Influence of hydrotropic coions on the shape transitions of sodium dioctylsulfosuccinate aggregates in an aqueous medium†
U. Thapaa,
D. Rayb,
J. Deya,
N. Sultanaa,
V. K. Aswalb and
K. Ismail*a
aDepartment of Chemistry, North-Eastern Hill University, Shillong-793022, India. E-mail: kismail@nehu.ac.in; kinehu@hotmail.com; Fax: +91-364-2550486; Tel: +91-364-2722610
bSolid State Physics Division, Bhabha Atomic Research Centre, Trombay, Mumbai-400085, India
First published on 15th May 2015
Abstract
The effect of hydrotropic organic coions on the physicochemical behavior of ionic surfactants has not yet been understood clearly, although as counterions they are known to have a remarkable effect on the microstructures of ionic surfactants. In this paper therefore the aggregation behavior of sodium dioctylsulfosuccinate (AOT) has been studied in aqueous media in the presence of sodium benzoate (NaB)/sodium salicylate (NaSa)/sodium meta-hydroxybenzoate (Na-mHB)/sodium para-hydroxybenzoate (Na-pHB). Surface tension and small angle neutron scattering (SANS) techniques are mainly used. AOT is known to have a special counterion binding behavior (SCB) with respect to sodium counterion, viz., at about 0.015 mol kg−1 of added NaCl the counterion binding constant of AOT increases suddenly by two fold which is attributed to the shape change of the AOT micelle. The SCB of AOT is found to exist in the presence of benzoate, m-hydroxybenzoate and p-hydroxybenzoate coions, but not in the presence of o-hydroxybenzoate (salicylate) coions. The SANS data, on the other hand, showed that shape transition of the AOT aggregate takes place in the presence of all the four hydrotropic coions; the concentration of NaSa required to induce the shape transition was however higher. A sharp increase in the value of the hydrodynamic diameter determined by the dynamic light scattering (DLS) experiments also indicates the shape transition of AOT aggregates in the presence of the salts. Chemical shift values of benzoate and hydroxybenzoates confirmed the binding of these coions to an AOT micelle. The binding of the salicylate coion is shown to be stronger due to its low hydration energy and high pKh (Kh is the hydrolysis constant) values.
Introduction
In aqueous solution, surfactants self-assemble into a variety of spatially organized nanostructures such as micelles, vesicles and lamellae. In the case of an ionic surfactant, the area per polar head group at the surface of a surfactant aggregate depends strongly on the nature of the counterion and more specifically on its ability to bind to the surfactant aggregate. Therefore, an easy method to induce morphological transition in surfactant aggregates is by adding counterions, since added counterions decrease the effective area per head group by screening the repulsive interaction between the head groups. Organic counterions,1–3 acting mostly as hydrotropes, are found to be more effective than inorganic counterions4–6 in promoting surfactant aggregation as well as morphological transition in surfactant aggregates. The structural features of hydrotropic salts are similar to surfactants since an aromatic group (hydrophobic) and an ionic group (hydrophillic) coexist within a molecule of a hydrotrope. The hydrophobicity of a hydrotrope is however generally much less than that of a surfactant. Some examples of common hydrotropic salts are benzoate, salicylate, tosylate, napthalenesulfonate, etc. The basic hydrotropic effect refers to the property of hydrotropes to enhance the solubility of organic/biological molecules in water.7,8 Studies are still being made on the physical chemistry of solutions of hydrotropes in order to obtain a better understanding of the hydrotropic effect.9 Apart from studying the solubility enhancing effect of hydrotropes, study on the effect of hydrotropes on surfactant solutions is also being carried out because hydrotropes find their use in surfactant formulations. Most of the studies on the effect of hydrotropes on surfactant solutions were mainly focussed on investigating the formation of viscoelastic wormlike/entangled micelles10,11 for the very fact that such highly viscous solutions have versatile applications.12,13 Studying the effect of organic/hydrophobic/hydrotropic counterions on the surfactant solutions has been gaining more importance in recent years.14–34Both electrostatic and hydrophobic interactions contributed to the binding of organic counterions to micelles. Hydrophobic interaction can as well cause binding of organic coions to ionic micelles and as a consequence organic coions can also be expected to influence the characteristics of surfactant aggregates significantly. However, fewer studies have been made on the influence of organic coions on surfactant systems.35–41 Therefore, our objective in this paper is to investigate the effect of organic coions on the aggregation and adsorption behavior of ionic surfactants.
Salicylate ion is known to have interesting effects on surfactant aggregates. Cetylpyridinium chloride/bromide (CPC/CPB) + sodium salicylate (NaSa) and cetyltrialkylammonium chloride/bromide (CTAAC/CTAAB) + NaSa systems are reported42–45 to exhibit viscoelasticity in aqueous medium due to the formation of wormlike micelles. On the other hand, sodium benzoate (NaB), sodium meta-hydroxybenzoate (Na-mHB) and sodium para-hydroxybenzoate (Na-pHB) do not induce viscoelasticity in CPC/CTAAC/CTAAB system. Thus, the specificity of the hydroxyl group at ortho position in NaSa plays a decisive role in forming wormlike micelles. Sodium dioctylsulfosuccinate (AOT), a double chained anionic surfactant, has a special counterion binding behavior (SCB)46 in aqueous NaCl solution, which is attributed to the shape change of the AOT micelle.47 Interestingly, in the presence of salicylate ion AOT does not exhibit SCB.47,48
In view of these interesting observations, we have chosen for the present study AOT as the ionic surfactant and benzoate, meta-, para- and ortho-hydroxybenzoate as the coions.
Experimental section
AOT (Sigma, 99% assay), NaSa (Fluka, 99.5% assay), NaB (Fluka, 99.5% assay) were used without further purification. Na-mHB and Na-pHB were prepared from their corresponding acids (meta-hydroxybenzoic acid and para-hydroxybenzoic acid, Himedia, 99.0% assay) by neutralising them with NaHCO3. Stock solutions of AOT and the salts were prepared in Milli-Q water, and the required concentrations were obtained by dilution.Surface tension measurements were made by the Wilhelmy plate method using a Krüss K11 tensiometer attached with a thermostat (Haake DC 10). EMF measurements were made using Orion Dual Star ion meter and Orion 8611 BNWP sodium selective combined electrode. All measurements were taken at 25 °C. The SANS measurements were carried out at Dhruva reactor, Bhabha Atomic Research Center, Mumbai, India. In this facility, the SANS diffractometer uses a beryllium oxide filtered neutron beam of mean wavelength (λ) 5.2 Å and the data were collected within the wave vector transfer (Q = 4π![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sin(θ/2)/λ, where θ is the scattering angle) range of 0.017–0.35 Å−1. To access low-Q data some of the measurements were done at SANS-I facility, Swiss Spallation Neutron source SINQ, Paul Scherrer Institut, Switzerland. Data were collected at two sample-to-detector distances (2 and 8 m) to cover a Q range of 0.006–0.25 Å−1. Here, the wavelength of neutron beam was 6 Å and the scattered neutrons from the samples were detected using two dimensional 96 cm × 96 cm detector.49 At both the facilities, the solutions were prepared in D2O (99.4 atom% D). The samples were placed in a quartz cell having a tightly fitting Teflon stopper. The concentration of AOT was fixed at 15 mmol kg−1 (mM) while the concentrations of the salts were varied. The measured SANS data were corrected to the background, the empty cell scattering and the sample transmission, and the corrected data were then normalized to the absolute cross-sectional unit using standard protocols.
sin(θ/2)/λ, where θ is the scattering angle) range of 0.017–0.35 Å−1. To access low-Q data some of the measurements were done at SANS-I facility, Swiss Spallation Neutron source SINQ, Paul Scherrer Institut, Switzerland. Data were collected at two sample-to-detector distances (2 and 8 m) to cover a Q range of 0.006–0.25 Å−1. Here, the wavelength of neutron beam was 6 Å and the scattered neutrons from the samples were detected using two dimensional 96 cm × 96 cm detector.49 At both the facilities, the solutions were prepared in D2O (99.4 atom% D). The samples were placed in a quartz cell having a tightly fitting Teflon stopper. The concentration of AOT was fixed at 15 mmol kg−1 (mM) while the concentrations of the salts were varied. The measured SANS data were corrected to the background, the empty cell scattering and the sample transmission, and the corrected data were then normalized to the absolute cross-sectional unit using standard protocols.
The analysis of the SANS data was done according to the procedure described elsewhere.47,50 In this method of data analysis, the differential scattering cross section per unit volume (dΣ/dΩ) is given by the expression
| dΣ/dΩ = n(ρm − ρs)2V2[〈F2(Q)〉 + 〈F(Q)〉2(S(Q) − 1)] + B | (1) |
In eqn (1), ρm and ρs denote the scattering length densities of the micelle and the solvent, respectively, n is the number density of the micelles, V is the volume of the micelle, F(Q) is the single particle form factor, S(Q) is the inter-particle structure factor and B is a constant term. F(Q) depends on the shape and size of the particles and S(Q) is decided by the spatial distribution of the particles. B accounts for the incoherent scattering background that occurs in the case of neutrons mainly due to the presence of hydrogen in the sample. The relations used for prolate ellipsoidal micelles are
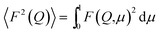 | (2) |
 | (3) |
F(Q,![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) μ) = 3(sin μ) = 3(sin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) x − x x − x![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) cos cos![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) x)/x3 x)/x3
| (4) |
| x = Q[a2μ2 + b2(1 − μ2)]1/2 | (5) |
In eqn (5), a and b are the semimajor and semiminor axes of the ellipsoidal micelle, respectively. The term μ in the above equations refer to the cosine of the angle between the directions of a and Q. The expression for S(Q) is given by the Fourier transform of the radial distribution function g(r). g(r) gives the probability of finding the centre of another micelle at a distance r from the centre of a reference micelle. S(Q) is calculated using the mean spherical approximation developed by Hayter and Penfold.51 For vesicles having inner radius R and thickness dR, the relations used are
 | (6) |
For a rod-like structure of radius R and length 2l, F(Q) is given by the relation
 | (7) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) sin
sin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) β) is the first order Bessel function and β is the angle between the axis of the rod and Q.
β) is the first order Bessel function and β is the angle between the axis of the rod and Q.
Dynamic light scattering (DLS) measurements were made at 25 °C using a Malvern Zetasizer Nano ZS instrument operating at 633 nm (4 mW He–Ne laser was used) and 90° scattering angle. Samples were filtered through a 0.22 μm membrane filter prior to measurements. The scattering intensity data were processed using the instrumental software to obtain the hydrodynamic diameter (dH) of aggregates in each sample.
1H NMR spectra of the salt solutions in the absence and presence of AOT in D2O (Aldrich, 99.9% D atom) were recorded at 25 °C on a Bruker Advance II-400 Spectrometer operating at 400 MHz.
Results and discussion
Representative plots of the surface tension (γ) versus AOT concentration in the presence of varying concentrations of NaB are shown in Fig. S1–S4.† The values of critical micelle concentration (cmc) determined from the surface tension isotherms are shown in Fig. 1 (Table S1†) as a function of the salt concentration.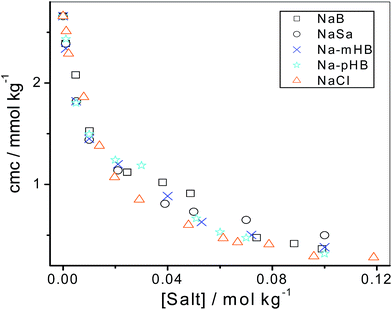 | ||
| Fig. 1 Variation of cmc of AOT as a function of salt concentration. cmc values of AOT in the presence of NaCl were taken from ref. 46. | ||
Counterion binding
From Fig. 1, it is apparent that the effect of the benzoate and hydroxybenzoate coions on the cmc of AOT is not very significant. To determine the counterion binding constant (β) of AOT in the aqueous salt media, we have used the Corrin–Harkins (CH) equation,
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) c0 = A − β[ln(c0 + ce)] c0 = A − β[ln(c0 + ce)]
| (8) |
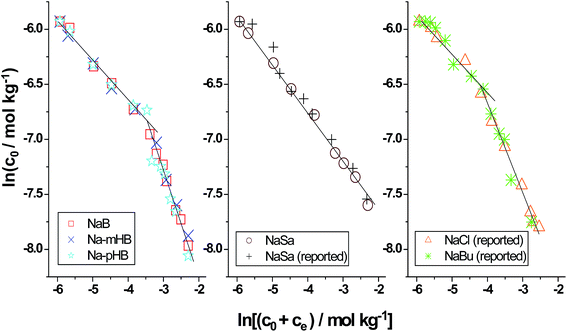 | ||
| Fig. 2 CH plots of AOT in different salt medium. The data for AOT + NaCl and AOT + NaBu (sodium butyrate) are taken from ref. 46 and those for AOT + NaSa are taken from ref. 48. | ||
| Salt | c* | β | |
|---|---|---|---|
| Below c* | Above c* | ||
| NaCl | 15.0 | 0.39 | 0.82 |
| NaB | 30.0 | 0.41 | 0.71 |
| NaSa | — | 0.45 | — |
| Na-mHB | 30.0 | 0.37 | 0.92 |
| Na-pHB | 30.0 | 0.32 | 0.95 |
In order to measure the amount of sodium ions bound to AOT micelle, we also determined the cmc from the EMF method using sodium ion-selective electrode. The experimental EMF data are shown for AOT in the presence of NaB and Na-pHB in Fig. S5 and S6,† respectively. These data were analyzed in the fashion described earlier46 and the value of β was determined using an iteration method. We could determine the values of cmc and β only up to 30 mM salt concentration (values shown in Fig. S5 and S6†), which are comparable to the values obtained above from the surface tension data and the CH plot. However, the values of cmc and β could not be determined accurately above 30 mM salt concentration because the EMF data were found to be scattered and not reproducible. Probably high background concentration of sodium counterion in the salt solution is responsible for this, because addition of low concentration of AOT to such a salt solution may not produce much change in the EMF.
As stated in the introduction section, in CPC/CPB/CTAAC/CTAAB surfactants viscoelasticity is induced by the salicylate counterion, but not by the benzoate, m-hydroxybenzoate and p-hydroxybenzoate counterions. Analogously, there is a distinction between the influence exerted by benzoate, m-hydroxybenzoate and p-hydroxybenzoate coions on the SCB of AOT on one hand and that by salicylate coion on the other hand. Therefore, with respect to inducing viscoelasticity in CPC/CPB/CTAAC/CTAAB surfactants and influencing the SCB of AOT, the salicylate ion (as counterion or coion) behaves differently compared to other isomeric hydroxybenzoates. The present study therefore reveals that the effect of ortho-hydroxybenzoate (salicylate) on the binding of sodium counterion to anionic AOT micelle is different from that of meta- and para-isomers. The neutron reflectivity study52 on the adsorption of hydroxybenzoate and chlorobenzoate counterions at the CTAB monolayer–water interface also revealed that the degree of counterion binding and the location of the counterions are not same for the different isomeric forms of these counterions.
The difference in the counterion binding behavior of AOT in aqueous NaSa as compared to that in aqueous NaB, Na-mHB and Na-pHB reveals that the location and orientation of salicylate coion bound to the AOT micelle is different from those of the other three coions. This may be explained in the following manner:
The pKa (Ka is the dissociation constant) values for the different benzoates53 are: NaB, 4.19; NaSa, 2.97; Na-mHB, 4.06; Na-pHB, 4.48. Since we are dealing with the salts of these acids, the benzoate and the hydroxybenzoate ions undergo hydrolysis in aqueous medium. The pKh (Kh is the hydrolysis constant) values of salicylate, benzoate, m-hydroxybenzoate and p-hydroxybenzoate ions are therefore equal to 11.03, 9.81, 9.94 and 9.52, respectively. Salicylate ions thus undergo least hydrolysis, which may be correlated to the presence of strong intramolecular hydrogen bonding in this ion. Shapley et al.53 showed that there is a strong intramolecular hydrogen bonding in salicylate ion due to which it has lowest solvation energy, about 8–10 kcal mol−1 lower than that of benzoate, which is in agreement with its high value of pKh. Low hydration energy and high pKh value render salicylate ion more hydrophobic than benzoate, m-hydroxybenzoate and p-hydroxybenzoate, and this characteristic of salicylate ion is considered to be responsible for its strong binding to cationic (as counterion) as well as to anionic (as coions) micelles.
SANS measurements
It was shown in the earlier study47,50,54 that the shape change of AOT micelle is responsible for the abrupt change in the value of β in aqueous NaCl, NH4Cl and tetraalkylammonium bromide solutions. The effect of the four organic coions on the shape/morphological transition of AOT aggregates is discussed below on the basis of the SANS data. In SANS experiments, the differential scattering cross section per unit volume (dΣ/dΩ) as a function of Q is measured. The different equations used in the determination of the shape of aggregates are given elsewhere.47,50| Salt | Salt conc. (mM) | Semi-major axis (Å) | Semi-minor axis (Å) | Fractional charge | Aggregation number |
|---|---|---|---|---|---|
| NaB | 10 | 29.1 ± 0.7 | 12.6 ± 0.4 | 0.33 | 32 |
| Na-mHB | 10 | 31.2 ± 0.7 | 12.6 ± 0.4 | 0.34 | 34 |
| Na-pHB | 10 | 30.1 ± 0.7 | 12.6 ± 0.4 | 0.29 | 31 |
| Salt | Salt conc. (mM) | Structure | Thickness of vesicle (Å) | Radius of rod (Å) |
|---|---|---|---|---|
| NaB | 15 | Rods | — | 12.6 ± 0.4 |
| NaB | 20 | Rods and ULV | 17.6 ± 0.6 | 12.6 ± 0.4 |
| NaB | 30 | Rods and ULV | 17.6 ± 0.6 | 12.6 ± 0.4 |
| NaB | 50 | Rods and ULV | 17.7 ± 0.6 | 12.6 ± 0.4 |
| NaB | 70 | Rods and ULV | 17.8 ± 0.6 | 12.6 ± 0.4 |
| Na-mHB | 15 | Rods | — | 12.6 ± 0.4 |
| Na-mHB | 20 | Rods and ULV | 17.5 ± 0.6 | 12.6 ± 0.4 |
| Na-mHB | 50 | Rods and ULV | 17.6 ± 0.6 | 12.6 ± 0.4 |
| Na-mHB | 70 | Rods and ULV | 17.7 ± 0.5 | 12.6 ± 0.4 |
| Na-pHB | 15 | Rods and ULV | 17.6 ± 0.5 | 12.6 ± 0.4 |
| Na-pHB | 20 | Rods and ULV | 17.7 ± 0.5 | 12.6 ± 0.4 |
| Na-pHB | 50 | Rods and ULV | 17.7 ± 0.5 | 12.6 ± 0.4 |
| Na-pHB | 70 | Rods and ULV | 17.9 ± 0.5 | 12.6 ± 0.4 |
| NaSa | 20 | Rods and ULV | 17.7 ± 0.5 | 12.6 ± 0.4 |
| NaSa | 50 | ULV | 18.0 ± 0.5 | — |
| NaSa | 100 | ULV | 18.2 ± 0.6 | — |
Thus, SANS data clearly reveal that the AOT aggregates in the presence of added NaB/NaSa/Na-mHB/Na-pHB undergo a shape transition irrespective of the fact that whether –OH group is absent or present or the position of the –OH group is different. Referring to our previous SANS data of AOT in presence of added NaCl, the shape transition of AOT aggregate takes place at 15 mM of added NaCl and a sudden two-fold change in the slope of the CH plot also takes place at almost the same concentration, i.e., 15 mM NaCl. Such a correlation found earlier47 between the shape change of AOT aggregate and the sudden change in the slope of the CH plot appears to have a dependence on the coions present in the system. In the cases of NaB, Na-mHB and Na-pHB, AOT has two values of β and SANS data too shows morphology change of AOT aggregate. From the SANS data, the shape change of AOT aggregate appears to occur at a lower salt concentration (15 mM) than expected from the c* (30 mM of salt concentration) value. However, this discrepancy disappears when we consider the total sodium ion (counterion) concentration at the region of shape change. The total sodium ion concentration is the sum of sodium ions contributed by the surfactant and the salt. The results of the CH plot refer to the behavior of the micellar system at the cmc (because the CH equation is valid at the cmc). At c* = 30 mM, the total sodium counter ion concentration is equal to about 31 mM (sodium ion contributed by AOT is about 1 mM since this is the cmc value of AOT in the presence of 30 mM of added salt). In SANS measurements the concentration of AOT needs to be kept high to get good neutron scattering, because at the cmc the scattering of neutron is very poor. Therefore, the AOT concentration is kept 15 mM in SANS experiments and the total sodium counter ion concentration at the region where micellar shape change takes place in the presence of benzoate and hydroxybenzoates is nearly equal to 30 mM (35 mM in the presence of NaSa). Thus, there is a good correlation between the CH plot result and SANS result with respect to total sodium ion concentration in presence of benzoate, m-hydroxybenzoate and p-hydroxybenzoate. In the case of NaSa, major discrepancy is however observed since the CH plot of AOT in presence of added NaSa shows only one region of counterion binding indicating thereby no shape change of AOT micelle. On the contrary, SANS data show two different morphologies of AOT aggregate in the presence of 20 mM or more of NaSa. Thus, NaSa is an exception as far as the correlation between slope change of the CH plot and shape change of AOT aggregate is concerned.
As we add sodium salt to a micellar solution of AOT, more counterions bind to a micelle (number of bound counterions, m, increases) which reduces the repulsion between the head groups at the micellar surface thereby increasing the aggregation number (n). Increase in aggregation number increases the size of the micelle, but the value of β remains almost unchanged since it is equal to the ratio (m/n) of the bound counterions to the aggregation number. Thus, although the values of both m and n increases when sodium salt is added, fortuitously the ratio m/n or β remains constant. However, as n increases size increases and at some point the shape also changes due to geometric requirement.47 At this point, the ratio m/n takes a different value and β changes. This is one explanation for correlating change in β to the shape change of micelle.47 In the light of this, it may be visualized that in the presence of NaSa although AOT micelle changes shape at about 20 mM concentration of NaSa, the value of β does not change because even after the micellar shape change the values of m and n may still be changing in such a manner that no change in the value of the ratio m/n occurs unlike in the case of NaCl, NaB, Na-mHB and Na-pHB.
The salicylate, benzoate, m-hydroxybenzoate and p-hydroxybenzoate ions after binding to the micelle may be considered to reside at the Stern layer. Stern layer now becomes a compact layer of polar headgroups (–SO3− and –COO−). In the case of salicylate the –OH group may also reside near the Stern layer. The surface potential thus increases. Because of the intramolecular hydrogen bonding salicylate may bind more strongly compared to other ions as already mentioned above. However, in the case of benzoate, m-hydroxybenzoate and p-hydroxybenzoate the surface potential increase will be more and consequently more sodium counterions will bind. Such differences in the binding of salicylate ion due to intramolecular hydrogen bonding may be responsible for the different counterion ion binding behavior in the presence of salicylate.
DLS measurements
In order to confirm further the variation of the morphology of AOT aggregate at some critical concentrations of added counterions, we performed DLS measurements of 15 mM AOT solutions with varying amounts of NaB and NaSa. In Fig. 6 the variation of dH of AOT aggregate is shown as a function of concentration of NaB and NaSa. A sudden increase in dH (about 1.6 times) occurs at a salt concentration which is comparable to the salt concentration where morphological transition has been found to occur from the SANS data. We expect same behavior with AOT + Na-mHB and Na-pHB also. The sudden change in dH confirms the change in the morphology of AOT at a particular concentration of the added salt as revealed by the SANS data.1H NMR spectra
The chemical shift (δ) values of the benzoate and hydroxybenzoates are listed in Table 4. Whatever the isomer may be, the trend in the δ value with increase in the salt concentration as we go from pre-micellar to micellar region is the same. In presence of micelle, the δ values remain constant irrespective of the concentration of the salt above c*. For all the protons of benzoate, an overall increase in the chemical shift (deshielding) is observed in AOT micellar medium irrespective of the positions of the protons. The deshielding of the benzoate protons suggest that the benzoate ions move from polar solvent medium (D2O) to less polar stern layer region of the AOT micelle.55 Similar trends in the variation of δ were observed in the case of other coions also. Thus, 1H NMR spectra confirm the binding of benzoate and hydroxybenzoate coions to the AOT micelle.Conclusions
In aqueous AOT solution, added organic coions like benzoate, ortho-hydroxybenzoate (salicylate), meta-hydroxybenzoate and para-hydroxybenzoate bind to the AOT micelle. This binding of coions is attributed to hydrophobic interaction and it affects significantly the binding of sodium counterion to AOT micelle and also the morphological transition of AOT micelles. The CH plots revealed the SCB of AOT existing in the presence of NaB, Na-mHB and Na-pHB and its disappearance in the presence of NaSa, whereas the SANS data showed the shape transition of AOT aggregate taking place in the presence of all the four salts (NaB, NaSa, Na-mHB and Na-pHB). In the presence of NaB, Na-mHB and Na-pHB the shape transition occurred from prolate ellipsoids to rods to rods + vesicles as the concentration of the salt increased. The concentration of NaB/Na-mHB/Na-pHB at which prolate ellipsoid to rod transition of AOT aggregate takes place is 15 mM which is same as that found in the presence of NaCl.47 On the other hand, slope change of the CH plots of AOT takes place in the presence of 30 mM (c*) of NaB, Na-mHB and Na-pHB. Thus, benzoate, meta-hydroxybenzoate and para-hydroxybenzoate coions increase the value of c* to 30 mM, which was otherwise 15 mM in the presence of NaCl. In other words, these three coions suppress the SCB of AOT to a certain extent, but not completely (complete suppression of SCB can be considered as equivalent to c* → ∞). Unlike these three coions, salicylate coion suppresses the SCB of AOT completely. But, SANS data showed occurrence of shape change of AOT micelle in the presence of NaSa also when the amount of NaSa is ≥20 mM instead of 15 mM as in the case of NaB, Na-mHB and Na-pHB. The present study therefore provides further support to the inference that slope change of the CH plot is an indication of the occurrence of shape change of AOT micelle, but also reveals that salicylate coion behaves differently by causing shape change of AOT micelle without manifesting any change in the slope of CH plot.Acknowledgements
We acknowledge the financial support received from the DST, New Delhi and UGC, New Delhi. U.T. and N.S. acknowledge the UGC, New Delhi for the research fellowships. J.D. acknowledges the CSIR, New Delhi for the Research Associateship.References
- P. A. Hassan, R. R. Srinivasa and E. W. Kaler, Langmuir, 2002, 18, 2543–2548 CrossRef CAS.
- P. A. Hassan, G. Fritz and E. W. Kaler, J. Colloid Interface Sci., 2003, 257, 154–162 CrossRef CAS.
- G. Garg, P. A. Hassan, V. K. Aswal and S. Kulshreshtha, J. Phys. Chem. B, 2005, 109, 1340–1346 CrossRef CAS PubMed.
- M. Bergström and J. S. Pedersen, Phys. Chem. Chem. Phys., 1999, 1, 4437–4446 RSC.
- M. Almgren, J. C. Gimel, K. Wang, G. Karlsson, K. Edwards, W. Brown and K. Mortensen, J. Colloid Interface Sci., 1998, 202, 222–231 CrossRef CAS.
- L. J. Magid, Z. Li and P. D. Butler, Langmuir, 2000, 16, 10028–10036 CrossRef CAS.
- H. S. Booth and H. E. Everson, Ind. Eng. Chem., Ind. Ed., 1948, 40, 1491–1493 CrossRef CAS.
- A. M. Saleh and L. K. El-Khordagui, Int. J. Pharm., 1985, 24, 231–238 CrossRef CAS.
- J. Eastoe, M. H. Hatzopoulos and P. J. Dowding, Soft Matter, 2011, 7, 5917–5925 RSC.
- M. E. Cates and S. J. Candau, J. Phys.: Condens. Matter, 1990, 2, 6869–6892 CrossRef CAS.
- T. M. Clausen, P. K. Vinson, J. R. Minter, H. T. Davis, Y. Talmon and W. G. Miller, J. Phys. Chem., 1992, 96, 474–484 CrossRef CAS.
- L. M. Walker, Curr. Opin. Colloid Interface Sci., 2001, 6, 451–456 CrossRef CAS.
- M. J. Rosen and M. Dahanayake, Industrial Utilization of Surfactants: Principles and Practice, AOCS Press, Champaign, Illinois, 2000 Search PubMed.
- R. Abdel-Rahem, Adv. Colloid Interface Sci., 2008, 141, 24–36 CrossRef CAS PubMed.
- W. Ge, E. Kesselman, Y. Talmon, D. J. Hart and J. L. Zakin, J. Non-Newtonian Fluid Mech., 2008, 154, 1–12 CrossRef CAS PubMed.
- J. Penfold, I. Tucker, E. Staples and R. K. Thomas, Langmuir, 2004, 20, 8054–8061 CrossRef CAS PubMed.
- D. Yu, M. Tian, Y. Fan, G. Ji and Y. Wang, J. Phys. Chem. B, 2012, 116, 6425–6430 CrossRef CAS PubMed.
- W.-J. Kim and S.-M. Yang, J. Colloid Interface Sci., 2000, 232, 225–234 CrossRef CAS PubMed.
- J. Zhang, B. Dong, L. Zheng and G. Li, Colloids Surf., A, 2006, 290, 157–163 CrossRef CAS PubMed.
- R. Abdel-Rahem, M. Gradzielski and H. Hoffmann, J. Colloid Interface Sci., 2005, 288, 570–582 CrossRef CAS PubMed.
- G. C. Kalur, B. D. Frounfelker, B. H. Cipriano, A. I. Norman and S. R. Raghavan, Langmuir, 2005, 21, 10998–11004 CrossRef CAS PubMed.
- T. Mukhim, J. Dey, S. Das and K. Ismail, J. Colloid Interface Sci., 2010, 350, 511–515 CrossRef CAS PubMed.
- G. Garg, P. A. Hassan, V. K. Aswal and S. K. Kulshreshtha, J. Phys. Chem. B, 2005, 109, 1340–1346 CrossRef CAS PubMed.
- G. Garg, V. K. Aswal, S. K. Kulshreshtha and P. A. Hassan, Pramana, 2004, 63, 351–355 CrossRef CAS.
- P. A. Hassan, S. N. Sawant, N. C. Bagkar and J. V. Yakhmi, Langmuir, 2004, 20, 4874–4880 CrossRef CAS.
- S. Kumar, Z. A. Khan and Kabir-ud-Din, J. Surfactants Deterg., 2004, 7, 367–371 CrossRef CAS.
- V. K. Aswal, J. Phys. Chem. B, 2003, 107, 13323–13328 CrossRef CAS.
- S. Kumar, D. Sharma and Kabir-ud-Din, Langmuir, 2003, 19, 3539–3541 CrossRef CAS.
- S. Kumar, A. Z. Naqvi, V. K. Aswal, P. S. Goyal and Kabir-ud-Din, Curr. Sci., 2003, 84, 1346–1349 CAS.
- Kabir-ud-Din, S. Kumar and D. Sharma, J. Surfactants Deterg., 2002, 5, 131–134 CrossRef CAS PubMed.
- M. Akram, S. Yousuf, T. Sarwar and Kabir-ud-Din, Colloids Surf., A, 2014, 441, 281–290 CrossRef CAS PubMed.
- J. Aslam, U. S. Siddiqui, W. H. Ansari and Kabir-ud-Din, J. Surfactants Deterg., 2013, 16, 693–707 CrossRef CAS PubMed.
- M. Ali, M. Jha, S. K. Das and S. K. Saha, J. Phys. Chem. B, 2009, 113, 15563–15571 CrossRef CAS PubMed.
- R. A. Abdel-Rahem, J. Surfactants Deterg., 2014, 17, 353–362 CrossRef CAS.
- S. Ikeda, S. Hayashi and T. Imae, J. Phys. Chem., 1981, 85, 106–112 CrossRef CAS.
- B. C. Paul, S. S. Islam and K. Ismail, J. Phys. Chem. B, 1998, 102, 7807–7812 CrossRef CAS.
- R. Ranganathan, L. T. Okano, C. Yihwa, E. O. Alonso and F. H. Quina, J. Phys. Chem. B, 1999, 103, 1977–1981 CrossRef CAS.
- T. Narita, N. Hirota, J. P. Gong and Y. Osada, J. Phys. Chem. B, 1999, 103, 6262–6266 CrossRef CAS.
- I. M. Umlong and K. Ismail, Colloids Surf., A, 2007, 299, 8–14 CrossRef CAS PubMed.
- M. A. Bhat, A. A. Dar, A. Amin and G. M. Rather, J. Dispersion Sci. Technol., 2008, 29, 514–520 CrossRef CAS PubMed.
- K. T. Nguyen and A. V. Nguyen, Soft Matter, 2014, 10, 6556–6563 RSC.
- T. Shikata, H. Hirata and T. Kotaka, Langmuir, 1988, 4, 354–359 CrossRef CAS.
- N. Memoto and M. Kuwahara, Langmuir, 1993, 9, 419–423 CrossRef.
- U. R. K. Rao, C. Manohar, B. S. Valaulikar and R. M. Iyer, J. Phys. Chem., 1987, 91, 3286–3291 CrossRef CAS.
- N. C. Das, H. Cao, H. Kaiser, G. Warren, J. Gladden and P. E. Sokol, Langmuir, 2012, 28, 11962–11968 CrossRef CAS PubMed.
- I. M. Umlong and K. Ismail, J. Colloid Interface Sci., 2005, 291, 529–536 CrossRef CAS PubMed.
- J. Dey, J. Bhattacharjee, P. A. Hassan, V. K. Aswal, S. Das and K. Ismail, Langmuir, 2010, 26, 15802–15806 CrossRef CAS PubMed.
- I. M. Umlong and K. Ismail, J. Surf. Sci. Technol., 2006, 22, 101–117 CrossRef CAS PubMed.
- J. Kohlbrecher and W. Wagner, J. Appl. Crystallogr., 2000, 33, 804–806 CrossRef CAS.
- U. Thapa, J. Dey, S. Kumar, P. A. Hassan, V. K. Aswal and K. Ismail, Soft Matter, 2013, 9, 11225–11232 RSC.
- J. B. Hayter and P. Penfold, Mol. Phys., 1981, 42, 109–118 CrossRef CAS PubMed.
- J. Penfold, I. Tucker, E. Staples and R. K. Thomas, Langmuir, 2004, 20, 8054–8061 CrossRef CAS PubMed.
- W. A. Shapley, G. B. Bacskay and G. G. Warr, J. Phys. Chem. B, 1998, 102, 1938–1944 CrossRef CAS.
- J. Dey, U. Thapa and K. Ismail, J. Colloid Interface Sci., 2012, 367, 305–310 CrossRef CAS PubMed.
- M. Vermathen, P. Stiles, S. J. Bachofer and U. Simonis, Langmuir, 2002, 18, 1030–1042 CrossRef CAS.
Footnote |
| † Electronic supplementary information available: Plots of experimental surface tension values (Fig. S1–S4), experimental EMF data (Fig. S5–S6) and critical micelle concentration values (Table S1). See DOI: 10.1039/c5ra04151a |
| This journal is © The Royal Society of Chemistry 2015 |





