Fabrication and characterization of KGM-based FMBO-containing aerogels for removal of arsenite in aqueous solution†
Shuxin Yea,
Weiping Jina,
Qing Huanga,
Ying Hua,
Bakht Ramin Shaha,
Shilin Liuab,
Yan Liab and
Bin Li*ab
aCollege of Food Science and Technology, Huazhong Agriculture University, Wuhan, 430070, China. E-mail: libinfood@mail.hzau.edu.cn; Fax: +86-27-87282966; Tel: +86-27-63730040
bKey Laboratory of Environment Correlative Dietology, Huazhong Agricultural University, Ministry of Education, China
First published on 1st May 2015
Abstract
Hybrid materials were obtained by immobilizing Fe and Mn oxides (FMBO) into a konjac glucomannan (KGM) based aerogel matrix to remove arsenite from water. Composite adsorbents were prepared through a coupling sol–gel process with a suitable freeze-drying technique. The KGM aerogels were employed as the framework and FMBO as the coating materials. Scanning electron microscopy (SEM), thermo gravimetric analysis (TGA), X-ray diffraction (XRD), Fourier transform infrared spectroscopy (FTIR) and X-ray photoelectron spectroscopy (XPS) were used for the characterization of the hybrid aerogels. The adsorption of As(III) by the composite aerogels decreased with increasing pH. Isotherms were well predicted by Freundlich behavior, implying the heterogeneous nature of the As(III) adsorption. The maximum As(III) uptake capacity reached 30.33 mg g−1 at the FMBO/KGM ratio of 1.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 at pH 7 and 323 K. The effects of coexisting anions including Cl−, NO3−, SO42−, SiO32−, PO43− as well as natural organic matter, which possibly exist in natural water, on As(III) removal were also investigated. The hybrid adsorbents could be easily regenerated by using NaOH solution, exhibiting excellent practicability and reusability. Furthermore, XPS analysis of composite aerogels before and after the reaction confirmed the oxidation–sorption mechanism for As(III) removal. This research extends the potential applicability of KGM-based aerogels and provides an eco-friendly and convenient approach to efficiently remove trace As(III) from aqueous solutions.
1 at pH 7 and 323 K. The effects of coexisting anions including Cl−, NO3−, SO42−, SiO32−, PO43− as well as natural organic matter, which possibly exist in natural water, on As(III) removal were also investigated. The hybrid adsorbents could be easily regenerated by using NaOH solution, exhibiting excellent practicability and reusability. Furthermore, XPS analysis of composite aerogels before and after the reaction confirmed the oxidation–sorption mechanism for As(III) removal. This research extends the potential applicability of KGM-based aerogels and provides an eco-friendly and convenient approach to efficiently remove trace As(III) from aqueous solutions.
1. Introduction
Arsenic in groundwater has attracted extensive attention since long-term ingestion of arsenic-contaminated water could result in cancer and other severe health problems.1 There are two predominant species of soluble arsenic in natural water, arsenate [As(V)] and arsenite [As(III)]. As(III) acts as the predominant chemical state of arsenic in groundwater.2 More importantly, As(III) is 25–60 times more toxic than As(V)3 and therefore deserves more attention.Among the possible As(III) treatment strategies, adsorption is considered to be the most applicable technology, because it is less expensive than membrane filtration, easier and safer to handle compared with the contaminated sludge produced by precipitation, and more versatile than ion exchange.4 Previous research5 has shown that iron and manganese oxides (FMBO) were effective adsorbents for arsenic removal due to large surface area coupling with high activities caused by a size-qualification effect. Moreover, FMBO could achieve the simultaneous removal of arsenite [As(III)] without dosing oxidants such as chlorine or ozone.6 However, additional post-treatment was needed after adsorption process to separate FMBO from water because of small particle size.7 Second pollution and recycling difficulty caused by FMBO were main defects inhibiting their adhibition in practice. Therefore, researchers recently have focused on doping FMBO within the solid host media like diatomite8 and anion exchanger9 to improve their applicability.
As renewable resources, polysaccharide aerogels possess tremendous potential for adsorbent framework material due to their superior surface porosities, favorable microporous structure and eco-friendly character. To date, aerogels adsorbents such as carbon nanofiber aerogels10 and cellulose-based hydrophobic carbon aerogels11 have been fabricated to remove heavy metals from wastewater. Konjac glucomannan (KGM) is a kind of natural polysaccharide from the tuber of the amorphophallus konjac plant. It was known that a thermo irreversible gel was formed from polymer monomer through sol–gel by the removal of acetyl groups on the molecular chains of KGM with addition of alkaline and heating.12 These hydrogels could be turned into aerogels by using unidirectional freeze-drying method (UFDM).13 Our previous work has documented that the addition of montmorillonite (MMT) into deacetylated konjac glucomannan (Da-KGM) could greatly improve mechanical properties and thermal stability of the aerogels.14 If KGM and Na+-MMT connected with each other and formed macroscopic three-dimensional (3D) materials while immobilizing the FMBO, they could not only effectively remove As(III) but also made them easily separated from the solution.
To the best of our knowledge, polysaccharide based aerogels modified with FMBO have never been fabricated and utilized to remove trace As(III) from aqueous solution. Hence, this research aimed to synthesis a novel KGM-based FMBO-containing aerogels and investigate the mechanism of As(III) removal. The strong oxidation and adsorption abilities of composite aerogels were determined through batch equilibration experiments. The effect of various parameters such as time, pH, temperature, initial concentration, co-existing anions, natural organic matter (NOM) and desorption agent on As(III) adsorption efficiency and adsorption mechanism were also studied.
2. Material and methods
2.1. Raw materials
The raw KGM was purchased from Hubei Konjac Gum Co., Ltd (Wuhan, China). Na+-MMT was purchased from NANOCOR Co., Ltd (Beijing, China). The other chemicals were of analytical grade and were purchased from Sinopharm Chemical Reagent Co., Ltd (Beijing, China). The As(III) stock solutions were prepared with deionized water using NaAsO2. Arsenite working solutions were freshly prepared by diluting arsenite solutions with deionized water. The concentrations of arsenite species were always given as elemental arsenic concentration in this study.2.2. Preparation of the composites
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 Mn/Fe molar ratio according to the method described by Zhang et al.15 The reaction of this co-precipitation process was as follows:
3 Mn/Fe molar ratio according to the method described by Zhang et al.15 The reaction of this co-precipitation process was as follows:| 3Fe2+ + MnO4− + 4OH− + 3H2O = 3Fe(OH)3 + MnO2 + H+ | (1) |
2.3. Adsorbent characterizations
The surface morphologies of FMBO and aerogels samples were examined by using scanning electron microscopy (SEM) (S-3000 N, Hitachi Co., Japan) with energy-dispersive X-ray analysis (EDX) (EDAX Co., USA). The morphological images were determined with transmission electron microscopy (Philips CM12/STEM, Eindhoven, Netherlands). Thermogravimetric measurements were carried out with a SDTQ600 Thermo-gravimetric analyses (TGA) apparatus (TA Instruments, USA), by heating the samples at a rate of 10 °C min−1 from room temperature to 600 °C. X-ray diffraction patterns were recorded in the range of 2θ = 5–85° (interval of 0.02°) on a Bruker D8 Advance X-ray Diffractometer, using Ni-filtered Cu Kα (λ = 0.154 nm) radiation operated at 40 kV and 40 mA. FTIR spectra were collected on a FTIR spectrometer (Nexus 470, Nicolet, USA) in a transmission mode. Samples for FTIR determination were ground with spectral grade KBr in an agate mortar.2.4. Batch As(III) adsorption tests
The kinetics experiments were conducted in batch mode. Defined amount of As(III) stock solution was added in a 500 mL glass vessel containing As(III) at a concentration of 10 mg L−1. After adjusting pH to 6.9 by adding 0.1 mol L−1 HCl or NaOH, 0.7 g of composite aerogels were added to obtain 1.4 g L−1 adsorbent content. The suspension was mixed with a magnetic stirrer, and the pH was maintained at 6.9 throughout the experiment by the addition of dilute acid and/or base solutions. During the reaction, approximately 5 mL aliquots were taken from the suspension at the following intervals: 0, 5, 10, 15, 30, 60, 120, 180, 330, 540, 720 and 1440 min. After the predetermined adsorption time, arsenic concentrations in the liquid phase were measured with an atomic fluorescence spectrophotometer (AFS-8220, Jitian Beijing).The effects of pH on arsenite removal and As(III) adsorption isotherms were also determined. The aerogels in small pieces with the same weight approximately 0.14 g were added into 100 mL glass vessels containing the As(III) solutions at the pH range of 4–10, which were shaken by using a rotary shaker at 303 K temperature for 24 h. Adsorption isotherms of As(III) by composite aerogels were determined at the equilibrium pH values of 6.9 ± 0.1. Initial arsenite concentrations were varied from 1 mg L−1 to 50 mg L−1 at 303 K, 313 K and 323 K with an adsorbent content of 1.4 ± 0.1 g L−1. All the composite aerogels were shaken on an orbit shaker for 24 h. To investigate the influence of coexisting anions including chloride, sulfate, nitrate, silicate and phosphate, the corresponding sodium salts were introduced into the As(III) solution. Similarly, humic acid and alginic acid sodium salts were also involved to investigate the influence of natural organic matter (NOM).
The adsorption capacity, q (mg g−1), was calculated using the following mass balance equation:
 | (2) |
2.5. Desorption and regeneration tests
For desorption studies, As(III) was firstly adsorbed by composite aerogels of A1.5 in a 100 mL glass vessel containing As(III) at a concentration of 10 mg L−1 for 24 h at 303 K. Then As(III) loaded composite aerogels (A1.5) were placed in 0.1, 0.5 or 1.0 mol L−1 of NaOH (100 mL) and shaken for 24 h at 303 K for desorption, followed by repeatedly rinsing with deionized water to neutralize pH. The above procedure was repeated 4 times to investigate the reusability of the adsorbents of A1.5.2.6. Zeta potential and FTIR analysis of FMBO
Zeta potential analyzer (Zetasizer 2000, Malvern, UK) and Fourier transform infrared spectroscopy (FTIR) (Nexus 470, Nicolet, USA) were used to analyze the zeta potential and FTIR spectra of FMBO particles before and after arsenite adsorption.2.7. XPS spectra of FMBO loaded aerogels (A1.5)
Surface structure of composite aerogels were analyzed by XPS before and after As(III) binding adsorption. XPS data process and peak fitting were performed using a nonlinear least-squares fitting program (XPS peak software 4.1, Raymund W. M. K. work).3. Results and discussion
3.1. Characterization
Table 1 showed the composition of FMBO in Da-KGM hydrogels with different weight ratio to get four different types. SEM image (Fig. 1a) of FMBO revealed that there were many micropores on the FMBO surface. Moreover, FMBO surfaces were rough and irregular in shape. TEM images (Fig. 1b) of FMBO showed that the microparticles of FMBO appeared to have a nanostructure, ranging in several tens of nanometers, and tiny particles aggregated together in a regular way. SEM micrographs of all composite aerogels showed a homogeneous, porous, interconnected 3D network (Fig. 1c–f). As the initial contents of Na+-MMT and KGM were constant, the additive amount of FMBO influenced the microstructure of aerogels. The roughness of aerogels surface increased with the increasing amount of FMBO, which could be proven by magnifying the scale bar from 500 μm to 10 μm. The EDX analysis (Fig. 1g) showed that the percentages of Fe and Mn atoms were 19.02% and 6.53% at the near surface of FMBO. It indicated that Fe/Mn molar ratio was about 2.91, a little lower than the bulk, which was in the range of 2.93–3.02. The similar Fe/Mn molar ratio (2.83) can be seen in composite aerogels of A1.5 (Fig. 1h).| KGM gel | FMBO/KGM ratio | KGM (wt%) | Na2CO3 (wt%) | Na+-MMT (wt%) | FMBO (wt%) |
|---|---|---|---|---|---|
| A0 (control) | 0.0 | 2 | 0.24 | 2 | — |
| A0.5 | 0.5 | 2 | 0.24 | 2 | 1 |
| A1.0 | 1.0 | 2 | 0.24 | 2 | 2 |
| A1.5 | 1.5 | 2 | 0.24 | 2 | 3 |
 | ||
| Fig. 1 SEM images of FMBO (a) and composite aerogel: (c) A0, (d) A0.5, (e) A1.0, (f) A1.5. (b) TEM image of FMBO and EDX analysis of (g) FMBO, (h) composite aerogel of A1.5. | ||
The thermal stability of four groups of composite aerogels was evaluated by TGA (Fig. 2a) and DTG analysis (Fig. 2b). It was found that thermal evaporation-induced weight loss occurred in all the samples. The thermal weight loss decreased from A0 to A1.5, indicating that the increase of FMBO amount can reduce the rate of thermal degradation of KGM. When the temperature reached 600 °C, the weight loss of A0, A0.5, A1.0, A1.5 were 42.68%, 37.13%, 33.13% and 31.76% respectively. There was a significant negative correlation between FMBO/KGM ratio and weight loss at 600 °C (significant at the 0.05 level. *). The results meant that addition of FMBO greatly improved the thermostability of aerogels adsorbent.
 | ||
| Fig. 2 (a) Thermo gravimetric analysis (TGA) and (b) differential thermal gravimetry (DTG) of Da-KGM aerogels: A0, A0.5, A1.0 and A1.5. | ||
To understand composite aerogels more, X-ray diffraction (XRD) patterns (Fig. S1†) of the FMBO, KGM, Na+-MMT and composite aerogels (A0, A1.5) were investigated. Addition of KGM and FMBO showed no shift in the peak position indicating no structural change of Na+-MMT in the composite aerogels. Moreover, the successful loading of FMBO in the Da-KGM aerogels was also clearly confirmed by FTIR analysis (Fig. S2†).
3.2. Adsorption studies
Pseudo-second-order (3) was used for correlation of adsorption data to examine the controlling mechanism involved in As(III) onto composite aerogels.
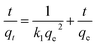 | (3) |
The kinetics data in Fig. 3 of As(III) adsorption of composite aerogels (A0.5, A1.0, A1.5) were fitted in pseudo-second-order kinetic model, and the calculated parameters were listed in Table 2. In terms of calculated pseudo-second-order rate constants k1, the sequence was as follows: k1 (A1.5) < k1 (A1.0) < k1 (A0.5). It suggested that although the adsorption rate of A1.5 was lower than A1.0 and A0.5, A1.5 still displayed significantly higher adsorption capacity than A1.0 and A0.5.
| Parameter | Pseudo-second-order kinetic model | |||
|---|---|---|---|---|
| qe(exp) (mg g−1) | qe (mg g−1) | k2 (g mg−1 min−1) | R2 | |
| a Initial As(III) concentration: 10 mg L−1; aerogels weight = 1.4 ± 0.1 g L−1; room temperature; pH = 6.9 ± 0.1. | ||||
| A0.5 | 3.38642 | 4.11391 | 0.000916598 | 0.9917 |
| A1.0 | 4.89886 | 6.5666 | 0.000335322 | 0.99785 |
| A1.5 | 5.81179 | 6.93888 | 0.0000600675 | 0.99498 |
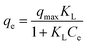 | (4) |
| qe = KFCen | (5) |
 | ||
| Fig. 5 Adsorption isotherms of As(III) on aerogels of A1.5 adsorbent at 303 K, 313 K and 323 K (experimental condition: aerogels weight = 1.4 ± 0.1 g L−1; pH = 6.9 ± 0.1 and contact time = 24 h). | ||
The adsorption data from the adsorption isotherms of the As(III) by using composite aerogels (A1.5) as adsorbents were also analyzed with both Langmuir and Freundlich equations. From Table 3, the linear plot of the Langmuir equation gave the R2 values close to unity while the R2 values from the Freundlich equation were lower, indicating the formation of monolayer adsorption of As(III) on the surface of composite aerogels (A1.5) in the studied concentrations. Similar studies were previously reported for adsorption of Cu2+, Fe2+, Pb2+ by polybenzoxazine aerogel.19 It was found that the adsorption capacity increased with increasing temperature implying that the adsorption process was endothermic in nature. KL was the Langmuir adsorption equilibrium constant which was a measure of the adsorption energy. A greater KL value indicated a steep initial slope of an isotherm implying a high affinity of the adsorbent towards the metal ions under the dilute conditions.20
| T/K | Langmuir | Freundlich | ||||
|---|---|---|---|---|---|---|
| KL/(L mg−1) | qmax/(mg g−1) | R2 | KF | n | R2 | |
| a Experimental condition: aerogels weight = 1.4 ± 0.1 g L−1; pH = 6.9 ± 0.1. | ||||||
| 303 | 0.07495 | 27.34481 | 0.99692 | 2.8218 | 1.70176 | 0.9904 |
| 313 | 0.08864 | 33.52488 | 0.99042 | 3.70763 | 1.66359 | 0.98201 |
| 323 | 0.21525 | 30.32912 | 0.98572 | 6.4697 | 2.1141 | 0.93142 |
ΔG = −RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ke Ke
| (6) |
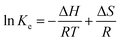 | (7) |
The negative ΔG values were 21.75, 22.90 and 26.02 kJ mol−1 for 303, 313 and 323 K respectively, implying the spontaneous nature of As(III) adsorption by composite aerogels of A1.5 adsorbent. However, the magnitude of ΔG decreased along with increasing temperature, implying a reduced degree of spontaneity at a higher temperature.22 From the van't Hoff plot (ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ke versus 1/T; plot not shown), the positive ΔH value (43.248 kJ mol−1) confirmed the endothermic nature of As(III) adsorption, while the negative value of ΔS [213.4 J (mol K)−1] revealed the decreased randomness (orderliness) at the solid/solution interface.
Ke versus 1/T; plot not shown), the positive ΔH value (43.248 kJ mol−1) confirmed the endothermic nature of As(III) adsorption, while the negative value of ΔS [213.4 J (mol K)−1] revealed the decreased randomness (orderliness) at the solid/solution interface.
Humic acid and alginate with concentration from 1 to 8 mg L−1 as total organic carbon (TOC) were used to model humic substances and polysaccharides, two major components of natural organic matter (NOM), respectively.23 As displayed in Fig. 6b, both humic acid and sodium alginate had slightly decreased As(III) removal efficiency of A1.5 aerogels. It was previously reported that humic acid with concentration up to 6.3 mg L−1 had insignificant effect on As(III) removal by FMBO.15 The above observation showed that composite aerogels synthesized in our study could be good adsorbents for As(III) removal regardless of absence of NOM (humic acid and alginate) in solutions up to 8 mg L−1.
3.3. Regeneration of composite aerogels
The reusability of adsorbent materials is important for effective water treatment.25 In order to investigate the regeneration potential of composite aerogels (A1.5) adsorbent, solutions with various concentration (0.1, 0.5 and 1.0 mol L−1) of NaOH were used as the desorption agent. Fig. 7a displayed that after 4 cycles of adsorption–regeneration process, the adsorption capacity just decreased from 4.62 mg g−1 to 2.92, 2.63 and 2.57 mg g−1 using 0.1, 0.5 and 1.0 mol L−1 NaOH respectively. Therefore, NaOH (0.1 mol L−1) was found to be more effective for regeneration of the composite aerogels. More interestingly, the 3D network of composite aerogels could be well kept during 4 adsorption–regeneration cycles. For most adsorbent materials, usually repeated high-speed centrifugation or filtration process had to be applied for their separation after adsorption.25 But for composite aerogels adsorbent in this study (Fig. 7b), their robust and stable macroscopic structure made the separation process very easy, indicating composite aerogels could be employed as an effective, convenient, and cost effective adsorbent for the trace As(III) removal from water with less separation procedure.3.4. Adsorption mechanism
FTIR spectra of the FMBO before and after reaction with As(III) has been shown in Fig. S3b.† For the FMBO sample, the band at 1627 cm−1 was assigned to the deformation of water molecules and indicated the presence of physisorbed water on the oxides. Three peaks at 1125, 1049 and 970 cm−1 were primarily due to the bending vibration of hydroxyl groups of iron oxides (Fe–OH) vibration.27 After adsorbing As(III), new band which corresponded to As–O stretching vibration27 appeared at 810 cm−1. This indicated that the As(III) was bound as a surface complex and not as a precipitated solid phase.26 From the FTIR analysis, it could be concluded that specific adsorption must occur at the aqueous arsenic/FMBO interface and the replacement of surface hydroxyl groups by the As(III) species was the main arsenic adsorption mechanism.
 | ||
| Fig. 8 XPS spectra of composite aerogels of A1.5 adsorbent before and after reaction with As(III) in the (a) survey scan; (b) Mn 2p region; (c) O 1s; and (d) As 3d energy regions. | ||
The oxidation states of manganese and adsorbed arsenite in composite aerogels of A1.5 adsorbent were measured by X-ray photoelectron spectroscopy. Binding energies of Mn 2p (Fig. 8b) for A1.5 composite aerogels were 640.5 eV and 651 eV, which assigned to the Mn 2p1/2 and Mn 2p3/2 transitions of Mn4+ (MnO2) in FMBO. However, the Mn 2p spectrum of the composite aerogels of A1.5 adsorbent after reaction with As(III) can be fitted with two components having binding energies at 641 eV and 639.5 eV, respectively. This indicated that both Mn(IV) and Mn(II) species were on the surface of adsorbent resulting from the reductive dissolution of MnO2. Mn(IV) and Mn(II) were found to account for 50.9% and 49.1% of Mn species. A similar phenomenon was also confirmed by other researchers.15 The binding energies of Fe 2p (figure not shown) were 711 eV and 748 eV (Fe 2p1/2 and 2p2/3), interpreting contributions from Fe(III) species only.28 Meanwhile, comparison of Fe 2p peak after reaction almost overlapped with the original one, implying that the chemical state of Fe was not changed.
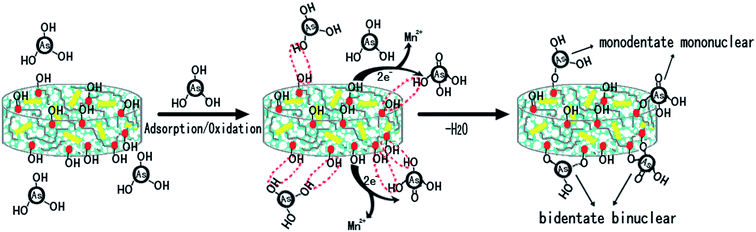
The O 1s peak (Fig. 8c) was fitted into three components. The peak with a binding energy of 530 eV was attributed to O2− (or M–O bonds), while the peak at 532 eV was assigned to OH− and the peak at 533.28 eV to H2O (bound water).29 The O 1s peaks at binding energy of 530.84 eV moved to 530.76 eV after reaction with As(III), which indicated that the M–OH groups participated in arsenite adsorption and M–O groups on the adsorbent surface formed after reaction. Fig. 8d depicted the fitted peak shapes of the As 3d spectra for the adsorbent after reaction with As(III). The binding energies of Ad 3d core level for As(III) and As(V) were 44.3 to 44.5 eV and 45.2 to 45.6 eV, respectively. As(III) and As(V) were found to account for 56.4% and 43.6% of As species, which indicated the process of As(III) oxidation–sorption on the surface of aerogels (Scheme 1).
4. Conclusion
This study demonstrated KGM-based composite aerogels with FMBO immobilization for the effective removal of As(III). The results indicated that the obtained aerogels possess good adsorption and oxidation properties for the removal of arsenite species in wide conditions. What is more, the composite aerogels (A0.5, A1.0 and A1.5) could easily separate as a whole after As(III) adsorption compare with nano-size FMBO particles alone. Involved with As(III) oxidation, the adsorption kinetics and isotherm data were better fitted by a pseudo-second-order model and a Langmuir model, respectively. The maximum binding capacity of composite aerogels A1.5 to As(III) according to Langmuir isotherm was 30.33 mg g−1 at pH 7 and 323 K, which was comparable to most of composite adsorbents before. The removal of As(III) by composite aerogels were achieved by the adsorption on Fe-oxides followed by the oxidation by Mn-oxides. The composite aerogels were multifunctional materials with good oxidation, adsorption, and separation properties and may have the potential application in water treatment. This study not only provides an alternative adsorbent for As(III) removal but also opens up new avenues for the application of KGM as the renewable framework materials in adsorption, separation and environment remediation field.Acknowledgements
This work got financial support by the Key Project of Chinese Ministry of Education (Grant no. 113047A) and the National Natural Science Foundation of China (Grant no. 31371841).References
- M. Bissen and F. H. Frimmel, Acta Hydrochim. Hydrobiol., 2003, 31, 9–18 CrossRef CAS PubMed.
- P. Smedley and D. Kinniburgh, Appl. Geochem., 2002, 17, 517–568 CrossRef CAS.
- N. E. Korte and Q. Fernando, Crit. Rev. Environ. Sci. Technol., 1991, 21, 1–39 CAS.
- J. L. Schnoor, Environmental modeling: fate and transport of pollutants in water, air, and soil, John Wiley and Sons, 1996 Search PubMed.
- A. Henglein, Chem. Rev., 1989, 89, 1861–1873 CrossRef CAS.
- K. Wu, R. Liu, H. Liu, F. Chang, H. Lan and J. Qu, J. Environ. Eng., 2011, 137, 1122–1127 CrossRef CAS.
- R. Jamshidi Gohari, W. Lau, T. Matsuura and A. Ismail, Sep. Purif. Technol., 2013, 118, 64–72 CrossRef CAS PubMed.
- F. Chang, J. Qu, H. Liu, R. Liu and X. Zhao, J. Colloid Interface Sci., 2009, 338, 353–358 CrossRef CAS PubMed.
- X. Li, K. He, B. Pan, S. Zhang, L. Lu and W. Zhang, Chem. Eng. J., 2012, 193, 131–138 CrossRef PubMed.
- H. Bi, Z. Yin, X. Cao, X. Xie, C. Tan, X. Huang, B. Chen, F. Chen, Q. Yang and X. Bu, Adv. Mater., 2013, 25, 5916–5921 CrossRef CAS PubMed.
- H. Wang, Y. Gong and Y. Wang, RSC Adv., 2014, 4, 45753–45759 RSC.
- X. Du, J. Li, J. Chen and B. Li, Food Res. Int., 2012, 46, 270–278 CrossRef CAS PubMed.
- X. Mi, G. Huang, W. Xie, W. Wang, Y. Liu and J. Gao, Carbon, 2012, 50, 4856–4864 CrossRef CAS PubMed.
- J. Han, T. Lei and Q. Wu, Cellulose, 2013, 20, 2947–2958 CrossRef CAS PubMed.
- G. Zhang, J. Qu, H. Liu, R. Liu and R. Wu, Water Res., 2007, 41, 1921–1928 CrossRef CAS PubMed.
- J. Lu, H. Liu, X. Zhao, W. Jefferson, F. Cheng and J. Qu, Colloids Surf., A, 2014, 455, 11–18 CrossRef CAS PubMed.
- Z. Gu, J. Fang and B. Deng, Environ. Sci. Technol., 2005, 39, 3833–3843 CrossRef CAS.
- V. Vadivelan and K. V. Kumar, J. Colloid Interface Sci., 2005, 286, 90–100 CrossRef CAS PubMed.
- T. Chaisuwan, T. Komalwanich, S. Luangsukrerk and S. Wongkasemjit, Desalination, 2010, 256, 108–114 CrossRef CAS PubMed.
- Y. H. Chang, C. F. Huang, W. J. Hsu and F. C. Chang, J. Appl. Polym. Sci., 2007, 104, 2896–2905 CrossRef CAS PubMed.
- B. S. Inbaraj, C. Chiu, G. Ho, J. Yang and B. Chen, Bioresour. Technol., 2008, 99, 1026–1035 CrossRef CAS PubMed.
- B. S. Inbaraj, J. Wang, J. Lu, F. Siao and B. Chen, Bioresour. Technol., 2009, 100, 200–207 CrossRef CAS PubMed.
- C. Shan and M. Tong, Water Res., 2013, 47, 3411–3421 CrossRef CAS PubMed.
- W. Xu, H. Wang, K. Wu, R. Liu, W. Gong and J. Qu, Water Environ. Res., 2012, 84, 521–528 CrossRef CAS.
- X. He, L. Cheng, Y. Wang, J. Zhao, W. Zhang and C. Lu, Carbohydr. Polym., 2014, 111, 683–687 CrossRef CAS PubMed.
- G. Zhang, H. Liu, R. Liu and J. Qu, J. Hazard. Mater., 2009, 168, 820–825 CrossRef CAS PubMed.
- Y. Zhang, M. Yang, X.-M. Dou, H. He and D.-S. Wang, Environ. Sci. Technol., 2005, 39, 7246–7253 CrossRef CAS.
- A. Glisenti, J. Mol. Catal. A: Chem., 2000, 153, 169–190 CrossRef CAS.
- X. Zhu and Y. Zhang, Appl. Surf. Sci., 1998, 125, 11–16 CrossRef CAS.
- H. Zhu, Y. Jia, X. Wu and H. Wang, J. Hazard. Mater., 2009, 172, 1591–1596 CrossRef CAS PubMed.
- K. Wu, T. Liu, W. Xue and X. Wang, Chem. Eng. J., 2012, 192, 343–349 CrossRef CAS PubMed.
- S. Kong, Y. Wang, H. Zhan, S. Yuan, M. Yu and M. Liu, Water Environ. Res., 2014, 86, 147–155 CrossRef CAS.
- B. A. Manning, S. E. Fendorf, B. Bostick and D. L. Suarez, Environ. Sci. Technol., 2002, 36, 976–981 CrossRef CAS.
- S. Wang and C. N. Mulligan, Environ. Int., 2008, 34, 867–879 CrossRef CAS PubMed.
- C. Escudero, N. Fiol, I. Villaescusa and J.-C. Bollinger, J. Hazard. Mater., 2009, 164, 533–541 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra03757c |
| This journal is © The Royal Society of Chemistry 2015 |