Monitoring of 2-butanone using a Ag–Cu bimetallic alloy nanoscale electrochemical sensor
Latif-ur Rahmanab,
Afzal Shah*a,
Suzanne Kay Lunsfordd,
Changseok Hanbe,
Mallikarjuna N. Nadagoudae,
Endalkachew Sahle-Demessief,
Rumana Qureshia,
Muhammad Saleem Khang,
Heinz-Bernhard Kraatzc and
Dionysios. D. Dionysiou*b
aDepartment of Chemistry, Quaid-i-Azam University, Islamabad, 45320, Pakistan. E-mail: afzal.shah@utoronto.ca; afzals_qau@yahoo.com
bEnvironmental Engineering and Science Program, Department of Biomedical, Chemical and Environmental Engineering, University of Cincinnati, Cincinnati, OH 45221-0012, USA. E-mail: dionysios.d.dionysiou@uc.edu
cDepartment of Physical and Environmental Sciences, University of Toronto, Scarborough, 1265 Military Trail, Toronto, M1C 1A4, Canada
dDepartment of Chemistry Wright State University, 3640 Colonel Glenn Hwy., Dayton, OH 45435, USA
eWater Quality Management Branch, Water Supply and Water Resources Division, National Risk Management Research Laboratory, The United States Environmental Protection Agency, Cincinnati, OH 45268, USA
fSustainable Technology Division, National Risk Management Research Laboratory, The United States Environmental Protection Agency, Cincinnati, OH 45268, USA
gNational Center of Excellence in Physical Chemistry, University of Peshawar, Khyber Pakhtunkhwa, Pakistan
First published on 12th May 2015
Abstract
Spherical shaped silver–copper alloy nanoparticles of 10–15 nm size were synthesized by an aqueous polymer solution method. The synthesized nanoscale Ag–Cu alloy was characterized by UV-Visible spectroscopy, X-ray diffraction spectroscopy, transmission electron microscopy (TEM), high resolution TEM, and energy dispersive spectroscopy. The XRD pattern showed high crystallinity and phase formation of the nanoparticles. The bimetallic alloy nanoparticles were coated over a well-polished glassy carbon electrode and the designed sensor was applied for the detection of a highly carcinogenic carbonyl compound, 2-butanone. Electrochemical studies revealed that PEI used as a capping agent also enhances the sensing of the modified electrode for the recognition of 2-butanone. The sensor developed from Ag–Cu (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) alloy nanoparticles showed the best sensing properties for the detection of 2-butanone as evidenced by electrochemical impedance spectroscopy and a 0.1 μM detection limit.
1) alloy nanoparticles showed the best sensing properties for the detection of 2-butanone as evidenced by electrochemical impedance spectroscopy and a 0.1 μM detection limit.
1. Introduction
Some intermetallic compounds having complex and stiff structures provide useful guidelines for the development of new material systems.1 Among such new material systems, bimetallic alloys of Ag and Cu are of utmost technological importance. The electronic conduction of Cu is significantly affected on coupling with Ag. Therefore, Ag–Cu alloy nanoparticles are synthesized for many industrial power applications.2 A literature survey reveals that Ag and Cu alloy formation is more spontaneous at 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 due to the homogeneous character of the crystalline structure.3 It has been reported that bimetallic alloys of Ag and Cu possess characteristic features and most of their properties such as free energy of mixing and electrical properties vary asymmetrically as a function of concentration.4 Such an asymmetry is generally attributed to size effect and the interactions between solute and solvent atoms or both. Different metals have different catalytic, surface, and magnetic properties.5 If their solid solution or alloy is prepared, their properties can be utilized more efficiently for the sensing of some toxic organic compounds.6 Furthermore, if the size of the alloy is brought to the nanoscale range, they will display unique physiochemical properties.5,6 Changing one of the alloy's metals, it is possible to change the spectroscopic, catalytic, thermal and electrochemical properties.6 Bimetallic alloy nanoparticles of noble metals such as Ag and Cu play a key role in the sensing of toxic organic compounds. Recent research on bimetallic alloy nanoparticles shows that three transition metals such as Cu, Ag and Au mostly form bimetallic alloy nanoparticles due their unique physiochemical nature.7 They can be used as a basic material for fabricating sensors to detect environmental toxins and carcinogenic substances.8
1 due to the homogeneous character of the crystalline structure.3 It has been reported that bimetallic alloys of Ag and Cu possess characteristic features and most of their properties such as free energy of mixing and electrical properties vary asymmetrically as a function of concentration.4 Such an asymmetry is generally attributed to size effect and the interactions between solute and solvent atoms or both. Different metals have different catalytic, surface, and magnetic properties.5 If their solid solution or alloy is prepared, their properties can be utilized more efficiently for the sensing of some toxic organic compounds.6 Furthermore, if the size of the alloy is brought to the nanoscale range, they will display unique physiochemical properties.5,6 Changing one of the alloy's metals, it is possible to change the spectroscopic, catalytic, thermal and electrochemical properties.6 Bimetallic alloy nanoparticles of noble metals such as Ag and Cu play a key role in the sensing of toxic organic compounds. Recent research on bimetallic alloy nanoparticles shows that three transition metals such as Cu, Ag and Au mostly form bimetallic alloy nanoparticles due their unique physiochemical nature.7 They can be used as a basic material for fabricating sensors to detect environmental toxins and carcinogenic substances.8
Sensing properties of Ag can be improved by making its alloy with metals such as Cu of nanoscale range. Hence, Ag–Cu alloy nanoparticles attract research attention due to their surface, optical, and electrochemical properties.8 Among bimetallic alloy nanoparticles, Ag–Au or Ag–Pt etc., Ag–Cu alloy nanoparticles are the preferred choice for sensor development as the presence of Cu content not only lowers the cost but also activates Ag for sensing the target compounds. Moreover, Ag–Cu alloy nanoparticles enhance the sensitivity of conducting polymer due to their electrocatalytic affect. In the presence of aqueous solution of polyethylene imine (PEI), the sensing properties of alloy nanoparticles can be significantly improved for the detection of carcinogenic organic compounds such as aldehydes and ketones.8 Previously, we have demonstrated the synthesis of Ag–Cu alloy nanoparticles using a polyol method.8 The other method available uses aqueous solution of hydrazine, which is not an environmentally friendly reducing agent. For the synthesis of bimetallic alloy nanoparticles, some organic plant extracts can also be used.9 But this method involves tedious experimental procedures such as preparation of medium and complicated synthesis steps.9 The most simple and convenient method is synthesis by aqueous polymer solution method using poly ethylene imine polymer.
Interestingly, PEI has dual functions, acting both as a capping agent and as a good nucleophile for the carbonyl compounds. However, it is more selective for ketones.10 The composite of Ag–Cu alloy nanoparticles and PEI has the ability of sensing ketone, even if present in trace amount. For achieving this objective, glassy carbon electrode (GCE) must be modified with PEI followed by Ag–Cu bimetallic alloy nanoparticles. In the absence of the conducting polymer, the bimetallic alloy nanoparticle-coated glassy carbon electrode cannot detect the target compound due to higher concentration of graphite. The conducting polymer provides a substrate for immobilization of the synthesized bimetallic nanoparticles on the anode that can easily oxidize the compound to be detected.10 In the current study, our point of interest is to detect one of the most hazardous and carcinogenic compound, 2-butanone, which mostly causes gastric and lung cancer.11 Hence, a sensor of Ag–Cu alloy nanoparticles trapped in PEI has been developed that can be utilized for the detection of the early stages of gastric and lung cancer. Since 2-butanone is volatile, it can be detected in mouth exhaled breath by PEI along with bimetallic alloy nanoparticles of Ag–Cu.12
2. Experimental
2.1. Materials and reagents
Silver nitrate (AgNO3), copper chloride (CuCl2), polyethylene imine (PEI), and ascorbic acid were purchased from Thermo Fisher Scientific Inc. Lithium perchlorate (LiClO4), potassium chloride (KCl), 2-butanone, acetonitrile and K4[Fe(CN)6] were obtained from ACROS Organics. All of the chemicals were analytical grade reagents and used as received. For the synthesis of Ag, Cu, and Ag–Cu alloy nanoparticles, a microwave oven made by LG company (model: MG 605 AP) was used.13 The entire solution of nanoparticles was kept in the microwave oven that was operated at 100% power of 1850 W and 3000 MHz frequency.2.2. Synthesis of mono metallic Ag and Cu nanoparticles
Ag nanoparticles were prepared by using a mixture of 0.01 M AgNO3 and 0.01 M PEI solutions. To prepare the mixture, 15 mL AgNO3 solution was added to 15 mL of PEI solution and the mixture was kept in the microwave oven for 30 minutes in a conical flask of 50 mL capacity. After the microwave irradiation, 15 mL ascorbic acid (0.1 M) was added and the solution turned dark brown, indicating the formation of Ag nanoparticles that were confirmed by UV-Visible spectroscopy.14 Cu nanoparticles were prepared following the same protocol but prior to PEI addition, 0.01 M copper chloride solution was purged with argon to avoid the formation of copper oxide. The heating time in the microwave oven for Cu nanoparticles was one hour. After 1 hour of microwave irradiation, the mixture turned bluish black indicating the formation of Cu nanoparticles.2.3. Synthesis of Ag–Cu bimetallic alloy nanoparticles
Ag–Cu bimetallic alloy nanoparticles were prepared by mixing 15 mL AgNO3 (0.01 M), 15 mL copper chloride (0.01 M) and 15 mL of 0.01 M PEI. Before using, the CuCl2 solution was purged with argon and heated with microwave irradiation for one hour. After the addition of AgNO3 to the mixture of CuCl2 and PEI, 15 mL of 0.1 M ascorbic acid was immediately added to the mixture followed by irradiation in microwave oven for 15 minutes. After the microwave irradiation, dark black color appeared showing the formation of Ag–Cu bimetallic alloy nanoparticles. Ag–Cu alloy nanoparticles of various compositions were synthesized by changing the molar concentration of Ag and Cu precursor solutions. Hence, Ag–Cu alloys with different ratios (i.e., 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and 1
2 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
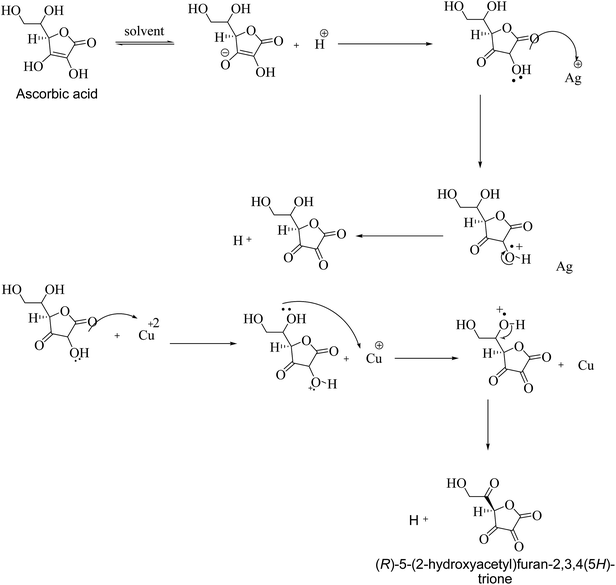
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) were synthesized. The whole process was carried out under controlled conditions with zero percent tolerance against contamination. They were confirmed by taking their UV-Visible spectra.15 During the synthetic process the precursor salts left Ag+ and Cu2+ that were reduced to Ag and Cu by ascorbic acid. Scheme 1 shows the reduction of Ag and Cu ions by ascorbic acid.
3) were synthesized. The whole process was carried out under controlled conditions with zero percent tolerance against contamination. They were confirmed by taking their UV-Visible spectra.15 During the synthetic process the precursor salts left Ag+ and Cu2+ that were reduced to Ag and Cu by ascorbic acid. Scheme 1 shows the reduction of Ag and Cu ions by ascorbic acid.
2.4. Synthesis of composite electrode
A glassy carbon electrode was cleaned with alumina slurry followed by washing with acetone and rinsing with deionized water. The polished electrode was inserted in the already prepared suspension of Ag–Cu alloy nanoparticles and was kept for 15 minutes in the suspension. After careful drying it was used as working electrode. During synthesis of nanoparticles and development of composite electrode, extreme care was taken for avoiding any sort of contamination because small quantities of impurities can lead to drastic changes in properties.2.5. Electrochemical studies
A 0.1 M lithium perchlorate solution was prepared in acetonitrile solvent (HPLC grade provided by ACROS Organics) and was used as a supporting electrolyte. Conventional electrode system of modified was used with glassy carbon electrode, platinum wire and Ag/AgCl containing freshly prepared KCl as working, auxiliary, and reference electrodes respectively. The electrochemical properties of the composite electrode were investigated using Bio analytical System Epsilon Potentiostat (BAS100B). The composite electrode was characterized by cyclic voltammetry (CV) in 0.02 M K4[Fe(CN)6] solution along with 0.1 M lithium perchlorate solution as supporting electrolyte.16,17 CV was carried out using well-polished glassy carbon electrode as well as GCE modified with bimetallic alloy nanoparticles of Ag and Cu embedded in PEI polymer.3. Results and discussion
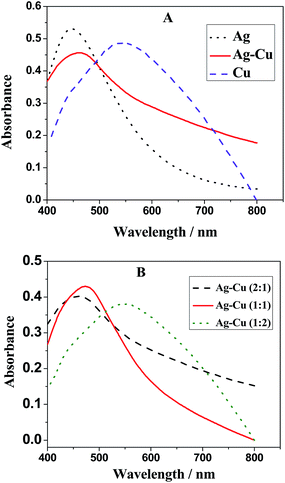
UV-Visible spectroscopy gave a single absorption band around 475 nm for Ag–Cu alloy nanoparticles, which is a strong evidence of the successful synthesis process.18 Fig. 1A shows a single plasmon band of Ag–Cu alloy nanoparticles at 475 nm that is at the middle of the maximum absorption spectra of Ag (at 425 nm) and Cu (at 545 nm) nanoparticles. It is also confirmed that the bimetallic nanoparticles are an alloy and not physically embedded monometallic nanoparticles of Ag and Cu metals.18 This also disregards the core–shell structure of bimetallic nanoparticles.19 Fig. 1B shows UV-Visible spectra of Ag–Cu alloy nanoparticles prepared in various ratios (i.e., 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 1
1 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2). A red shift of the light absorption peak was observed with an increase of Cu content in Ag–Cu alloy nanoparticles. This result suggests maximum absorption for Ag–Cu 1
2). A red shift of the light absorption peak was observed with an increase of Cu content in Ag–Cu alloy nanoparticles. This result suggests maximum absorption for Ag–Cu 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 at shorter wavelength. The maximum absorbance (for 1 mM solution) and maximum wavelengths for monometallic and bimetallic nanoparticles are listed in Table 1.
1 at shorter wavelength. The maximum absorbance (for 1 mM solution) and maximum wavelengths for monometallic and bimetallic nanoparticles are listed in Table 1.
| S. no | Nanoparticles | Maximum absorbance | λmax. | Average size (nm) |
|---|---|---|---|---|
| 1 | Ag | 0.54 | 425 | 11 |
| 2 | Cu | 0.48 | 545 | 10 |
| 3 | Ag–Cu (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
0.40 | 455 | 15 |
| 4 | Ag–Cu (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
0.45 | 475 | 12 |
| 5 | Ag–Cu (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
0.38 | 540 | 13 |
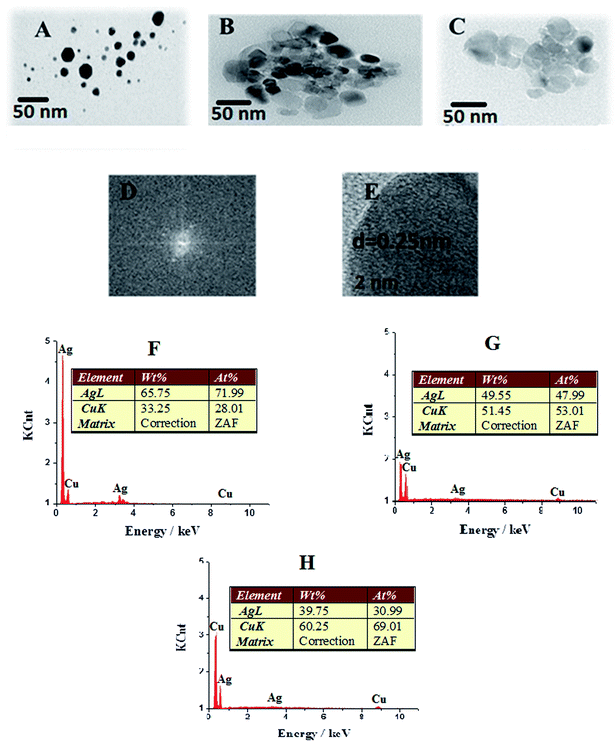
Fig. 2 shows transmission electron microscopy (TEM, Philips CM20) of Ag–Cu 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (A), Ag–Cu 1
1 (A), Ag–Cu 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (B), Ag–Cu 1
1 (B), Ag–Cu 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (C) and high resolution TEM (HR-TEM, JEM-2010F, JEOL) images of Ag–Cu 1
2 (C) and high resolution TEM (HR-TEM, JEM-2010F, JEOL) images of Ag–Cu 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (D and E) as well as the result of EDS analysis for Ag–Cu 2
1 (D and E) as well as the result of EDS analysis for Ag–Cu 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (F), Ag–Cu 1
1 (F), Ag–Cu 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (G) and Ag–Cu 1
1 (G) and Ag–Cu 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 (H). Fig. 2D corresponds to selected area electron diffraction (SAED) pattern of HR-TEM, while Fig. 2H is the HR-TEM image of single Ag–Cu alloy nanoparticle. TEM images indicated that Ag–Cu alloy nanoparticles are spherical with diameters of 10–15 nm. The crystal size of the samples was also evaluated using TEM images. Both particle sizes using HR-TEM and TEM are very similar to the particle size calculated from XRD.20 Fig. 2H shows the energy dispersive spectroscopy of Ag–Cu alloy nanoparticles at the ratio of 1
2 (H). Fig. 2D corresponds to selected area electron diffraction (SAED) pattern of HR-TEM, while Fig. 2H is the HR-TEM image of single Ag–Cu alloy nanoparticle. TEM images indicated that Ag–Cu alloy nanoparticles are spherical with diameters of 10–15 nm. The crystal size of the samples was also evaluated using TEM images. Both particle sizes using HR-TEM and TEM are very similar to the particle size calculated from XRD.20 Fig. 2H shows the energy dispersive spectroscopy of Ag–Cu alloy nanoparticles at the ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. Such figure signifies maximum composition of Ag and Cu in Ag–Cu alloy nanoparticles. The energy dispersive spectra as shown in Fig. 2F–H indicate purity of alloy nanoparticles with their respective elemental percentage compositions. The inset in Fig. 3 (XRD) shows electron diffraction pattern for the alloy nanoparticles in which the radii of the four main fringe patterns are in the ratio of 5/2
1. Such figure signifies maximum composition of Ag and Cu in Ag–Cu alloy nanoparticles. The energy dispersive spectra as shown in Fig. 2F–H indicate purity of alloy nanoparticles with their respective elemental percentage compositions. The inset in Fig. 3 (XRD) shows electron diffraction pattern for the alloy nanoparticles in which the radii of the four main fringe patterns are in the ratio of 5/2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9/2
9/2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 13/2. They show matching with (111), (200), (220), (222), and (311) planes which confirms that crystalline structure of Ag–Cu alloy nanoparticles is face-centered cubic (fcc).21
13/2. They show matching with (111), (200), (220), (222), and (311) planes which confirms that crystalline structure of Ag–Cu alloy nanoparticles is face-centered cubic (fcc).21
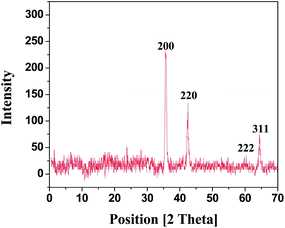 | ||
Fig. 3 XRD pattern of Ag–Cu (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) alloy nanoparticles synthesizes by aqueous polymer solutions from AgNO3, CuCl2 in the present of PEI as a capping agent. 1) alloy nanoparticles synthesizes by aqueous polymer solutions from AgNO3, CuCl2 in the present of PEI as a capping agent. | ||
Fig. 3 shows XRD spectra of two peaks corresponding to (200) and (220) planes for Ag and Cu appearing at 2θ of 36° and 42°, respectively. They were taken as desired peaks and the size of the nanoparticles was calculated using Debye Scherrer formula. The calculated particle sizes are summarized in Table 1. Five characteristic peaks corresponding to (111), (200), (220), (222), and (311) planes appearing at 2θ = 18°, 36°, 42°, 60°, and 64°, respectively, confirming that Ag–Cu alloy nanoparticles have fcc structure.21 One of the peaks corresponding to (200) plane is more intense as compared to others which clarify that (200) plane is more exposed having maximum orientation. This further demonstrates that Ag–Cu alloy nanoparticles are highly crystalline as evidenced by HR-TEM and XRD analyses.22
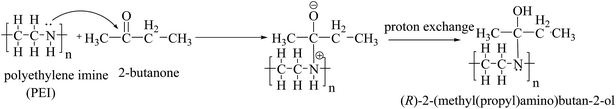
FTIR analysis was performed to verify the binding ability and stabilization of Ag–Cu bimetallic alloy nanoparticles by polyethylene imine after the alloy nanoparticles were used for the detection of 2-butanone. Before using the prepared bimetallic alloy nanoparticles for the sensing application, they were washed four times with acetone to remove (R)-5-(2-hydroxyacetyl)furan-3,3,4-(5-H)-trione and (R)-2-oxo-2-(3,4,5-trioxotetrahydrofuran-2-yl)acetaldehyde which are byproducts during the reduction of Ag and Cu ion by ascorbic acid (see Scheme 1). Fig. 4A and B show FTIR analysis of PEI and Ag–Cu (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) alloy nanoparticles, respectively. The intense band centered at 1650.68 cm−1 for both PEI and Ag–Cu alloy nanoparticles (stabilized by PEI) appeared due to stretching and bending vibration of NH as shown by Scheme 2.23 Two other bands appearing at 1496.12 and 1464.53 cm−1 were assigned to methylene (sym/antisym) vibration of hydrocarbons present at PEI. Two other less intense bands are reported at 1443.86 and 1424.59 cm−1 for the N–C bond in PEI.24 The prominent band appeared at 1293.28 cm−1 was due to adjacent NH corresponding to stretching and bending vibration. Fig. 4B shows the change in band at 1073.58 cm−1 which is due to the reduction of Ag and Cu ions by NH group. The appearance of band at 576.12 cm−1shows not only the reduction of Ag and Cu ions by the hydroxyl group of ascorbic acid, but also indicates PEI as a capping agent. PEI acts as nucleophile and makes a coordinate covalent bond with Ag and Cu ions that gives intense band centered at 576 cm−1 as shown in Fig. 4B.25 Sensing of 2-butanone by PEI giving (R)-2-(methyl (propyl) amino) butan-2-ol compound can be shown by the chemical reaction depicted in Scheme 2.
1) alloy nanoparticles, respectively. The intense band centered at 1650.68 cm−1 for both PEI and Ag–Cu alloy nanoparticles (stabilized by PEI) appeared due to stretching and bending vibration of NH as shown by Scheme 2.23 Two other bands appearing at 1496.12 and 1464.53 cm−1 were assigned to methylene (sym/antisym) vibration of hydrocarbons present at PEI. Two other less intense bands are reported at 1443.86 and 1424.59 cm−1 for the N–C bond in PEI.24 The prominent band appeared at 1293.28 cm−1 was due to adjacent NH corresponding to stretching and bending vibration. Fig. 4B shows the change in band at 1073.58 cm−1 which is due to the reduction of Ag and Cu ions by NH group. The appearance of band at 576.12 cm−1shows not only the reduction of Ag and Cu ions by the hydroxyl group of ascorbic acid, but also indicates PEI as a capping agent. PEI acts as nucleophile and makes a coordinate covalent bond with Ag and Cu ions that gives intense band centered at 576 cm−1 as shown in Fig. 4B.25 Sensing of 2-butanone by PEI giving (R)-2-(methyl (propyl) amino) butan-2-ol compound can be shown by the chemical reaction depicted in Scheme 2.
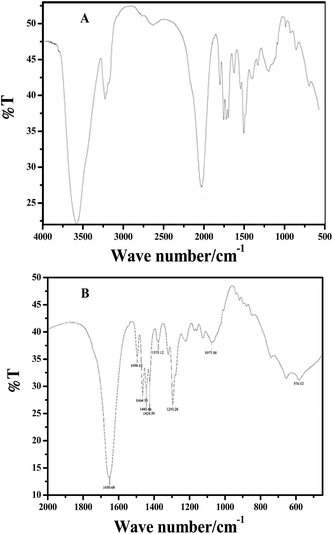 | ||
| Fig. 4 FTIR spectra of (A) PEI and (B) Ag–Cu alloy nanoparticles scanning at the range of 2000 cm−1 to 450 cm−1. | ||
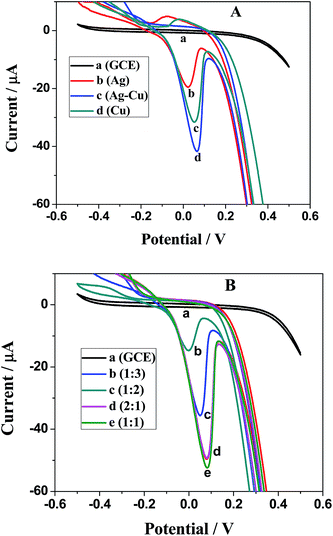
Electrochemical studies were carried out at constant pH using 0.1 M KCl solution. The highest current response and the good stability of working electrode were two eminent properties for the selection of this electrolyte for buffer preparation.26 Such electrolyte was also acting as a supporting electrolyte along with lithium perchlorate. Cyclic voltammograms were taken for monometallic and bimetallic alloy nanoparticles of Ag and Cu prepared by aqueous solution methods using PEI as a capping agent.27 The applied potential range was from −0.5 V to +0.5 V (vs. SCE). Fig. 5A shows CVs of Ag, Cu and Ag–Cu alloy nanoparticles, indicating prominent oxidation peak of Ag–Cu alloy nanoparticles at the potential of 90 mV as listed in Table 2.28 Fig. 5B shows cyclic voltammograms of Ag–Cu alloy nanoparticles prepared at various molar proportions of Ag![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Cu (i.e., 2
Cu (i.e., 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and 1
2 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3). By comparing the amperometric response of different working electrodes modified with Ag–Cu (1
3). By comparing the amperometric response of different working electrodes modified with Ag–Cu (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, 1
2, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 and 2
3 and 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) alloy nanoparticles, the most prominent anodic peak was obtained by Ag–Cu (1
1) alloy nanoparticles, the most prominent anodic peak was obtained by Ag–Cu (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) at the potential range of 80–100 mV due to its more uniform particle size.29 The data can be seen in Table 3. Hence, a glassy carbon electrode modified with Ag–Cu (1
1) at the potential range of 80–100 mV due to its more uniform particle size.29 The data can be seen in Table 3. Hence, a glassy carbon electrode modified with Ag–Cu (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) alloy nanoparticles prepared by the aqueous polymer solution method using PEI as a capping agent was used for selective detection of the carcinogenic organic compound, 2-butanone.30
1) alloy nanoparticles prepared by the aqueous polymer solution method using PEI as a capping agent was used for selective detection of the carcinogenic organic compound, 2-butanone.30
| S. no | Nanoparticles | Epa (mV) | Ipa (A) |
|---|---|---|---|
| 1 | Ag | 40 | −1.50 × 10−5 |
| 2 | Cu | 60 | −2.55 × 10−5 |
| 3 | Ag–Cu | 90 | −4.50 × 10−5 |
| S. no | Ag–Cu alloy nanoparticles | Epa (mV) | Ipa (A) |
|---|---|---|---|
| 1 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
90 | −5.2 × 10−5 |
| 2 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 2 |
30 | −2.6 × 10−5 |
| 3 | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 3 |
10 | −1.2 × 10−5 |
| 4 | 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
60 | −4.8 × 10−5 |
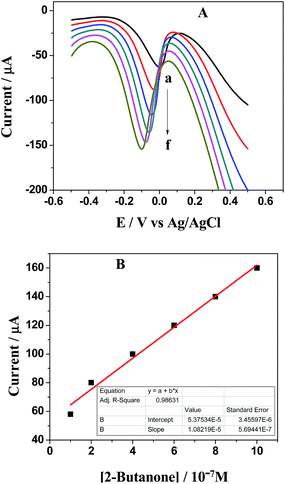
An observation of cyclic voltammograms shown in Fig. 6 reveals that anodic peak current increases with increase in concentration of 2-butanone.31 This figure shows change in oxidation peak current from 0.2 × 10−5 to 0.4 × 10−5 A with an increase in concentration of 2-butanone. The sensing was applicable for the lowest concentration of 2-butanone i.e., 1.5 μM. Square wave voltammetry was carried out to detect the lowest possible concentration of 2-butanone which was our point of interest. Interestingly, Fig. 7A shows the lowest sensing concentration of 0.1 μM which is significant achievement in this work. The plot in Fig. 7B shows a direct correlation between oxidation peak current (with negative value) and concentration of 2-butanone with negative shift in the electrode potential.32 Anodic peak current increases from 0.6 × 10−5 to 1.6 × 10−5 A with increase in the concentration from 0.1 to 1 μM at the potential range of 90 to 60 mV. This work is important and novel in the sense that instead of Au, the less expensive metal Cu was used along with Ag and no extra compound was required other than PEI.
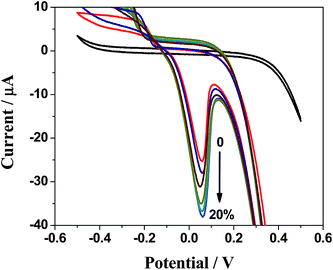 | ||
| Fig. 6 CV of Ag–Cu alloy nanoparticles modified electrode when exposed to various concentration of 2-butanone at the range of 0 to 20% v/v in 0.1 M KCl solution at scan rate of 100 mV s−1. | ||
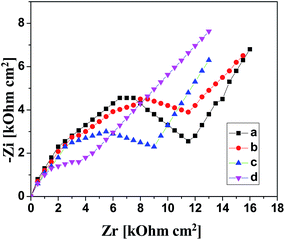
Electrochemical impedance spectroscopy (EIS) was also carried out as it provides more informative results about the composite and NPs modified electrodes. Fig. 8 shows a distinctive change in the value of charge transfer resistance (Rct) for unmodified and modified GCE with polymer PEI, Ag–Cu alloy nanoparticles and their composite (PEI/Ag–Cu 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 NPs/GCE) for the oxidation of 2-butanone. Higher value of Rct (8.49 × 103 Ω) shows inefficient electron transfer process on bare GCE. For GCE modified with PEI, Fig. 8b shows a decrease in the Rct value (8.12 × 103 Ω) that can be attributed to the negative charge on PEI which decreases the conductivity due to the possible resistance offered by the layer to ferrocyanide anions.33 Fig. 8c shows a further decrease in Rct value (4.82 × 103 Ω) when GCE is coated with Ag–Cu 1
1 NPs/GCE) for the oxidation of 2-butanone. Higher value of Rct (8.49 × 103 Ω) shows inefficient electron transfer process on bare GCE. For GCE modified with PEI, Fig. 8b shows a decrease in the Rct value (8.12 × 103 Ω) that can be attributed to the negative charge on PEI which decreases the conductivity due to the possible resistance offered by the layer to ferrocyanide anions.33 Fig. 8c shows a further decrease in Rct value (4.82 × 103 Ω) when GCE is coated with Ag–Cu 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 alloy nanoparticles due to electron transfer process accelerated by NPs. For the composite electrode a further decrease in Rct value (2.25 × 103 Ω) can be seen in Fig. 8d. This decrease caused by PEI along with Ag–Cu alloy nanoparticles demonstrates that PEI and Ag–Cu alloy nanoparticles facilitate in accelerating the electrons transfer process. The information resulted from EIS strongly supports the results of CV and SWV.
1 alloy nanoparticles due to electron transfer process accelerated by NPs. For the composite electrode a further decrease in Rct value (2.25 × 103 Ω) can be seen in Fig. 8d. This decrease caused by PEI along with Ag–Cu alloy nanoparticles demonstrates that PEI and Ag–Cu alloy nanoparticles facilitate in accelerating the electrons transfer process. The information resulted from EIS strongly supports the results of CV and SWV.
The reproducibility of the newly developed sensor was examined by processing it for the same concentration of 2-butanone. For the sensing of 2-butanone five equally modified glassy carbon electrodes having different compositions of Ag and Cu (in Ag–Cu alloy nanoparticles) were prepared. These precise values i.e., 35.2, 33.5, 36.6, 34.5, and 35.9 V were obtained. The stability of these nanosensors was also investigated by testing the same sensor after two weeks for the same concentration of 2-butanone. A slight deviation was observed which may be due to the electron transfer process between nanoparticles and polymer used.
4. Conclusions
An environmental friendly method was developed for the synthesis of bimetallic Ag–Cu alloy nanoparticles. They were developed as a sensor for the detection of 2-butanone. PEI was used as capping as well as sensing detector. The electrochemical catalytic behavior of Ag–Cu alloy nanoparticles was found to depend on the size of nanoparticles and the Ag![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Cu molar ratio. Bimetallic nanoparticles exhibited higher catalytic activities than the monometallic nanoparticles. It is anticipated that the designed sensor can successfully be applied for the detection of early stage of gastric and lung cancer caused by 2-butanone. At early stages of such diseases, a very small amount of 2-butanone is evaporated (less than in 1 μM) which can easily be detected by the sensor developed from Ag–Cu alloy nanoparticles.
Cu molar ratio. Bimetallic nanoparticles exhibited higher catalytic activities than the monometallic nanoparticles. It is anticipated that the designed sensor can successfully be applied for the detection of early stage of gastric and lung cancer caused by 2-butanone. At early stages of such diseases, a very small amount of 2-butanone is evaporated (less than in 1 μM) which can easily be detected by the sensor developed from Ag–Cu alloy nanoparticles.
5. Notice
The U.S. Environmental Protection Agency, through its Office of Research and Development, funded and managed, or partially funded and collaborated in, the research described herein. It has been subjected to the Agency's administrative review and has been approved for external publication. Any opinions expressed in this paper are those of the author(s) and do not necessarily reflect the views of the agency, therefore, no official endorsement should be inferred. Any mention of trade names or commercial products does not constitute endorsement or recommendation for use.Acknowledgements
This work was jointly supported by the Chemistry Department of Quaid-i-Azam University, Islamabad, Pakistan, Department of Physical and Environmental Sciences, University of Toronto, Canada and Department of Biomedical, Chemical and Environmental Engineering (DBCEE), University of Cincinnati, Cincinnati, Ohio, USA. The financial support of Higher Education Commission of Pakistan is also acknowledged.References
- S. Mansour, N. Boutarek and S. E. Amar, Adv. Mater. Lett., 2014, 5, 356–359 CAS.
- M. A. AL-Jalali, Adv. Mater. Lett., 2014, 5, 14–16 CAS.
- A. M. Vora, Adv. Mater. Lett., 2013, 4, 146–154 CAS.
- A. Kumar, I. S. Jha and B. P. Singh, Adv. Mater. Lett., 2013, 4, 155–159 Search PubMed.
- M. Valodkar, S. Mode, A. Pall and S. Thakore, Mater. Res. Bull., 2011, 46, 384–389 CrossRef CAS PubMed.
- A. Shah, L. Rahman, R. Qureshi and Z. Rahman, Rev. Adv. Mater. Sci., 2012, 30, 133–149 CAS.
- M. N. K. Chowdhury and M. D. H. Beg, Mater. Lett., 2013, 98, 26–29 CrossRef CAS PubMed.
- L. Rahman, R. Qureshi, A. Shah and M. M. Yasinzai, C. R. Chim., 2012, 15, 533–538 CrossRef CAS PubMed.
- A. Ceylan, K. Jastrezmsky and S. I. Shah, Metall. Mater. Trans. A, 2006, 37, 2033–2039 CrossRef.
- F. Dugonjic and J. Plank, J. Appl. Polym. Sci., 2011, 121, 1262–1275 CrossRef PubMed.
- Y. Zhang, G. Gao, Q. Qian and D. Cui, Nano Lett., 2012, 7, 461–475 CrossRef PubMed.
- B. Michell and J. Boromo, J. Clin. Pharmacol., 2000, 14, 303–312 Search PubMed.
- M. Valodkar, S. Modi, A. Pal and S. Thakore, Mater. Res. Bull., 2011, 46, 384–389 CrossRef CAS PubMed.
- A. Pal, S. Shah and S. Devi, Colloids Surf., A, 2007, 30, 2483–2487 Search PubMed.
- W. Robert, J. Scott, M. O. Wilson and M. Richard, J. Phys. Chem. B, 2005, 109, 692–704 CrossRef PubMed.
- J. Wang, J. Li, A. J. Baca, J. Hu, F. Zhou, W. Yan and D. W. Pang, J. Anal. Chem., 2003, 75, 3941–3945 CAS.
- J. Han, Y. Zahou, Y. Q. Chai, L. Mao, Y. L. Yuan and D. W. Pang, Talanta, 2011, 85, 130–135 CrossRef CAS PubMed.
- A. Pal, S. Shah and S. Devi, Mater. Chem. Phys., 2009, 114, 530–532 CrossRef CAS PubMed.
- Y. F. Huang and H. Lie, J. Colloid Interface Sci., 2006, 301, 145–154 CrossRef CAS PubMed.
- A. Pal, S. Shah and S. Devi, Colloids Surf., A, 2007, 302, 51–57 CrossRef CAS PubMed.
- X. Liu, A. Wang, X. Wang, C. Yuan and T. Zhang, Chem. Commun., 2008, 3187–3189 RSC.
- X. Liu and T. Jan, Catal. Today, 2011, 160, 103–108 CrossRef CAS PubMed.
- P. Rajasekharreddy, P. Rani and B. Sreedhar, J. Nanopart. Res., 2010, 12, 1711–1721 CrossRef CAS.
- M. A. Faramarzi and H. Farootanfar, Colloids Surf., B, 2011, 87, 23–27 CrossRef CAS PubMed.
- M. Shakibali, H. Farootanfar, M. K. Mulazadeh, Z. Bagherzadeh, V. N. Nafissi, A. R. Shaverdi and M. A. Faramarzi, J. Appl. Biomater., 2010, 57, 71–75 Search PubMed.
- R. Imani, A. Iglic, A. P. F. Turner and A. Tiwari, Electrochem. Commun., 2014, 40, 84–87 CrossRef CAS PubMed.
- H. Cai, N. Zhu, Y. Jiang, P. He and Y. Fang, Biosens. Bioelectron., 2003, 18, 1311–1319 CrossRef CAS.
- N. Karimian, M. Vagin, M. Hossein, A. Zavar, M. Chamsaz, A. P. F. Turner and A. Tiwari, Biosens. Bioelectron., 2013, 50, 492–498 CrossRef CAS PubMed.
- Y. W. Cao, R. C. Jin and C. A. Mirkin, J. Am. Chem. Soc., 2001, 123, 7961–7962 CrossRef CAS.
- A. Kamyabi, M. Hajari, N. Turner and A. Tiwari, Talanta, 2013, 116, 801–808 CrossRef PubMed.
- O. Parlak, A. Tiwari, A. P. F. Turner and A. Tiwari, Biosens. Bioelectron., 2013, 49, 53–62 CrossRef CAS PubMed.
- X. Liu, A. Wang, X. Wang, C. Yuan, T. Zhang and J. F. Lee, Prog. Mater. Sci., 2013, 23, 317–325 Search PubMed.
- L. Rahman, A. Shah, S. B. Khan, A. M. Asiri, H. Hussain, C. Han, R. Qureshi, M. N. Ashiq, M. A. Zia and M. Ishaq, J. Appl. Electrochem., 2015, 45, 463–472 CrossRef.
| This journal is © The Royal Society of Chemistry 2015 |