Phosphonium-based tetrakis dibenzoylmethane Eu(III) and Sm(III) complexes: synthesis, crystal structure and photoluminescence properties in a weakly coordinating phosphonium ionic liquid†
C. M. Malbaa,
F. Enrichibc,
M. Facchina,
N. Demitrid,
J. R. Plaisierd,
M. M. Natilee,
M. Selvaa,
P. Rielloa,
A. Perosa *a and
A. Benedetti*a
*a and
A. Benedetti*a
aDipartimento di Scienze Molecolari e Nanosistemi, Università Ca' Foscari di Venezia, INSTN and Centro di Microscopia Elettronica “Giovanni Stevanato”, via Torino 155/b, 30170 Venezia-Mestre, Italy. E-mail: alvise@unive.it; benedett@unive.it; Fax: +39-041-234-8594; Tel: +39-041-234-8544
bLaboratorio Nanofab, Veneto Nanotech SCpA, via delle Industrie 5, 30175 Venezia-Marghera, Italy
cCNR-IFN, Istituto di Fotonica e Nanotecnologie, Labortorio CSMFO, via alla Cascata 56/C, 38123 35 Povo, Trento, Italy
dElettra – Sincrotrone Trieste S.C.p.A., S.S. 14 Km 163.5 in Area Science Park, 34149 Basovizza – Trieste, Italy
eCNR-IENI, INSTM, Dipartimento di Scienze Chimiche – Università di Padova, via F. Marzolo 1, 35131 Padova, Italy
First published on 2nd July 2015
Abstract
Highly luminescent anionic Ln(III) β-diketonate complexes of the formula [P8,8,8,1][Ln(dbm)4], with Ln = Eu3+ and Sm3+, [P8,8,8,1] = trioctylmethylphosphonium and dbm = 1,3-diphenylpropane-1,3-dione were synthesized. The single crystal X-ray structure of the samarium and europium complexes showed that the metal ion was surrounded by four ligands and that no water or solvent molecules were coordinated. The solid complexes showed good thermal stability up to 250 °C. The complexes easily dissolved in the ionic liquid trioctylmethylphosphonium bis(trifluoromethylsulfonyl)imide [P8,8,8,1][Tf2N], due to the presence of a common phosphonium countercation in the ionic liquid and in the Eu(III) and Sm(III) complexes. The photoluminescence of the complexes was studied in the solid state and in an ionic liquid as well as in acetonitrile (MeCN) as a solvent.
The past three decades have witnessed continuing interest in the field of lanthanide based materials research. Lanthanide(III) β-diketonate complexes have been widely studied either as tris (LnL3 type) or tetrakis (β-diketonate) complexes [C]+[Ln(L)4]− type.1,2 Because of the lower coordination number of tris complexes (generally six), they usually contain water or other solvent molecules in the first coordination sphere, which is detrimental for obtaining high luminescence intensities. In the latter case, all the coordination positions are filled up by four β-diketonate ligand donor atoms and the charge neutrality is provided by an organic cation. An additional ligand in the tetrakis complexes provides better shielding of the Ln3+ ion which can lead to significant enhancement of the luminescence properties of the complex by avoiding the coordination of strongly quenching solvent molecules.3,4 Even though tetrakis complexes usually show much higher luminescence intensities than the corresponding hydrated tris complexes,2,5–7 there are few reports on the lanthanides complexes of the [C]+[Ln(L)4]− type as compared to the tris complexes of the LnL3 type and the number of crystal structures available for tetrakis β-diketonate complexes is limited as compared to the tris complexes.1 However, these ligands exhibit lower photostability for the corresponding complexes in conventional organic solvents.
Recent studies have successfully demonstrated the use of room-temperature ionic liquids (RTILs) in photochemistry and spectroscopy.8–17 Lanthanide metal containing ionic liquids are being viewed as new types of promising soft materials which can combine the advantageous properties of the ionic liquids with additional intrinsic magnetic, optical and catalytic properties of the incorporated metal complex anion.13 Therefore, it is attractive and desirable to develop ionic liquids containing high concentrations of lanthanides. Most of the studies on the lanthanide compounds in ionic liquids have been performed with ionic liquids based on imidazolium cations and on the emission of Eu3+ due to high-luminescence intensity combined with relatively easy-to-interpret emission spectra of the Eu3+ ion.3,10,11,14–16
For example, a high quantum yield and an enhanced photostability have been reported for a Eu(III) tetrakis complex after dissolving in a weakly coordinating imidazolium ionic liquid (IIL).3 The improvement of the photophysical properties of tetrakis complex in ionic liquid [HMIM][Tf2N] were explained based on hydrogen bonding between the cation and ligands.
Even though Sm3+ has, intrinsically, lower optical performances than Eu, as reported in a few papers,17–19 it is an interesting lanthanide emitter. In fact, in addition to visible luminescence, it also shows a large luminescence contribution in the near-infrared region (NIR). Due to the poor absorbing abilities (molar extinction coefficients (ε) less than 3 L mol−1 cm−1) and smaller energy gap between the emitting level and the next lower energy level (7500 cm−1) of Sm3+, it shows weaker luminescence intensity compared to other lanthanide ions,17 that can be enhanced by using efficient sensitizing ligands such as β-diketonates.
Phosphonium ionic liquids (PILs) have been available in large scale for about two decades and their use has gained great interest as reaction media.20,21 They were found to have a wider electrochemical window than the imidazolium salts. Unlike imidazolium ionic liquids, PILs will not be reduced by the electropositive f-elements and therefore these are suitable for study of the electrochemical properties and electrodeposition of f-elements. Moreover, PILs are found to be thermally more stable and often easier to obtain colourless than nitrogen-based analogues.22 Recently we have also reported the synthesis and optical properties of metal-free dansyl-based luminescent ILs.23 When the lanthanide complexes are dissolved in imidazolium ILs, the acidic hydrogen atom on the imidazolium ring forms hydrogen bonds with the diketonate anion oxygens.17 Even though in our case the α-protons on the phosphonium cation are poorly acidic and poorly hydrogen bonding towards the dibenzoylmethane ligand, nonetheless previous reports indicate strong ion pairing with the anion.24
The aim of this paper was to prepare new phosphonium lanthanide complexes with: (1) a good shielding of the lanthanide ion accomplished by an efficient light harvesting antenna such as 1,3-diphenylpropane-1,3-dione, (2) good solubility in the weakly coordinating ionic liquid, trioctylmethylphosphonium bis(trifluoromethanesulfonyl)imide [P8,8,8,1][Tf2N].
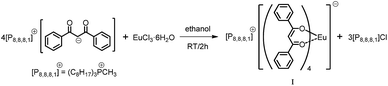
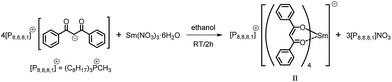
We here report a straightforward and efficient synthesis of trioctylmethylphosphonium tetrakis (dibenzoylmethane) europate(III) [P8,8,8,1][Eu(dbm)4] (I) and trioctylmethylphosphonium tetrakis (dibenzoylmethane) samarate(III) [P8,8,8,1][Sm(dbm)4] (II) complexes and their photoluminescence properties in the [P8,8,8,1][Tf2N] phosphonium ionic liquid. The introduction of the trioctylmethylphosphonium countercation [P8,8,8,1] in the Eu(III) and Sm(III) complexes ensured their solubility in the ionic liquid [P8,8,8,1][Tf2N]. The photophysical properties of solutions of tetrakis europium and samarium complexes in [P8,8,8,1][Tf2N] were investigated systematically. The visible luminescence of (I) and (II) in solid state and in [P8,8,8,1][Tf2N] has been studied along with the near infrared emission (NIR) of (II). [P8,8,8,1][Tf2N] IL was used in this study, since the [Tf2N] anions are known to coordinate very weakly to the lanthanide ions.
Experimental section
All reagents used were ACS grade and were used as received without any further purification. [P8,8,8,1][CH3OCO2] and [P8,8,8,1][Tf2N] ionic liquids were synthesized and purified according to previously reported procedures.25 1H-NMR, 13C-NMR and HMQC spectra were collected on a Varian Unity spectrometer operating at 400 MHz for 1H and at 100 MHz for 13C in CDCl3 solution at 25 °C with TMS as internal standard. The 1H-NMR spectrum of [P8,8,8,1][dbm] was acquired at 60 °C as a neat compound, i.e. without dissolving in a deuterated solvent. A calibrated coaxial glass capillary inserted into the NMR tube filled with [D6]-DMSO was used to lock the magnetic field.Photoluminescence excitation, emission and time-resolved analysis were carried out using a Horiba-Jobin Yvon Fluorolog 3–21 spectrofluorimeter. A Xenon arc lamp was used as a continuous-spectrum source selecting the excitation wavelength by a double Czerny–Turner monochromator. The detection system was constituted by a iHR300 single grating monochromator coupled to an R928 PMT (range 185–900 nm) or to an R5509-73 PMT (range 300–1700 nm). The excitation spectra were recorded with 1 nm band pass resolution, dividing the PMT signal by the intensity of the lamp, measured by using a calibrated photodetector. The emission spectra were recorded with 1 nm band-pass resolution and corrected for the response of the instrument, keeping into account the wavelength dependent efficiencies of the optical elements and detectors.
Time-resolved analyses were performed in multi-channel scaling modality (MCS) by using a tunable pulsed Nd:YAG laser system as excitation source (wavelength selection from 210 nm to 2300 nm, 10 Hz repetition rate, 6 ns pulse duration). The decay profiles were fitted with the least squares method, by using a single exponential equation. External quantum efficiency Φext values were obtained according to eqn (1) by using an integrating sphere coupled to the same Fluorolog-3 system and by measuring the rate between the number of emitted photons and the number of absorbed photons. The internal quantum efficiency Φint of the lanthanide ion can be estimated on the basis of eqn (2), where τ is the measured luminescence lifetime and τrad is the radiative lifetime. For europium τrad can be estimated from the emission spectrum by using eqn (3), where n indicates the refractive index of the sample and I(5D0 → 7FJ)/I(5D0 → 7F1) is the ratio between the total integrated emission from the Eu 5D0 level to the 7FJ manifold (J = 0–4) and the integrated intensity of the transition 5D0 → 7F1.26
Other research groups have investigated theoretically the radiative transition probability for samarium and a general conclusion seems to be in the order of 3 ms, which was here taken for comparison.27,28
 | (1) |
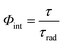 | (2) |
 | (3) |
TGA–DTG thermogravimetric analyses were performed in air from 30 °C to 900 °C with a heating rate of 20 °C min−1 with a Netzsch STA 409 thermobalance.
All the details of the XRD measurements are reported in the ESI.†
[P8,8,8,1][CH3OCO2]
Trioctylphosphine (TOP, 25 mL, 20.8 g, 56 mmol), DMC (30 mL, 32.1 g, 356 mmol) and methanol (30 mL) were combined (two phases) in a sealed 200 mL steel autoclave fitted with a pressure gauge and a thermocouple for temperature control. Three freeze–pump–thaw cycles were carried out to ensure complete degassing of the mixture. The empty volume was then filled with nitrogen. The autoclave was heated for 20 h at 140 °C with magnetic stirring, after which time it was cooled and vented. Methanol and residual DMC were removed from the mixture by rotary evaporation, to give [P8,8,8,1][CH3OCO2] (27.5 g, 100%) as a viscous clear colourless liquid. 1H NMR (neat, 60 °C, 400 MHz, DMSO-d6): δ (ppm) = 3.15 (s, 3H; CH3OCO2), 2.33 (br t, 6H; P–CH2), 1.88 (d, J(P,H) = 14 Hz, 3H; P–CH3), 1.42 (br, 6H), 1.27 (br, 6H), 1.11 (br, 24H), 0.70 (br t, 9H); 13C{1H} NMR (neat, 60 °C, 100 MHz, DMSO-d6), CH2 assigned by 2D INADEQUATE: δ (ppm) = 155.8 (1C; C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 50.0 (1C; CH3O), 30.9 (3C; C6), 29.9 (d, J(P,C) = 15 Hz, 3C; C3), 28.2 (3C; C5), 28.0 (3C; C4), 21.6 (3C; C7), 20.7 (d, J(P,C) = 4 Hz, 3C; C2), 19.0 (d, J(P,C) = 48 Hz, 3C; C1), 12.8 (3C; C8), 2.6 ppm (d, J(P,C) = 53 Hz, 3C; P–CH3); FTIR (neat): 2900, 2856, 1669 cm−1.
O), 50.0 (1C; CH3O), 30.9 (3C; C6), 29.9 (d, J(P,C) = 15 Hz, 3C; C3), 28.2 (3C; C5), 28.0 (3C; C4), 21.6 (3C; C7), 20.7 (d, J(P,C) = 4 Hz, 3C; C2), 19.0 (d, J(P,C) = 48 Hz, 3C; C1), 12.8 (3C; C8), 2.6 ppm (d, J(P,C) = 53 Hz, 3C; P–CH3); FTIR (neat): 2900, 2856, 1669 cm−1.
[P8,8,8,1][Tf2N]
The [P8,8,8,1][Tf2N] ionic liquid was prepared by a metathesis reaction between [P8,8,8,1][CH3OCO2] (8.0 g, 16.26 mmol) and bis(trifluoromethane)sulfonimide lithium salt [LiNTf2] (4.66 g, 16.26 mmol) in water. The mixture was stirred at 70 °C for 2 h. The product was extracted with 3 aliquots (50 mL) of CH2Cl2, dried over Na2SO4, and the solvent was removed under reduced pressure to afford pure, and dry [P8,8,8,1][Tf2N] as a clear liquid. (6.10 g, 60%) 1H NMR (neat, 60 °C, 400 MHz, DMSO-d6): δ (ppm) = 1.96 (br, 6H; P–CH2), 1.55 (d, J(P,H) = 13 Hz, 3H; P–CH3), 1.34 (br, 6H), 1.24 (br, 6H), 1.09 (br, 24H), 0.68 (br t, 9H); 13C{1H} NMR (neat, 60 °C, 100 MHz, DMSO-d6): δ (ppm) = 119.1 (q, J(F,C) = 321 Hz, 2C; CF3), 30.6 (3C; C6), 29.3 (d, J(P,C) = 15 Hz, 3C; C3), 27.8 (3C; C5), 27.5 (3C; C4), 21.4 (3C; C7), 20.2 (d, J(P,C) = 5 Hz, 3C; C2), 19.0 (d, J(P,C) = 48 Hz, 3C; C1), 12.6 (3C; C8), 2.3 (d, J(P,C) = 52 Hz, 3C; P–CH3); FTIR (neat): = 2930, 2859, 1468, 1352 cm−1.[P8,8,8,1][dbm]
In a 50 mL round bottomed flask were combined equimolar amounts of 1,3-diphenyl-1,3-propanedione (dbm) (2.27 g, 10.14 mmol) and [P8,8,8,1][CH3OCO2] (5 g, 10.14 mmol). The mixture was stirred at RT for 2 h, and then stirred under vacuum to eliminate co-products MeOH and CO2. The resulting viscous red ionic liquid was obtained in quantitative yield (6.134 g, 99.5%). 1H NMR (neat, 60 °C, 400 MHz, DMSO-d6) δ (ppm): 8.48 (m, 4H), 8.03–7.83 (m, 4H), 7.83–7.75 (m, 2H), 7.09 (s, 1H), 2.73 (m, 6H), 2.42 (d, J = 13.9 Hz, 3H, CH3–P), 2.03–1.59 (m, 36H), 1.42 (t, J = 6.8, 5.6 Hz, 9H); 13C{1H} NMR (neat, 60 °C, 400 MHz, DMSO-d6) δ (ppm): 180.49 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 126.64, 126.18, 89.55 (C−), 31.08, 29.99, 28.35, 21.87, 20.95, 19.20 (CH2–P, J = 48.5 MHz), 3.21, 2.90 (CH3–P, J = 51.3 MHz) FTIR: 3055, 3028, 2955, 2925, 2854, 2731, 1608, 1565, 1493, 1465, 1431, 722.
O), 126.64, 126.18, 89.55 (C−), 31.08, 29.99, 28.35, 21.87, 20.95, 19.20 (CH2–P, J = 48.5 MHz), 3.21, 2.90 (CH3–P, J = 51.3 MHz) FTIR: 3055, 3028, 2955, 2925, 2854, 2731, 1608, 1565, 1493, 1465, 1431, 722.
[P8,8,8,1][Eu(dbm)4] (I)
An aqueous solution (6 mL) of EuCl3·6H2O (0.57 g, 1.58 mmol) was added dropwise to [P8,8,8,1][dbm] (3.88 g, 6.32 mmol) in ethanol (10 mL). The reaction mixture was stirred for 2 h during which time the complex precipitated, it was then was filtered and dried under high vacuum (10−1 mbar/1 h). The crystals of [P8,8,8,1][Eu(dbm)4] (I) were suitable for single-crystal X-ray structure determination (C85H98EuO8P: Mr: 1430.62) yield (1.56 g, 69%), m.p.: 194.7–198.5. 1H NMR (400 MHz, CDCl3-d1) δ (ppm): 16.89 (s, 1H), 8.02–8.00 (m, 16H), 7.65–7.44 (m, 24H), 2.60–2.45 (m, 6H), 2.2 (d, J = 13.3 Hz, 3H, CH3–P), 1.64–1.21 (m, 36H), 0.89 (m, 9H), °C FTIR: 3060 νs(C–H)ar, 3029, 2954, 2925, 2856 νs(C–H); 1957, 1896, 1600, 1552, 1515 νs(C–O); 1470, 1425, 1310 νs(C–O⋯M); 1280, 1220, 1180 νs(C–P); 1066 νs(C–O); 1030 ν(ring deformation), 940, 780, 720 cm−1, Raman: 1595 ν(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C)ar; 1556; 1488, 1443 w ν(C
C)ar; 1556; 1488, 1443 w ν(C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C)ar; 1309 s ν(C–H)ar; 1274, 1178, 1059, 1000 (ring deformation); 940, 788, 682 (ring deformation); 617, 460 m ν(Eu–O); 240, 190 ν(Eu–O chelate ring deformation); 133, anal. calcd: C, 71.2%; H, 6.85%. Found: C, 69.94%; H, 6.70%.
C)ar; 1309 s ν(C–H)ar; 1274, 1178, 1059, 1000 (ring deformation); 940, 788, 682 (ring deformation); 617, 460 m ν(Eu–O); 240, 190 ν(Eu–O chelate ring deformation); 133, anal. calcd: C, 71.2%; H, 6.85%. Found: C, 69.94%; H, 6.70%.
[P8,8,8,1][Sm(dbm)4] (II)
An aqueous solution (6 mL) of Sm(NO3)3·6H2O (0.96 g, 2.16 mmol) was added dropwise to [P8,8,8,1][dbm] (5.263 g, 8.64 mmol) in ethanol (6 mL). The reaction mixture was stirred for 2 h during which time the complex precipitated, it was then was filtered and dried under high vacuum (10−1 mbar/4 h). It was purified by recrystallization from ethanol (2.59 g, 84%). The complex showed red/orange luminescence under the UV lamp (366 nm). (C85H98SmO8P: Mr: 1429.016) m.p.: 232.4–240.7 °C; 1H NMR (400 MHz, CDCl3-d1) δ (ppm): 17.03 (s, 1H), 8.35–8.24 (m, 16H), 7.51–7.42 (m, 8H), 7.41, 7.34 (m, 16H), 1.34–1.25 (m, 6H), 1.11–1.035 (m, 6H), 1.01–0.97 (m, 6H), 0.90–0.81 (m, 6H), 0.58–0.49 (m, 6H), 0.41–0.29 (m, 6H), 0.06–0.2 (m, 6H), −0.48 (d, J = 12.5 Hz, 3H, CH3–P) FTIR (KBr): = 3062, 2927, 2854 (C–H), 1760 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), 1616, 1596, 1554, 1515, 1465, 1423, 1307, 1276, 1218, 1176, 1068 (C–P), 1025, 941, 717, 690 cm−1; Raman: 1595, 1488, 1443, 1312, 1274, 1179, 1060, 997, 938, 785, 670, 617, 402, 241, 196, 127. Anal. calcd: C, 71.37%; H, 6.85%. Found: C, 71.35%; H, 6.80%.
O), 1616, 1596, 1554, 1515, 1465, 1423, 1307, 1276, 1218, 1176, 1068 (C–P), 1025, 941, 717, 690 cm−1; Raman: 1595, 1488, 1443, 1312, 1274, 1179, 1060, 997, 938, 785, 670, 617, 402, 241, 196, 127. Anal. calcd: C, 71.37%; H, 6.85%. Found: C, 71.35%; H, 6.80%.
Results and discussion
Both complexes I and II were prepared starting from the parent colourless ionic liquid [P8,8,8,1][CH3OCO2], by exchanging the methylcarbonate anion with dibenzoylmethane to yield the corresponding pure phosphonium dibenzoylmethanate [P8,8,8,1][dbm] (Scheme 1), accompanied by only methanol and CO2 as by-products. Addition of EuCl3·6H2O or Sm(NO3)3·6H2O according to the procedure reported by Melby et al. yielded the desired lanthanide complexes.29 Complex I was prepared by dissolving 4 molar equivalents of the ionic liquid [P8,8,8,1][dbm] in ethanol, followed by the dropwise addition of 1 equiv. of the EuCl3·6H2O in water (Scheme 2).Complex II was prepared by dissolving 4 molar equivalents of the ionic liquid [P8,8,8,1][dbm] in ethanol, followed by the dropwise addition of 1 equiv. of the Sm(NO3)3·6H2O in water (Scheme 3).
Previous reports on the synthesis of lanthanide ionic liquid complexes involved deprotonating a diketone with a base, adding a halide ionic liquid, and a lanthanide salt, followed by isolation of the product.17 In the present instance, the diketonate ionic liquid [P8,8,8,1][dbm] was formed spontaneously by mixing the parent [P8,8,8,1][CH3OCO2] with dibenzoylmethane. Then the ionic liquid–lanthanide complexes were generated directly from [P8,8,8,1][dbm] by the addition of the lanthanide salt.
Crystal structures of (I) and (II)
Attempts to determine the crystal structure of the newly synthesized compounds were done starting from crystals obtained from slow evaporation of ethanol solutions. Important disorder contribution have been encountered in all the Eu crystals. The crystallographic data and refinement details are summarized in the ESI.† [P8,8,8,1][Eu(dbm)4] and [P8,8,8,1][Sm(dbm)4] complexes show monoclinic cells, containing two crystallographically independent molecules for the europium complex (Fig. 1) and one for samarium one (Fig. 2). No crystallization solvent molecules have been found trapped in the crystal packing. | ||
| Fig. 1 Preponderant conformation found in the asymmetric unit of [P8,8,8,1][Eu(dbm)4] crystal (50% probability ellipsoids). | ||
Both the complexes show a mean bond length of 2.40(4) Å around the metal atom and mean bond angles of 75(5)°. The overall coordination geometry (Fig. 3) resembles a distorted square antiprism as previously reported in literature for similar tetrakis complexes.30
 | ||
| Fig. 3 Ball and stick representation of metal coordination sphere. Both [P8,8,8,1][Ln(dbm)4] (Ln = Eu, Sm) show a distorted square antiprism coordination geometry. | ||
Thermal analyses
Thermal analyses of the complexes I and II were carried out under pure nitrogen and in a 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 N2/air atmosphere in the temperature range from 40 to 900 °C, at a heating rate 10 °C min−1.
1 N2/air atmosphere in the temperature range from 40 to 900 °C, at a heating rate 10 °C min−1.
Photoluminescence properties
 | ||
| Fig. 6 Room temperature excitation spectra of complex (I) in solid state (λem = 611 nm), in ionic liquid [P8,8,8,1][Tf2N] (λem = 614 nm) and in MeCN (λem = 611 nm). | ||
The emission spectra of the (I) in solid state, dissolved in acetonitrile (MeCN) and dissolved in the ionic liquid [P8,8,8,1][Tf2N] obtained by excitation at 415, 350 and 390 nm respectively, are presented in Fig. 7. Dissolving complex (I) in MeCN and in the ionic liquid [P8,8,8,1][Tf2N] was found to affect differently the spectroscopic fine structure of the emission spectrum of the compound compared to the solid state.
 | ||
| Fig. 7 Room temperature emission spectra of complex (I) in solid state (λexc = 415 nm), in MeCN (λexc = 350 nm) and in ionic liquid [P8,8,8,1][Tf2N] (λexc = 390 nm). | ||
The excitation energy was absorbed by the ligands and transferred to the Eu3+ emitting centre in a subsequent step (antenna effect). The emission spectrum for the solid state consisted of several transitions 5D0 → 7FJ (J = 0, 1, 2, 3, 4) related to the radiative de-excitation from the 5D0 emitting level of Eu3+ [579 (5D0 → 7F0), 591 (5D0 → 7F1), 611, 613 (5D0 → 7F2), 651 (5D0 → 7F3) and 691, 702 nm (5D0 → 7F4)]. The spectrum was dominated by the so-called “europium red” 5D0 → 7F2 emission as the dominant band (hypersensitive transition) peaking at 611 nm, giving an intense red luminescence output for the complex (I). The intensity ratio R(5D0 → 7F2)/I(5D0 → 7F1) was 18.9. Such a high ratio is typical of the Eu3+ ion without inversion symmetry.34
Unlike the solid state, the emission bands related to the 5D0 → 7FJ (J = 0, 1, 2, 3, 4) transitions for the complex (I) dissolved in acetonitrile (MeCN) and in the ionic liquid [P8,8,8,1][Tf2N] were broadened. It is well known that rare earths luminescence is sensitive to the local environment and can experience a line broadening passing from an ordered state to a more disordered or dynamic system.35 The broadening of the photoluminescence emission peaks passing from the pure complex in solid state to the complex in liquid (either in molecular solvent or ionic liquid) is thus commonly observed for rare earth complexes in solution, due to the electrostatic interactions and collisions with the surrounding molecules. The emission band at 611 nm due to the 5D0 → 7F2 transition was shifted to 614 nm in the ionic liquid. This band broadening is particularly evident for complex (I) dissolved in the ionic liquid as well as its shift to 614 nm. This effect could perhaps be attributed to the interactions existing between the phosphonium cation and the ligand.24 In our case, these interactions are revealed by the change of the 1H-NMR chemical shifts of the P–CH3 protons in the presence of different anions. This effect may be analogous to the one observed for encapsulated lanthanide complexes,36 as previously suggested by others.3 It should be noted that the emission spectrum for all the samples showed only one peak due to 5D0 → 7F0 transition (579 nm), which implies that all the Eu3+ ions occupy a site of the same symmetry.37,38 The hypersensitive transition (5D0 → 7F2) for the emission at 614 nm revealed changes of the coordination sphere of the Eu3+ ion. The symmetry and strength of the ligand field around Eu3+ could be determined from the ratio of the relative intensities between the 5D0 → 7F2/5D0 → 7F1 transitions (Table 1). In contrast to the magnetic dipole 5D0 → 7F1 transition, the intensity of the electric dipole 5D0 → 7F2 transition changed with the variation of the ligand field strength. The R value for the complex (I) dissolved in MeCN and [P8,8,8,1][Tf2N] was 19.2 and 9.2 respectively.
| Solid | MeCN | [P8,8,8,1][Tf2N] | |
|---|---|---|---|
| I(5D0 → 7F2)/I(5D0 → 7F1) | 18.9 | 19.2 | 9.2 |
| τrad (ms) | 0.63 | 1.29 | 2.09 |
| τmeas (ms) | 0.58 | 0.16 | 0.69 |
| Φint (%) | 92 | 12 | 33 |
| Φext (%) | 58 | 7 | 20 |
By using eqn (3) the radiative lifetime of Eu3+ in the three different environments was estimated. The refractive index for MeCN is 1.344, for [P8,8,8,1][Tf2N] it was estimated based on the reported value of [P6,6,6,14][Tf2N] at 20 °C as 1.45 (ref. 39) and for the solid state was assumed to be 1.5 for comparison purposes. The obtained radiative lifetime values are reported in Table 1.
The luminescence decay curves of Eu3+ related to the 5D0 → 7F2 emission in the pure europium complex (I) in solid state, dissolved in acetonitrile (MeCN) and dissolved in the ionic liquid [P8,8,8,1][Tf2N] are shown in Fig. 8. The lifetime measurements were performed at room temperature at 611 nm (solid state and MeCN) or 614 nm (ionic liquid [P8,8,8,1][Tf2N]) emission wavelength under pulsed laser excitation at 390 nm. The decay curves were fitted by a single exponential and the lifetime values for all the samples are listed in Table 1. The lifetime of the 5D0 excited state of the pure complex was 0.58 ms. This value was, as expected, higher than lifetimes of several tris complexes with dibenzoylmethane ligands40 and tetrakis europium complexes.41
 | ||
| Fig. 8 Photoluminescence decay curves for complex (I) in solid state, in the MeCN and in ionic liquid [P8,8,8,1][Tf2N]. | ||
When dissolved in ionic liquid the complex (I) showed higher lifetime as compared to the lifetime value of the complex in solid state and the one dissolved in MeCN solvent.
The internal quantum efficiency calculated from eqn (2) is reported in Table 1. Φint reached a value as high as 92% for the pure complex in solid state while it was 12% and 33% in MeCN and [P8,8,8,1][Tf2N], respectively, an indication of significant nonradiative recombination pathways. The measurements of the external quantum efficiency corresponding to the rate between absorbed and emitted photons followed a similar trend. It is worth observing that the rate between Φext and Φint was an indication of the transfer efficiency between the antenna ligand and the rare earth. For complex (I) this value was estimated at about 60%. The longer lifetime in ionic liquid compared to the solid state is not commonly observed and can be rationalized based on the following two considerations. The first concerns the radiative lifetime of each system which in the case of Eu complex was evaluated directly from the experimental spectrum and resulted much higher in the ionic liquid than in the solid state. The other concerns the non-radiative recombinations which are related to the vibrational energy losses and other quenching mechanisms. In this case, as expected, the liquid environment results more dissipative than the solid state and this is reflected in the internal Φint (33% vs. 92%). Globally, in our system the balance resulted in favour of the ionic liquid, as verified by the direct lifetime measurement.
 | ||
| Fig. 9 Room temperature excitation spectrum (λem = 650 nm) of the (II) complex in solid state (black line), in MeCN (red line), and in ionic liquid (blue line). | ||
The emission spectra of the complex (II) in solid state, dissolved in acetonitrile (MeCN) and dissolved in the ionic liquid [P8,8,8,1][Tf2N] obtained by excitation at 418, 350 and 390 nm respectively, are presented in Fig. 10 for the visible range and in Fig. 11 for the NIR range. The emission properties of complex II are different from those of I as they are related to the different rare earth (Sm vs. Eu). The assignments of lines are given in Table 2. The dibenzoyl methane ligands (dbm) absorbed the excitation energy and transferred it to the Sm3+ emitting centre. The emitting level for all the samarium emissions was the 4G5/2 level. The emission spectrum exhibited four emission transitions in the visible at 563, 608, 650 and 709 nm due to 4G5/2 → 6H5/2, 4G5/2 → 6H7/2, 4G5/2 → 6H9/2 and 4G5/2 → 6H11/2 transitions. The 4G5/2 → 6H9/2 transition was the hypersensitive transition and responsible for the most intense line (c) in the visible region (650 nm). The complex showed the characteristic orange colour of the Sm3+ ion. A peak located at 563 nm, due to 4G5/2 → 6H5/2 transition, had predominant magnetic dipolar character. For the Sm3+ ion, the ratio between the integrated areas of the 4G5/2 → 6H9/2 and 4G5/2 → 6H5/2 (R) transitions, in the emission spectrum, was used as a measure for the polarizability of its chemical environment. The intensity ratio R calculated for the complex (II) in solid state was 13.3. This value was quite high for a Sm3+ complex17 and was similar to Eu(III) β-diketonate complexes.
 | ||
| Fig. 10 Emission spectra of complex (II) in solid state (λexc = 418 nm), dissolved in MeCN (λexc = 350 nm) and ionic liquid Tf2N (λexc = 390 nm). | ||
 | ||
| Fig. 11 NIR luminescence of complex (II) in solid state (λexc = 418 nm), dissolved in MeCN (λexc = 350 nm) and ionic liquid [P8,8,8,1][Tf2N] (λexc = 390 nm). | ||
| Line | Line assignment | Wavenumber (cm−1) | Wavelength (nm) |
|---|---|---|---|
| a | 4G5/2 → 6H5/2 | 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 762 762 |
563 |
| b | 4G5/2 → 6H7/2 | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 447 447 |
608 |
| c | 4G5/2 → 6H9/2 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 384 384 |
650 |
| d | 4G5/2 → 6H11/2 | 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 104 104 |
709 |
| e | 4G5/2 → 6H13/2 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 579 579 |
795 |
| f | 4G5/2 → 6F1/2 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 325 325 |
883 |
| g | 4G5/2 → 6H15/2 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 061 061 |
904 |
| h | 4G5/2 → 6F3/2 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 810 810 |
925 |
| i | 4G5/2 → 6F5/2 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 526 526 |
950 |
| j | 4G5/2 → 6F7/2 | 9652 | 1036 |
| k | 4G5/2 → 6F9/2 | 8475 | 1180 |
In addition to visible emission, complex (II) also displayed emission in the NIR region of the electromagnetic spectrum (Fig. 11). The main line in the NIR area was at 950 nm due to 4G5/2 → 6F5/2 transition.
The emission spectrum of the Sm complex (II) dissolved in MeCN or in ionic liquid [P8,8,8,1][Tf2N] was similar to the one in the solid state. Nevertheless, as previously observed also for complex (I), transitions were broadened with respect to the solid state as typically observed in solution, due to the electrostatic interactions and collisions with the surrounding molecules.35
The luminescence decay curves for the complex (II) in solid state, in MeCN and in ionic liquid [P8,8,8,1][Tf2N] are shown in Fig. 12. The lifetime measurements were performed at 650 nm under pulsed laser excitation at 390 nm. The decay curves were fitted by a single exponential and the lifetime values are reported in Table 3. The lifetime values for the complex (II) in solid state, in MeCN and in ionic liquid [P8,8,8,1][Tf2N] were respectively 63.5 μs, 3.1 μs and 19.1 μs. The complex (II) in ionic liquid showed a higher lifetime as compared to the complex in MeCN solvent, in agreement with what observed for complex (I). However, in this case the longest lifetime was observed in the solid state.
 | ||
| Fig. 12 The decay curve for 4G5/2 → 6H9/2 emission in the samarium complex (II) in solid state, in MeCN and in ionic liquid [P8,8,8,1][Tf2N]. | ||
| Solid | MeCN | [P8,8,8,1][Tf2N] | |
|---|---|---|---|
| I(4G5/2 → 6H9/2)/I(4G5/2 → 6H5/2) | 13.3 | 12.2 | 4.8 |
| τrad (μs) | 3000 (ref. 26 and 27) | 3000 (ref. 26 and 27) | 3000 (ref. 26 and 27) |
| τmeas (μs) | 63.5 | 3.1 | 19.1 |
| Φint (%) | 2.1 | 0.1 | 0.6 |
| Φext (%) | 2.7 | <0.1 | 0.8 |
As a general observation, these values were lower than the radiative lifetime reported for this kind of ion in theoretical works, which is about 3 ms, indicating an important role of nonradiative recombinations. Other lifetimes on Sm complexes in different solvents and also in ionic liquids have been reported to range from a few microseconds to hundreds of microseconds.18,43
A rough evaluation of the internal quantum efficiency calculated from eqn (2) by using a radiative lifetime of 3 ms is shown in Table 3. Unfortunately, for the samarium complex II it is not possible to give an evaluation of the radiative lifetime by using a simple formula based on the experimental emission spectra, as was done for europium using eqn (3). Therefore, the results obtained based on literature studies on similar complexes are just a rough indication, which can be useful for comparison. Φint was about 2% for the pure complex in solid state while it was 0.1% and 0.6% in MeCN and [P8,8,8,1][Tf2N] respectively. The measurements of the external quantum efficiencies were obtained by using an integrating sphere, which in our case is limited to the visible spectral region, and are therefore slightly underestimated. However, it is worth observing that they follow the same trend of the internal efficiency values. Significant uncertainty on the internal efficiency related to the unknown radiative lifetime and a 15% uncertainty on the measurement of external efficiency should be kept in mind in comparing these numbers.
Conclusions
Tetrakis rare earth complexes (Eu3+ and Sm3+) with 1,3-diphenylpropane-1,3-dione ligand were synthesized. The crystal structures of complexes (I) and (II) consists of [P8,8,8,1]+ cations and complex anions. It shows that the Eu3+ and Sm3+ ions are surrounded by four 1,3-diphenylpropane-1,3-dione ligands. The coordination number of the Eu3+ and Sm3+ ions is eight. No solvent molecules are coordinated to the central metal ions. Unlike imidazolium cation based lanthanide tetrakis complexes, H-bonding between the [P8,8,8,1] cation and the complex anion is impossible in our case, rather the phosphonium cation is thought to ion-pair with the ligands thus contributing to the stability of the complexes in the present case. Both the Eu and Sm tetrakis complexes showed thermal stability up to 250 and 285 °C, respectively. The photoluminescence of complexes (I) and (II) in phosphonium ionic liquid [P8,8,8,1][Tf2N] was studied and the changes in the fine structure of emission spectra were observed. Complex (I) showed a lifetime value of 0.58 ms in solid state that increased to 0.69 ms when dissolved in ionic liquid [P8,8,8,1][Tf2N], while a value of 0.16 ms was observed in acetonitrile solvent. The longer emission lifetime observed for I in the IL compared to the solid state is not commonly observed and may indicate that matching the cation of the IL and of the complex might contribute to stabilize the complex. Internal quantum efficiencies as high as 92% in solid state, 33% in ionic liquid but only 12% in acetonitrile were found, indicating an important contribution of nonradiative recombination paths. Moreover, external quantum efficiencies of 58% (solid state), 20% (ionic liquid) and 7% (acetonitrile) indicate a ligand to emitter transfer efficiency of the order of 60%. The time resolved analysis of complex (II) showed a lifetime value of 63.5 μs in solid state which decreased to 19.1 μs when dissolved in ionic liquid and again a much shorter lifetime of 3.1 μs in MeCN. For Sm the internal quantum efficiencies were much lower than Eu. From a value of about 2% in solid state and 1% in ionic liquid, it decreases drastically to 0.1% in acetonitrile, indicating an important contribution of nonradiative recombination paths. These phenomena can justify the difference between our results and previously reported ones.3 External quantum efficiencies follow the same trend and similar values. The optical properties of the phosphonium ionic liquids synthesized by the procedure here described, along with their ability to dissolve the lanthanide tetrakis β-diketonate complexes open new potential applications for these materials, e.g. for study of the electrochemical properties and electrodeposition of f-elements or for photovoltaic cells. In the case of photovoltaic cells, the emission wavelength of complex II at 950 nm would be better compared to the emission of I at 612 nm, but the lower efficiency of II observed in our case also, still make I preferable.Acknowledgements
M. M. N. acknowledges the Italian MIUR under the project FIRB RBAP114AMK “RINAME”.References
- K. Binnemans, in Handbook on the Physics and Chemistry of Rare Earths, ed. K. A. J. Gschneidner, J.-C. G. Bünzli and V. K. Pecharsky, Elsevier, Amsterdam, 2005, vol. 35, pp. 107–272 Search PubMed.
- K. Binnemans, Chem. Rev., 2009, 109, 4283–4374 CrossRef CAS PubMed.
- P. Nockemann, E. Beurer, K. Driesen, R. van Deun, K. van Hecke, L. van Meervelt and K. Binnemans, Chem. Commun., 2005, 4354–4356 RSC.
- W. D. Horrocks Jr, J. P. Bolender, W. D. Smith and R. M. Supkowski, J. Am. Chem. Soc., 1997, 119, 5972–5973 CrossRef.
- R. van Deun, D. Moors, B. de Fré and K. Binnemans, J. Mater. Chem., 2003, 13, 1520–1522 RSC.
- S. M. Bruno, R. A. S. Ferreira, F. A. Almeida Paz, L. D. Carlos, M. Pillinger, P. Ribeiro-Claro and I.-S. Goncalves, Inorg. Chem., 2009, 48, 4882–4895 CrossRef CAS PubMed.
- R. G. Charles and E. P. Riedel, J. Inorg. Nucl. Chem., 1966, 28, 3005–3018 CrossRef CAS.
- K. Driesen, P. Nockemann and K. Binnemans, Chem. Phys. Lett., 2004, 395, 306–310 CrossRef CAS PubMed.
- S. Arenz, A. Babai, K. Binnemans, K. Driesen, R. Giernoth, A.-V. Mudring and P. Nockemann, Chem. Phys. Lett., 2005, 402, 75–79 CrossRef CAS PubMed.
- I. Billard, S. Mekki, C. Gaillard, P. Hesemann, G. Moutiers, C. Mariet, A. Labet and J.-C. G. Bünzli, Eur. J. Inorg. Chem., 2004, 6, 1190–1197 CrossRef PubMed.
- E. Guillet, D. Imbert, R. Scopelliti and J.-C. G. Bünzli, Chem. Mater., 2004, 16, 4063–4070 CrossRef CAS.
- L. N. Puntus, K. J. Schenk and J.-C. G. Bünzli, Eur. J. Inorg. Chem., 2005, 4739–4744 CrossRef CAS PubMed.
- K. Binnemans, Chem. Rev., 2007, 107, 2592–2614 CrossRef CAS PubMed.
- C. Gaillard, I. Billard, A. Chaumont, S. Mekki, A. Ouadi, M. A. Denecke, G. Moutiers and G. Wipff, Inorg. Chem., 2005, 44, 8355–8367 CrossRef CAS PubMed.
- S. Samikkanu, K. Mellem, M. Berry and P. S. May, Inorg. Chem., 2007, 46, 7121–7128 CrossRef CAS PubMed.
- R. Nagaishi, M. Arisaka, T. Kimura and Y. Kitatsuji, J. Alloys Compd., 2007, 431, 221–225 CrossRef CAS PubMed.
- K. Lunstroot, P. Nockemann, K. van Hecke, L. van Meervelt, C. Gorller-Walrand, K. Binnemans and K. Driesen, Inorg. Chem., 2009, 48, 3018–3026 CrossRef CAS PubMed.
- H. F. Brito, O. L. Malta, M. C. F. C. Felinto, E. E. S. Teotonio, J. F. S. Menezes, C. F. B. Silva, C. S. Tomiyama and C. A. A. Carvalho, J. Alloys Compd., 2002, 344, 293–297 CrossRef CAS.
- B. Yan and Q. Y. Xie, Chem. Pap., 2004, 58, 316–319 CAS.
- J. D. Holbrey and K. R. Seddon, Clean Products and Processes, 1999, 1, 223–236 Search PubMed.
- M. R. Netherton and G. C. Fu, Org. Lett., 2001, 26, 4295–4298 CrossRef PubMed.
- C. J. Bradaric, A. Downard, C. Kennedy, A. J. Robertson and Y. Zhou, Green Chem., 2003, 5, 143–152 RSC.
- G. Fiorani, M. Selva, A. Perosa, A. Benedetti, F. Enrichi, P. Licence and T. L. Easun, Green Chem., 2015, 17, 538–550 RSC.
- (a) K. J. Fraser, E. I. Izgorodina, M. Forsyth, J. L. Scott and D. R. MacFarlane, Chem. Commun., 2007, 3817–3819 RSC; (b) J. Ammer, C. Nolte, K. Karaghiosoff, S. Thallmair, P. Mayer, R. de Vivie-Riedle and H. Mayr, Chem.–Eur. J., 2013, 19, 14612–14630 CrossRef CAS PubMed.
- M. Fabris, V. Lucchini, M. Noè, A. Perosa and M. Selva, Chem.–Eur. J., 2009, 15, 12273–12282 CrossRef CAS PubMed.
- M. H. V. Werts, R. T. F. Jukes and J. W. Verhoeven, Phys. Chem. Chem. Phys., 2002, 4, 1542–1548 RSC.
- X. Y. Chen, M. P. Jensen and G. K. Liu, J. Phys. Chem. B, 2005, 109, 13991–13999 CrossRef CAS PubMed.
- Z. Li, J. Yu, L. Zhou, H. Zhang and R. Deng, Inorg. Chem. Commun., 2008, 11, 1284–1287 CrossRef CAS PubMed.
- L. R. Melby, N. J. Rose, E. Abramson and J. C. Caris, J. Am. Chem. Soc., 1964, 86, 5117–5125 CrossRef CAS.
- L. M. Sweeting and A. L. Rheingold, J. Am. Chem. Soc., 1987, 109, 2652–2658 CrossRef CAS.
- B. Tong, S. Wang, Y. Meng and B. Wang, Photochem. Photobiol. Sci., 2007, 6, 519–520 CAS.
- D. Zhao, W. Qin, C. Wu, J. Zhang, G. Qin and H. Lin, J. Rare Earths, 2004, 22, 49–52 Search PubMed.
- X. Guo, J.-L. Canet, D. Boyer, P. Adumeau and R. Mahiou, J. Sol-Gel Sci. Technol., 2012, 64, 404–410 CrossRef CAS.
- C. Gorrler-Walrand and K. Binnemans, Spectral Intensities of f–f Transitions, in Handbook on the Physics and Chemistry of Rare Earths, Elsevier, Amsterdam, 1998, vol. 25, ch. 167, pp. 101–264 Search PubMed.
- R. S. Meltzer, Line Broadening Mechanisms and Their Measurement, in Spectroscopic Properties of Rare Earths in Optical Materials, ed. G. Liu and B. Jacquier, Springer-Verlag Berlin Heidelberg, 2005, Ch. 4 Search PubMed.
- Q. Xu, L. Li, B. Li, J. Yu and R. Xu, Microporous Mesoporous Mater., 2000, 38, 351–358 CrossRef CAS.
- K. Binnemans, P. Lenaerts, K. Driesen and C. Görller-Walrand, J. Mater. Chem., 2004, 14, 191–195 RSC.
- W. D. Horrocks Jr and M. Albin, Prog. Inorg. Chem., 1984, 31, 1–104 CrossRef PubMed.
- H. F. D. Almeida, J. A. Lopes-da-Silva, M. G. Freire and J. A. P. Coutinho, J. Chem. Thermodyn., 2013, 57, 372–379 CrossRef CAS PubMed.
- B. Chen, Y. Luo, H. Liang, J. Xu, F. Guo, Y. Zhang, A. Lin and X. Liu, Spectrochim. Acta, Part A, 2008, 70, 1203–1207 CrossRef PubMed.
- J. P. Duignan, I. D. H. Oswald, I. C. Sage, L. M. Sweeting, K. Tanaka, T. Ishihara, K. Hirao and G. Bourhill, J. Lumin., 2002, 97, 115–126 CrossRef CAS.
- G. H. Dieke, Spectra and Energy Levels of Rare Earth Ions in Crystals, Interscience Publishers, New York, 1968 Search PubMed.
- K. Lunstroot, P. Nockermann, K. van Hecke, L. van Meervelt, C. Görrler-Walrand, K. Binnemans and K. Driese, Inorg. Chem., 2009, 48, 3018–3026 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. CCDC 1051982 and 1051983. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c5ra03947a |
| This journal is © The Royal Society of Chemistry 2015 |