Combination of ultrasound and Fenton treatment for improving the hydrolysis and acidification of waste activated sludge
Hongxu Bao*ab,
Lei Jianga,
Chunxiao Chena,
Chunxue Yangb,
Zhangwei Heb,
Yaodong Fenga,
Weiwei Caib,
Wenzong Liubc and
Aijie Wang*bc
aSchool of Environmental Science, Liaoning University, Shenyang 110036, China. E-mail: baohongxu555@163.com; Fax: +86 024 62204818; Tel: +86 024 62202248
bState Key Laboratory of Urban Water Resources and Environments, Harbin Institute of Technology, Harbin 150090, China. E-mail: waj0578@hit.edu.cn; Fax: +86 451 86282195; Tel: +86 451 86282195
cKey Laboratory of Environmental Biotechnology, Research Center for Eco-Environmental Sciences, Chinese Academy of Sciences, Beijing 100085, China
First published on 18th May 2015
Abstract
The effects of ultrasound (US), Fenton treatment, and combined US/Fenton treatment as physicochemical pretreatment processes for waste activated sludge (WAS) hydrolysis and acidification were investigated in this study. The soluble carbohydrate concentrations after US/Fenton treatment were 2.36-fold, 2.17-fold and 17.11-fold more than that obtained by the Fenton treatment, US and control tests, respectively. The corresponding concentrations of soluble protein after US/Fenton treatment were 2.43-fold, 1.47-fold and 13.37-fold more than that obtained by the Fenton treatment, US and control tests, respectively. The degree of sludge disintegration (DDCOD) for the US/Fenton treatment was 15.4%, whereas it was only 9.97% and 3.18% for the US and Fenton tests. The maximum accumulation of volatile fatty acids (VFAs) was obtained by the US/Fenton pretreatment (4594 mg COD L−1), which presented obvious advantages over US (3485 mg COD L−1) and Fenton (2700 mg COD L−1) treatments. The combination of the US and Fenton treatments had a synergetic effect on improving the hydrolysis and subsequent acidification of WAS.
1. Introduction
Recently, with increasing municipal wastewater along with urbanization, more and more waste activated sludge (WAS) is produced in municipal wastewater treatment plants (WWTPs). Sludge disposal is becoming a serious problem, representing almost 50% of the current operating cost of a WWTP.1 WAS contains a substantial amount of nontoxic organic compounds such as carbohydrates, proteins and lipids. Some methods have been used for WAS treatment.2,3 Anaerobic digestion (AD) is a sustainable and cost-effective sludge treatment method because it can not only achieve sludge reduction, but also produce resources with high added value.4 As one of the by-products of AD, volatile fatty acids (VFAs) are an important substrate for the production of bioenergy and nutrient removal and have attracted a great deal of attention.5 However, their production has often been limited by their long retention time and low degradation efficiency, which are generally associated with the hydrolysis efficiency of sludge.6 In order to accelerate hydrolysis, WAS pretreatments such as mechanical,7 chemical,8 biological9 and combined10 methods have been developed to improve anaerobic fermentation efficiency for VFAs accumulation.Ultrasound (US) is an effective mechanical pretreatment method for enhancing the biodegradability of sludge, and Pilli et al. reported that US would be very effective for sludge disposal and treatment in all WWTPs.11 US can be applied to disrupt sludge flocs and microbial cell walls, which could disintegrate sludge and release organic substances into the liquid phase. Hydro-mechanical shear forces are predominantly responsible for WAS disintegration, and the oxidizing effect of ˙OH can contribute to WAS disintegration when ultrasonic intensity increases to a certain extent.12 High specific energy input (>5000 kJ kg TS−1), high power density/intensity and long sonication time were found suitable for sludge disintegration.13
Fenton reaction, an environmentally friendly technique, is one of the most effective advanced oxidation processes. In the Fenton reaction process, ferrous iron (Fe2+) catalyzes the decomposition of H2O2. This can accelerate the generation of hydroxyl radicals, which are powerful and non-selective oxidizing agents.14 Hydroxyl radicals can pass easily through the microbial cell membrane to discharge intracellular organic substances into the liquid phase. Meanwhile, the hydroxyl radicals can oxidize the recalcitrant compounds into biodegradable ones, which can serve as nutrients for further biological treatment.15
Some researchers have studied the combination of US and Fenton processes for sludge treatment.16,17 Qiu et al. (2013) investigated ultrasonic irradiation as a pretreatment for Fenton-acclimation treatment to enhance the efficiency of sludge reduction.16 Ning et al. (2014) studied the effect of US-assisted Fenton (US/Fenton) treatment on the physicochemical properties of textile dyeing sludge.17 However, little work has explored the effects of combined treatments on the hydrolysis and further acidification processes of WAS. US can enhance the oxidation rate of the Fenton process by generating more ˙OH because of the cavitation within US irradiation.17 Thus, the US/Fenton treatment may have a synergetic effect on WAS pretreatment. The aim of the present work is to investigate the influence of US, Fenton and US/Fenton pretreatment on WAS hydrolysis and further acidification processes during anaerobic fermentation.
2. Material and methods
2.1 Sludge properties
WAS was collected from the secondary sedimentation tank of the Taiping WWTP (Harbin City, Heilongjiang Province, China). The sludge was firstly concentrated by gravity settling for 24 h at 4 °C and then filtered through 1.0 mm sieves to eliminate the larger fraction and provide homogeneous digested sludge. The filtered WAS was stored at 4 °C for later use. WAS should be diluted before using, and its main characteristics are listed in Table 1.| Parameter | Average valuea |
|---|---|
| a All values are expressed in mg L−1 except pH. | |
| pH | 6.85 ± 0.5 |
| TSS (total suspended solids) | 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 381 ± 368 381 ± 368 |
| VSS (volatile suspended solids) | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 124 ± 142 124 ± 142 |
| Soluble chemical oxygen demand | 213 ± 7 |
| Total chemical oxygen demand | 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 333 ± 225 333 ± 225 |
| Total SCFAs (as COD) | 21 ± 2 |
| Solute carbohydrate (as COD) | 25 ± 3 |
| Solute protein (as COD) | 154 ± 13 |
2.2 Pretreatment methods
US treatment was performed in an US reactor at a frequency of 40 kHz (KQ2200DA, Kunshan Shumei Inc., China). The US power density (P) was adjusted to 0.4 kW L−1, and the treatment time (t) was 10 min. Fenton treatment was conducted with a magnetic stirrer (JB-2A, Shanghai Leici Inc., China). Sludge samples were initially adjusted to pH 3 by sulfuric acid (H2SO4), and the concentration of Fe2+ was adjusted to 0.04 g L−1 by adding ferrous sulfate (FeSO4·7H2O). The reaction was then initiated 90 min after adding 10 g L−1 H2O2 (30 wt%).16For US/Fenton treatment, sludge was first pretreated by the US method and then pretreated with Fenton reaction. The treatment conditions were the same as mentioned above. The soluble organic matter content and particle size were measured after each pretreatment process.
2.3 Batch-scale anaerobic fermentation
Batch anaerobic fermentation of the control (untreated) and the US-, Fenton- and US/Fenton-pretreated samples (350 mL) were conducted in 500 mL serum bottles. Nitrogen gas was flushed for 10 min to remove oxygen from the system, and then all bottles were sealed with butyl rubber stoppers and stirred in an air-bath shaker (SHZ-82, Zhiborui Inc., China) at 35 ± 1 °C and 100 rpm for 16 day. The variations in carbohydrate, protein and VFA contents were measured. Simultaneously, biogas was collected using Cali-5-Bond™ gas-sampling bags (1 L). Batch tests were carried out in duplicate for each test.2.4 Analytical methods
Supernatant was used to analyze soluble chemical oxygen demand (SCOD) along with carbohydrate, protein and VFA contents. Sludge samples were centrifuged at a speed of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 10 min, and the supernatant was then filtered through a 0.45 μm cellulose nitrate membrane filter. The supernatant was stored at 4 °C prior to analysis. Measurements of SCOD, total chemical oxygen demand (TCOD), total suspended solids (TSS) and volatile suspended solids (VSS) were conducted according to the standard methods.18 The pH value was measured with a pH meter (Seven Multi, Mettler Toledo, Switzerland). Carbohydrate content was measured by the phenol-sulfuric method with glucose as the standard.19 Soluble protein content was determined by the bicinchoninic acid method with bull serum albumin as the standard.20 VFAs were analyzed using gas chromatography (GC; Agilent 7890), and gas composition was determined by GC (Agilent 4890D).21
000 rpm for 10 min, and the supernatant was then filtered through a 0.45 μm cellulose nitrate membrane filter. The supernatant was stored at 4 °C prior to analysis. Measurements of SCOD, total chemical oxygen demand (TCOD), total suspended solids (TSS) and volatile suspended solids (VSS) were conducted according to the standard methods.18 The pH value was measured with a pH meter (Seven Multi, Mettler Toledo, Switzerland). Carbohydrate content was measured by the phenol-sulfuric method with glucose as the standard.19 Soluble protein content was determined by the bicinchoninic acid method with bull serum albumin as the standard.20 VFAs were analyzed using gas chromatography (GC; Agilent 7890), and gas composition was determined by GC (Agilent 4890D).21
The degree of sludge disintegration (DDCOD) was determined by measuring the variation in chemical oxygen demand (COD) according to eqn (1):
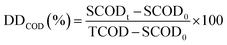 | (1) |
In this study, VFA content was determined as the sum of acetic (HAc), propionic (HPr), n-butyric (n-HBu), iso-butyric (iso-HBu), n-valeric (n-HVa) and iso-valeric (iso-HVa) acids. The COD conversion factors were 1.06 g COD/g carbohydrate (assumed as C6H12O6), 1.50 g COD/g protein (assumed as (C4H6.1O1.2N)x), 1.07 g COD/g HAc, 1.51 g COD/g HPr, 1.82 g COD/g HBu, and 2.04 g COD/g HVa.21
The particle size distributions of the sludge samples were determined by a Malvern particle size analyzer (Mastersizer-APA2000, UK) with a measurement range from 0.02 to 2000 μm.
3. Results and discussion
3.1 Effect of pretreatment on WAS disintegration
Sludge contains large amounts of organic matter, mostly in the form of solid particles. WAS pretreatment can enhance the release of soluble organics, which can be evaluated by DDCOD, soluble carbohydrate and protein contents. As shown in Fig. 1, DDCOD was 9.97%, 3.18% and 15.4%, after US, Fenton and US/Fenton pretreatments, respectively. Meanwhile, soluble carbohydrates and proteins increased with increasing DDCOD. The soluble carbohydrate concentration after US/Fenton treatment was 441 mg COD L−1, which was 2.36-fold, 2.17-fold and 17.11-fold more than those obtained for the Fenton, US and control tests, respectively. The corresponding concentration of soluble protein after US/Fenton treatment was 2059 mg COD L−1, which was 2.43-fold, 1.47-fold and 13.37-fold more than those obtained for the Fenton, US and control tests, respectively. These results are similar with those of previous studies and suggest that intracellular microbial organic matter in WAS can be effectively released into the extracellular environment by US/Fenton pretreatment.16 WAS disintegration occurred mainly as a result of hydro-mechanical shear forces produced by US cavitation bubbles,12 and longer treatment time or higher US intensity broke more microbial cell walls, releasing intracellular material into the liquid phase.4 Hydroxyl radicals produced by Fenton could also pass easily through the microbial cell membrane to discharge intracellular organic substances into the liquid phase.15 The release of substantial components after sludge pretreatment is beneficial for the subsequent sludge anaerobic fermentation. No significant increases in VFAs concentration were observed in any of the samples.Sludge disintegration was closely related to sludge particle size. The particle size distributions before and after treatment are shown in Fig. 2. For untreated WAS, the particle size profile presented a maximum representing the highest volume percentage (6.88%) at 50.24 μm, while the other three peaks were centered at 25.18 μm (5.54%, US), 44.77 μm (6.66%, Fenton) and 22.44 μm (5.3%, US/Fenton). The d50 (μm), the value that 50% of the volumes distribute below, was 44.01 μm for the untreated sludge; this was a little higher than the d50 for the Fenton-treated sludge (41.53 μm) and much higher than those of the US-treated (24.52 μm) and US/Fenton-treated (22.63 μm) sludges. The smaller particle size indicated that US/Fenton pretreatment significantly disrupts sludge floc structure and converts organic matter into soluble phases.17
Shock waves generated by US could disrupt large particles, leading to an increase in the number of smaller particles. Thus, US resulted in a greater decrease in particle size. In contrast, Fenton oxidation resulted in insignificant effects on particle size. This may be explained by the acidic pH and generation of ferric hydroxide and ferric hydroxo complexes.22
3.2 Consumption of soluble organics
Protein and carbohydrate accounted for almost 70–80% of the main constituents of WAS.11 Fig. 3 shows the changes in soluble carbohydrate and protein contents after different pretreatments with increasing anaerobic fermentation time. Soluble carbohydrate and protein concentrations in the pretreated sludge were higher than those in the untreated sludge, and both decreased with increasing fermentation time. In addition, the combined treatment presented obvious advantages for the release of organic matter compared to US and Fenton alone, while there was little variation for the untreated sludge.The maximum concentrations of soluble carbohydrate were 507 ± 40 mg COD L−1, 300 ± 18 mg COD L−1 and 204 ± 5 mg COD L−1, respectively, for US/Fenton-treated sludge, Fenton-treated sludge and US-treated sludge, respectively. Nevertheless, the soluble carbohydrate concentration was only 57 ± 6 mg COD L−1 in the control (Fig. 3a). In contrast, the peak concentrations of soluble protein were 2719 ± 127 mg COD L−1, 1650 ± 28 mg COD L−1 and 1399 ± 10 mg COD L−1 for US/Fenton-, Fenton-, and US-treated sludges, respectively, while it was only 684 ± 56 mg COD L−1 in the control (Fig. 3b). The change in protein concentration was stable after the third day because the release rate and degradation rate attained a balance; the protein concentration then decreased after the twelfth day because the degradation rate exceeded the release rate.23 The protein degradation rate was lower than the carbohydrate degradation rate, which may be due to the poor biodegradability of proteins compared to carbohydrates and lipids.24
3.3 VFAs production and composition
More hydrolysis products provided more substrates for acidification, and more VFAs could be obtained.25 As shown in Fig. 4, during fermentation, the accumulated VFAs concentration increased during the first three days and then decreased in the US-treated, Fenton-treated and untreated sludge. However, in the US/Fenton-treated sludge, the concentration of accumulated VFAs increased during the first eight days and then decreased. Meanwhile, the VFAs concentrations in all the pretreated sludges were higher than that in untreated sludge. The highest VFAs production was 3485 mg COD L−1 and 2700 mg COD L−1 for the US and Fenton treatments on the third day, while it was 4594 mg COD L−1 for the US/Fenton treatment on the eighth day. The time lag in VFAs production in the combined treatment test may be attributed to a lower acidogenic bacteria activity or lower biomass of acidogenic bacteria caused by the combined pretreatment; however, there was a remarkable increase in VFAs production when the acidogenic bacteria became enriched with increasing fermentation time.The comparison of VFAs production under different pretreatments is presented in Table 2. As been presented, US combined Fenton pretreatment for VFAs production from WAS was first mentioned in this study. US combined with acid or alkaline treatment (nos 1, 2 and 3) enhanced VFAs accumulation compared with US alone, regardless of whether the pH was adjusted initially or throughout the entire fermentation time.26–28 Meanwhile, US combined with biological methods (no. 4) also accelerated VFAs accumulation.29 Hydroxyl radicals produced by Fenton treatment could easily discharge intracellular organic substances into the liquid phase, which was beneficial for VFAs accumulation.15 Thus, the combined US/Fenton pretreatment can be seen as an effective way to promote VFAs accumulation.
| No. | Pretreated method and parameter | Fermentation pH | Fermentation time (day) | Temp. (°C) | Maximum VFAs production | Reference | |
|---|---|---|---|---|---|---|---|
| 1 | Ultrasound-alkaline | 20 kHz, 10 min, 1.0 kW L−1 | 10 | 3 | 21 ± 1 | 3110 mg COD L−1 | 26 |
| 2 | Ultrasound-acid | 28 kHz, 60 min, pH 3 | 6 | 10.5 | 35 ± 2 | 1700 mg L−1 | 27 |
| Ultrasound-alkaline | 28 kHz, 60 min; pH 12 | 6 | 10.5 | 35 ± 2 | 3700 mg L−1 | ||
| 3 | Ultrasound-alkaline | 20 kHz, 30 min, 2.0 kW L−1; pH 11 | Uncontrolled | 15 | 20 ± 2 | 1751 mg L−1 | 28 |
| 4 | Ultrasound-thermophilic bacteria | 40 kHz, 0.5 kW L−1, 10 min; 60 °C, 6 h, 10% Geobacillus sp. G1 | Uncontrolled | 4 | 35 | 4437 mg COD L−1 | 29 |
| 5 | Ultrasound-Fenton | 40 kHz, 0.4 kW L−1, 10 min; 0.04 g L−1 Fe2+, 10 g L−1 H2O2, pH 3 | Uncontrolled | 8 | 35 | 4594 mg COD L−1 | This study |
Short-chain fatty acids (C2 to C5), which can be formed directly from the fermentation of carbohydrates, proteins and lipids, are usually the main products of the acidogenic digestion of sludge.23 The VFAs composition and percentage corresponding to the maximum VFAs yield are detailed in Table 3 and Fig. 5. HAc is the most suitable substrate for many bioprocesses such as biogas production and biopolymer production.29 As shown in Table 3, for all tested pretreatments, when the maximum VFAs yield was achieved, HAc had the highest concentration among all acids. The highest HAc concentration after US/Fenton treatment was 2039 mg COD L−1, which was 1.72-fold and 1.78-fold those obtained in the Fenton and US tests, respectively. VFAs composition analysis (Fig. 5) revealed that HAc and HPr were the two main products, irrespective of the treatment. However, the VFAs composition was different among the pretreated tests and the control test. The order of VFAs abundance in the pretreated tests was HAc > HPr, while it was HPr > HAc in the control test. Therefore, the WAS pretreatment affected not only the types of particulates being hydrolyzed, but also the types of VFAs produced. This result was different from that of Yang et al., who detected that iso-HBu was the second major VFA component during the fermentation process.9 This difference may be caused by the different effects of the different pretreatment methods on the microbial communities in WAS.
| HAc | HPr | i-HBu | n-HBu | i-HVa | n-HVa | |
|---|---|---|---|---|---|---|
| a The units of individual VFA is mg COD L−1. The values are means of two replicates ± SD. | ||||||
| Control | 182 ± 5 | 237 ± 27 | 79 ± 4 | 55 ± 6 | 161 ± 12 | 36 ± 4 |
| US | 1146 ± 57 | 824 ± 18 | 252 ± 3 | 501 ± 25 | 413 ± 2 | 350 ± 9 |
| Fenton | 1188 ± 31 | 453 ± 21 | 198 ± 9 | 332 ± 21 | 296 ± 15 | 233 ± 16 |
| US/Fenton | 2039 ± 32 | 754 ± 24 | 348 ± 9 | 447 ± 11 | 524 ± 2 | 482 ± 10 |
3.4 Ultimate methane production
During sludge fermentation, the effect of pretreatment on methanogen activity expressed by ultimate methane production is shown in Fig. 6. Compared with untreated sludge, ultimate methane production was enhanced by the US and Fenton pretreatment. Meanwhile, the US/Fenton treatment produced the highest ultimate methane yield (232.80 mL/L-sludge), which was 1.4-fold, 1.17-fold and 5.2-fold of those obtained in the Fenton, US and control tests, respectively. The small variation in methane production between the US and US/Fenton pretreatments may be due to redundant H2O2, which could decrease the rate of methane production as well as the ultimate methane production of WAS via advanced oxidation.30 Thus, a higher VFAs accumulation was observed using the US/Fenton pretreatment. Meanwhile, the higher VFAs concentration induced a lower pH value, which had a negative effect on methanogenic bacteria.4. Conclusions
The effects of US, Fenton, and US/Fenton pretreatments on WAS hydrolysis and acidification were experimentally investigated in this study. The results showed that:(1) WAS organics could be effectively released from microbial intracellular environments into the extracellular environment by US/Fenton pretreatment.
(2) WAS acidification could also be enhanced by US/Fenton pretreatment.
(3) The ultimate methane production obtained in pretreated sludge was higher than that obtained in untreated sludge.
(4) The US/Fenton pretreatment had a synergetic effect on improving the hydrolysis and subsequent acidification process, making it more effective than individual US and Fenton treatments.
Acknowledgements
This work was supported by the National Natural Science Foundation (30470054); the Scientific and Technological Project of Liaoning Province (2001304024); the Natural Science Foundation of Liaoning Province (no. 20120132); Liaoning Province Science and the Cause of Public Research Fund (no. 20111012); the Bureau of Shenyang City Science and Technology Research Foundation (no. F12-277-1-39); and the City State Key Laboratory of Water Resources and Water Environment of open fund (no. HC201214).References
- L. Appels, J. Baeyens, J. Degreve and R. Dewil, Prog. Energy Combust. Sci., 2008, 34, 755–781 CrossRef CAS PubMed.
- X. Zhou, G. M. Jiang, Q. L. Wang and Z. G. Yuan, RSC Adv., 2014, 4, 50644–50652 RSC.
- S. S. Yang, W. Q. Guo, Y. D. Chen, X. J. Zhou, H. S. Zheng, X. H. Feng, R. L. Yin and N. Q. Ren, RSC Adv., 2014, 4, 52892–52897 RSC.
- A. Tiehm, K. Nickel, M. Zellhorn and U. Neis, Water Res., 2001, 35, 2003–2009 CrossRef CAS.
- W. S. Lee, A. S. M. Chua, H. K. Yeoh and G. C. Ngoh, Chem. Eng. J., 2014, 235, 83–99 CrossRef CAS PubMed.
- D. H. Kim, E. Jeong, S. E. Oh and H. S. Shin, Water Res., 2010, 44, 3093–3100 CrossRef CAS PubMed.
- A. J. Zhou, C. X. Yang, F. Y. Kong, D. D. Liu, Z. B. Chen, N. Q. Ren and A. J. Wang, J. Environ. Biol., 2013, 34, 381–389 CAS.
- M. Tokumura, M. Sekine, M. Yoshinari, H. T. Znad and Y. Kawase, Process Biochem., 2007, 42, 627–633 CrossRef CAS PubMed.
- C. X. Yang, W. Z. Liu, Z. W. He, S. Thangavel, L. Wang, A. J. Zhou and A. J. Wang, Bioresour. Technol., 2014, 175, 509–516 CrossRef PubMed.
- A. M. Yeneneh, S. Chong, T. K. Sen, H. M. Ang and A. Kayaalp, Water, Air, Soil Pollut., 2013, 224, 1559–1567 CrossRef.
- S. Pilli, P. Bhunia, S. Yan, R. J. LeBlanc, R. D. Tyagi and R. Y. Surampalli, Ultrason. Sonochem., 2011, 18, 1–18 CrossRef CAS PubMed.
- F. Wang, Y. Wang and M. Ji, J. Hazard. Mater., 2005, 123, 145–150 CrossRef CAS PubMed.
- V. K. Tyagi, S. L. Lo, L. Appels and R. Dewil, Crit. Rev. Environ. Sci. Technol., 2014, 44, 1220–1288 CrossRef PubMed.
- E. Neyens and J. Baeyens, J. Hazard. Mater., 2003, 98, 33–50 CrossRef CAS.
- J. L. de Morais and P. P. Zamora, J. Hazard. Mater., 2005, 123, 181–186 CrossRef PubMed.
- S. Qiu, M. J. Xia and Z. S. Li, Water Sci. Technol., 2013, 67, 1701–1707 CrossRef CAS PubMed.
- X. A. Ning, H. Chen, J. R. Wu, Y. J. Wang, J. Y. Liu and M. Q. Lin, Chem. Eng. J., 2014, 242, 102–108 CrossRef CAS PubMed.
- APHA, The Standard Methods for the Examination of Water and Wastewater, American Public Health Association, Washington, DC, USA, 21st edn, 2005 Search PubMed.
- D. Herbert, P. J. Phipps and R. E. Strange, in Methods in Microbiology, ed. J. R. Norris and D. W. Ribbons, Academic Press, 1971, part B, pp. 209–344 Search PubMed.
- P. K. Smith, R. I. Krohn, G. T. Hermanson, A. K. Mallia, F. H. Gartner, M. D. Provenzano, E. K. Fujimoto, N. M. Goeke, B. J. Olson and D. C. Klenk, Anal. Biochem., 1985, 150, 76–85 CrossRef CAS.
- A. J. Zhou, J. W. Du, C. Varrone, Y. Z. Wang, A. J. Wang and W. Z. Liu, Process Biochem., 2014, 49, 283–289 CrossRef CAS PubMed.
- T. T. H. Pham, S. K. Brar, R. D. Tyagi and R. Y. Surampalli, Ultrason. Sonochem., 2010, 17, 38–45 CrossRef CAS PubMed.
- Y. G. Chen, S. Jiang, H. Y. Yuan, Q. Zhou and G. W. Gu, Water Res., 2007, 41, 683–689 CrossRef CAS PubMed.
- Y. Miron, G. Zeeman, J. B. Van Lier and G. Lettinga, Water Res., 2000, 34, 1705–1713 CrossRef CAS.
- Z. Y. Ji, G. L. Chen and Y. G. Chen, Bioresour. Technol., 2010, 101, 3457–3462 CrossRef CAS PubMed.
- Y. Y. Yan, L. Y. Feng, C. J. Zhang, C. Wisniewski and Q. Zhou, Water Res., 2010, 44, 3329–3336 CrossRef CAS PubMed.
- X. L. Liu, H. Liu, J. H. Chen, G. C. Du and J. Chen, Waste Manage., 2008, 28, 2614–2622 CrossRef CAS PubMed.
- X. R. Kang, G. M. Zhang, L. Chen, W. Y. Dong and W. D. Tian, Ind. Eng. Chem. Res., 2011, 50, 12372–12378 CrossRef CAS.
- C. X. Yang, A. J. Zhou, Z. W. He, L. Jiang, Z. C. Guo, A. J. Wang and W. Z. Liu, Environ. Sci. Pollut. Res., 2015, 1–10 Search PubMed.
- C. Eskicioglu, A. Prorot, J. Marin, R. L. Droste and K. J. Kennedy, Water Res., 2008, 42, 4674–4682 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2015 |






