Label-free and turn-on fluorescent cyanide sensor based on CdTe quantum dots using silver nanoparticles
Ali A. Ensafi*,
N. Kazemifard and
B. Rezaei
Department of Analytical Chemistry, College of Chemistry, Isfahan University of Technology, Isfahan 84156-83111, Iran. E-mail: Ensafi@cc.iut.ac.ir; Ensafi@yahoo.com; Fax: +98-31-33912350; Tel: +98-31-33913269
First published on 17th April 2015
Abstract
Silver nanoparticles were used to develop a simple turn-on fluorescent assay based on glutathione-capped CdTe quantum dot for the determination of trace amounts of the lethal poison, cyanide. It was found that the fluorescence intensity of glutathione-capped CdTe quantum dots increased with increasing cyanide concentration. Several experimental variables, such as pH and amounts of quantum dots and silver nanoparticles and their effect on the analytical signals, were optimized. Using this optical sensor under optimum conditions, cyanide was measured in the range of 0.01–2.5 μg mL−1 with a detection limit as low as 0.004 μg mL−1. Relative standard deviations of 2.0% (for 0.5 μg mL−1, n = 10) and 1.8% (for 2.0 μg mL−1, n = 10) were obtained. Investigation of the effects of potential interfering anions on the response of the sensor revealed its high selectivity for the detection of cyanide in real samples. High sensitivity, superior selectivity, low detection limit (0.004 μg mL−1) and ease of production are the most important advantages of the present sensor. Finally, the sensor was applied for the determination of cyanide in real samples.
1. Introduction
Quantum dots (QDs) are brightly luminescent semiconductor nanoparticles that have found wide applications in bioanalysis and bioimaging in recent decades. This is due to their unique photoproperties such as a wide UV-Vis absorption spectrum and a narrow photoluminescence spectrum.1 Compared to organic dyes, QDs have about 10–20 times brighter fluorescence and 100–200 times better photostability.2 A lot of studies in a variety of fields have been devoted over decades for the interesting optical properties of nanoparticles made with silver and gold (SNPs and GNPs, respectively).3Plasmon resonance absorption of SNPs and GNPs have molar extinction coefficients (∼3 × 1011 M−1 cm−1) that make them good energy acceptors so that they often serve the role of quenchers in fluorimetric methods.4–8 The fluorescence of CdTe-QDs is significantly reduced by SNPs to the extent that their self-quenching effect could be eliminated.
Cyanide is one of the most lethal toxins; moreover, 300 μg mL−1 of this poison would be enough to kill a man quickly. Cyanide's toxicity to humans lies in its ability to suppress oxygen transfer via its binding to the active sites of cytochrome C oxidase that results in hypoxia.9 Long-term exposure to low amounts of cyanide also affects the central nervous system.10 Cyanide poisoning may also occur through inhaling emissions from residential applications, metal plating, metal mining, plastic manufacturing, and metal processing industries.11
Several analytical methods have been developed for the detection of cyanide, including liquid chromatography-mass spectrometry,12 fluorimetric13–15 chemiluminescence,9,16 colorimetric17,18 and electrochemical methods.19 A few methods have also been reported for the fluorimetric determination of cyanide using QDs.20–24 Among these, fluorescent sensors enjoy many advantages. They are highly sensitive, inexpensive, easy to use, and especially suitable as a diagnostic device for analytical purposes. Moreover, they can be employed hands-free via a remote control.
Quantum dots based on organic dyes as the fluorophore have attractive properties and several advantages.25 Cyanide could enhance the fluorescence of CdTe QDs; however, the linear range was narrow and the selectivity was poor. To resolve this problem, Shang and et al. used copper ion-modified CdTe quantum dots.20 The fluorescence of CdTe QDs was quenched by the copper ions. However, in the presence of CN−, copper ions could be desorbed from the surface of the QDs and therefore the fluorescence of CdTe QDs increased. In a similar report for CN− detection, copper ion was used as a carbon dot fluorescence quencher.24 Cyanide is well-known to be capable of dissolving nanometals such as Ag in the presence of oxygen and in a basic solution.26 In addition, the fluorescence of CdTe QDs could be quenched by silver nanoparticles.
Herein, a novel and simple fluorimetric sensor is reported for cyanide detection based on glutathione-capped cadmium telluride quantum dot (GSH-capped CdTe QDs). One limitation of the fluorescence detection at high fluorescence intensity is self-quenching. In this study, SNPs were used as a quencher to overcome the problem. The method was observed to be highly selective and sensitive for the detection of cyanide contents as low as 0.004 μg mL−1. Moreover, it was found that the detection limit and dynamic range of the proposed sensor was comparable with other fluorescence methods reported in the literature for measuring cyanide (Table 1).
| Fluorophore | Linear dynamic range (μg mL−1) | Detection limit (μg mL−1) | Mode of assay | Ref. |
|---|---|---|---|---|
| Poly[2-methoxy-5-(3,7-dimethyloctyloxy)-1,4 phenylenevinylene] | 0.026–15.6 | 0.0156 | Enhancement | 13 |
| Rhodamine B | 0.01–3.12 | 0.001 | Enhancement | 14 |
| Copper ion-modified CdTe-QD | 0.0078–0.312 | 0.0039 | Enhancement | 20 |
| 2-Mercaptoethane sulfonate-modified CdSe-QD | Up to 6.5 | 0.029 | Quenching | 21 |
| 2-Mercaptoethanol-capped ZnS-QD | 0.063–0.67 | 0.0044 | Quenching | 23 |
| Glutathione-capped CdTe-QD | 0.01–2.50 | 0.004 | Enhancement | This work |
2. Experimental
2.1. Chemicals
Glutathione, sodium tellurite, CdCl2 and NaBH4 were purchased from Aldrich. AgNO3 and trisodium citrate were purchased from Merck. All other analytical reagent grade chemicals (with the highest degree of purity available), 0.01 mol L−1 carbonate buffer solution and deionized water were used throughout.A 50.0 μg mL−1 stock solution of cyanide was prepared by dissolving an appropriate amount of NaCN into a 100 mL standard flask.
A 0.040 mol L−1 CdCl2 and Na2TeO3 (0.010 mol L−1) as precursors were prepared with highest purity available chemicals using deionized water.
2.2. Apparatus
UV-VIS absorption spectra were obtained using a Jasco V-570 UV/Vis/NIR spectrophotometer.The luminescence spectra were obtained on a Jasco FP-750 spectrofluorometer. The slit widths for the excitation and emission were fixed at 10.0 nm.
Transmission electron microscopy (TEM) experiments were carried out with a Philips CM30 300 kV TEM.
2.3. Preparation of GSH-capped CdTe QDs
The GSH-capped CdTe QDs were prepared according to the protocol reported in the literature.27 Briefly, 2.0 mL of 0.04 mol L−1 CdCl2 was diluted to 50 mL. Then, 0.050 g trisodium citrate dihydrate, 0.025 g glutathione, 2.0 mL of 0.010 mol L−1 Na2TeO3, and 0.025 g NaBH4 were added to the CdCl2 solution with stirring at room temperature. After 2 h, the mixture was refluxed for 12 h at 90 °C. The resulting solution was transferred to a dark container and kept at 4 °C.2.4. Preparation of citrate-stabilized SNPs
SNPs were prepared via a previously reported method.26 Briefly, 0.125 mL of 0.10 mol L−1 AgNO3 and 0.125 mL of 0.10 mol L−1 sodium citrate were added to 50 mL of water with continued stirring. Then, 3 mL of a freshly prepared 5.0 mmol L−1 NaBH4 was added into the abovementioned aqueous solution and stirred for 30 min. The resulting yellow colloidal SNPs solution was stored at 4 °C overnight before use.2.5. Measurement procedure
For the determination of cyanide, a freshly prepared mixture containing 0.45 nmol L−1 of GSH-capped CdTe QDs and 8.0 nmol L−1 of SNPs in carbonate buffer (pH 10.0), plus an appropriate volume of sample solution was mixed. Then, the fluorescence spectrum of the solution was recorded at 550–700 nm upon excitation at 400 nm. The slit widths of both the excitation and the emission were set to 10.0 nm. The response function (F − F0) values of the sensor were obtained with different concentrations of cyanide, where F and F0 are the fluorescence intensities at 615 nm in the presence and absence of cyanide, respectively.2.6. Sample preparation
Real samples of human serum and wastewater were filtered to remove any particles before analysis. The standard addition method was used for the determination of cyanide. 1.0 mL of each real sample (with and without standard solution) was transferred into a vial containing 0.45 nmol L−1 of GSH-capped CdTe QDs and 8.0 nmol L−1 of SNPs at pH 10.0. Then, the fluorescence spectrum was recorded at 550–700 nm upon excitation at 400 nm.3. Results and discussion
3.1. TEM and UV-Vis absorption
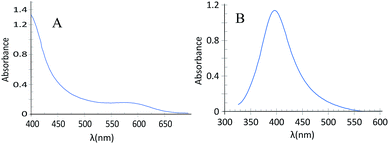
TEM was used to characterize the GSH-capped CdTe QDs and SNPs. The image of the colloidal GSH-capped CdTe QDs solution (Fig. 1A) shows that the nanoparticles were mostly round in shape with an average particle size of ∼8 nm. The UV-Vis absorption spectrum of the GSH-capped CdTe QDs (Fig. 2A) shows a wide UV-Vis absorption spectrum as expected for the quantum dots. The spectrum was employed to determine the GSH-capped CdTe QDs content.28 The amount of GSH-capped CdTe QDs was calculated to be 0.34 μmol L−1.TEM analysis was also used to verify the size of the SNPs (Fig. 1B). Based on the TEM image of the colloidal SNPs solution, the nanoparticles were mostly round in shape with an average particle size of ∼11 nm. The UV-Vis absorption spectrum of the synthesized SNPs is shown in Fig. 2B, as can be seen the absorption maximum wavelength is ∼400 nm.
3.2. Operating principles
The fluorescence intensity of GSH-capped CdTe QDs increased in the presence of cyanide. Given the fact that the photoluminescence of QDs arises from electron–hole recombination, it is logical to expect that changes in the surface charges or QD components caused by chemical or physical interactions between ions or small molecules and the QDs might affect the efficiency of the electron–hole recombination and, thereby the luminescent emission.29At high cyanide concentrations (above 0.5 μg mL−1), the fluorescence intensity of GSH-capped CdTe QDs (in the absence of SNPs) was reduced because of the self-absorption effect of GSH-capped CdTe QDs (Fig. 3). In this study, this technical problem (self-absorption) was resolved by adding SNPs.
 | ||
| Fig. 3 Effect of CN− on the fluorescence intensity of 0.45 nmol L−1 GSH-capped CdTe QDs at pH 10.0 in the absence of SNPs. | ||
The energy transfer or inner filter effect (IFE) of fluorescence refers to the absorption of the excitation and/or emission of light by absorbers in the detection system. As can be seen in Fig. 2B (from the absorption spectrum of CdTe-QDs and that of SNPs), there is no obvious overlap between the spectrum of CdTe-QDs and that of the SNPs. Therefore, it does not reflect any evidence of an IFE. However, as can be seen in this figure, the maximum wavelength of the SNPs was about 400 nm, i.e., equal to the excitation wavelength of CdTe-QDs. Therefore, the photoluminescence emission intensity of GSH-capped CdTe QDs was reduced in the presence of SNPs. After addition of cyanide ions, the luminescent emission of GSH-capped CdTe QDs was increased. This is due to the fact that in the presence of cyanide, the SNPs are dissolved in the form of Ag(CN)n(n−1)−, therefore the plasmon absorption band of the SNPs decreased in intensity.26 As observed by absorption spectroscopy (Fig. 4), the absorption band of the SNPs decreased gradually.
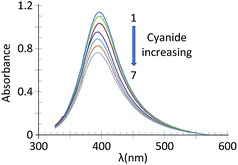 | ||
| Fig. 4 UV-Vis absorption spectra of SNPs at pH 10.0 containing different concentrations of cyanide: (1) 0.00; (2) 0.015; (3) 0.5; (4) 0.15; (5) 1.00; (6) 1.50; (7) 2.00; and (8) 2.50 μg mL−1. | ||
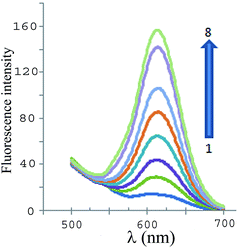
Scheme 1 shows the trend of the fluorescence signal of the sensor by the addition of cyanide ions. The zeta potential of CdTe-QDs was found to be negative at pH 10. Moreover, citrate-stabilized SNPs were negatively charged and, thus no obvious interactions (e.g., electrostatic binding) were expected to occur with GSH-capped CdTe QDs. Fig. 5 shows the fluorescence emission spectra of a solution containing different amounts of cyanide (under the optimum conditions). As can be seen in Fig. 5, the proposed sensor works in a ‘turn-on’ mode, which is generally more sensitive than the turn-off assay mode because the enhanced fluorescence of the signal transduction gives a much better signal-to-noise ratio for the sensing scheme.30
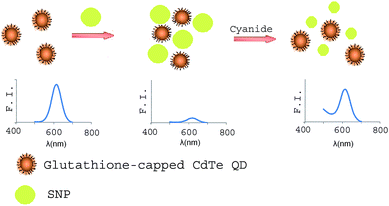 | ||
| Scheme 1 A turn-on fluorescent cyanide sensor based on GSH-capped CdTe QDs using silver nanoparticles. | ||
3.3. Effect of sample solution pH
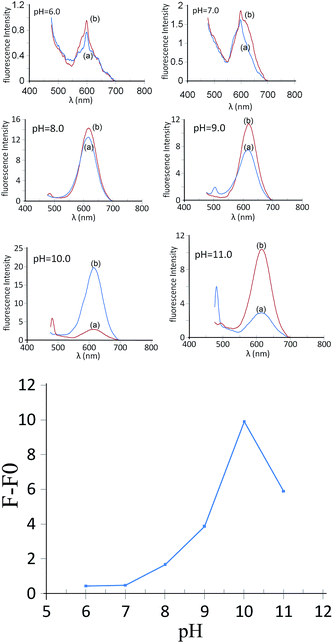
One method most commonly used for dispersing QDs in an aqueous solution is to modify their outer surface with anionic carboxylate groups. In this experiment, the fluorescence of the CdTe solution was depended strongly on the solution pH. This means that a suitable buffer should be used to maintain the pH of the aqueous solution at a constant level. For this purpose, the response of the sensor was measured at different pH values (6.0–11.0) using solutions containing 0.28 nmol L−1 of GSH-capped CdTe QDs, 8.0 nmol L−1 of SNPs and 1.0 μg mL−1 cyanide (Fig. 6A and B). As can be seen, the best pH value for the determination of cyanide by the sensor was 10.0. Since the pKa of the –COOH group in glutathione is 3.6, the zeta potential of the GSH-capped CdTe QDs was negative at pH 10.0. At an adequately basic pH, the electrostatic repulsion between GSH-capped CdTe QDs afforded a stable colloidal suspension, whereas an acidic pH yielded insoluble aggregates of GSH-capped CdTe QDs.1 At higher pH levels, however, GSH-capped CdTe QDs may dissolve and the response may also be decreased simultaneously in higher pH medium solutions (pH > 10). This is due to the fact that production of too many –OH groups on the surface of QDs would hinder cyanide and GSH-capped CdTe QDs interactions. Therefore, pH 10.0 was selected as the optimum pH for further experiments.3.4. Effect of the amount of GSH-capped CdTe QDs
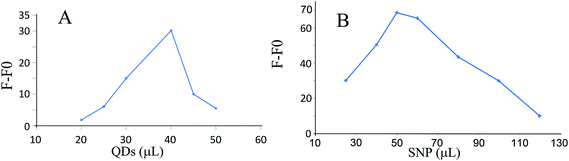
In order to optimize the amount of GSH-capped CdTe QDs in solution, six solutions were prepared with different volumes of GSH-capped CdTe QDs (15 to 50 μL of 34.0 nmol L−1) in the presence of 8.0 nmol L−1 of SNPs and 1.0 μg mL−1 cyanide at pH 10.0. The results are shown in Fig. 7A and clearly confirm that a solution containing 0.45 nmol L−1 of GSH-capped CdTe QDs yielded the best response to cyanide. Moreover, the sensitivity of the sensor obviously declined with decreasing the amounts of GSH-capped CdTe QDs. A high GSH-capped CdTe QDs content may lead to the self-absorption of the GSH-capped CdTe QDs fluorescence.3.5. Effect of the amount of SNPs
SNPs were used to inhibit self-absorption during the fluorescence measurements. The influence of the amount of SNPs on the sensor response was investigated by preparing several solutions with different volumes (25 to 120 μL) of 0.24 μmol L−1 SNPs in the presence of 0.45 nmol L−1 of GSH-capped CdTe QDs and 1.0 μg mL−1 cyanide at pH 10.0. Based on the results (Fig. 7B), a solution containing 4.0 nmol L−1 of SNPs yielded the best response to cyanide. Higher amounts of SNPs were found to reduce the sensor sensitivity.4. Analytical figures of merit
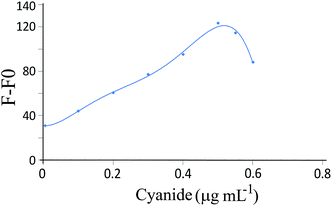
Under optimized conditions, the response function (F − F0) values of the sensor were obtained for different cyanide concentrations. Fig. 8 shows the calibration curve under the optimum conditions. The detection limit (3Sb/m, where S is the blank standard deviation (n = 10) and m is the slope of the calibration curve) was obtained to be 0.004 μg mL−1.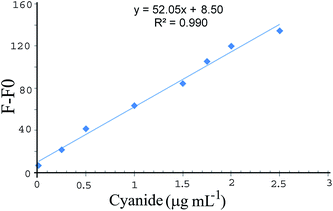 | ||
| Fig. 8 Calibration graph for the cyanide determination at the optimum conditions. Conditions: pH, 10.0; 0.45 nmol L−1 of GSH-capped CdTe QDs and 4.0 nmol L−1 SNPs. | ||
To consider the repeatability of the sensor, 0.5 and 2.0 μg mL−1 of cyanide solution were measured ten times. The results showed RSD% values of 2.0% and 1.8% for cyanide solutions of 0.5 and 2.0 μg mL−1, respectively.
5. Selectivity
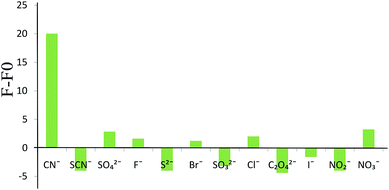
The potential interference of common anions on the selectivity of the sensor was investigated under optimum conditions. For this purpose, the optical sensor responses to several anions were examined. The tested anions included 20.0 μg mL−1 Br−, Cl−, ClO3−, C2O42−, F−, I−, NO2−, SO32−, SO42−, S2−, NO3− and SCN−, and 0.20 μg mL−1 CN−. As can be seen in Fig. 9, the proposed sensor exhibits a better selectivity for cyanide than for the other ions examined.6. Application
The applicability of the sensor for real sample analysis was investigated to analyze spiked human serum samples. In addition, to check the accuracy of the sensor, a potentiometric standard method (using CN− selective electrode) was used to measure the cyanide content in a wastewater sample. The results are given in Table 2 confirm the acceptable recovery and accuracy of the sensor.| Samples | CN− added, (μg mL−1) | CN− founda, proposed sensor (μg mL−1) | Recovery (%) | CN− founda, potentiometric method (μg mL−1) |
|---|---|---|---|---|
| a Average values of six determinations ± standard deviations. | ||||
| Human serum sample | 0.50 | 0.49 ± 0.01 | 98.0 | — |
| 1.50 | 1.52 ± 0.03 | 101.3 | — | |
| 2.00 | 1.97 ± 0.02 | 98.5 | — | |
| Wastewater | — | 2.38 ± 0.02 | — | 2.30 ± 0.13 |
7. Conclusion
A new optical sensor based on GSH-capped CdTe QDs was developed for the detection of cyanide at ultra-trace levels. The fluorescence intensity of the sensor was considerably enhanced in the presence of cyanide. The sensor was capable of determining cyanide content in the range of 0.01–2.5 μg mL−1. The sensor was also found to work in a ‘turn-on’ mode, which is usually more sensitive than a “turn-off” assay. Higher sensitivity, superior selectivity, low detection limit (0.004 μg mL−1) and ease of production are the most important advantages of the proposed sensor. Finally, the sensor was successfully employed for the determination of cyanide in real samples.Acknowledgements
The authors wish to thank Isfahan University of Technology (IUT) Research Council and Center of Excellence in Sensor and Green Chemistry for their support.References
- E. Petryayeva, W. R. Algar and I. L. Medintz, Appl. Spectrosc., 2013, 67, 215–252 CrossRef CAS PubMed.
- A. M. Smith and S. Nie, Analyst, 2004, 129, 672–677 RSC.
- M. R. Hormozi Nezhad, M. A. Karimi and F. Shahheydari, Nanotechnology, 2010, 17, 148–153 Search PubMed.
- M. A. El-Sayed, Acc. Chem. Res., 2001, 34, 257–264 CrossRef CAS PubMed.
- A. C. Templeton, W. P. Wuelfing and R. W. Murray, Acc. Chem. Res., 2000, 33, 27–36 CrossRef CAS PubMed.
- Y. Sun and Y. Xia, Analyst, 2003, 128, 686–691 RSC.
- A. D. Mc-Farland and R. P. Van Duyne, Nano Lett., 2003, 3, 1057–1062 CrossRef CAS.
- T. R. Jensen, M. Duval Malinsky, C. L. Haynes and R. P. Van Dune, J. Phys. Chem. B, 2000, 104, 10549–10556 CrossRef CAS.
- M. Amjadi, J. Hassanzadeh and J. L. Manzoori, Microchim. Acta, 2014, 181, 1851–1856 CrossRef CAS.
- The Agency for Toxic Substances and Disease Registry, Toxicological Profile For Cyanide, US Department of Health and Human Services, Atlanta, GA, 2006, pp. 221–228 Search PubMed.
- R. Badugu, J. R. Lakowicz and C. D. Geddes, Dyes Pigm., 2005, 64, 49–55 CrossRef CAS PubMed.
- C. Lacroix, E. Saussereau, F. Boulanger and J. P. Goullé, J. Anal. Toxicol., 2011, 35, 143–147 CrossRef CAS PubMed.
- L. Shang and S. Dong, Anal. Chem., 2009, 81, 1465–1470 CrossRef CAS PubMed.
- Y. Zhai, L. Jin, P. Wang and S. Dong, Chem. Commun., 2011, 47, 8268–8270 RSC.
- Y. Sun, S. Fan, D. Zhao, L. Duan and R. Li, J. Fluoresc., 2013, 23, 1255–1261 CrossRef CAS PubMed.
- A. V. Gavrilov, A. A. Druzhinin, K. I. Zakharov, V. A. Ishutin, S. A. Nemkov and I. A. Pushkin, J. Anal. Chem., 2005, 60, 1029–1034 CrossRef CAS PubMed.
- A. R. Surleva and G. Drochioiu, J. Chem. Educ., 2013, 90, 1654–1657 CrossRef CAS.
- A. R. Surleva, S. Bancila and E. V. Todorova, Sci. J. Anal. Chem., 2014, 2, 1–6 CrossRef.
- A. Safavi, N. Maleki and H. R. Shahbaazi, Anal. Chim. Acta, 2004, 503, 213–221 CrossRef CAS PubMed.
- L. Shang, L. Zhang and S. Dong, Analyst, 2009, 134, 107–113 RSC.
- W. J. Jin, M. T. Fernandez-Arguelles, J. M. Costa-Fernandez, R. Pereiro and A. Sanz-Medel, Chem. Commun., 2005, 15, 883–885 RSC.
- W. J. Jin, J. M. Costa-Fernandez, R. Pereiro and A. Sanz-Medel, Anal. Chim. Acta, 2004, 522, 1–8 CrossRef CAS PubMed.
- M. Shamsipur and H. R. Rajabi, Mater. Sci. Eng., C, 2014, 36, 139–145 CrossRef CAS PubMed.
- Y. Dong, R. Wang, W. Tian, Y. Chi and G. Chen, RSC Adv., 2014, 4, 3701–3705 CAS.
- U. Resch-Genger, M. Grabolle, S. Cavaliere-Jaricot, R. Nitschke and T. Nann, Nat. Methods, 2008, 5, 763–775 CrossRef CAS PubMed.
- L. Shang, C. Qin, L. Jin, L. Wang and S. Dong, Analyst, 2009, 134, 1477–1482 RSC.
- X. Cui, M. Liu and B. Li, Analyst, 2012, 137, 3293–3299 RSC.
- W. W. Yu, L. Qu, W. Guo and X. Peng, Chem. Mater., 2003, 15, 2854–2860 CrossRef CAS.
- D. E. Moore and K. Patel, Langmuir, 2001, 17, 2541–2544 CrossRef CAS.
- R. Badugu, J. R. Lakowicz and C. D. Geddes, J. Am. Chem. Soc., 2005, 127, 3635–3641 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2015 |