57 | 15.6 | 0.66 | 0.32 | 0.52 | 0.33 | 0.87 | 0.26 | 99.74 |
| VR | 0.9999 | 15.6–250 | 2.38 | 15.6 | 0.98 | 0.59 | 0.92 | 0.37 | 0.97 | 0.98 | 99.43 |
| ORT | 0.9992 | 18.7–299 | 1.42 | 18.7 | 0.38 | 0.82 | 0.32 | 0.78 | 0.29 | 0.75 | 99.67 |
| RT | 0.9997 | 14.7–235 | 1.43 | 14.7 | 0.35 | 0.87 | 0.18 | 0.80 | 0.28 | 0.99 | 98.97 |
| VIT | 0.9999 | 18.8–301 | 3.34 | 18.8 | 0.25 | 0.57 | 0.33 | 0.42 | 0.14 | 0.67 | 98.68 |
| HYP | 0.9997 | 19.3–309 | 2.21 | 19.3 | 0.23 | 0.64 | 0.26 | 0.46 | 0.19 | 0.56 | 100.30 |
The precision variations of retention time and peak area were less than 0.98% and 0.87%, which indicates that the proposed method is accurate (Table 2). Good repeatability was obtained for the seven phenolic standards, the RSDs of the peak areas and retention times were 0.62 and 0.47% for CA, 0.33 and 0.52% for VG, 0.37 and 0.92% for VR, 0.78 and 0.32% for ORT, 0.80 and 0.18% for RT, 0.42 and 0.33% for VIT, 0.46 and 0.26% for HYP, respectively (Table 2). For the stability test, retention time and peak area of sample solution (ethanol extracts) were analyzed every 6 h within 48 h, and the sample solution was found to be rather stable within 48 h (RSD < 1.0%). Analyses recovery was determined by standard addition at three different concentrations. Recovery experiments were conducted in triplicate and evaluated. The average recoveries of CA, VG, VR, ORT, RT, VIT and HYP were 99.9%, 100.74%, 99.9%, 100.47%, 99.07%, 99.18% and 100.71% (n = 5), respectively (Table 2).
The above data indicated the proposed method provides good linearity, sensitivity, selectivity, accuracy as well as precision for the simultaneous analysis of seven phenolic compounds in C. Pinnatifida leaves. These results differed from the data reported by Shibata et al. (2004). In that report, although E. bicyclis was collected every four months and the quantification of the phlorotannins was performed with dried samples, no clear seasonal variation of the phlorotannins was observed.
3.2. Optimization of UAE conditions
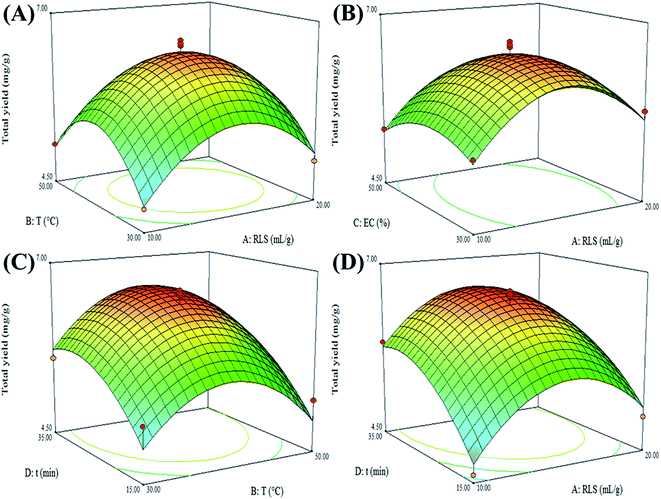
Based on results of preliminary studies (data not shown), four key parameters including ratio of liquid/solid, extraction temperature, ethanol concentration and extraction time were picked out for the investigation in this study. As results of single factor experiments shown in Fig. 3, ratio of liquid/solid (10–20), extraction temperature (30–50 °C), ethanol concentration (30–50%) and extraction time (15–35 min) were chosen for the further optimization by BBD combined with RSM. All the experiment data were shown in Table 1.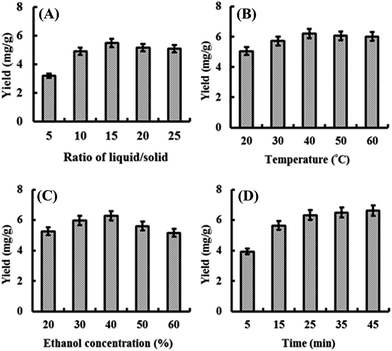 | ||
| Fig. 3 Effect of ratio of liquid/solid (A), extraction temperature (B), ethanol concentration (C) and extraction time (D) on the total yields (mg g−1) of seven phenolic compounds. | ||
| Y = 6.48 + 0.11X1 + 0.045X2 − 0.073X3 + 0.45X4 − 0.049X1X2 − 0.005X1X3 − 0.070X1X4 − 0.020X2X3 + 0.058X2X4 + 0.011X3X4 − 0.72X12 − 0.71X22 − 0.29X32 − 0.37X42 |
| Source | Sum of squaresa | DFb | Mean squarec | F valued | P-value prob. > Fe | Significancef |
|---|---|---|---|---|---|---|
| a Sum of the squared differences between the average values and the overall mean.b Degrees of freedom.c Sum of squares divided by DF.d Test for comparing term variance with residual variance.e Probability of seeing the observed F-value if the null hypothecs is true.f P-value less than 0.05 indicate model term is significant.g Consists of terms used to estimate experimental error.h Variation of the data around the fitted model. | ||||||
| Model | 8.45 | 14 | 0.60 | 14.59 | <0.0001 | Significant |
| X1 | 0.16 | 1 | 0.16 | 3.84 | 0.0704 | Significant |
| X2 | 0.025 | 1 | 0.025 | 0.60 | 0.4529 | Not significant |
| X3 | 0.064 | 1 | 0.064 | 1.53 | 0.2357 | Not significant |
| X4 | 2.43 | 1 | 2.43 | 58.79 | <0.0001 | Significant |
| X1X2 | 9.409 × 10−3 | 1 | 9.409 × 10−3 | 0.23 | 0.6408 | Not significant |
| X1X3 | 1.102 × 10−4 | 1 | 1.102 × 10−4 | 2.664 × 10−3 | 0.9596 | Not significant |
| X1X4 | 0.020 | 1 | 0.020 | 0.48 | 0.5010 | Not significant |
| X2X3 | 1.560 × 10−3 | 1 | 1.560 × 10−3 | 0.038 | 0.8488 | Not significant |
| X2X4 | 0.014 | 1 | 0.014 | 0.33 | 0.5759 | Not significant |
| X3X4 | 4.622 × 10−4 | 1 | 4.622 × 10−4 | 0.011 | 0.9173 | Not significant |
| X12 | 3.37 | 1 | 3.37 | 81.33 | <0.0001 | Significant |
| X22 | 3.24 | 1 | 3.24 | 78.42 | <0.0001 | Significant |
| X32 | 0.56 | 1 | 0.56 | 13.59 | 0.0024 | Significant |
| X42 | 0.91 | 1 | 0.91 | 22.03 | 0.0003 | Significant |
| Residualg | 0.58 | 14 | 0.041 | Significant | ||
| Lack of fith | 0.36 | 10 | 0.036 | 0.67 | 0.7220 | Not significant |
| R2 | 0.9358 | |||||
3.3. Comparison of different extraction procedures
ME, HRE and UAE methods were compared for their performances of extracting seven phenolic compounds from C. pinnatifida leaves under the optimized conditions. The extraction yields of CA, VG, VR, ORT, RT, VIT and HYP are summarized in Table 4. Overall, the total extraction yields of seven phenolic compounds using UAE were close to ME but higher than HRE which need a long-term extraction (4 h) at high extraction temperature (80 °C). The possible reason was that VG and VR were more susceptible to high extraction temperature and they would be degraded under the condition of long-term heating. In addition, although ME method possessed the comparable yield as against UAE, it cost much more time (ME 12 h) than UAE (30 min). Moreover, the ultrasonic cavitation of UAE could create strong forces that could break cell walls mechanically, which allow high extraction yield under moderate conditions. Consequently, it was concluded that the UAE method is an alternative technique for phenolic compound extraction from C. pinnatifida leaves, which could offer shorter time, lower extraction temperature and lower energy input.| Yield (mg g−1) | Method | ||
|---|---|---|---|
| UAE | HRE | ME | |
| a * and ** denote significant (P < 0.05) and very significant (P < 0.01), respectively. | |||
| CA | 0.058 ± 0.003 | 0.037 ± 0.007 | 0.055 ± 0.013 |
| VG | 0.22 ± 0.007 | 0.15 ± 0.011 | 0.20 ± 0.005 |
| VR | 4.09 ± 0.005** | 2.75 ± 0.015 | 3.03 ± 0.008 |
| ORT | 0.023 ± 0.006 | 0.019 ± 0.008 | 0.017 ± 0.014 |
| RT | 0.086 ± 0.011 | 0.072 ± 0.013 | 0.080 ± 0.003 |
| VIT | 0.90 ± 0.010* | 0.30 ± 0.003 | 0.88 ± 0.012 |
| HYP | 0.64 ± 0.013 | 0.61 ± 0.006 | 0.60 ± 0.009 |
3.4. Antioxidant activities
It is known that phenolic compounds are hydroxylated derivatives of benzoic and cinnamic acids and contribute to overall antioxidant activity in the plants. Many reports have showed that phenolic compounds, especially flavonoids exhibit extensive free radical scavenging activities through their reactivity as hydrogen or electron-donating agents, and metal ion chelating property.41,42 It is reported that CA, VG, VR, ORT, RT, VIT and HYP exhibit important contributions to the antioxidant activities of extracts from several medical plants. From this point of view, C. pinnatifida leaf extracts may be the potential antioxidant ingredients or additives in food industry due to their high contents of these phenolic compounds. Hence, the antioxidant activities of extracts from C. pinnatifida leaves were comprehensively evaluated by several in vitro antioxidant methods including DPPH radical scavenging, ABTS radical scavenging and reducing power tests. Results are shown in Table 5.| Analyte | DPPH test IC50 (mg mL−1) | ABTS test (mmoL g−1 Trolox) | Reducing power test IC50 (mg mL−1) |
|---|---|---|---|
| a * Denote significant (P < 0.05). | |||
| UAE | 0.69 ± 0.003* | 0.86 ± 0.001* | 0.24 ± 0.013* |
| HRE | 2.34 ± 0.011 | 0.20 ± 0.004 | 0.89 ± 0.011 |
| ME | 1.04 ± 0.007 | 0.28 ± 0.002 | 0.43 ± 0.015 |
| VC | 0.074 ± 0.015 | 1.021 ± 0.014 | — |
| BHT | — | — | 0.125 ± 0.005 |
Obviously, UAE extracts exhibited notable DPPH radical scavenging activity with an IC50 value (0.69 mg mL−1), which was superior to those of HRE (2.34 mg mL−1) and ME (1.04 mg mL−1). Moreover, the antioxidant activities of UAE extracts with an IC50 of 0.24 mg mL−1 in the reducing power test was stronger than those of HRE (0.89 mg mL−1) and ME (0.43 mg mL−1). Furthermore, ABTS radical scavenging activity values of UAE extracts was 0.86 mmol g−1 Trolox, which was higher than those for HRE (0.20 mmol g−1 Trolox) and ME (0.28 mmol g−1 Trolox). These results indicated that UAE extracts had better antioxidant activity as compared to ME and HRE, which may due to their higher levels of total phenolic compounds. Overall, the antioxidant activity screening results are indicative of C. pinnatifida leaf extracts as potential antioxidant ingredients or additives in food and pharmaceutical industries.
4. Conclusion
In this work, a simple and accurate HPLC method was established for the simultaneous determination of CA, VG, VR, ORT, RT, VIT and HYP from C. pinnatifida leaves. The extraction conditions were optimized by BBD combined with RSM as follows: extraction temperature 41 °C, extraction time 31 min, ratio of liquid/solid 15, and ethanol concentration 39%. The optimal yields of CA, VG, VR, ORT, RT, VIT and HYP were 0.46, 0.38, 4.37, 0.033, 0.036, 0.30 and 1.19, mg g−1, respectively. In comparison of conventional ME and HRE methods, UAE offered higher extraction yields of seven phenolic compounds. Moreover, UAE extracts exhibited better antioxidant activities than those of other methods. Overall, we can conclude that UAE is an efficient and alternative method for the extraction of phenolic compounds form C. pinnatifida leaves and its extracts can be considered as a kind of potential health-promoting ingredients or additives with the antioxidant activity for the pharmaceutical and healthy food industry.Abbreviations
| CA | Chlorogenic acid |
| VG | Vitexin-4′′-o-glucoside |
| VR | Vitexin-2′′-o-rhamnoside |
| ORT | Orientoside |
| RT | Rutin |
| VIT | Vitexin |
| HYP | Hyperoside |
| BBD | Box–Behnken design |
| RSM | Response surface methodology |
| HPLC | High pressure liquid chromatography |
| UAE | Ultrasound-assisted extraction |
| ME | Maceration extraction |
| HRE | Heat-refluxing extraction |
| C. pinnatifida | Crataegus pinnatifida |
| RLS | Ratio of liquid/solid |
| T | Extraction temperature |
| EC | Ethanol concentration |
| t | Extraction time |
Acknowledgements
The authors gratefully acknowledge the financial supports by Fundamental Research Funds for the Central Universities (2572014CA08, 2572014EY0103), the Assisted Project by Heilong Jiang Postdoctoral Funds for Scientific Research Initiation (LBH-Q13003) and Youth Fund of National Natural Science Foundation of China (21403032).References
- The State Pharmacopoeia Commission of PR China, The Pharmacopoeia of People's Republic of China, Chemical Industry Publishing House, Beijing, 2010, p. 30 Search PubMed.
- T. X. Liu, Y. N. Cao and M. M. Zhao, Food Chem., 2010, 119, 1656–1662 CrossRef CAS PubMed.
- P. Z. Liu, B. R. Yang and H. Kallio, Food Chem., 2010, 121, 1188–1197 CrossRef CAS PubMed.
- E. S. Kao, C. J. Wang, W. L. Lin, Y. F. Yin, C. P. Wang and T. H. Tseng, J. Agric. Food Chem., 2005, 53, 430–436 CrossRef CAS PubMed.
- T. Cui, J. Z. Li, H. Kayahara, L. Ma, L. X. Wu and K. Nakamura, J. Agric. Food Chem., 2006, 54, 4574–4581 CrossRef CAS PubMed.
- Q. Chang, M. Zhu, Z. Zou, M. Chow and W. K. K. Ho, J. Chromatogr. B: Biomed. Sci. Appl., 2001, 760, 227–235 CrossRef CAS.
- P. Y. Gao, L. Z. Li, Y. Peng, F. F. Li, C. Niu, X. X. Huang, M. Ming and S. J. Song, Biochem. Syst. Ecol., 2010, 38, 988–992 CrossRef CAS PubMed.
- S. J. Song, L. Z. Li, P. Y. Gao, Y. Peng, J. Y. Yang and C. F. Wu, Food Chem., 2011, 129, 933–939 CrossRef CAS PubMed.
- T. Bahorun, E. Aumjaud, H. Ramphul, M. Rycha, A. Luximon-Ramma, F. Trotin and O. I. Aruoma, Nahrung, 2003, 47, 191–198 CrossRef CAS PubMed.
- J. P. Brown, Mutat. Res., Rev. Genet. Toxicol., 1980, 75, 243–277 CrossRef CAS.
- A. Kirakosyan, E. Seymour, P. B. Kaufman, S. Warber, S. Bolling and S. C. Chang, J. Agric. Food Chem., 2003, 51, 3973–3976 CrossRef CAS PubMed.
- W. H. Frishman, P. Beravol and C. Carosella, Dis.-Mon., 2009, 55, 121–192 CrossRef PubMed.
- J. Xu, L. Wu, W. Chen and A. C. Chang, J. Chromatogr. A, 2008, 1202, 189–195 CrossRef CAS PubMed.
- M. Yao, H. E. Ritchie and P. D. Brown-Woodman, J. Ethnopharmacol., 2008, 118, 127–132 CrossRef PubMed.
- X. P. Ding, X. T. Wang, L. L. Chen, J. Qi, T. Xu and B. Y. Yu, Food Chem., 2010, 120, 929–933 CrossRef CAS PubMed.
- S. Cheng, F. Qiu, J. Huang and J. Q. He, J. Sep. Sci., 2007, 30, 717–721 CrossRef CAS PubMed.
- W. Q. Li, Q. P. Hu and J. G. Xu, Food Chem., 2015, 175, 50–56 CrossRef CAS PubMed.
- X. X. Ying, R. X. Wang, J. Xu, W. J. Zhang, H. B. Li, C. S. Zhang and F. M. Li, J. Chromatogr. Sci., 2009, 43, 201–205 Search PubMed.
- J. Q. Liao, B. D. Qu and B. G. Xu, Anal. Methods, 2015, 7, 336–341 RSC.
- B. Mandana, A. R. Russly, S. T. Farah, G. Ali, M. S. Liza, S. Jinap, H. Azizah and I. S. M. Zaidul, Food Bioprod. Process., 2011, 89, 67–72 CrossRef PubMed.
- L. F. Zhang and Z. L. Liu, Ultrason. Sonochem., 2008, 15, 731–737 CrossRef CAS PubMed.
- W. Huang, A. Xue, H. Niu, Z. Jia and J. W. Wang, Food Chem., 2009, 114, 1147–1154 CrossRef CAS PubMed.
- H. F. Zhang, X. H. Yang, L. D. Zhao and Y. Wang, Innovative Food Sci. Emerging Technol., 2009, 10, 54–60 CrossRef CAS PubMed.
- M. Chaharlangi, H. Parastar and A. Malekpour, RSC Adv., 2015, 5, 26246–26254 RSC.
- A. K. Shriwas and P. R. Gogate, Sep. Purif. Technol., 2011, 79, 1–7 CrossRef CAS PubMed.
- J. Wang, B. G. Sun, Y. P. Cao, Y. X. Tian and H. Li, Food Chem., 2008, 106, 804–810 CrossRef CAS PubMed.
- N. Wu, K. Fu, Y. J. Fu, Y. G. Zu, F. R. Chang, Y. H. Chen, X. L. Liu, Y. Kong, W. Liu and C. B. Gu, Molecules, 2009, 14, 1032–1043 CrossRef CAS PubMed.
- G. Ma, X. H. Jiang, Z. Chen, J. Ren, C. R. Li and T. M. Liu, J. Pharm. Biomed. Anal., 2007, 44, 243–249 CrossRef CAS PubMed.
- C. H. Wang, Y. X. Wang and H. J. Liu, J. Pharm. Anal., 2011, 1, 291–296 CrossRef CAS PubMed.
- J. E. Cacace and G. Mazza, J. Food Eng., 2003, 59, 379–389 CrossRef.
- M. González and R. Lobo, Sep. Purif. Technol., 2010, 71, 347–355 CrossRef PubMed.
- E. Dorta, M. G. Lobo and M. González, LWT–Food Sci. Technol., 2012, 45, 261–268 CrossRef CAS PubMed.
- C. Liyana-Pathirana and F. Shahidi, Food Chem., 2005, 93, 47–56 CrossRef CAS PubMed.
- E. M. Silva, H. Rogez and Y. Larondelle, Sep. Purif. Technol., 2007, 55, 381–387 CrossRef CAS PubMed.
- S. Sahin and R. Samli, Ultrason. Sonochem., 2013, 20, 595–602 CrossRef CAS PubMed.
- J. N. He, H. T. Yan and C. L. Fan, RSC Adv., 2014, 4, 42608–42616 RSC.
- G. Y. Pan, G. Y. Yu, C. H. Zhu and J. L. Qiao, Ultrason. Sonochem., 2012, 19, 486–490 CrossRef CAS PubMed.
- D. B. Muñiz-Márquez, G. C. Martínez-Ávila, J. E. Wong-Paz, R. Belmares-Cerda, R. Rodríguez-Herrera and C. N. Aguilar, Ultrason. Sonochem., 2013, 20, 1149–1154 CrossRef PubMed.
- Y. Wen, H. G. Chen, X. Zhou, Q. F. Deng, Y. Zhao, C. Zhao and X. J. Gong, RSC Adv., 2015, 5, 19686–19695 RSC.
- R. Chirinos, H. Rogez, D. Campos, R. Pedreschi and Y. Larondelle, Sep. Purif. Technol., 2007, 55, 217–225 CrossRef CAS PubMed.
- N. Martins, L. Barros, M. Dueñas, C. Santos-Buelga and I. C. F. R. Ferreira, RSC Adv., 2015, 5, 26991–26997 RSC.
- J. Montes-Avila, F. Delgado-Vargas, S. P. Díaz-Camacho and G. A. Rivero, RSC Adv., 2012, 2, 1827–1834 RSC.
| This journal is © The Royal Society of Chemistry 2015 |