Fabrication and stabilization of nanoscale emulsions by formation of a thin polymer membrane at the oil–water interface
Abstract
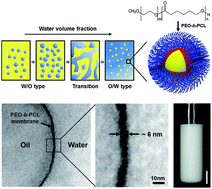
This study introduces a robust approach for the fabrication of extremely stable oil-in-water nanoemulsions in which the interface is stabilized by assembly of amphiphilic poly(ethylene oxide)-block-poly(ε-caprolactone) (PEO-b-PCL) copolymers. Phase inversion emulsification, induced by variation of the water volume fraction, facilitated effective assembly of the block copolymers at the oil–water interface. Subsequent application of simple probe-type sonication reduced the droplet size of the precursor emulsions to approximately 200 nm. The prepared nanoemulsions were surprisingly stable against drop coalescence and aggregation, as confirmed by analysis of changes in the droplet size after repeated freeze–thaw cycling and by monitoring the creaming kinetics under conditions of high ionic strength and density mismatch. The results highlight that good structural assembly of the PEO-b-PCL block copolymers at the oil–water interface generated a mechanically flexible but tough polymer film, thereby remarkably improving the emulsion stability.

 Please wait while we load your content...
Please wait while we load your content...