Microwave-assisted in situ synthesis of reduced graphene oxide/Mn3O4 composites for supercapacitor applications
Haiyan Zhang*,
Zidong Huang,
Yunyong Li*,
Yiming Chen,
Wenguang Wang,
Yipeng Ye and
Peng Deng
Guangdong Provincial Key Laboratory of Functional Soft Condensed Matter, School of Material and Energy, Guangdong University of Technology, Guangzhou, 510006, China. E-mail: hyzhang@gdut.edu.cn; yunyong207@163.com; Fax: +86 2039322570; Tel: +86 2039322570
First published on 14th May 2015
Abstract
Reduced graphene oxide/Mn3O4 (GM) composites were prepared by a simple and convenient strategy, that is, microwave irradiation of the hydrothermal product of reduced graphene oxide–Mn(NO3)2 mixtures. Mn3O4 nanoparticles with sizes of 20–50 nm were uniformly distributed on the surface of reduced graphene oxides. The GM composites exhibited good electrochemical performance with a specific capacitance of 344.8 F g−1 at the discharge current density of 1 A g−1 using 5 M NaOH as the electrolyte. The energy density of the GM composites was as high as 47.8 W h kg−1 with a power density of 1000 W kg−1. After 5000 cycles of charge/discharge experiments, a high level retaining specific capacitance of 342.1 F g−1 was obtained with 99.2% retention of the initial capacitance at 1 A g−1 and the equivalent series resistance of the GM composites system was much lower than that of pure Mn3O4. Therefore, the GM composites with large capacitance, good cycling performance and reversibility can be used as a promising electrode material for supercapacitor applications.
1. Introduction
Supercapacitors have drawn tremendous attention in the field of electronic devices and electric vehicles owing to their higher power densities compared with secondary batteries and higher energy densities than electrostatic and electrolytic capacitors.1–4 Supercapacitors can charge and discharge instantaneously, so they usually work as an electric vehicle power unit to instantaneously provide a large current for electric vehicles, which can improve the ability of acceleration and climbing of the electric vehicles. At the same time, they can reduce the damage from the large discharge current for the power battery and extend the cycle life of the battery. So many countries have invested a lot of manpower, materials and financial resources to study supercapacitors. There are two kinds of supercapacitors involving electric double-layer capacitors (EDLCs) and pseudocapacitors depending on the charge conversion/storage mechanism. The electrostatic forces in the electrical double layers formed at the interface between electrode and electrolyte play an important role in the energy storage of EDLCs.5 Generally, carbon materials, such as activated carbon, carbon nanotubes, and graphene, possesses large surface area and readily accessible mesopores, which have been widely used as electrode materials in EDLCs. Meanwhile, the charge storage mechanism of pseudocapacitors results from the fast Faradaic redox reaction between the electrolyte and electrode.6 The electrode materials of pseudocapacitors generally involve various metal oxides and conductive polymers. However, EDLCs have high power density but suffer from low energy density, while pseudocapacitors possess a high specific capacitance but encounter poor cycle performance. Therefore, the combination of carbon materials and pseudo-capacitive materials is supposed to achieve the supercapacitors with high energy density and excellent cycling stability.In recent years, transitions metal oxides have been extensively studied as pseudo-capacitive materials due to their higher capacitance than carbon materials and better cycling stability than conductive polymers.7–11 Among various transition metal oxides, Mn3O4 is a potentially interesting material for supercapacitors due to its low cost, environmental benignity and relatively broad work potential window in aqueous solution.12–14 However, the poor electronic conductivity (10−5 to 10−6 S cm−1)15 and cycling stability of Mn3O4 limit its further application for supercapacitors. Graphene, a flat monolayer of carbon atoms that tightly pack into a two-dimensional honeycomb lattice, has emerged as a promising material for supercapacitor application owing to its excellent electronic properties, such as high carrier mobility (200![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cm2 V−1 S−1),16 high thermal conductivity (5300 W m−1 K−1)17 and large theoretical specific surface area (2600 m2 g−1).18 Graphene/Mn3O4 composites have been widely used as a kind of electrode material for high-performance supercapacitors since graphene nanosheets can serve as a highly conductive matrix for anchoring metal oxide nanoparticles.19 Wang et al.19 synthesized graphene/Mn3O4 nanocomposites by mixing graphene suspension in ethylene glycol with MnO2 organosol, followed by subsequent ultrasonication processing and heat treatment. The specific capacitance of supercapacitors using graphene/Mn3O4 nanocomposites as electrode materials is up to 175 F g−1 in 1 M Na2SO4 aqueous solution and 256 F g−1 in 6 M KOH aqueous solution, respectively. Lee et al.20 prepared graphene/Mn3O4 nanorods composites by a simple hydrothermal process from KMnO4 using ethylene glycol as reducing agent. The specific capacitance of the graphene/Mn3O4 nanorods composites was 121, 115, 107, 97, 88, 85 and 83 F g−1 at current density of 0.5, 1, 2, 5, 10, 15 and 20 A g−1, respectively. Zhang et al.21 synthesized nanocomposites with Mn3O4 nanoparticles anchored on the graphene nanosheets by one-step solvothermal process, and the resulted graphene/Mn3O4 nanocomposites exhibited a specific capacitance of 147 F g−1 and the capacitance loss of 5% after 500 consecutive cycles in 1 M Na2SO4 aqueous solution at a current density of 0.1 A g−1. Wu et al.22 prepared graphene/Mn3O4 composites via a simple solvothermal process by mixing Mn(AC)2·4H2O with graphite oxide suspension in ethanol–H2O mixture. Under a potential window of −0.2 to 0.8 V, the composites delivered an initial specific capacitance of 161 F g−1 at 1 A g−1 and increased to 230 F g−1 after 1000 cycles.
000 cm2 V−1 S−1),16 high thermal conductivity (5300 W m−1 K−1)17 and large theoretical specific surface area (2600 m2 g−1).18 Graphene/Mn3O4 composites have been widely used as a kind of electrode material for high-performance supercapacitors since graphene nanosheets can serve as a highly conductive matrix for anchoring metal oxide nanoparticles.19 Wang et al.19 synthesized graphene/Mn3O4 nanocomposites by mixing graphene suspension in ethylene glycol with MnO2 organosol, followed by subsequent ultrasonication processing and heat treatment. The specific capacitance of supercapacitors using graphene/Mn3O4 nanocomposites as electrode materials is up to 175 F g−1 in 1 M Na2SO4 aqueous solution and 256 F g−1 in 6 M KOH aqueous solution, respectively. Lee et al.20 prepared graphene/Mn3O4 nanorods composites by a simple hydrothermal process from KMnO4 using ethylene glycol as reducing agent. The specific capacitance of the graphene/Mn3O4 nanorods composites was 121, 115, 107, 97, 88, 85 and 83 F g−1 at current density of 0.5, 1, 2, 5, 10, 15 and 20 A g−1, respectively. Zhang et al.21 synthesized nanocomposites with Mn3O4 nanoparticles anchored on the graphene nanosheets by one-step solvothermal process, and the resulted graphene/Mn3O4 nanocomposites exhibited a specific capacitance of 147 F g−1 and the capacitance loss of 5% after 500 consecutive cycles in 1 M Na2SO4 aqueous solution at a current density of 0.1 A g−1. Wu et al.22 prepared graphene/Mn3O4 composites via a simple solvothermal process by mixing Mn(AC)2·4H2O with graphite oxide suspension in ethanol–H2O mixture. Under a potential window of −0.2 to 0.8 V, the composites delivered an initial specific capacitance of 161 F g−1 at 1 A g−1 and increased to 230 F g−1 after 1000 cycles.
In this paper, we prepared reduced graphene oxide/Mn3O4 (GM) composites via a simple and convenience strategy, that is, microwave-assisted in situ synthesis of Mn3O4 nanoparticles on the graphene sheets. Reduced graphene oxides (RGO) were obtained by microwave radiation of graphite oxide (GO). GM composites were prepared by hydrothermal synthesis of aqueous mixture of RGO and Mn(NO3)2, followed by microwave irradiation of the hydrothermal synthesis product. Compared with the solvothermal methods, which need a long time to heat or dry the hydrothermal product to obtain Mn3O4/graphene composites, our strategy is more efficient and energy-saving. Additionally, the most important point to be noted is that, compared with the conventional microwave hydrothermal method which need in a condition of strong alkali or hydrazine hydrate (toxic), our strategy is green and environmentally friendly. The supercapacitors based on the as-prepared GM composites showed a far higher specific capacitance than RGO and more excellent cycling stability than pure Mn3O4.
2. Experimental
2.1 Preparation of RGO
Graphite oxide was prepared by a modified Hummers method.23 Reduced graphene oxide was obtained by microwave radiation of GO in a microwave oven at 1000 W for 2 min in ambient condition and a large volume expansion was observed.2.2 Preparation of GM composites
RGO was dispersed into distilled water under ultrasonic condition to form aqueous suspension with a concentration of 0.1 g L−1. Then 100 mL of as-obtained RGO aqueous suspension was mixed with a Mn(NO3)2 aqueous solution. The mixture was transferred into an autoclave and heated at 180 °C for 24 h. After cooling down to room temperature, the intermediate product was collected and dried at 30 °C. Finally, the GM composites were obtained through microwave radiation at 1000 W for 2 min in ambient condition once again.2.3 Characterization of samples
X-Ray diffraction spectroscopy (XRD; DMAX-Ultima IV, Rigaku Corporation, Japan) and Fourier transform infrared spectroscopy (FTIR; Nicolet 380, Nicolet, USA) were employed to identify the crystal structure and the chemical bands of GO, RGO and GM composites. The molecular vibration modes and defects of samples were measured by Raman spectroscopy (Renishaw 2000 Confocal Raman Microprobe; Renishaw Instruments, England). The morphology and structure of the samples were investigated by field emission scanning electron microscopy (FESEM; Quanta 200F, FEI, Holand) and transmission electron microscopy (TEM; JEM-2010 HR, Jeol, Japan). Thermogravimetric measurements (TGA; SDT 2960, TA Instruments, American) were carried out to determine the weight percentage of Mn3O4 in GM composites of the RGO and GM composites.2.4 Electrochemical measurements
The supercapacitor electrodes were prepared by pressing the mixture of as-prepared GM composites, acetylene black and polytetrafluoroethlene at a weight ratio of 8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 onto the Ni foam and dried at 80 °C for 12 h. The electrodes were sliced into a 16.2 mm diameter wafer and packed into button-type supercapacitors using 5 M of NaOH aqueous solution as electrolyte. The electrochemical properties of packaged button cell were studied by cyclic voltammetry, galvanostatically charge/discharge measurements and AC impedance test. The Nyquist plots and CV curves were measured by an electrochemical work station (IM6) with the potential from 0 to 1.0 V at different scan rates. The cycle performance and the charge/discharge efficiency were tested by a LAND battery tester (CT2001A, Wuhan LAND Corporation, China) under the various current densities of charge/discharge. Meanwhile, RGO and pure Mn3O4 electrodes were also prepared and tested under the same condition for comparison.
1 onto the Ni foam and dried at 80 °C for 12 h. The electrodes were sliced into a 16.2 mm diameter wafer and packed into button-type supercapacitors using 5 M of NaOH aqueous solution as electrolyte. The electrochemical properties of packaged button cell were studied by cyclic voltammetry, galvanostatically charge/discharge measurements and AC impedance test. The Nyquist plots and CV curves were measured by an electrochemical work station (IM6) with the potential from 0 to 1.0 V at different scan rates. The cycle performance and the charge/discharge efficiency were tested by a LAND battery tester (CT2001A, Wuhan LAND Corporation, China) under the various current densities of charge/discharge. Meanwhile, RGO and pure Mn3O4 electrodes were also prepared and tested under the same condition for comparison.
3. Results and discussion
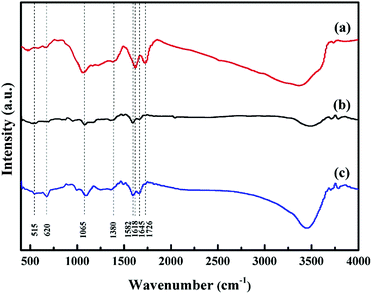
Fig. 1 shows the XRD patterns of GO, RGO and GM composites. As shown in Fig. 1a, the diffraction peaks at 10.46° and 42.6° correspond to the (001) and (111) reflection of GO, indicating that graphite had been fully oxidized.24 The interlayer spacing of GO was 0.87 nm, which was much larger than that of pristine graphite (0.34 nm) due to the introduction of oxygen-containing functional groups on the graphite sheets.22 After microwave radiation treatment of GO, the (001) peak disappeared, while a new peak emerged at 25°, as shown in Fig. 1b. The peak with weaker intensity and wider shape corresponded to the disorderly stack and amorphous structure of RGO, resulting from the decomposition of oxygen-containing functional groups during the reduction of GO.23 Fig. 1c shows the XRD pattern of GM composites. All diffraction peaks can be indexed to typical Hausmannite Mn3O4 (JCPDS card no. 24-0734). In addition, the peak of RGO at 25° was shifted to around 26°, indicating that the residual oxygen-containing functional groups were further decomposed simultaneously during the final microwave irradiation.FTIR spectroscopy was used to analysis the existence of oxygen-containing functional groups and their changes after microwave radiation treatment. Fig. 2 shows the FTIR spectra of GO, RGO and GM composites, respectively. As shown in Fig. 2a, a broad and intense absorption band appeared in the range of 3000–3700 cm−1 derived from the stretching vibration of –OH. Two typical peaks at 1726 and 1618 cm−1 can be attributed to the stretching vibration of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O in carboxy and the deformation vibration absorption peak of water molecules, respectively. Another two weak peaks at 1380 and 1065 cm−1 corresponded to C–O and the vibration adsorption peak of C–O–C, respectively.23 Compared with GO, the intensity of all absorption peaks related to oxygen-containing functional groups in RGO (Fig. 2b) obviously decreased. The peaks at 1618 and 1726 cm−1 were almost disappeared, but the peaks at 1065, 1380 and 3364 cm−1 still existed, indicating that part of oxygen-containing functional groups were decomposed during microwave irradiation. Furthermore, a new absorption peak located at 1582 cm−1, corresponding to the aromatic skeletal of C
O in carboxy and the deformation vibration absorption peak of water molecules, respectively. Another two weak peaks at 1380 and 1065 cm−1 corresponded to C–O and the vibration adsorption peak of C–O–C, respectively.23 Compared with GO, the intensity of all absorption peaks related to oxygen-containing functional groups in RGO (Fig. 2b) obviously decreased. The peaks at 1618 and 1726 cm−1 were almost disappeared, but the peaks at 1065, 1380 and 3364 cm−1 still existed, indicating that part of oxygen-containing functional groups were decomposed during microwave irradiation. Furthermore, a new absorption peak located at 1582 cm−1, corresponding to the aromatic skeletal of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching vibration, was observed. The result further demonstrated the reduction of GO.25 A new band at 1645 cm−1 originated from the O–H bending vibration of absorbed water molecules. For the GM composites, the absorption peaks at 1065, 1380, 1618, 1645 and 3364 cm−1 corresponded to RGO (Fig. 2c). Two extra broad absorption peaks at 515 and 620 cm−1 were associated with the coupling mode between octahedral sites and Mn–O stretching modes of tetrahedral.22 The FTIR results further confirmed the reduction of GO and the formation of Mn3O4.
C stretching vibration, was observed. The result further demonstrated the reduction of GO.25 A new band at 1645 cm−1 originated from the O–H bending vibration of absorbed water molecules. For the GM composites, the absorption peaks at 1065, 1380, 1618, 1645 and 3364 cm−1 corresponded to RGO (Fig. 2c). Two extra broad absorption peaks at 515 and 620 cm−1 were associated with the coupling mode between octahedral sites and Mn–O stretching modes of tetrahedral.22 The FTIR results further confirmed the reduction of GO and the formation of Mn3O4.
Raman spectroscopy is a powerful non-destructive technique to gain information of the ordered and disordered crystal structures of the samples. Fig. 3 shows the Raman spectra of natural graphite, RGO and GM composites. As shown in Fig. 3a, the natural graphite exhibits two Raman bands. The strong G band at 1581 cm−1 corresponded to the first-order scattering of E2g mode, while the weak D band at 1354 cm−1 corresponded to graphite edges.26 For the RGO and GM composites, two strong Raman peaks for G band and D band were also observed respectively, as shown in Fig. 3b and c. The ratio of the intensity of D and G band (ID/IG) for RGO was 0.96, which was dramatically higher than that of natural graphite (0.03), indicating much defects and high disorder in RGO resulted from the oxygen-containing defects due to the fiercely oxidation.27 After hydrothermal treatment and microwave irradiation, the ID/IG of GM composites was increased to 1.12, indicating that more defects and disorder existed in GM composites than RGO due to the presence of Mn3O4 crystallites on graphene sheets.
The surface morphologies of RGO and GM composites were studied by FE-SEM. RGO was crumpled into a curly and wavy sheet, and a few layers of RGO were stacked with each other, as shown in Fig. 4a. In the case of GM composites, the Mn3O4 nanoparticles were uniformly distributed on the surface of the RGO sheets (Fig. 4b). Further observation indicated that no stacking of graphene in the composites occurred, leading to a larger available surface area for energy storage.
Fig. 5 shows the TEM images of RGO and GM composites. As shown in Fig. 5a, RGO was transparent and a few layers of RGO were stacked with each other, which was consistent with the FE-SEM characterization. We can also observe in Fig. 5b that Mn3O4 nanoparticles with sizes of 20–50 nm were uniformly distributed on the transparent RGO sheets.
TGA measurements were employed to investigate the weight percentage of Mn3O4 in GM composites of RGO and the graphene in GM composites, as shown in Fig. 6. All samples were heated in a temperature range of 30–800 °C with a ramp rate of 20 °C min−1 in air. The weight loss of ca. 2.4% below 100 °C was due to the evaporation of the absorbed moisture. For the RGO, the weight loss was reached to 100% when the temperature reached to 800 °C, indicating that the RGO was completely burn off in air at 800 °C. For the GM composite, there was a weight loss of 65.7% at the temperature range of 100–800 °C, which can be attributed to the combustion of the graphene in the composites. Therefore, the content of Mn3O4 in GM composites was ∼32.7 wt% (31.9%![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 97.6%).
97.6%).
The as-prepared samples were fabricated into supercapacitor electrodes and tested by cyclic voltammetry measurement (CV) in a two-electrode symmetrical cell. The CV curves of the electrodes at a scan rate of 10 mV s−1 in the voltage range of 0–1.0 V using 5 M of NaOH aqueous solution as electrolyte was shown in Fig. 7a. The CV curves of RGO and GM composite were both near-rectangular and symmetric, indicating that they both possessed a perfect capacitive behavior and electrochemical performance. For the pure Mn3O4, a distorted CV curve was observed, which was probably caused by the poor electrical conductivity of pure Mn3O4. In addition, it is clearly seen that the CV loop of GM composite is much larger than those of RGO and Mn3O4. Fig. 7b shows the CV curves of GM measured at scan varying from 10 to 100 mV s−1, which are also near-rectangular and symmetric, indicating that the GM exhibits good charge and discharge reversibility.
Fig. 7c shows the galvanostatic charge/discharge curves of pure Mn3O4, RGO and GM composites electrodes between 0 V and 1.0 V at a current density of 1 A g−1. And the galvanostatic charge/discharge curves at different current densities as shown in Fig. 7d. The curves in the total range of 0–1.0 V were linear and symmetrical, indicating a good capacitive behavior. In the galvanostatically charge/discharge measurement, the specific capacitance of single electrode should be twice the supercapacitor in that the inner circuit of supercapacitor is equal to the series connection of two capacitors. The specific capacitance of single electrode (Csingle) was calculated according to the following eqn (1):23
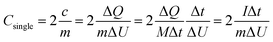 | (1) |
Fig. 8a shows the specific capacitance of samples at different current density. When the current density increased to 10 A g−1, the specific capacitance of GM composites still kept at a high value of 258.1 F g−1 (∼74.9% capacitance retention), which is far larger than those of the RGO (115.0 F g−1, ∼73.6% capacitance retention) and pure Mn3O4 (29.5 F g−1, ∼22.9% capacitance retention). The result indicated that GM composites had a more excellent rate performance than pure Mn3O4. The enhanced capacitance and the excellent rate performance of GM composites were mainly attributed to the synergistic effect of the graphene and Mn3O4 nanoparticles. Firstly, uniform distribution of Mn3O4 nanoparticles on graphene sheets can eliminate restacking and agglomeration of graphene, and offer a high active surface for taking a full advantage of Mn3O4-based pseudocapacitance and graphene-based electric double-layer capacitance. Secondly, graphene with high electrical conductivity can serve as the electron superhighway, which facilitate a rapid and efficient charge transport, thus to improve the electronic conductivity of the overall system. Thirdly, the graphene can inhibit the aggregation of the Mn3O4 nanoparticles, thus to preserve the high-surface-area interface between the Mn3O4 nanoparticles and electrolytes.
 | ||
| Fig. 8 Effects of current density on the specific capacitance of electrodes (a), and Ragone plots (b) of the supercapacitors based on pure Mn3O4, RGO and GM composites. | ||
Fig. 8b shows the Ragone plots of the supercapacitors based on the RGO, pure Mn3O4 and GM composites. The energy density (E) and specific power density (P) of electrodes were calculated according to eqn (2)28 and (3):28
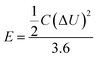 | (2) |
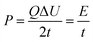 | (3) |
To investigate the transport characteristics of the charge carries in RGO, pure Mn3O4 and GM composites electrodes, the electrochemical impedance spectroscopy (EIS) of the supercapacitors was measured and the corresponding Nyquist plot was shown in Fig. 9a. Nyquist plot is a plot of the imaginary component (Z′′) of the impedance against the real component (Z′) reflecting the frequency response of electrode/electrolyte system. Apparently, a semicircle portion was observed at higher frequencies corresponding to the charge transfer limiting process. The slope portion of 45° of the curve is called the Warburg resistance resulting from the frequency dependence of ions diffusion/transport in the electrolyte.6
 | ||
| Fig. 9 EIS (a) and cycle performance measured at 1 A g−1 (b) for the supercapacitors based on pure Mn3O4, RGO and GM composites. | ||
At higher frequencies, the relationship between Z′ and Z′′ can be described as eqn (4), while at low frequencies, a straight line of the slope of 45° can be obtained according to eqn (5):29
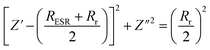 | (4) |
| Z′′ = Z′ − RESR − Rr + 2σ2Cd | (5) |
The Nyquist plot in Fig. 9a showed that equivalent series resistance (ESR) of the RGO, pure and GM composite systems were 0.33, 0.38 and 0.63 Ω, respectively, illustrating that the combination of high electrical conductive graphene with Mn3O4 resulted in a good conductivity of the overall system. This behavior was attributed to the good conductivity of graphene matrix and the excellent contact between the graphene and Mn3O4 nanoparticles. The almost straight line of the slope of 45° in the low-frequency region and the low RESR for GM composite system demonstrated that the electrodes based on GM composite can be used as ideal capacitors.
Cycling stability, one of most important requirements in practical application, was investigated in the range of 0–1 V at 1 A g−1 in 5 M of NaOH aqueous solution. Fig. 9b presents the cycling performance of RGO, pure Mn3O4 and GM composites with cycle times over 5000. It is found that the specific capacitance of pure Mn3O4 electrodes retained only about 39.2% after 5000 cycles. However, the curves of RGO and GM composites were almost smooth horizontal lines, showing an excellent cycle performance. The specific capacitance of GM composites electrodes slightly decreased from 344.8 to 342.1 F g−1, retaining about 99.2% of initial capacitance after 5000 cycles. Compared with pure Mn3O4, the excellent cycling stability of GM composites was attributed to the excellent interconnection of Mn3O4 nanoparticles at the surface and interior of the electrodes. The uniform dispersion of Mn3O4 nanoparticles on the graphene sheets and the large distance between neighboring graphene sheets provided enough space to buffer the volume change of Mn3O4 nanoparticles during charge/discharge redox reaction. Table 1 summarizes electrochemical performance reported in literature for Mn3O4/graphene composites as a supercapacitor electrode material. It is clear that our as-papered electrodes are superior to those of most recent counterparts in performance.
| Preparation method | Electrolyte | Measurement protocol | Maximum capacitance | Capacitance retention after cycle test | Ref. (year) |
|---|---|---|---|---|---|
| Ultrasound assisted | 1 M Na2SO4 | 5 mV s−1 | 175 F g−1 | — | (ref. 19) 2010 |
| 6 M KOH | 256 F g−1 | ||||
| Solvothermal | 1 M Na2SO4 | 1 A g−1 | 115 F g−1 | 100% after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles 000 cycles |
(ref. 20) 2012 |
| Microwave-assisted | 0.5 M Na2SO4 | 25 mV s−1 | 193 F g−1 | — | (ref. 30) 2012 |
| Solvothermal | 1 M Na2SO4 | 1 A g−1 | 147 F g−1 | 95% after 500 cycles | (ref. 21) 2012 |
| Solvothermal | 1 M Na2SO4 | 5 mV s−1 | 225 F g−1 | 82.1% after 900 cycles | (ref. 22) 2013 |
| Solvothermal | 0.5 M Na2SO4 | 1 A g−1 | 205.5 F g−1 | 98.7% after 2000 cycles | (ref. 31) 2013 |
| 5 mM NaNCO3 | |||||
| Solvothermal | 1 M Na2SO4 | 0.1 A g−1 | 171 F g−1 | 92.9% after 50 cycles | (ref. 32) 2013 |
| Dielectric barrier discharge (DBD) plasma-assisted | Saturated K2SO4 | 50 mA g−1 | 260 F g−1 | 92% after 800 cycles | (ref. 33) 2013 |
| Chemical decomposition | 1 M Na2SO4 | 0.5 A g−1 | 131 F g−1 | 99% after 500 cycles | (ref. 34) 2014 |
| Combined modified Hummers | Saturated K2SO4 | 50 mA g−1 | 260 F g−1 | 94% after 1000 cyles | (ref. 35) 2014 |
| Ultrasound assisted | 1 M Na2SO4 | 0.5 mA cm−2 | 312 F g−1 | 76% after 1000 cycles | (ref. 36) 2015 |
| Microwave-assisted | 5 M NaOH | 1 A g−1 | 344.8 F g−1 | 99.2% after 5000 cycles | This work |
4. Conclusion
GM composites have been successfully fabricated through microwave-assisted in situ hydrothermal synthesis of Mn3O4 nanoparticles on the RGO. The supercapacitors based on GM composites electrodes showed excellent electrochemical performance. With 5 M of NaOH as the electrolyte, supercapacitor electrodes based on GM composites material showed a specific capacitance as high as 344.8 F g−1 at the current density of 1 A g−1 in the voltage range of 0–1.0 V and a high energy density up to 47.8 W h kg−1 at the power density of 1000 W kg−1. Furthermore, the GM composites exhibited good cycling stability with little decay after 5000 cycles. The supercapacitor electrodes based on GM composites with excellent electrochemical properties and stability are expected to find widely promising applications in high-performance supercapacitors.Acknowledgements
This work is supported by the link project of the National Natural Science Foundation of China and Guangdong Province (no. U1401246), the National Natural Science Foundation of China (Grant no. 51276044), and the Natural Science Foundation of Guangdong Province of China (no. 2014A030310382).References
- J. R. Miller and P. Simon, Science, 2008, 321, 651–652 CrossRef CAS PubMed.
- M. Noked, A. Soffer and D. Aurbach, J. Solid State Electrochem., 2011, 15, 1563–1578 CrossRef CAS.
- T. Brezesinski, J. Wang, S. H. Tolbert and B. Dunn, Nat. Mater., 2010, 9, 146–151 CrossRef CAS PubMed.
- H. Wang, Z. Liu, X. Chen, P. Han, S. Dong and G. Cui, J. Solid State Electrochem., 2011, 15, 1179–1184 CrossRef CAS PubMed.
- R. H. Baughman, A. A. Zakhidov and W. A. de Heer, Science, 2002, 297, 787–792 CrossRef CAS PubMed.
- M. D. Stoller, S. Park, Y. Zhu, J. An and R. S. Ruoff, Nano Lett., 2008, 8, 3498–3502 CrossRef CAS PubMed.
- A. G. Pandolfo and A. F. Hollenkamp, J. Power Sources, 2006, 157, 11–27 CrossRef CAS PubMed.
- S. Sarangapani, B. Tilak and C. P. Chen, J. Electrochem. Soc., 1996, 143, 3791–3799 CrossRef CAS PubMed.
- J. Li, X. Wang, Q. Huang, S. Gamboa and P. Sebastian, J. Power Sources, 2006, 158, 784–788 CrossRef CAS PubMed.
- X. Cao, Y. Shi, W. Shi, G. Lu, X. Huang, Q. Yan, Q. Zhang and H. Zhang, Small, 2011, 7, 3163–3168 CrossRef CAS PubMed.
- G. Yu, L. Hu, N. Liu, H. Wang, M. Vosgureitchian, Y. Yang, Y. Cui and Z. Bao, Nano Lett., 2011, 11, 4438–4442 CrossRef CAS PubMed.
- B. Jiang, Y. Liu and Z. Wu, J. Hazard. Mater., 2009, 162, 1249–1254 CrossRef CAS PubMed.
- S.-B. Ma, K.-W. Nam, W.-S. Yoon, S.-M. Bak, X.-Q. Yang, B.-W. Cho and K.-B. Kim, Electrochem. Commun., 2009, 11, 1575–1578 CrossRef CAS PubMed.
- X. Q. Yu, Y. He, J. P. Sun, K. Tang, H. Li, L. Q. Chen and X. J. Huang, Electrochem. Commun., 2009, 11, 791–794 CrossRef CAS PubMed.
- D. Bélanger, L. Brousse and J. W. Long, Electrochem. Soc. Interface, 2008, 17, 49 Search PubMed.
- S. V. Morozov, K. S. Novoselov, M. I. Katsnelson, F. Schedin, D. C. Elias, J. A. Jaszczak and A. K. Geim, Phys. Rev. Lett., 2008, 100, 016602 CrossRef CAS.
- A. A. Balandin, S. Ghosh, W. Bao, I. Calizo, D. Teweldebrhan, F. Miao and C. N. Lau, Nano Lett., 2008, 8, 902–907 CrossRef CAS PubMed.
- S. Stankovich, D. A. Dikin, G. H. B. Dommett, K. M. Kohlhaas, E. J. Zimney, E. A. Stach, R. D. Piner, S. T. Nguyen and R. S. Ruoff, Nature, 2006, 442, 282–286 CrossRef CAS PubMed.
- B. Wang, J. Park, C. Wang, H. Ahn and G. Wang, Electrochim. Acta, 2010, 55, 6812–6817 CrossRef CAS PubMed.
- J. W. Lee, A. S. Hall, J.-D. Kim and T. E. Mallouk, Chem. Mater., 2012, 24, 1158–1164 CrossRef CAS.
- X. Zhang, X. Sun, Y. Chen, D. Zhang and Y. Ma, Mater. Lett., 2012, 68, 336–339 CrossRef CAS PubMed.
- Y. Wu, S. Liu, H. Wang, X. Wang, X. Zhang and G. Jin, Electrochim. Acta, 2013, 90, 210–218 CrossRef CAS PubMed.
- J. Ye, H. Zhang, Y. Chen, Z. Cheng, L. Hu and Q. Ran, J. Power Sources, 2012, 212, 105–110 CrossRef CAS PubMed.
- B. Zhao, J. Song, P. Liu, W. Xu, T. Fang, Z. Jiao, H. Zhang and Y. Jiang, J. Mater. Chem., 2011, 21, 18792–18798 RSC.
- Z. Wu, W. Ren, L. Gao, J. Zhao, Z. Chen, B. Liu, D. Tang, B. Yu, C. Jiang and H.-M. Cheng, ACS Nano, 2009, 3, 411–417 CrossRef CAS PubMed.
- F. Tuinstra and J. L. Koenig, J. Chem. Phys., 1970, 53, 1126–1130 CrossRef CAS PubMed.
- A. Ferrari, J. Meyer, V. Scardaci, C. Casiraghi, M. Lazzeri, F. Mauri, S. Piscanec, D. Jiang, K. Novoselov and S. Roth, Phys. Rev. Lett., 2006, 97, 187401 CrossRef CAS.
- J. Yan, Z. Fan, T. Wei, J. Cheng, B. Shao, K. W. L. Song and M. Zhang, J. Power Sources, 2009, 194, 1202–1207 CrossRef CAS PubMed.
- Z. Huang, H. Zhang, Y. Chen, W. Wang, Y. Chen and Y. Zhong, Electrochim. Acta, 2013, 108, 421–428 CrossRef CAS PubMed.
- C. Liu, K. Chang, C. Hu and W. Wen, J. Power Sources, 2012, 217, 184–192 CrossRef CAS PubMed.
- S. Yang, X. Song, P. Zhang and L. Gao, J. Mater. Chem., 2013, 1, 14162 RSC.
- Y. Fan, X. Zhang, Y. Liu, Q. Cai and J. Zhang, Mater. Lett., 2013, 95, 153–156 CrossRef CAS PubMed.
- J. Qu, F. Gao, Q. Zhou, Z. Wang, H. Hu, B. Li, W. Wan, X. Wang and J. Qiu, Nanoscale, 2013, 5, 2999–3005 RSC.
- K. Subramani, D. Jeyakumara and M. Sathish, Phys. Chem. Chem. Phys., 2014, 16, 4952–4961 RSC.
- F. Gao, J. Qu, Z. Zhao, Q. Zhou, B. Li and J. Qiu, Carbon, 2014, 80, 640–650 CrossRef CAS PubMed.
- B. G. S. Raj, R. N. R. Ramprasad, A. M. Asiri, J. J. Wu and S. Anandan, Electrochim. Acta, 2015, 156, 27–137 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2015 |