Synergistic effect in bimetallic copper–silver (CuxAg) nanoparticles enhances silicon conversion in Rochow reaction†
Zailei
Zhang
*a,
Yongjun
Ji
a,
Jing
Li
a,
Ziyi
Zhong
b and
Fabing
Su
*a
aState Key Laboratory of Multiphase Complex Systems, Institute of Process Engineering, Chinese Academy of Sciences, Beijing, China 100190. E-mail: zhangzl@ipe.ac.cn; fbsu@ipe.ac.cn; Fax: +86-10-82544851; Tel: +86-10-82544850
bInstitute of Chemical Engineering and Sciences, A*star, 1 Pesek Road, Jurong Island, Singapore 627833
First published on 16th June 2015
Abstract
The oleylamine thermal reduction process was employed to prepare bimetallic copper–silver (CuxAg (0 ≤ x ≤ 50)) nanoparticles, such as Cu, Cu50Ag, Cu20Ag, Cu10Ag, Cu5Ag, CuAg, CuAg2, and Ag, by using Cu(CH3COO)2 and AgNO3 as the precursors. The samples were characterized by X-ray diffraction, transmission electron microscopy, thermogravimetric analysis, and X-ray photoelectron spectroscopy. The CuxAg hybrid nanostructure showed good particle dispersion, and Cu and Ag metals were well mixed. The catalytic properties of these bimetallic CuxAg nanoparticles as model catalysts for the Rochow reaction were explored. Compared to monometallic Cu and Ag nanoparticles, bimetallic CuxAg nanoparticles resulted in a much higher silicon conversion, which is attributed to the synergistic electronic effect between Cu and Ag metals. For example, the Cu atom was observed to have a lower electron density in the CuxAg bimetallic nanoparticle than that in monometallic Cu nanoparticles, which enhanced the formation of methylchlorosilanes on the silicon surface with chloromethane, demonstrating the significance of the CuxAg bimetallic catalysts in catalytic reactions during organosilane synthesis. The insights gained in this study should be conducive to the design of good Cu-based catalysts for the Rochow reaction.
1. Introduction
Recently, the fabrication of bimetallic nanoparticles has been of great interest because of the ample opportunities provided to modify and enhance their optical and electronic properties via engineering of the particle composition, structure and even geometric morphology.1–8 Many bimetallic nanoparticles, such as AuCu,9 AgCu,10 RuCu,11 PdCu,12 PtCu,13 FeCu,14 NiCu,15 PdAu,16 PtPd,17 PdRu,18 PtCo,19 and PtNi20 have been applied in aerobic oxidation,9 the oxygen reduction reaction,10,20 hydrogenation,11,16 the Sonogashira reaction,12 oxidation of formic acid,13 degradation of organic contaminants,14 hydrogen production,15 ethanol electro-oxidation,17 methanol electro-oxidation,18 and hydrogenolysis.19 In particular, several reports revealed that Cu-based bimetallic catalysts exhibited much higher activity than monometallic Cu particles in catalytic reactions, e.g., CuAu improved the reduction of carbon dioxide,1 CuPd enhanced the oxidation of cyclohexane by hydrogen peroxide,21 CuAg enhanced the oxidation of methanol to CO2,22 and CuRu lowered the activation energy for the hydrolytic dehydrogenation of ammonia borane,23etc.Rochow reaction was discovered in 1940s as the direct synthesis route to produce methylchlorosilanes via the reaction of silicon (Si) with chloromethane on Cu-based catalysts.24 Over the past few decades, many researches have indicated that the Cu-based catalysts are the main catalysts applicable to Rochow reaction,25–28 and Zn and ZnO are effective in promoting the Cu-based catalyst by increasing the dimethyldichlorosilane selectivity and Si conversion.29,30 In general, the Cu-based catalysts are mixed with Zn or ZnO promoters both in the organosilane industry and in fundamental studies. In the past five years, our group has studied the Cu,31 Cu2O,32,33 Cu@Cu2O,34 CuO,35,36 Cu–Cu2O–CuO composites,37,38 and CuCl39 as active components for this reaction, and found that the catalyst composition, structure, size, and shape have significant effect on it. However, the above catalysts still suffer from low Si conversion, limiting their practical application. Because of the presence of the synergistic effect in some bimetallic catalysts,22,40 and the vast opportunities for engineering the particle composition, size and shape, etc., it is expected that the bimetallic catalysts may have high potentiality for Rochow reaction.1,41
Herein, we synthesize the bimetallic CuxAg (0 ≤ x ≤ 50) nanoparticles via a oleylamine thermal reduction process using Cu(CH3COO)2 and AgNO3 as the precursors. The composite of CuxAg nanoparticles can be easily controlled by adjusting the ratio of metal precursors. These bimetallic CuxAg (such as Cu50Ag, Cu20Ag, Cu10Ag, Cu5Ag, CuAg, and CuAg2) nanoparticles are used as model catalysts for Rochow reaction. Compared with the Si conversion for monometallic Cu (35.4% for Cu) and Ag (0% for Ag) nanoparticles, these bimetallic CuxAg nanoparticles exhibit much enhanced Si conversion (65.4% for Cu50Ag, 68.6% for Cu20Ag, 71.1% for Cu10Ag, 73.0% for Cu5Ag, and 70.3% for CuAg), demonstrating the synergistic electronic effect between Cu and Ag atoms in the AgCu bimetallic nanoparticles, which led to the lower electron density for Cu atoms, in the Rochow reaction. This work provides useful clues for improving the Cu-based catalysts to achieve higher Si conversion in Rochow reaction.
2. Experimental
2.1. Synthesis of CuxAg (0 ≤ x ≤ 50) nanoparticles
A modified thermal decomposition process was used to synthesize the CuxAg bimetallic nanoparticles. AgNO3 (A.R., Sinopharm Chemical Reagent Co., Ltd, China) was used as the Ag precursor, Cu(CH3COO)2 (A.R., Sinopharm Chemical Reagent Co., Ltd, China) as the Cu precursor, and oleylamine (C18H37N, Sigma-Aldrich) as the solvent, surfactant and reducing agent.42,43 All chemicals were used as received without further purification. In a typical synthesis for CuAg nanoparticles, 10 mmol of Cu(CH3COO)2 and 10 mmol of AgNO3 were added to 80 mL of oleylamine in a three neck flask, which was then heated to 180 °C for 8 h with stirring. The nanoparticles were washed with ethanol (A.R., Sinopharm Chemical Reagent Co., Ltd, China) and hexamethylene (A.R., Sinopharm Chemical Reagent Co., Ltd, China), and separated by several rounds of centrifugation. The purified nanoparticles were then dried at 60 °C for more than 24 h in a vacuum oven. Similarly, Cu, Cu50Ag, Cu20Ag, Cu10Ag, Cu5Ag, CuAg2, and Ag nanoparticles were prepared by varying the ratio of Cu and Ag precursors. Meanwhile, Cu–Ag composites were prepared as a comparison in the following way: 10 mmol of Cu(CH3COO)2 was added to 80 mL of oleylamine in a three-neck flask, which was then heated to 180 °C for 8 h with stirring. Subsequently, 10 mmol of AgNO3 was added to the three-neck flask and the solution was stirred for another 8 h. The nanoparticles were washed with ethanol and hexamethylene via centrifugation, and dried in a vacuum oven at 60 °C for 24 h. In addition, Cu + Ag composites were prepared by mixing the same weight of Cu nanoparticles and Ag nanoparticles.2.2. Materials characterization
X-ray diffraction patterns (XRD) were recorded on a PANalytical X'Pert PRO MPD using the Cu Kα radiation of (λ = 1.5418 Å). The microscopic feature of the samples was characterized by field-emission scanning electron microscopy (FESEM) with an energy-dispersive X-ray spectrometer (EDX) (JSM-7001F, JEOL, Tokyo, Japan) and transmission electron microscopy (TEM) with an EDX (JEM-2010F, JEOL, Tokyo, Japan) operated at 300 kV. Thermogravimetric (TG) analysis was carried out on an EXSTAR TG/DTA 6300 (Seiko Instruments, Japan) at a heating rate of 2 °C min−1 in air. X-ray photoelectron spectroscopy (XPS) analysis was carried out on an ESCALAB 250Xi from Thermo Scientific Corporation using AlKa X-ray radiation. The amount of carbon deposited was measured using a CS-344 Infrared Analyzer (Leco, US).2.3. Catalytic measurement
The evaluation of catalysts was carried out with a typical lab fixed-bed reactor.32 The prepared CuxAg catalysts were thermal treatment at 350 °C before the catalytic reaction. The Si powder (20–50 mesh) and prepared CuxAg catalyst together with Zn as a promoter with a ratio of 200![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 were mixed homogeneously to form a contact mass, which was loaded in the glass reactor. The reactor system was purged with purified N2 for 0.5 h followed by heating to 325 °C within 1 h under a N2 flow rate of 25 mL min−1. Subsequently, N2 was turned off, and CH3Cl with a flow rate of 25 mL min−1 was introduced into the reactor to react with Si followed by heating to 325 °C. After a given period of 24 h, the reaction was stopped. The gas product was cooled into a liquid phase with the circulator bath controlled at −5 °C by a programmable thermal circulator (Ningbo Xinzhi biological technology Co., LTD). The waste contact mass (solid residue after reaction), containing unreacted Si powder, Cu, Ag, and Zn compounds were weighed for calculating Si conversion. The products in the liquid solution were quantitative analyzed on an Agilent Technologies (GC-7890A) gas chromatograph equipped with KB-201 column and TCD detector. Gas chromatography system was used for identification of the products, which was mainly comprised of methyltrichlorosilane (CH3SiCl3, M1), dimethyldichlorosilane ((CH3)2SiCl2 M2), trimethylchlorosilane ((CH3)3SiCl, M3), methyldichlorosilane (CH3SiHCl2, M1H), dimethylchlorosilane ((CH3)2SiHCl, M2H), low boiler (LB) and high boiler (HB). The selectivity of the products was calculated by the peak area ratio (in percentage). The Si conversion was that the ratio of weight difference of contact mass (before and after reaction) and weight of Si (before reaction) (formula (1)).
1 were mixed homogeneously to form a contact mass, which was loaded in the glass reactor. The reactor system was purged with purified N2 for 0.5 h followed by heating to 325 °C within 1 h under a N2 flow rate of 25 mL min−1. Subsequently, N2 was turned off, and CH3Cl with a flow rate of 25 mL min−1 was introduced into the reactor to react with Si followed by heating to 325 °C. After a given period of 24 h, the reaction was stopped. The gas product was cooled into a liquid phase with the circulator bath controlled at −5 °C by a programmable thermal circulator (Ningbo Xinzhi biological technology Co., LTD). The waste contact mass (solid residue after reaction), containing unreacted Si powder, Cu, Ag, and Zn compounds were weighed for calculating Si conversion. The products in the liquid solution were quantitative analyzed on an Agilent Technologies (GC-7890A) gas chromatograph equipped with KB-201 column and TCD detector. Gas chromatography system was used for identification of the products, which was mainly comprised of methyltrichlorosilane (CH3SiCl3, M1), dimethyldichlorosilane ((CH3)2SiCl2 M2), trimethylchlorosilane ((CH3)3SiCl, M3), methyldichlorosilane (CH3SiHCl2, M1H), dimethylchlorosilane ((CH3)2SiHCl, M2H), low boiler (LB) and high boiler (HB). The selectivity of the products was calculated by the peak area ratio (in percentage). The Si conversion was that the ratio of weight difference of contact mass (before and after reaction) and weight of Si (before reaction) (formula (1)).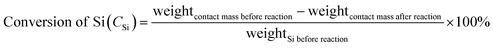 | (1) |
3. Results and discussion
3.1. Catalyst characterization
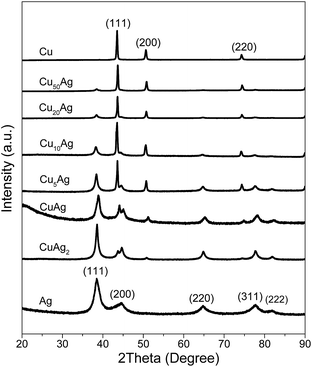
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. TEM image of Fig. S1a† shows that no obvious Ag nanoparticles are attached on the surface of Cu nanoparticles for Cu50–Ag nanoparticles after only adding 0.2 mmol AgNO3 precursor. When the AgNO3 precursor amount is increased to 0.5 mmol and 1.0 mmol, small Ag nanoparticles are attached on the surface of Cu nanoparticles for Cu20–Ag (Fig. S1b†) and Cu10–Ag (Fig. S1c†). With increase of the AgNO3 precursor amount to 2.0 mmol (Fig. S1d†), large Ag nanoparticles are attached on the surface of Cu nanoparticles. With further increase of the AgNO3 precursor amount to 10.0 mmol, the TEM image of Cu–Ag nanoparticles in Fig. 2e reveals that the larger Ag nanoparticles are highly dispersed on the surface of the large Cu nanoparticles. The atomic ratio of Cu
1. TEM image of Fig. S1a† shows that no obvious Ag nanoparticles are attached on the surface of Cu nanoparticles for Cu50–Ag nanoparticles after only adding 0.2 mmol AgNO3 precursor. When the AgNO3 precursor amount is increased to 0.5 mmol and 1.0 mmol, small Ag nanoparticles are attached on the surface of Cu nanoparticles for Cu20–Ag (Fig. S1b†) and Cu10–Ag (Fig. S1c†). With increase of the AgNO3 precursor amount to 2.0 mmol (Fig. S1d†), large Ag nanoparticles are attached on the surface of Cu nanoparticles. With further increase of the AgNO3 precursor amount to 10.0 mmol, the TEM image of Cu–Ag nanoparticles in Fig. 2e reveals that the larger Ag nanoparticles are highly dispersed on the surface of the large Cu nanoparticles. The atomic ratio of Cu![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ag is also about 1
Ag is also about 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 from the TEM-EDX spectrum of Cu–Ag nanoparticles (Fig. 2f). In addition, the SEM image of Cu–Ag2 nanoparticles indicates Cu and Ag nanoparticles are aggregated together (not shown here). In the preparation processes, the Cu nanoparticles act as a spacer separating the Ag nanoparticles, and the size of Ag nanoparticles is increasing with the rising amount of AgNO3 precursor. In case with fixed amount of Cu nanoparticles, it will have the fixed adsorption sites for the given amounts of Cu nanoparticles. After adding less amount of AgNO3 precursor, the formed Ag nanoparticles with smaller in size are attached on the fixed adsorption sites of Cu nanoparticles. Upon further adding more AgNO3 precursor, the formed smaller Ag nanoparticles can grow larger with the addition AgNO3 precursor.
1 from the TEM-EDX spectrum of Cu–Ag nanoparticles (Fig. 2f). In addition, the SEM image of Cu–Ag2 nanoparticles indicates Cu and Ag nanoparticles are aggregated together (not shown here). In the preparation processes, the Cu nanoparticles act as a spacer separating the Ag nanoparticles, and the size of Ag nanoparticles is increasing with the rising amount of AgNO3 precursor. In case with fixed amount of Cu nanoparticles, it will have the fixed adsorption sites for the given amounts of Cu nanoparticles. After adding less amount of AgNO3 precursor, the formed Ag nanoparticles with smaller in size are attached on the fixed adsorption sites of Cu nanoparticles. Upon further adding more AgNO3 precursor, the formed smaller Ag nanoparticles can grow larger with the addition AgNO3 precursor.
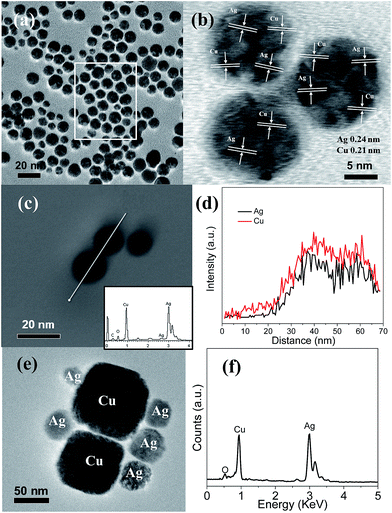 | ||
| Fig. 2 TEM (a), HRTEM (b), and TEM-EDX images (c) (inset is its EDX spectrum), the line scan along the white line in (c) (d), TEM image (e) and its TEM-EDX spectrum (f) of Cu–Ag nanoparticles. | ||
A similar morphology is observed for Cu5Ag nanoparticles. The HRTEM image of Cu5Ag nanoparticles shown in Fig. 3a indicates that Cu and Ag have a spacing distance of about 0.21 nm and 0.24 nm, respectively, corresponding to the interplanar distance of the (111) plane of the Cu and Ag phases. The line scan (Fig. 3c) along the white line in Fig. 3b also shows that Cu and Ag are mixed fairly in the nanoparticles. Fig. 3d reveals the TEM-EDX spectrum of Cu5Ag, indicating the presence of Cu and Ag elements with an approximate atomic ratio of 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, consistent with the initial atomic ratio of Cu(CH3COO)2 and AgNO3 precursors.
1, consistent with the initial atomic ratio of Cu(CH3COO)2 and AgNO3 precursors.
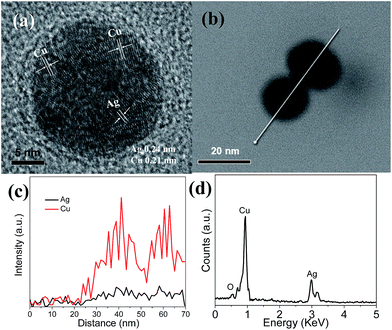 | ||
| Fig. 3 HRTEM image (a), TEM-EDX image (b), the line scan along the direction denoted by the white line in (b) (c), and its TEM-EDX spectrum (d) of Cu5Ag nanoparticles. | ||
The TEM image of Fig. 4a shows that Cu nanoparticles with a mean size of 73.6 nm and a standard deviation of 8.2 are synthesized after addition of Cu(CH3COO)2 precursor only. The HRTEM image in Fig. 4b indicates a spacing distance of 0.21 nm for these Cu nanoparticles, which corresponds to the interplanar distance of the (111) plane. After addition of 0.2 mmol, 0.5 mmol, and 1.0 mmol AgNO3 precursor together, the obtained Cu50Ag (Fig. S1a†), Cu20Ag (Fig. S1b†), and Cu10Ag (Fig. S1c†) nanoparticles have the similar size of pure Cu nanoparticles (Fig. 4a). However, upon further increasing the AgNO3 precursor amount to 5.0 mmol, 10.0 mmol, and 20.0 mmol, it is clearly seen that the size of Cu5Ag (Fig. 3a), CuAg (Fig. 2a), and CuAg2 (Fig. S1d†) nanoparticles are similar to the pure Ag nanoparticles (Fig. 4c). Compared with Cu (Fig. 4a), Cu50Ag (Fig. S1a†), Cu20Ag (Fig. S1b†), and Cu10Ag (Fig. S1c†) nanoparticles, the prepared Cu5Ag (Fig. 3a), CuAg (Fig. 2a), CuAg2 (Fig. S1d†), and Ag (Fig. 4c) nanoparticles are smaller in size, consistent with the above XRD calculated results using the classical Scherrer equation. Fig. 4d indicates that the lattice fringe spacing of Ag is about 0.24 nm, corresponding to the interplanar distance of the (111) plane. But we failed to obtain a clear HRTEM image, probably because of the surface covering of the smaller Ag nanoparticles by oleylamine.45
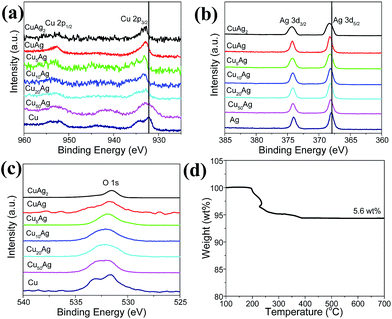 | ||
| Fig. 5 XPS spectra for all the samples: Cu 2p (a), Ag 3d (b), and O 1s (c), and TG curve for CuAg nanoparticles in air (d). | ||
In addition, compared with the pure Cu and Ag nanoparticles, a peak shift is observed for bimetallic nanoparticles. Ag has a higher redox potential than Cu, thus the former has strong attraction than Cu for the electron in the CuxAg bimetallic nanoparticles, leading to lower electron densities of Cu atoms in the CuxAg bimetallic nanoparticles than those in the pure Cu nanoparticles. Indeed, from Mulliken charge analysis, the atomic charge of Cu in the CuxAg bimetallic nanoparticle has a positive value.48 The peak shift in the XPS analysis clearly shows the lower electron density of Cu in the CuxAg bimetallic nanoparticle than that of pure Cu nanoparticles, suggesting the electronic interaction between the Ag and Cu.49 The increase of binding energy of Cu 2p3/2 and Cu 2p1/2 in CuxAg bimetallic nanoparticles (Fig. 5a) indicates the electron transfer from Cu to Ag, which may due to the size and the ratio of Cu![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ag in the CuxAg bimetallic nanoparticles. The peak increase for Ag 3d5/2 and Ag 3d3/2 in bimetallic samples (Fig. 5b) is caused by the strong attraction for electron by Ag than Cu. This can be attributed to the synergistic electronic effect between Cu and Ag. Fig. 5c displays the O1s spectra of the samples. There is one peak for bimetallic nanoparticle, and a relative strong peak for pure Cu nanoparticles at 531.7 eV, which can be ascribed to copper oxide species due to the partial oxidation of the Cu nanoparticles on the surface, further confirming that pure Cu nanoparticles are more susceptible to oxidation than Cu in CuxAg nanoparticles. In addition, the atomic ratio of Cu and Ag elements is also approximately 1
Ag in the CuxAg bimetallic nanoparticles. The peak increase for Ag 3d5/2 and Ag 3d3/2 in bimetallic samples (Fig. 5b) is caused by the strong attraction for electron by Ag than Cu. This can be attributed to the synergistic electronic effect between Cu and Ag. Fig. 5c displays the O1s spectra of the samples. There is one peak for bimetallic nanoparticle, and a relative strong peak for pure Cu nanoparticles at 531.7 eV, which can be ascribed to copper oxide species due to the partial oxidation of the Cu nanoparticles on the surface, further confirming that pure Cu nanoparticles are more susceptible to oxidation than Cu in CuxAg nanoparticles. In addition, the atomic ratio of Cu and Ag elements is also approximately 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 from XPS analysis. Fig. 5d shows the TG curve for CuAg nanoparticles in air, the weight loss of about 5.6 wt% may be derived from the trace amount of organic residue on their surface. However, there is no further weight increase for CuAg nanoparticles in air at higher temperatures, probably because of formation of the CuAg alloy, which is more resistant to oxidation even annealed them in air at higher temperatures.
1 from XPS analysis. Fig. 5d shows the TG curve for CuAg nanoparticles in air, the weight loss of about 5.6 wt% may be derived from the trace amount of organic residue on their surface. However, there is no further weight increase for CuAg nanoparticles in air at higher temperatures, probably because of formation of the CuAg alloy, which is more resistant to oxidation even annealed them in air at higher temperatures.
3.2. Formation process of CuxAg (0 ≤ x ≤ 50) nanoparticles
The possible formation process of the CuxAg (0 ≤ x ≤ 50) nanoparticles is proposed according to the above XRD, TEM and XPS analysis, and is illustrated in Fig. 6. In the first stage, the Cu2+ and Ag2+ ions are reduced and nucleated to form Cu and Ag nanoclusters under solvothermal conditions with assistance of oleylamine (C18H37N), which acts as the solvent, surfactant as well as the reducing agent.42,43 In one control experiment, we also synthesized isolated Cu and Ag nanoparticles with a size range of about 1–5 nm at 180 °C by shortening the reaction time to 1.5 h (Fig. S3†), in which, the Ag and Cu particles were still not fused together. Furthermore, in another control experiment, we synthesized Cu–Ag sample (Fig. 2g) by adding and reducing Cu and Ag separately. Interestingly, the individual Cu and Ag particles were observed but they formed loosely combined assemblies (Fig. 2e). Obviously, C18H37N can be absorbed on the surface of the Cu and Ag nanoclusters and act as structure-directing agents to regulate their surface state, influence the nucleation and aggregation process of these nanoclusters, which are finally self-assembled to the CuxAg hybrid nanoparticles.3.3. Catalytic property
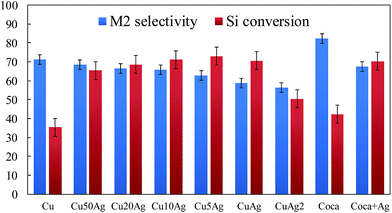
Fig. 7 and Table 1 reveal the catalytic performance of Cu, Ag, CuxAg bimetallic nanoparticles and commercial catalysts for Rochow reaction. It can be seen that much higher Si conversion are observed on Cu50Ag (65.4%), Cu20Ag (68.6%), Cu10Ag (71.1%), Cu5Ag (73.0%), CuAg (70.3%), and CuAg2 (50.4%) than that on the Cu nanoparticles (35.4%), Ag nanoparticles (0%) and on the several reported Cu-based catalysts (Table 2), which should be related to the synergistic electronic effect between the Cu and Ag nanoparticles with electron transfer from Cu to Ag. The Si conversion is first increased with the increase of Ag content (Cu/Ag from 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0 to 5
0 to 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), but decreased again with further increase of Ag content. It is understandable that the addition of excess Ag will lead to the final decrease of the Si conversion, as it is Cu, not Ag, that acts as the active sites. This can be evidenced in Table 1, the pure Ag has no activity in conversion of Si while the pure Cu works well for it. In addition, the top Si conversion are observed on Cu10Ag (71.1%) and CuAg (70.6%), which are obviously higher than that on Cu10–Ag (60.6%), Cu–Ag (57.6%), and Cu + Ag (48.2%), possibly because the Cu10Ag and CuAg nanoparticles have more proper synergistic electronic interaction and local structure for Rochow reaction than that of Cu10–Ag, Cu–Ag nanoparticles and Cu + Ag composites, although the details are still not clear. Comparing with pure Cu nanoparticles, the CuAg samples always have higher Si conversion but lower M2 selectivity. Similar phenomenon is observed for the commercial catalyst. After adding Ag nanoparticles, it shows a much higher Si conversion of 70.2%, a lower M2 of 72.6%, as compared with that of the commercial catalyst without adding Ag nanoparticles (42.3% and 82.3% respectively), indicating the importance of the Ag in the Cu-based catalyst. Since the high similarity in particle size in the prepared CuxAg and the commercial catalyst without Ag nanoparticles or added with Ag nanoparticles, the particle sizes of catalyst should not be the main reason for the difference in Si conversion. In other words, the structural factors such as the synergistic electronic effect of the CuxAg catalysts (Ag has a higher redox potential than Cu, and has strong attraction than Cu for the electron, leading to lower electron densities of Cu atoms in the CuxAg bimetallic nanoparticles) should have more important role for this catalytic reaction. As dimethyldichlorosilane (M2) is the most valuable precursor in organosilane industry, and the bimetallic CuxAg nanoparticles and commercial catalyst added with Ag nanoparticles always have lower selectivity of M2 than their counterparts without Ag. In the design of Rochow reaction catalysts, it should carefully balance the Si conversion and M2 selectivity by adjusting the ratio of Cu and Ag.
1), but decreased again with further increase of Ag content. It is understandable that the addition of excess Ag will lead to the final decrease of the Si conversion, as it is Cu, not Ag, that acts as the active sites. This can be evidenced in Table 1, the pure Ag has no activity in conversion of Si while the pure Cu works well for it. In addition, the top Si conversion are observed on Cu10Ag (71.1%) and CuAg (70.6%), which are obviously higher than that on Cu10–Ag (60.6%), Cu–Ag (57.6%), and Cu + Ag (48.2%), possibly because the Cu10Ag and CuAg nanoparticles have more proper synergistic electronic interaction and local structure for Rochow reaction than that of Cu10–Ag, Cu–Ag nanoparticles and Cu + Ag composites, although the details are still not clear. Comparing with pure Cu nanoparticles, the CuAg samples always have higher Si conversion but lower M2 selectivity. Similar phenomenon is observed for the commercial catalyst. After adding Ag nanoparticles, it shows a much higher Si conversion of 70.2%, a lower M2 of 72.6%, as compared with that of the commercial catalyst without adding Ag nanoparticles (42.3% and 82.3% respectively), indicating the importance of the Ag in the Cu-based catalyst. Since the high similarity in particle size in the prepared CuxAg and the commercial catalyst without Ag nanoparticles or added with Ag nanoparticles, the particle sizes of catalyst should not be the main reason for the difference in Si conversion. In other words, the structural factors such as the synergistic electronic effect of the CuxAg catalysts (Ag has a higher redox potential than Cu, and has strong attraction than Cu for the electron, leading to lower electron densities of Cu atoms in the CuxAg bimetallic nanoparticles) should have more important role for this catalytic reaction. As dimethyldichlorosilane (M2) is the most valuable precursor in organosilane industry, and the bimetallic CuxAg nanoparticles and commercial catalyst added with Ag nanoparticles always have lower selectivity of M2 than their counterparts without Ag. In the design of Rochow reaction catalysts, it should carefully balance the Si conversion and M2 selectivity by adjusting the ratio of Cu and Ag.
| Sample | Product composition (%) | C Si (%) | ||||||
|---|---|---|---|---|---|---|---|---|
| M1 | M2 | M3 | M1H | M2H | LB | HB | ||
| Cu | 18.3 | 71.3 | 2.8 | 2.7 | 1.1 | 0.1 | 3.7 | 35.4 |
| Cu50Ag | 19.4 | 68.4 | 2.1 | 6.4 | 0.9 | 0.2 | 2.6 | 65.4 |
| Cu20Ag | 20.8 | 66.5 | 2.2 | 6.5 | 0.8 | 0.2 | 3.0 | 68.6 |
| Cu10Ag | 22.4 | 65.9 | 1.9 | 6.9 | 0.8 | 0.1 | 2.0 | 71.1 |
| Cu10–Ag | 25.4 | 60.4 | 2.1 | 6.7 | 1.9 | 0.2 | 3.3 | 60.6 |
| Cu5Ag | 24.9 | 62.6 | 1.9 | 6.3 | 0.8 | 0.5 | 3.0 | 73.0 |
| CuAg | 28.3 | 58.6 | 2.0 | 6.9 | 0.8 | 0.3 | 3.1 | 70.6 |
| Cu–Ag | 33.2 | 52.4 | 2.1 | 6.7 | 1.9 | 0.2 | 3.5 | 57.6 |
| Cu + Ag | 30.3 | 56.9 | 2.0 | 6.4 | 1.2 | 0.2 | 3.0 | 48.2 |
| CuAg2 | 28.9 | 56.4 | 2.1 | 6.8 | 0.9 | 0.4 | 4.5 | 50.4 |
| Ag | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Coca | 12.2 | 82.3 | 2.4 | 0.1 | 0.2 | 0.4 | 2.4 | 42.3 |
| Coca + Ag | 19.7 | 72.6 | 2.6 | 0.1 | 0.2 | 2.5 | 2.3 | 70.2 |
| Sample | CSi (%) | Ref. |
|---|---|---|
a
Ref. 29 Si![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) catalyst catalyst![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Zn = 200 Zn = 200![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, temperature 300 °C; ref. 31–39 Si 1, temperature 300 °C; ref. 31–39 Si![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) catalyst catalyst![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Zn = 100 Zn = 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, temperature 325 °C; ref. 50 Si 1, temperature 325 °C; ref. 50 Si![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) catalyst catalyst![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Zn = 100 Zn = 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.06, temperature 300 °C; this work Si 0.06, temperature 300 °C; this work Si![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) catalyst catalyst![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Zn = 200 Zn = 200![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, temperature 325 °C. 1, temperature 325 °C.
|
||
| Cu5Ag nanoparticles | 73.0 | This work |
| CuAg nanoparticles | 70.6 | This work |
| Porous Cu microparticles | 33.5 | 31 |
| Mesoporous Cu2O microspheres | 33.2 | 32 |
| Hexahedron Cu2O microparticles | 40.4 | 33 |
| Core–shell Cu@Cu2O microspheres | 29.2 | 34 |
| Flower-like CuO microparticles | 38.8 | 35 |
| Dandelion-like CuO microparticles | 41.6 | 36 |
| CuO–Cu2O–Cu nanoparticles | 23.4 | 37 |
| Cu–Cu2O–CuO microparticles | 32.4 | 38 |
| Tetrahedra CuCl microparticles | 24.8 | 39 |
| CuCl particles | ∼12 | 50 |
| CuCl particles | ∼20 | 29 |
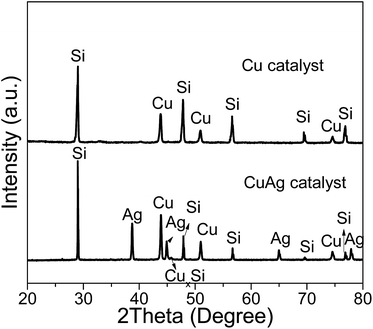
The compositions of contact masses including the deposited carbon after reaction with Cu and CuAg catalyst were further characterized by XRD, SEM, SEM-EDX. Fig. 8 displays XRD patterns of the contact masses after reaction with Cu and CuAg catalyst, in which the unreacted Si, Cu, and Ag phases with CuAg catalyst, and the unreacted Si, and Cu phases with Cu catalyst are detected. In Rochow reaction, CuxSi is normally suggested as the key catalytic active species, from which M2 is produced.51,52 CuxSi formed between Cu-based catalyst and Si interface is an indicator of the Cu-based catalyst activity.53 When Cu-based catalyst and Si are brought together at elevated temperatures, CuxSi is formed, from which methylchlorosilanes can be produced. In addition of Cu, Ag, and Si peaks, a small peak of CuxSi is observed in the XRD patterns of the reacted contact masses with CuAg catalyst, suggesting that the bimetallic CuAg nanoparticles are more active in generating CuxSi active species. We also found there is a shorter induction period for bimetallic CuAg nanoparticles (about 2 h) as compared to the monometallic Cu nanoparticles (about 10 h), possibly because of the higher activity of the bimetallic CuAg nanoparticles.
The scanning electron microscopy (SEM) and SEM-EDX analysis results for the contact masses after reaction with Cu and CuAg catalysts are shown in Fig. 9. Fig. 9a reveals the SEM image of the reacted contact mass with Cu catalyst, in which a large number of deposited carbon fibers are formed. The EDX spectrum (Fig. 9b) demonstrates that the contact mass after the reaction mainly consists of C, O, Si, and Cu, of which C and Si atoms are the most predominant. However, SEM image (Fig. 9c) and its EDX spectrum (Fig. 9d) of the reacted contact mass with CuAg catalyst shows it mainly consists of C, O, Si, Ag, and Cu, of which Si atoms are the major, and little C is observed. The formation of carbon deposit is due to the cleavage of the C–H and C–Cl bond in CH3Cl monolayers on the surface of contact mass, which can restrain the catalytic reaction. The content of carbon deposited on the surface of the reacted contact masses is measured using a CS-344 Infrared Analyzer, which revealed that CuAg catalyst with Si contains 0.262 wt% of carbon, much lower than the value of 1.206 wt% on Cu catalyst with Si, consistent with the above SEM-EDX analysis, indicating that the CuAg catalyst improve the anti-coking ability.
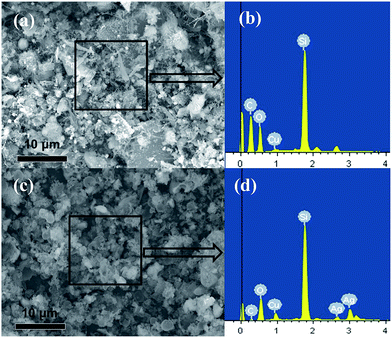 | ||
| Fig. 9 SEM images (a and c), and their EDX spectrum (b and d) of the contact masses after reaction for Cu catalyst (a and b), and CuAg catalyst (c and d). | ||
4. Conclusions
In conclusion, we have demonstrated a thermal reduction process to prepare bimetallic CuxAg (0 ≤ x ≤ 50) nanoparticles. These CuxAg nanoparticles with various compositions have diameters in the range of 5–100 nm. For Rochow reaction, higher Si conversion are always observed on the bimetallic CuxAg nanoparticles (such as 65.4% for Cu50Ag, 68.6% for Cu20Ag, 71.1% for Cu10Ag, 73.0% for Cu5Ag, 70.3% for CuAg) than that on monometallic Cu nanoparticles (35.4%), which is due to the synergistic electronic effect between Cu and Ag, which enable the active sites to be more active for the formation of methylchlorosilanes on the Si surface with CH3Cl. The highest Si conversion is observed on the Cu10Ag and CuAg catalysts. However, it should be pointed out that, although there is a significant increase in Si conversion, the M2 selectivity is decreased after addition of the Ag component. The work sheds light on the development of more effective Cu-based catalysts for Rochow reaction.Acknowledgements
The authors gratefully acknowledge the supports from the National Natural Science Foundation of China (no. 51402299 and 51272252) and Hundred Talents Program of the Chinese Academy of Sciences.Notes and references
- D. Kim, J. Resasco, Y. Yu, A. M. Asiri and P. D. Yang, Nat. Commun., 2014, 5, 4948 CrossRef CAS PubMed.
- A. M. Beale and B. M. Weckhuysen, Phys. Chem. Chem. Phys., 2010, 12, 5562 RSC.
- P. Felfer, P. Benndorf, A. Masters, T. Maschmeyer and J. M. Cairney, Angew. Chem., Int. Ed., 2014, 53, 11190 CrossRef CAS PubMed.
- N. J. Divins, I. Angurell, C. Escudero, V. Perez-Dieste and J. Llorca, Science, 2014, 346, 620 CrossRef CAS PubMed.
- U. Banin, Nat. Mater., 2007, 6, 625 CrossRef CAS PubMed.
- D. Asakura, C. H. Li, Y. Mizuno, M. Okubo, H. S. Zhou and D. R. Talham, J. Am. Chem. Soc., 2013, 135, 2793 CrossRef CAS PubMed.
- F. Studt, F. Abild-Pedersen, Q. X. Wu, A. D. Jensen, B. Temel, J. D. Grunwaldt and J. K. Norskov, J. Catal., 2012, 293, 51 CrossRef CAS.
- J. Yao, K. J. Koski, W. D. Luo, J. J. Cha, L. B. Hu, D. S. Kong, V. K. Narasimhan, K. F. Huo and Y. Cui, Nat. Commun., 2014, 5, 5670 CrossRef CAS PubMed.
- Y. Sugano, Y. Shiraishi, D. Tsukamoto, S. Ichikawa, S. Tanaka and T. Hirai, Angew. Chem., Int. Ed., 2013, 52, 5295 CrossRef CAS PubMed.
- K. Shin, D. H. Kim, S. C. Yeo and H. M. Lee, Catal. Today, 2012, 185, 94 CrossRef CAS.
- J. J. Liu, L. L. Zhang, J. T. Zhang, T. Liu and X. S. Zhao, Nanoscale, 2013, 5, 11044 RSC.
- D. Sengupta, J. Saha, G. De and B. Basu, J. Mater. Chem. A, 2014, 2, 3986 CAS.
- S. Chen, H. Su, Y. Wang, W. Wu and J. Zeng, Angew. Chem., Int. Ed., 2015, 54, 108 CrossRef CAS PubMed.
- Y. B. Wang, H. Y. Zhao and G. H. Zhao, Appl. Catal., B, 2015, 164, 396 CrossRef CAS.
- J. H. Lin and V. V. Guliants, ChemCatChem, 2012, 4, 1611 CrossRef CAS.
- X. Yang, D. Chen, S. J. Liao, H. Y. Song, Y. W. Li, Z. Y. Fu and Y. L. Su, J. Catal., 2012, 291, 36 CrossRef CAS.
- X. Yang, Q. D. Yang, J. Xu and C. S. Lee, J. Mater. Chem., 2012, 22, 25492 Search PubMed.
- J. H. Zeng, F. B. Su, Y. F. Han, Z. Q. Tian, C. K. Poh, Z. L. Liu, J. Y. Lin, J. Y. Lee and X. S. Zhao, J. Phys. Chem. C, 2008, 112, 15908 CAS.
- G. H. Wang, J. Hilgert, F. H. Richter, F. Wang, H. J. Bongard, B. Spliethoff, C. Weidenthaler and F. Schuth, Nat. Mater., 2014, 13, 294 Search PubMed.
- C. L. Zhang, S. Y. Hwang, A. Trout and Z. M. Peng, J. Am. Chem. Soc., 2014, 136, 7805 CrossRef CAS PubMed.
- Z. Q. Zhang, J. L. Huang, L. Zhang, M. Sun, Y. C. Wang, Y. Lin and J. Zeng, Nanotechnology, 2014, 25, 435602 CrossRef PubMed.
- J. Czaplinska, I. Sobczak and M. Ziolek, J. Phys. Chem. C, 2014, 118, 12796 CAS.
- N. Cao, K. Hu, W. Luo and G. Z. Cheng, J. Alloys Compd., 2014, 590, 241 CrossRef CAS.
- E. G. Rochow, J. Am. Chem. Soc., 1945, 67, 963 CrossRef CAS.
- J. Acker and K. Bohmhammel, J. Organomet. Chem., 2008, 693, 2483 CrossRef CAS.
- R. Voorhoeve, B. Geertsem and J. C. Vlugter, J. Catal., 1965, 4, 43 CrossRef CAS.
- J. M. Bablin, A. C. Crawford, D. C. DeMoulpied and L. N. Lewis, Ind. Eng. Chem. Res., 2003, 42, 3555 CrossRef CAS.
- W. Luo, G. Wang and J. Wang, Ind. Eng. Chem. Res., 2006, 45, 129 CrossRef CAS.
- L. N. Lewis and W. J. Ward, Ind. Eng. Chem. Res., 2002, 41, 397 CrossRef CAS.
- J. M. Bablin, A. C. Crawford, D. C. DeMoulpied and L. N. Lewis, Ind. Eng. Chem. Res., 2003, 42, 3555 CrossRef CAS.
- Z. Zhang, H. Che, Y. Wang, X. She, J. Sun, P. Gunawan, Z. Zhong and F. Su, ACS Appl. Mater. Interfaces, 2012, 4, 1295 CAS.
- Z. Zhang, H. Che, Y. Wang, J. Gao, L. Zhao, X. She, J. Sun, P. Gunawan, Z. Zhong and F. Su, Ind. Eng. Chem. Res., 2012, 51, 1264 CrossRef CAS.
- Z. Zhang, H. Che, J. Gao, Y. Wang, X. She, J. Sun, P. Gunawan, Z. Zhong and F. Su, Catal. Sci. Technol., 2012, 2, 1207 CAS.
- Z. Zhang, H. Che, Y. Wang, J. Gao, Y. Ping, Z. Zhong and F. Su, Chem. Eng. J., 2012, 211, 421 CrossRef.
- Z. Zhang, H. Che, Y. Wang, J. Gao, X. She, J. Sun, Z. Zhong and F. Su, RSC Adv., 2012, 2, 2254 RSC.
- Z. Zhang, H. Che, Y. Wang, L. Song, Z. Zhong and F. Su, Catal. Sci. Technol., 2012, 2, 1953 CAS.
- W. Liu, L. Jia, Y. Wang, L. Song, Y. Zhu, X. Chen, Z. Zhong and F. Su, Ind. Eng. Chem. Res., 2013, 52, 6662 CrossRef CAS.
- S. Liu, Y. Wang, Y. Zhu, G. Wang, Z. Zhang, H. Che, L. Jia and F. Su, RSC Adv., 2014, 4, 7826 RSC.
- X. Chen, L. Jia, Y. Wang, L. Song, Y. Zhu, W. Liu, Z. Zhong and F. Su, J. Colloid Interface Sci., 2013, 404, 16 CrossRef CAS PubMed.
- B. T. Meshesha, N. Barrabes, J. Llorca, A. Dafinov, F. Medina and K. Fottinger, Appl. Catal., A, 2013, 453, 130 CrossRef CAS.
- W. L. Zhu, R. Michalsky, O. Metin, H. F. Lv, S. J. Guo, C. J. Wright, X. L. Sun, A. A. Peterson and S. H. Sun, J. Am. Chem. Soc., 2013, 135, 16833 CrossRef CAS PubMed.
- H. Hiramatsu and F. E. Osterloh, Chem. Mater., 2004, 16, 2509 CrossRef CAS.
- N. R. Kim, K. Shin, I. Jung, M. Shim and H. M. Lee, J. Phys. Chem. C, 2014, 118, 26324 CAS.
- W. X. Zhang, X. N. Yang, Q. Zhu, K. Wang, J. B. Lu, M. Chen and Z. H. Yang, Ind. Eng. Chem. Res., 2014, 53, 16316 CrossRef CAS.
- M. Chen, Y. G. Feng, X. Wang, T. C. Li, J. Y. Zhang and D. J. Qian, Langmuir, 2007, 23, 5296 CrossRef CAS PubMed.
- C. K. Wu, M. Yin, S. O'Brien and J. T. Koberstein, Chem. Mater., 2006, 18, 6054 CrossRef CAS.
- E. Stathatos, P. Lianos, P. Falaras and A. Siokou, Langmuir, 2000, 16, 2398 CrossRef CAS.
- R. S. Mulliken, J. Chem. Phys., 1955, 23, 1833 CrossRef CAS.
- Z. Chen, D. Mochizuki, M. M. Maitani and Y. Wada, Nanotechnology, 2013, 24, 265602 CrossRef PubMed.
- A. D. Gordon, B. J. Hinch and D. R. Strongin, Catal. Lett., 2009, 133, 14 CrossRef CAS.
- W. F. Banholzer and M. C. Burrell, J. Catal., 1988, 114, 259 CrossRef CAS.
- T. C. Frank, K. B. Kester and J. L. Falconer, J. Catal., 1985, 91, 44 CrossRef CAS.
- W. F. Banholzer, N. Lewis and W. Ward, J. Catal., 1986, 101, 405 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra04575d |
| This journal is © The Royal Society of Chemistry 2015 |