Molecularly imprinted cryogel cartridges for the specific filtration and rapid separation of interferon alpha†
Mehmet Emin Çormanab,
Canan Armutcub,
Serpil Özkara*c,
Lokman Uzun*b and
Adil Denizlib
aSinop University, Department of Bioengineering, Sinop, Turkey
bHacettepe University, Department of Chemistry, Biochemistry Division, Beytepe, Ankara, Turkey. E-mail: lokman@hacettepe.edu.tr; Fax: +90 312 2992163; Tel: +90 312 297 7337
cAnadolu University, Department of Chemistry, Biochemistry Division, Eskişehir, Turkey. E-mail: soyavuz@anadolu.edu.tr; Tel: +90 222 335 0580
First published on 13th May 2015
Abstract
In this study, we synthesised specific filtration cartridges with selective recognition sites for target molecules and used them to separate interferon α-2b from aqueous solutions. We combined molecular imprinting technology with cryogel to achieve specific and rapid filtration of interferon α-2b through the macroporous structure of a cryogel network. Recombinant interferon α-2b-imprinted poly(2-hydroxyethyl methacrylate-N-methacryloyl-L-tryptophan) P(HEMATrp)/α-2bIFN cryogels were synthesised via free-radical bulk polymerisation under partially frozen conditions. The interferon α-2b filtration conditions were subsequently optimised with respect to factors such as pH, initial concentration, temperature, centrifugation speed, salt concentration and type and the amount of precomplex incorporated. Selectivity experiments were conducted in respect to isoelectric point as well as size of the competitor proteins under both uncompetitive and competitive conditions. The relative selectivity coefficients of the specific filtration cartridge in respect to isoelectronic points for interferon/IgG, interferon/HSA and interferon/insulin pairs were 3.72, 7.10 and 10.68 times greater than the coefficient of a non-imprinted [P(HEMATrp)] filtration cartridge, respectively. Similarly, the relative selectivity coefficients of the specific filtration cartridge in respect to competitor size for interferon/lysozyme, interferon/myoglobin and interferon/carbonic anhydrase pairs were calculated as 7.58, 10.40 and 11.68 under uncompetitive conditions whereas those values under competitive conditions were calculated as 1.08, 1.05 and 1.34, respectively. The results indicated that specific filtration cartridges developed could repeatedly adsorb interferon α-2b with a short separation time without any significant decrease in the adsorption capacity even if competitive conditions were conducted.
1. Introduction
Molecularly imprinted polymers (MIPs) are fabricated with a specific recognition region for a target molecule. Three-dimensional recognition sites can be formed by the polymerisation of functional monomers with a cross-linking agent in the presence of a target molecule; the removal of target molecules from the polymer results in the cavities selectively rebinding the target molecule.1,2 Therefore, MIPs exhibit selective high binding abilities with respect to the shape, size and chemical properties of the target molecules via the proper orientation of three-dimensional interaction points in their polymeric matrix.3 MIPs have some significant advantages, including molecular recognition ability, high selectivity, easy preparation, relatively low cost, durability and good mechanical and thermal stability.4,5 MIPs have been successfully used in analytic applications, including HPLC, GC, CEC and biosensors for recognising elements with diverse purposes, i.e. separation, purification and detection.2,6Biomolecules play significant roles in food, biological, chemical and medical industries. Numerous methods that involve filtration cartridges, such as liquid chromatography, capillary electrophoresis and membrane-based applications, have been developed for the separation and purification of biomolecules after combining with MIPs.7–9 In recent years, specific filtration-cartridge-based MIPs have become a promising method among separation and purification processes.7,10 Although the essential basis of separation via a filtration cartridge is molecular weight, secondary factors such as molecular shape, charge and electrostatic properties can also play important roles during separation processes.11,12 Specific filtration provides several important advantages, including high flux, precise selectivity, low fouling and good stability.7,13–15 Specific filtration cartridges are typically easy to reuse and run. In addition to these advantages, specific filtration consumes low amounts of energy; thus, it is a more effective method than other techniques used for the same purpose.16,17
Interferons are cytokines made by viral infected cells. They are very significant biological proteins because they exhibit antiviral, cytotoxic, immunomodulatory, antiproliferative and antitumor activities and also enhance the expression of the major histocompatibility antigens.18,19 Interferons are separated into three different major types according to their structural, biochemical and antigenic properties. First, alpha interferon (α-IFN) is mainly produced by monocytes and B-lymphocytes. α-IFN is usually used in the treatment of hairy-cell leukaemia, hepatitis C, chronic hepatitis B and different types of cancer.20 Second, beta interferon (β-IFN) is produced by fibroblasts after stimulation and by epithelium cells. β-IFN is used to treat multiple sclerosis. Third, gamma interferon (γ-IFN) is produced by T-lymphocytes and differs from α-IFN and β-IFN. Interferons represent an ideal treatment for numerous diseases, including hepatitis, cancers and tumours; thus, extensive efforts have been directed towards the purification of interferons. Human interferons have long been purified using techniques such as electrophoresis, cation–anion exchange and various chromatographic methods.21 As an alternative to these techniques, we present here a molecular-imprinting-based specific filtration cartridge for the purification of interferons from aqueous solutions. This approach provides high selectivity for the target molecules and allows a rapid separation process.
In this study, recombinant interferon α-2b-imprinted poly(2-hydroxyethyl methacrylate-N-methacryloyl-L-tryptophan) P(HEMATrp)/α-2bIFN specific filtration cartridges were prepared via the molecular imprinting technique. This study was composed of three main parts: first, N-methacryloyl-L-tryptophan (MATrp) was synthesised as a functional monomer. Second, P(HEMATrp)/α-2bIFN specific filtration cartridges were synthesised through free-radical polymerisation under partially frozen conditions. The MATrp monomer imparted hydrophobic character to the P(HEMATrp)/α-2bIFN cartridges due to well-established information about hydrophobic nature of interferons.21–24 Third, the P(HEMATrp)/α-2bIFN specific filtration cartridges were characterised by Fourier-transform infrared spectrophotometry (FTIR), scanning electron microscopy (SEM) and swelling tests and by recombinant interferon α-2b adsorption studies using different amounts of recombinant interferon α-2b at different pH values, centrifugation speeds, ionic strengths and temperatures. After recombinant interferon α-2b adsorption from aqueous solutions was optimised, selectivity experiments were designed in four different ways by using insulin, immunoglobulin G (IgG) and human serum albumin (HSA) as competitors for investigating isoelectric points whereas lysozyme, myoglobin and carbonic anhydrase were chosen as competitors for investigating size effect and complexity of the media (competitive or uncompetitive conditions). Subsequently, this combined technique provides highly selective and efficient purification of interferon.
2. Experimental
2.1. Materials
For the preparation of specific filtration cartridges, 2-hydroxyethyl methacrylate (HEMA), N,N′-methylenebis(acrylamide) (MBAAm), ammonium persulphate (APS) and N,N,N′,N'-tetramethylethylenediamine (TEMED), immunoglobulin G (IgG), human serum albumin (HSA), lysozyme (chicken egg white), myoglobin (equine skeletal muscle, 95–100%), carbonic anhydrase (bovine erythrocyte), L-tryptophan and methacryloyl chloride were obtained from Sigma Chemical Co. (St. Louis, MO, USA). Recombinant interferon α-2b (INTRON® A) was purchased from Merck & Co., Inc., USA whereas human insulin were acquired Novo Nordisk A/S (Bagsvaerd, Denmark). In this study, deionized water purified by Barnstead (Dubuque, IA, USA) ROpure LP® reverse osmosis unit was used.2.2. Synthesis of N-methacryloyl-L-tryptophan (MATrp)
MATrp, a polymerisable derivative of hydrophobic amino acid, tryptophan was synthesised according to the following experimental procedure25 in order to insert hydrophobicity through polymeric backbone. Briefly, L-tryptophan (5.0 g) and sodium nitrite (NaNO2; 0.2 g) were dissolved in potassium carbonate solution [K2CO3; 30 mL, 5% (w/v)]. The reaction mixture was cooled to 0 °C in an ice-water bath. Methacryloyl chloride (4.0 mL) was slowly added to the reaction mixture under nitrogen atmosphere, and the mixture was stirred using a magnetic stirrer at room temperature for 2 h. After the reaction was completed, the pH of the solution was adjusted to pH 7.0 and the solution was extracted with ethyl acetate. After the aqueous phase was evaporated using a rotary evaporator, the product, MATrp was recrystallised from an ether–cyclohexane mixture.2.3. Preparation of P(HEMATrp)/α-2bIFN filtration cartridges
To prepare P(HEMATrp)/α-2bIFN specific filtration cartridges, interferon α-2b (template) and MATrp monomer were first dissolved in 10.0 mL of deionised water; the ratio was tuned to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
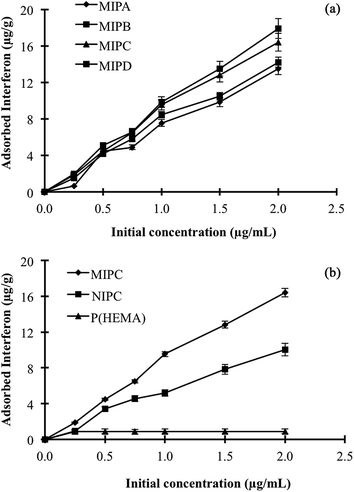
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25 (molar basis), and the obtained solution was stirred with a magnetic stirrer at room temperature for 3 h to form a complex (MATrp–α-2bIFN) between interferon α-2b and the MATrp monomer. Second, P(HEMATrp)/α-2bIFN specific filtration cartridges were prepared in the presence of the MATrp–α-2bIFN complex. Polymerisation syrup was prepared by dissolving HEMA (basic monomer, 1.3 mL), N,N′-methylenebis(acrylamide) (MBAAm, crosslinker, 0.283 g) in deionised water. The final monomer solution was adjusted to 12% (w/v) with respect to the total monomer concentration. This solution was cooled in an ice bath for 20 min before the initiators were added. The specific filtration cartridges were produced by free-radical polymerisation initiated by the redox initiator couple of TEMED (25 μL) and APS (20 mg). The polymerisation solutions were immediately poured into plastic syringes (2 mL) with closed outlets at the bottom and were frozen at −12 °C for 24 h. After the polymerisation was complete, the P(HEMATrp)/α-2bIFN specific filtration cartridges were thawed at room temperature. The specific filtration cartridges were then washed several times with deionised water in order to remove any unreacted monomer. After being cleaned, the specific filtration cartridges were stored in buffer containing 0.02% sodium azide at 4 °C until use. To examine the amount of imprinted cavities, different amounts of MATrp–α-2bIFN complex in the range 1.0–4.0 mL were included in the recipe (Table 1). The P(HEMATrp)/α-2bIFN specific filtration cartridges were designated as MIPA, MIPB, MIPC and MIPD in accordance with the volume of complex used (1, 2, 3 and 4 mL, respectively). The non-imprinted versions of cryogel cartridges [P(HEMATrp) and P(HEMA)] were also prepared using the same polymerisation reaction with/without functional monomer MATrp instead of the pre-polymerisation complex.
25 (molar basis), and the obtained solution was stirred with a magnetic stirrer at room temperature for 3 h to form a complex (MATrp–α-2bIFN) between interferon α-2b and the MATrp monomer. Second, P(HEMATrp)/α-2bIFN specific filtration cartridges were prepared in the presence of the MATrp–α-2bIFN complex. Polymerisation syrup was prepared by dissolving HEMA (basic monomer, 1.3 mL), N,N′-methylenebis(acrylamide) (MBAAm, crosslinker, 0.283 g) in deionised water. The final monomer solution was adjusted to 12% (w/v) with respect to the total monomer concentration. This solution was cooled in an ice bath for 20 min before the initiators were added. The specific filtration cartridges were produced by free-radical polymerisation initiated by the redox initiator couple of TEMED (25 μL) and APS (20 mg). The polymerisation solutions were immediately poured into plastic syringes (2 mL) with closed outlets at the bottom and were frozen at −12 °C for 24 h. After the polymerisation was complete, the P(HEMATrp)/α-2bIFN specific filtration cartridges were thawed at room temperature. The specific filtration cartridges were then washed several times with deionised water in order to remove any unreacted monomer. After being cleaned, the specific filtration cartridges were stored in buffer containing 0.02% sodium azide at 4 °C until use. To examine the amount of imprinted cavities, different amounts of MATrp–α-2bIFN complex in the range 1.0–4.0 mL were included in the recipe (Table 1). The P(HEMATrp)/α-2bIFN specific filtration cartridges were designated as MIPA, MIPB, MIPC and MIPD in accordance with the volume of complex used (1, 2, 3 and 4 mL, respectively). The non-imprinted versions of cryogel cartridges [P(HEMATrp) and P(HEMA)] were also prepared using the same polymerisation reaction with/without functional monomer MATrp instead of the pre-polymerisation complex.
| Polymer codes | HEMA (mL) | Complex (mL) | Monomer (mL) | MBAAm (g) | Water (mL) |
|---|---|---|---|---|---|
| a APS: 20 mg; TEMED: 25 μL; temperature/duration: −12 °C/24 h. | |||||
| P(HEMA) | 1.3 | — | — | 0.283 | 13.7 |
| NIPC | 1.3 | — | 3 | 0.283 | 10.7 |
| MIPA | 1.3 | 1 | — | 0.283 | 12.7 |
| MIPB | 1.3 | 2 | — | 0.283 | 11.7 |
| MIPC | 1.3 | 3 | — | 0.283 | 10.7 |
| MIPD | 1.3 | 4 | — | 0.283 | 9.7 |
2.4. Removal of the template molecule
A solution of 1.0 M ethylene glycol was used to remove the template molecules from P(HEMATrp)/α-2bIFN specific filtration cartridges; this procedure was performed at room temperature for 72 h. The specific filtration cartridges were treated with this solution until no interferon α-2b was detected in the washing solution. The removed interferon α-2b was monitored by UV spectrophotometry; the amount of interferon α-2b extracted from the specific filtration cartridges was quantified on the basis of it absorbance at 280 nm.2.5. Characterisation of the specific filtration cartridges
To characterise these cryogel cartridges, swelling tests of P(HEMA), NIPC and MIPC filtration cartridges were performed in distilled water and their water-uptake ratios were calculated. In these experiments, the specific filtration cartridges were first dried at room temperature and weighed (W0). The cartridges were subsequently placed in a 50 mL beaker containing distilled water (10 mL) for 2 h. After the cartridges reached an equilibrium swollen state, they were removed from the water, wiped using a piece of filter paper and reweighed (WS). The water content uptaken by specific filtration cartridges was determined by eqn (1):
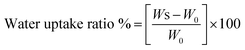 | (1) |
The surface morphologies of the specific filtration cartridges were examined using SEM. Initially, cartridges were dried in air at room temperature before analysis. A fragment from each of the specific filtration cartridges was mounted onto an SEM sample mount and subsequently sputter-coated with gold for 2 min. These samples were then loaded into a scanning electron microscope (JEOL JEM1200EX, Tokyo, Japan), and the sample surfaces were scanned at the desired magnification to display the physical appearance of the surfaces of the specific filtration cartridges. The characteristic functional groups of the P(HEMA), NIPC and MIPC specific filtration cartridges were analysed using a Fourier-transform infrared spectrophotometer (Shimadzu 8000 Series, Japan). After the specific filtration cartridges were dried under ambient conditions, the samples were crushed into free particles (2 mg) using a mortar; the particles were then mixed with KBr (IR Grade, 98 mg) and pressed into pellets. The FTIR spectra were recorded over wavenumber ranging from 4000 to 400 cm−1.
2.6. Adsorption studies
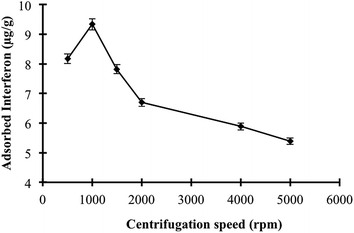
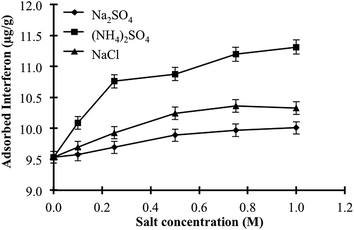
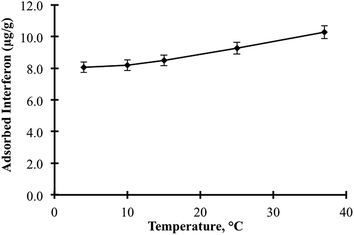
Interferon α-2b adsorption studies were performed using the P(HEMA), NIPC and P(HEMATrp)/α-2bIFN [MIP (A, B, C, D)] specific filtration cartridges (Scheme 1). The studies related to the optimisation of adsorption comprised three main parts. First specific filtration cartridges were equilibrated with 2.5 mL of proper buffer solution (acetate or phosphate buffer) for 10 min; these equilibrated solutions were then filtered through specific filtration cartridges within 10 min using centrifugation speeds between 500 and 5000 rpm. Second, interferon α-2b aqueous solutions were added to cartridges; the solutions and cartridges were then allowed to interact for 10 min. After adsorption, the specific filtration cartridges were filtered using the same procedure. Third, a desorbing solution (1.0 M ethylene glycol, 2.5 mL) was interacted with the specific filtration cartridges for 10 min; the cartridges were then washed out with water to prepare them for the next adsorption study. Numerous factors that affect adsorption, including the pH, interferon α-2b concentration, centrifugation speed and temperature, were investigated. In addition, the effects of the salt type and ionic strength on the interferon α-2b adsorption were investigated using the specific filtration cartridges because the salt type [NaCl, Na2SO4, (NH4)2SO4] and ionic strength are significant factors that govern hydrophobic interactions. All experiments (n = 3) and measurements (n = 3) were performed in triplicate.2.7. Selectivity studies
After optimisation of recombinant interferon α-2b adsorption from aqueous solutions, selectivity experiments were designed in four different ways by using insulin, immunoglobulin G (IgG) and human serum albumin (HSA), lysozyme, myoglobin and carbonic anhydrase as competitors. Firstly, we performed selectivity experiments with insulin, IgG and HSA due to their abundance and presence probability with interferon molecules in same media. Herein, we conducted selectivity experiments through two different approaches, normal proteins and dye-attached proteins. After that, we performed the selectivity experiments with lysozyme, myoglobin and carbonic anhydrase to assess the effect of the size on the recognition ability under both competitive and uncompetitive conditions.Actually, selectivity studies were performed for samples in aqueous solutions using the previously described procedure. The interferon α-2b adsorption capacity and selectivity coefficients for the NIPC and MIPC specific filtration cartridges were investigated from singular aqueous biomolecule solutions; the biomolecules were quantified via their absorbance at 280 nm, as measured using a UV spectrophotometer (UV-1601, Shimadzu, Tokyo, Japan).
The distribution coefficients (Kd) of interferon α-2b with respect to insulin, IgG and HSA were calculated using eqn (2):
| Kd = [(Ci − Cf)/Cf] × V/m | (2) |
The selectivity (k) and relative selectivity (k′) coefficients of the cartridges, which represent their binding tendency through interferon α-2b with respect to competitors (insulin, IgG and HSA), were calculated as follows:
| k = Kd,template protein/Kd,competitor | (3) |
| k′ = kimprinted/knon-imprinted | (4) |
Separately, in order to visualise the selectivity of specific filtration cartridge, we performed a set of experiment by using dye-modified protein samples as competing analytes from aqueous solutions as mentioned before. We have chosen different reactive dyes, reactive red 120, alkali blue 6B, reactive green HE4BD, and congo red for each protein molecule of four competitors, interferon, insulin, IgG, and HSA, respectively. Prior to attach dye molecules to proteins, we separately prepared the protein (in the concentration range of 0.25–4.0 μg mL−1) and dye solutions (250 mg L−1) at pH 10.0 (100 mM, carbonate buffer). Then, we mixed the solutions together in volume ratio as 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (dye
1 (dye![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) protein) and allowed to interact at 125 rpm for 2 h. After that, we dialysed the dye molecules unreacted overnight by using dialysis membrane (Serva GmbH, Heidelberg, Germany, with cutoff 12 kDa). We determined the proper wavelength for each dye by scanning wavelength in the range of 280–700 nm by using UV-vis spectrophotometer (Shimadzu UV-1601, Tokyo, Japan). Then, the solutions including dye attached protein molecules were interacted with MIPC and NIPC specific filtration cartridges as given as before. The quantification was achieved by applying proper mass balance in respect to data obtained from spectrophotometer at wavelengths 697 nm, 679 nm, 659 nm, and 677 nm for reactive red 120-interferon, alkali blue 6B-insulin, reactive green HE4BD-IgG, congo red-albumin, respectively. Finally, we also calculated distribution, selectivity and relative selectivity constants for MIPC and NIPC specific filtration cartridges in accordance with the data obtained.
protein) and allowed to interact at 125 rpm for 2 h. After that, we dialysed the dye molecules unreacted overnight by using dialysis membrane (Serva GmbH, Heidelberg, Germany, with cutoff 12 kDa). We determined the proper wavelength for each dye by scanning wavelength in the range of 280–700 nm by using UV-vis spectrophotometer (Shimadzu UV-1601, Tokyo, Japan). Then, the solutions including dye attached protein molecules were interacted with MIPC and NIPC specific filtration cartridges as given as before. The quantification was achieved by applying proper mass balance in respect to data obtained from spectrophotometer at wavelengths 697 nm, 679 nm, 659 nm, and 677 nm for reactive red 120-interferon, alkali blue 6B-insulin, reactive green HE4BD-IgG, congo red-albumin, respectively. Finally, we also calculated distribution, selectivity and relative selectivity constants for MIPC and NIPC specific filtration cartridges in accordance with the data obtained.
In addition, in order to assess selectivity of specific filtration cartridge in respect to size of competitors, we performed adsorption studies with NIPC and MIPC specific filtration cartridges not only from singular solution (uncompetitive conditions) but also from multi-protein solutions (competitive conditions) using the potential competitor biomolecules lysozyme (14.3 kDa), myoglobin (17.6 kDa), carbonic anhydrase (29.0 kDa) having the molecular weight closer to that of interferon α-2b (20 kDa). For this purpose, we adjusted the initial interferon α-2b concentration to 2 μg mL−1 while the other competitor proteins had initial concentration of 100 μg mL−1 at pH 6.0 (phosphate buffer). The interferon α-2b adsorption capacity and selectivity coefficients for the NIPC and MIPC specific filtration cartridges were investigated under uncompetitive and competitive conditions while quantifying the proteins via UV-vis spectrometry absorbance at four different wavelengths of 270 nm, 275 nm, 280 nm and 408 nm using a UV spectrophotometer (UV-1601, Shimadzu, Tokyo, Japan). These wavelength values were determined by scanning absorption spectrum for each protein molecules while calibrations curves at each wavelength were plotted to quantification purpose (ESI†). Herein, we have to mention that we used proper software to calculate each protein amount by applying matrices solution for four variables with four equations under competitive adsorption from multi-protein solution.
2.8. Desorption and reusability
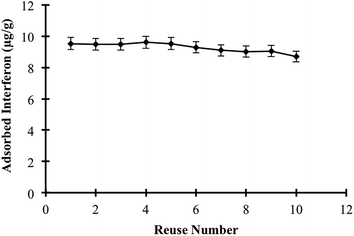
Adsorbed interferon α-2b was desorbed using 1.0 M ethylene glycol for an interaction time of 1 h. The final interferon α-2b concentration in the desorbing medium was also determined at 280 nm, and proper mass balances were used to calculate the achieved desorption ratio. To demonstrate the reusability of the specific filtration cartridges (MIPC), we performed ten consecutive interferon α-2b adsorption/desorption/regeneration cycles using the same specific filtration cartridges.3. Results and discussion
3.1. Characterisation of specific filtration cartridges
In this study, we developed specific filtration cartridges through molecularly imprinted cryogels. The cryogelation process has been thermodynamically driven process starting with formation of first ice crystal and their growth through the column. This process causes an incredible increase in monomer concentration called as cryo-concentration and helps researcher to synthesise monolithic and highly crosslinked 3D-polymeric network, in which ice crystals form interconnected large macropores meanwhile they force the monomers into unfrozen organic monomer phase. Even though the system looks like single ice block, it consists of frozen ice crystals and unfrozen organic monomer phase altogether. As you mentioned before, template molecules surrounded by functional monomer and crosslinker place at interface between organic and aqueous phase, therefore polymerisation occurs there to form imprinted cavities. By this way, cryogel could recognise the template molecules with fast adsorption kinetic in short interaction time. In addition, molecular imprinting of biomolecules like protein has some problems depending on their denaturation tendencies under harsh polymerisation conditions. Cryogelation is also excellent alternative in respect to not only mild polymerization condition but also enhancing polymer efficiency while limiting structural problems for proteins. Finally, cryogels are classified as powerful alternative to develop new adsorbent for selective, specific and fast protein separation via molecular imprinting technique besides their other physical, chemical, structural and economical features. As a result, we aimed to synthesise the cryogel-based cartridges for specific, fast and efficient filtration of interferons and characterise them via swelling test, SEM observation and FTIR spectroscopy.The water uptake ratios of the P(HEMA), NIPC and MIPC specific filtration cartridges were determined as 87.3%, 84.3% and 85.5%, respectively. In this respect, the equilibrium swelling ratios of the specific filtration cartridges were calculated as 6.86 g H2O/g for P(HEMA), 5.40 g H2O/g for NIPC and 5.85 g H2O/g for MIPC. The P(HEMA) filtration cartridge water-uptake ratio was the highest among the investigated filtration cartridges. Because tryptophan was used as a starting molecule to incorporate a functional monomer into the specific filtration cartridges' structure, the hydrophobicity of the polymeric network was increased. Consequently, water uptake decreased in comparison to P(HEMA).
SEM was used to investigate the porosity and surface morphology of the specific filtration cartridges. The SEM images of the porous structures of the P(HEMA), NIPC and MIPC specific filtration cartridges are shown in Fig. 1. All of the specific filtration cartridges have supermacropores and interconnected flow channels, which enable convective transport; therefore, the diffusional mass transfer resistance of these cartridges was substantially reduced. Moreover, these cartridges enable the use of high flowrates and centrifugation speeds due to their high specific internal surface area.
FTIR analysis was used to investigate the incorporation of MATrp into the polymeric backbone. As evident in Fig. 2a, the spectrum of the P(HEMA) filtration cartridge shows an O–H stretching vibration band at approximately 3287 cm−1. A CH alkyl stretching band was observed at approximately 2942 cm−1 in the same spectrum. In addition, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and C
O and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching bands were observed at approximately 1718 cm−1 and 1653 cm−1, respectively. The O–H stretching vibration bands in the spectra of both NIPC and MIPC were broader than those in the spectrum of the P(HEMA) filtration cartridge because of the presence of additional NH2 groups from the functional monomer. The FTIR spectra of the NIPC and MIPC cartridges exhibit characteristic NH bending vibration bands at approximately 1533 cm−1 and 1535 cm−1, respectively. As a result of the additional carbonyl groups, the characteristic stretching vibration band of the C
C stretching bands were observed at approximately 1718 cm−1 and 1653 cm−1, respectively. The O–H stretching vibration bands in the spectra of both NIPC and MIPC were broader than those in the spectrum of the P(HEMA) filtration cartridge because of the presence of additional NH2 groups from the functional monomer. The FTIR spectra of the NIPC and MIPC cartridges exhibit characteristic NH bending vibration bands at approximately 1533 cm−1 and 1535 cm−1, respectively. As a result of the additional carbonyl groups, the characteristic stretching vibration band of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O group at approximately 1710 cm−1 in the spectra of both NIPC and MIPC became sharper. Moreover, the sharpness of the C
O group at approximately 1710 cm−1 in the spectra of both NIPC and MIPC became sharper. Moreover, the sharpness of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bands increased because of an increase in the amount of MATrp in the MIP (A, B, C, D) specific filtration cartridges (Fig. 2b). The FTIR analysis results indicate that MATrp was successfully incorporated into the specific filtration cartridges.
O bands increased because of an increase in the amount of MATrp in the MIP (A, B, C, D) specific filtration cartridges (Fig. 2b). The FTIR analysis results indicate that MATrp was successfully incorporated into the specific filtration cartridges.
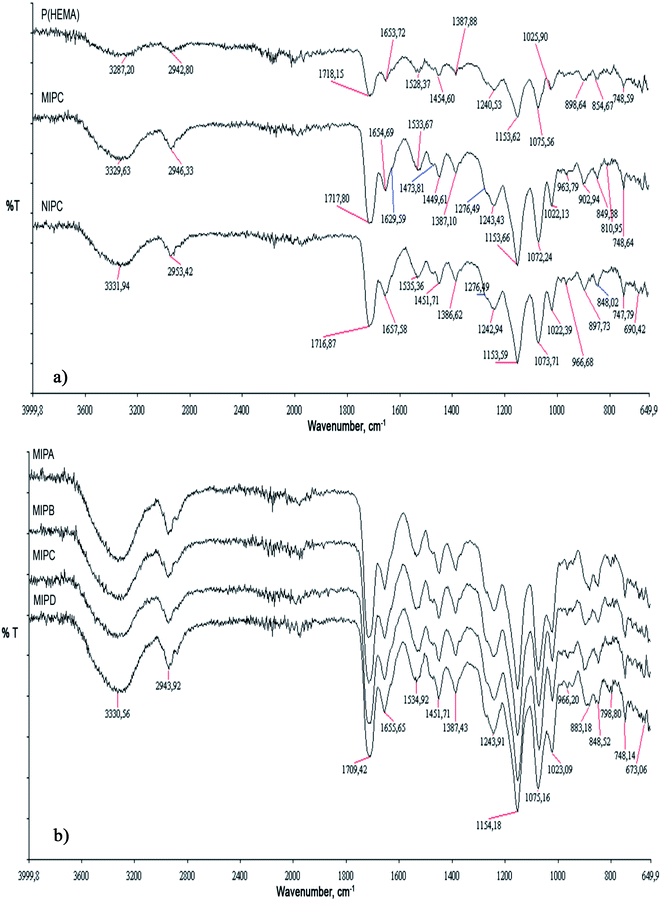 | ||
| Fig. 2 (a) FTIR analysis of P(HEMA), MIPC and NIPC specific filtration cartridges. (b) FTIR analysis of MIP (A, B, C, D) specific filtration cartridges. | ||
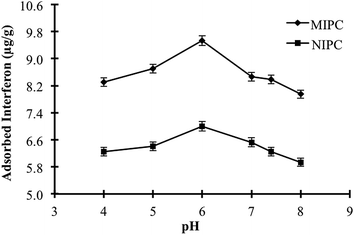
3.2. Optimisation of adsorption conditions
| Protein | MIPC | NIPC | k′ | ||
|---|---|---|---|---|---|
| Kd (mL g−1) | k | Kd (mL g−1) | k | ||
| Interferon | 61.523 | — | 13.296 | — | — |
| IgG | 24.277 | 2.534 | 19.545 | 0.680 | 3.72 |
| HSA | 29.460 | 2.088 | 45.172 | 0.294 | 7.10 |
| Insulin | 39.370 | 1.563 | 90.850 | 0.146 | 10.68 |
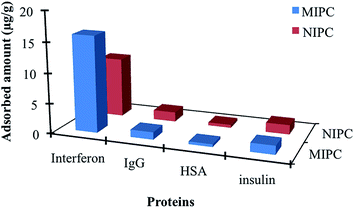
As mentioned in experimental section, we also performed selectivity experiment by using dye-modified protein samples. Herein, we just summarised the results without any figure to avoid misunderstanding with previous discussion, but figures, table and experimental steps were given in ESI† in more details. As well known, molecularly imprinted polymers have shape and size memory against template molecules and selectively recognise them via which structural modification applied on it before imprinting process. Therefore, the adsorption of template molecules after labelling may cause significant decrease in selectivity and specificity. That is reason why the selectivity constants calculated from dye-modified proteins were lower than the actual values as mentioned before. This depends on the changes in surface charge distribution and size of the proteins; but the selectivity tendency and specificity against template protein retained as the highest (ESI†). The affinity order in accordance to relative selectivity coefficients were calculated as interferon > insulin > HSA > IgG which was same as unmodified version. As a result we could concluded that the specific filtration cartridges retain their specificity against the template molecules in same order with free competitors even if dye molecules attached to proteins while some significant decrease in distribution and selectivity constant were also determined as expected.
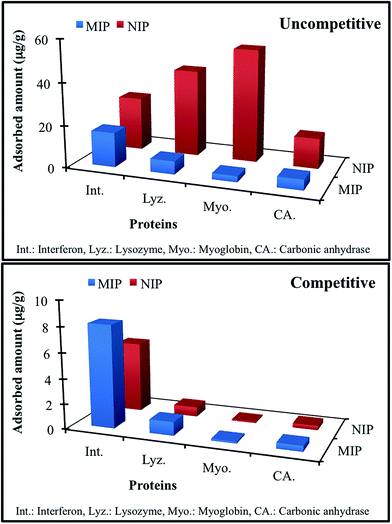
Besides isoelectric point, in other words surface charge density and distribution effect, the molecular size and concentration were also effective parameters on recognition ability of specific filtration cartridges. Therefore, we designed a set of selectivity experiments by using proteins that are potential competitors including lysozyme (14.3 kDa), myoglobin (17.6 kDa) and carbonic anhydrase (29.0 kDa) having the molecular weight close to that of interferon α-2b (20 kDa). In order to determine the effect of size, adsorption studies were performed from singular aqueous solution through uncompetitive manner. In addition, we performed the same experiment through competitive manner by using aqueous multi-protein solution while adjusting the concentration to 2 μg mL−1 for interferon α-2b and 100 μg mL−1 for others to assess the effect of concentration as well as molecular size. Due to fact that the pI values for interferon α-2b, lysozyme, myoglobin and carbonic anhydrase were 5.9, 11.35, 6.8–7.2 and 5.4, respectively, lysozyme and myoglobin have net negative charge, carbonic anhydrase has slightly net positive charge and interferon α-2b has almost no net charge at pH 6.0. Fig. 9 visualised the variation in molecular recognition abilities of specific filtration cartridges. As clearly seen in figure, imprinted cartridge selectively recognise the template protein, interferon α-2b under uncompetitive conditions and adsorption capacities were ordered as interferon α-2b > lysozyme > carbonic anhydrase > myoglobin whereas non-imprinted cartridge behave in different way in which adsorption capacities were ordered as myoglobin > lysozyme > interferon α-2b > carbonic anhydrase. The results confirmed the creation of specific recognition cavities due to molecular imprinting techniques. On the other hand, adsorption capabilities of both cartridges have been more similar under competitive conditions applied while capacities were ordered as interferon α-2b > lysozyme > myoglobin > carbonic anhydrase. Herein, two important parameters should be kept in mind: (i) the concentrations of proteins were adjusted to 2 μg mL−1 for interferon α-2b and 100 μg mL−1 for others and (ii) the adsorption capacity values were calculated by solving four equations with four variable via applying numerical solution for linear equation system to the data obtained from UV-vis absorption spectrum of multi-protein solution. These variations in adsorptive abilities of cartridges depend on both size and concentration of competitors as well as surface charge and charge distributions. In order to assess the functionality and specificity gained by molecular imprinting process, we have calculated the distribution (Kd, mL g−1), selectivity (k) and relative selectivity (k′) coefficients for the cartridges for both situations, uncompetitive and competitive (Table 3). As seen in table, the coefficients for relative selectivity gained by molecular imprinting were 7.58, 10.40 and 11.68 for interferon α-2b/lysozyme, interferon α-2b/myoglobin and interferon α-2b/carbonic anhydrase pairs. The threshold value for this coefficient is 1.0 to assess the selectivity of the system. How much this value is high means how much your system is selective. As conclusion, the proposed specific filtration cartridges showed really high selectivity against template protein under uncompetitive conditions. In case of competitive conditions, the values were calculated as 1.08, 1.05 and 1.34 for interferon α-2b/lysozyme, interferon α-2b/myoglobin and interferon α-2b/carbonic anhydrase pairs. Although they significantly decreased, they are still in acceptable limits. These decreases may be explained two ways: (i) the competitors in smaller size easily access to imprinted cavities and interact with functional monomer. In addition, surface charge distribution may significantly contribute to this adsorption behaviour due to proton donor character of indole ring found in functional monomer, MATrp. But, surface charge density and charge distribution of these smaller competitors are not fully complementary to orientation of functional monomer fixed in imprinted cavities. By this way, molecularly imprinted filtration cartridges show selectivity against template protein fully complementary to imprinted cavities even if under competitive conditions. (ii) As mentioned before, there is a huge concentration difference between interferon α-2b and other competitors. Each single interferon α-2b molecule has to compete with 150 competitors with different size and charge. Although this concentration disadvantages, specific filtration cartridges recognise the template and show selectivity in analytically acceptable levels. As conclusion, proposed specific filtration cartridges could be classified as alternative separation tool for selective, specific, reusable and fast interferon α-2b adsorption from not only singular adsorption media (uncompetitive) but also from multi-protein mixture (competitive) while challenging with the disadvantages such as charge density and distribution, size and concentration.
| Protein | MIPC | NIPC | k′ | |||
|---|---|---|---|---|---|---|
| Kd (mL g−1) | k | Kd (mL g−1) | k | |||
| a Int: interferon α-2b, Lyz: lysozyme, Myo: myoglobin, CA: carbonic anhydrase.b Initial protein concentration for interferon α-2b was 2 μg mL−1 whereas that for competitor proteins was 100 μg mL−1. | ||||||
| (a) Uncompetitive | Inta | 24.189 | — | 6.079 | — | — |
| Lyza | 5.854 | 4.131 | 11.119 | 0.546 | 7.58 | |
| Myoa | 4.804 | 5.036 | 12.554 | 0.484 | 10.40 | |
| CAa | 1.398 | 17.308 | 4.103 | 1.481 | 11.68 | |
| (b) Competitiveb | Inta | 11.385 | — | 5.543 | — | — |
| Lyza | 10.800 | 1.054 | 5.683 | 0.975 | 1.08 | |
| Myoa | 11.463 | 0.993 | 5.862 | 0.945 | 1.05 | |
| CAa | 6.766 | 1.682 | 4.402 | 1.259 | 1.34 | |
4. Conclusion
In recent years, specific filtration has become a promising technique among methods of separating and purifying biomolecules. Herein, the critical properties of specific filtration, such as high flux, low fouling and good stability with molecular imprinting techniques and cryogels, were combined to develop cryogel-based specific filtration cartridges. The obtained results clearly indicated that the proposed cartridge exhibits good selectivity towards interferon α-2b. Thus, the prepared specific filtration cartridges have been successfully used to adsorb interferon α-2b in an aqueous solution without any significant reduction in adsorption capacity. In conclusion, the developed cartridges could be classified as a promising, cost-friendly alternative for the specific, selective and rapid separation of interferon α-2b molecules.References
- D. Singabraya, L. Bultel, F. Sineriz, M. Mothere, D. Lesur, J. Kovensky and D. Papy-Garcia, Talanta, 2012, 99, 833–839 CrossRef CAS PubMed.
- M. C. Cela-Perez, A. Lasagabaster-Latorrea, M. J. Abad-Lopez, J. M. Lopez-Vilarino and M. V. Gonzalez-Rodriguez, Vib. Spectrosc., 2013, 65, 74–83 CrossRef CAS PubMed.
- F. G. Tamayo, E. Turiel and A. Martin-Esteban, J. Chromatogr. A, 2007, 1152, 32–40 CrossRef CAS PubMed.
- M. Andac, I. Y. Galaev and A. Denizli, Colloids Surf., B, 2013, 109, 259–265 CrossRef CAS PubMed.
- G. Diaz-Diaz, D. Antuna-Jimenez, M. C. Blanco-Lopez, M. J. Lobo-Castanon, A. J. Miranda-Ordieres and P. Tunon-Blanco, TrAC, Trends Anal. Chem., 2012, 33, 68–80 CrossRef CAS PubMed.
- P. Zhao and J. Hao, Food Chem., 2013, 139, 1001–1007 CrossRef CAS PubMed.
- M. Kumar and M. Ulbricht, Polymer, 2014, 55, 354–365 CrossRef CAS PubMed.
- H. Susanto, A. Roihatin, N. Aryanti, D. D. Anggoro and M. Ulbricht, Mater. Sci. Eng., C, 2012, 32, 1759–1766 CrossRef CAS PubMed.
- Q. Li, Q. Bi, H. H. Lin, L. X. Bian and X. L. Wang, J. Membr. Sci., 2013, 427, 155–167 CrossRef CAS PubMed.
- D. M. Bohonak and A. L. Zydney, J. Membr. Sci., 2005, 254, 71–79 CrossRef CAS PubMed.
- F. H. She, K. L. Tung and L. X. Kong, Robot Comput Integrated Manuf, 2008, 24, 427–434 CrossRef PubMed.
- J. Cho, G. Amy and J. Pellegrino, J. Membr. Sci., 2000, 164, 89–110 CrossRef CAS.
- C. Zhao, X. Zhou and Y. Yue, Desalination, 2000, 129, 107–123 CrossRef CAS.
- E. S. Hatakeyama, H. Ju, C. J. Gabriel, J. L. Lohr, J. E. Bara, R. D. Noble, B. D. Freeman and D. L. Gina, J. Membr. Sci., 2009, 330, 104–116 CrossRef CAS PubMed.
- B. K. Nandi, R. Uppaluri and M. K. Purkait, Appl. Clay Sci., 2008, 42, 102–110 CrossRef CAS PubMed.
- M. J. Luján-Facundo, J. A. Mendoza-Roca, B. Cuartas-Uribe and S. Álvarez-Blanco, Sep. Purif. Technol., 2013, 120, 275–281 CrossRef PubMed.
- A. W. Zularisam, A. F. Ismail and R. Salim, Desalination, 2006, 194, 211–231 CrossRef CAS PubMed.
- M. Cindric, N. Galic, M. Vuletic, M. Klaric and V. Drevenkar, J. Pharm. Biomed. Anal., 2006, 40, 781–787 CrossRef CAS PubMed.
- E. Kontsekova, H. Liptakova, V. Mucha and P. Kontsek, Int. J. Biol. Macromol., 1999, 24, 11–14 CrossRef CAS.
- Y. S. Wang, S. Youngster, M. Grace, J. Bausch, R. Bordens and D. F. Wyss, Adv. Drug Delivery Rev., 2002, 54, 547–570 CrossRef CAS.
- A. Dogan, S. Ozkara, M. M. Sari, L. Uzun and A. Denizli, J. Chromatogr. B: Anal. Technol. Biomed. Life Sci., 2012, 893–894, 69–76 CrossRef CAS PubMed.
- M. W. Davey, J. W. Huang, E. Sulkowski and W. A. Carter, J. Biol. Chem., 1975, 250, 348–349 CAS.
- Y. Saylan, M. M. Sari, S. Ozkara, L. Uzun and A. Denizli, Mater. Sci. Eng., C, 2012, 32, 937–944 CrossRef CAS PubMed.
- V. Karakoc, H. Yavuz and A. Denizli, Colloids Surf., A, 2004, 240, 93–99 CrossRef CAS PubMed.
- F. Yilmaz, N. Bereli, H. Yavuz and A. Denizli, Biochem. Eng. J., 2009, 43, 272–279 CrossRef CAS PubMed.
- A. R. Chaves, B. J. G. Silva, F. M. Lancas and M. E. C. Queiroz, J. Chromatogr. A, 2011, 1218, 3376–3381 CrossRef CAS PubMed.
- J. A. Queiroz, C. T. Tomaz and J. M. S. Cabral, J. Biotechnol., 2001, 87, 143–159 CrossRef CAS.
- R. L. Baldwin, Biophys. J., 1996, 71, 2056–2063 CrossRef CAS.
- S. Hjerten, K. Yao, K. O. Eriksson and B. Johansson, J. Chromatogr. A, 1986, 359, 99–109 CrossRef CAS.
- E. Yilmaz, L. Uzun, A. Y. Rad, U. Kalyoncu, S. Unal and A. Denizli, J. Biomater. Sci., Polym. Ed., 2008, 19, 875–892 CrossRef CAS PubMed.
- M. E. Corman, N. Bereli, S. Ozkara, L. Uzun and A. Denizli, Biomed. Chromatogr., 2013, 27, 1524–1531 CrossRef PubMed.
- C. Armutcu, N. Bereli, E. Bayram, L. Uzun, R. Say and A. Denizli, Colloids Surf., B, 2014, 114, 67–74 CrossRef CAS PubMed.
- J. Kufleitner, S. Wagner, F. Worek, H. V. Briesen and J. Kreuter, J. Microencapsulation, 2010, 27, 506–513 CrossRef CAS PubMed.
- T. J. Causon, A. Nordborg, R. A. Shellie and E. F. Hilder, J. Chromatogr. A, 2010, 1217, 3519–3524 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra07307c |
| This journal is © The Royal Society of Chemistry 2015 |