SiO2-confined silicon/carbon nanofiber composites as an anode for lithium-ion batteries
Mahmut Diricanab,
Yao Lua,
Kun Fua,
Huseyin Kizilb and
Xiangwu Zhang*a
aFiber and Polymer Science Program, Department of Textile Engineering, Chemistry and Science, North Carolina State University, Raleigh, NC 27695-8301, USA. E-mail: xiangwu_zhang@ncsu.edu; Fax: +1-919-515-6532; Tel: +1-919-515-6547
bNano-Science and Nano-Engineering Program, Graduate School of Science, Engineering and Technology, Istanbul Technical University, Istanbul 34469, Turkey
First published on 8th April 2015
Abstract
Because of its ultra-high theoretical capacity (4200 mA h g−1), Si is considered as the most promising anode material candidate for next-generation high-energy lithium-ion batteries. However, the practical use of Si based anodes is constrained by the high volume change (up to 400%) of the Si active material during cycling. Intensive volume change of Si causes severe pulverization, loss of electrical contact between Si particles and the carbon current collector, and unstable SEI formation on the electrode surface. Herein, we introduce nanoscale silica-coated silicon/carbon (Si@C–SiO2) nanofiber composites that can maintain their structural stability during repeated cycling. Results indicated that nanoscale SiO2 coating of Si@C nanofibers helped preserve the Si particles within the nanofiber structure, resulting in stable solid electrolyte interphase formation and improved cycling performance. Electrochemical performance results showed that the Si@C–SiO2 nanofiber composite anodes had good capacity retention of 89.8% and high coulombic efficiency of 97.2% at the 50th cycle. It is, therefore, demonstrated that nanoscale SiO2 coating is an effective method to improve the electrochemical performance of Si@C nanofiber composite anodes.
1. Introduction
Among the existing rechargeable battery technologies, lithium-ion batteries have drawn great attention in recent years because of their superior properties, including high energy density, durable cycle life and good power performance.1,2 With recent improvements in technologies of electric vehicles and portable electronic devices, development of high-capacity electrode materials for high-energy lithium-ion batteries has become critically important to meet their energy requirements.3–5 For most current commercial lithium-ion batteries, graphitic materials are used in the anode because of their low cost and long cycling performance. However, graphitic anode materials are not capable of meeting the ever-growing capacity requirements of future portable electronics and electric vehicles because of their low specific capacity of 372 mA h g−1.6,7 On the other hand, lithium storage capacities of alloy-type anodes (e.g., silicon, tin, germanium, and their oxides) are much higher than those of commercially-used intercalation-type graphite anodes.Among all alloy-type anodes, Si provides the highest theoretical specific capacity of 4200 mA h g−1. Because of its ultra-high theoretical capacity, Si is considered as the most promising candidate for next-generation high-energy lithium-ion batteries.8,9 However, similar to other alloy-type anode materials, the insertion of lithium ions into Si during charge and discharge causes high volumetric change (up to 400%), which results in intense pulverization of active Si material and significant loss of electrical contact between Si particles and carbon conductor. In addition, high volume expansion and intense pulverization of active Si particles triggers extra electrolyte consumption on the fresh surface of Si, leading to unstable solid-electrolyte-interphase (SEI) film formation.10,11 These drawbacks result in the severe capacity fading of Si-based anodes during repetitive lithiation and delithiation processes.12 Reducing the size of Si particles into the nanoscale range and embedding them into carbon structures are some of the accepted effective methods for minimizing the volume expansion problem of Si-based anodes. Nano-sized Si/carbon (Si/C) composite anodes combine the advantageous properties of Si (high capacity) and carbon (excellent electronic conductivity and structural stability).13,14
Carbon nanofibers are one of the best options for hosting active Si due to their high electronic conductivity, large surface area, and good electrochemical stability.15 However, electrochemical performance results of the reported Si/C nanofiber composites are well below the expected performance due to direct exposure of Si nanoparticles with the electrolyte on the fiber surfaces which leads to heavy SEI formation.16,17 The exposed Si nanoparticles could also be detached from the nanofiber surfaces during the repetitive cycling processes. To eliminate the aforementioned problems, Fu, et al., and Dirican, et al., introduced an additional nanoscale disordered carbon layer onto the surface of Si@C nanofibers by chemical vapor deposition (CVD) technique to prevent direct exposure of the Si nanoparticles with the electrolyte solution and increase the mechanical bonding strength between the Si nanoparticles and carbon nanofiber matrix.15,18 With the additional protective layer, not only stable SEI is formed but also the detachment of Si nanoparticles from the nanofiber surfaces can be prevented. Confinement of the Si nanoparticles can also be ensured by using an easy and low-cost sol–gel method to form nanoscale amorphous SiO2 coating on Si@C nanofibers. Similar to nanoscale carbon coating, nanoscale SiO2 coating can lead to stable SEI formation on coated nanofiber surfaces and increased mechanical bonding between Si nanoparticles and carbon nanofiber matrix.
In this study, we report nanoscale SiO2 coated Si@C (Si@C–SiO2) nanofiber composites as high-energy anodes for lithium-ion batteries. We introduced a feasible method to improve the electrochemical performance of previously reported Si@C nanofiber composite electrodes. The main target of this study is to propose a simple, but effective approach for the confinement of Si nanoparticles inside the Si@C nanofiber composite electrodes. Si@C nanofibers were produced by simple electrospinning and subsequent heat treatment processes. The resultant Si@C nanofibers were coated with thin amorphous SiO2 layers by using sol–gel TEOS solution to achieve further improvement on the cycling performance of the composite anodes. Nanoscale SiO2 coating helped maintain the Si nanoparticles within the nanofiber structure and stabilize the SEI formation, resulting in improved cycling performance. Electrochemical performance results show that Si@C–SiO2 nanofiber composite anodes exhibit capacity retention of 89.8% and coulombic efficiency of 97.2% after 50th cycles. Although better electrochemical performance (e.g., higher capacity retention or higher coulombic efficiency) has been reported in the literature,14,19 this work does present an easy and versatile approach for improving the electrochemical performance of previously reported Si@C nanofiber composite electrodes. Nevertheless, future study can be conducted to refine the structure of Si@C–SiO2 nanofiber composite and further improve their electrochemical performance. In addition, the method used in this work can be applied to most of the previously reported Si/C based composite anodes to improve their performance.
2. Experimental
2.1. Nanofiber preparation
Tetraethyl orthosilicate (TEOS, 99%), polyacrylonitrile (PAN, Mw = 150![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1), N,N-dimethylformamide (DMF), ammonium hydroxide solution (NH4OH, 28–30%), and ethanol were purchased from Sigma-Aldrich. Si nanoparticles (diameter: 30–50 nm) were purchased from Nanostructures & Amorphous Materials, Incorporated. All chemicals were used without further purification.
000 g mol−1), N,N-dimethylformamide (DMF), ammonium hydroxide solution (NH4OH, 28–30%), and ethanol were purchased from Sigma-Aldrich. Si nanoparticles (diameter: 30–50 nm) were purchased from Nanostructures & Amorphous Materials, Incorporated. All chemicals were used without further purification.
Fig. 1 schematically illustrates the entire fabrication approach of electrospun Si@PAN nanofibers, Si@C nanofibers, and Si@C–SiO2 nanofibers. For the preparation of Si@PAN nanofibers, PAN (8 wt%) was first dissolved in DMF at 60 °C and mechanically stirred for 24 h. Si nanoparticles (20 wt% with respect to PAN) were then added into the as-prepared PAN/DMF solution and stirred at room temperature for 24 h, followed by ultrasonic treatment for 1 h to obtain a homogenous dispersion. The as-prepared Si/PAN dispersion was then electrospun into Si@PAN nanofibers with a flow rate of 0.75 ml h−1, a voltage of 16 kV, and a tip-to-collector distance of 25 cm. Si@C nanofiber composites were prepared from Si@PAN nanofibers through stabilization in air environment at 280 °C for 5.5 h with a heating rate of 5 °C min−1, followed by carbonization at 700 °C for 2 h in argon atmosphere with a heating rate of 2 °C min−1, during which PAN was pyrolyzed to carbon.
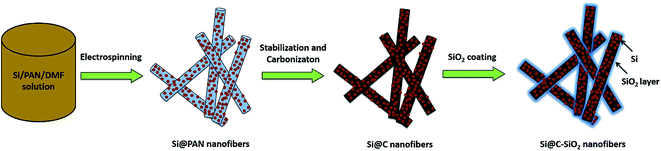 | ||
| Fig. 1 Schematic illustration of the fabrication process of electrospun Si@PAN nanofibers, Si@C nanofibers, and Si@C–SiO2 nanofibers. | ||
For the preparation of Si@C–SiO2 nanofiber composites, the as-prepared Si@C nanofibers were coated with nanoscale SiO2 by using a sol–gel TEOS solution. The sol–gel TEOS solution was prepared by adding 3 ml ammonium hydroxide into a mixture of 125 ml ethanol and 30 ml water, followed by the addition of 2.4 g TEOS dropwise under vigorous stirring. The reaction was left at room temperature under vigorous stirring for 30 minutes. During the synthesis of Si@C–SiO2 nanofiber composites, 100 mg Si@C nanofibers were immersed into the as-prepared sol–gel TEOS solution and treated for different duration times. Solution treatment time was varied from 0.5 h, 1 h to 2 h to investigate its effect on electrochemical performance. After solution treatment, the Si@C–SiO2 nanofiber composites were first dried in vacuum oven and later washed with deionized water for three times.
2.2. Structure characterization
The morphology of nanofiber composites was examined by field emission scanning electron microscope (FE-SEM, FEI Verios 460 L) and scanning transmission electron microscope (STEM, JEOL 2000FX). The structure of composite nanofibers was investigated by Fourier transform infrared spectroscopy (FTIR, Nicolet Nexus 470), wide angle X-ray diffraction (WAXD, Rigaku Smartlab), and Renishaw Raman microscope. Compositions of the nanofiber composites were examined by elemental analysis (Perkin Elmer 2400 Series II CHNS/O Elemental Analyzer).2.3. Electrochemical evaluation
The electrochemical properties of Si@C–SiO2 nanofiber composites were tested using CR 2032-type coin cells. The working electrodes were prepared by mixing 70 wt% grounded nanofiber composites with 20 wt% carbon black and 10 wt% sodium alginate binder. The slurry of each sample was coated on copper foil substrates and vacuum-dried for 12 h at 80 °C. The average mass loading of the electrodes used in this study was 1.2 mg cm−2. Lithium ribbon (99.9%, Aldrich) was used as the counter electrode and Celgard 2400 membrane was used as the separator. The electrolyte consisted of a 1 M solution of LiPF6 dissolved in ethylene carbonate (EC) + dimethyl carbonate (DMC) + diethyl carbonate (DEC) (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 by volume, MTI Corporation). Coin cells were assembled in a high-purity argon-filled glove box. The electrochemical performance was investigated by galvanostatic charge–discharge experiments at a constant current density of 100 mA g−1 (around C/10) with cut-off potentials between of 0.01 and 2.00 V on a LAND-CT 2001A battery test system.
1 by volume, MTI Corporation). Coin cells were assembled in a high-purity argon-filled glove box. The electrochemical performance was investigated by galvanostatic charge–discharge experiments at a constant current density of 100 mA g−1 (around C/10) with cut-off potentials between of 0.01 and 2.00 V on a LAND-CT 2001A battery test system.
3. Results and discussion
3.1. Morphology and structure
Fig. 1 shows schematically the fabrication approach of Si@PAN nanofibers, Si@C nanofibers, and Si@C–SiO2 nanofibers. Fabrication process of these nanofiber composites composed of three steps: electrospinning of Si/PAN solution into Si@PAN nanofibers, stabilization and carbonization of electrospun nanofibers to obtain Si@C nanofibers, and SiO2 coating of Si@C nanofibers to form Si@C–SiO2 nanofibers. Compositions of the nanofiber composites were calculated based on the elemental analysis data. Elemental analysis results revealed that Si@C nanofibers contained 24.05% Si, 10.30% SiO2, and 65.65% C. The SiO2 component of Si@C nanofibers might come from the surface oxidation of Si nanoparticles. The Si@C–SiO2 nanofiber composite with 0.5 h coating time had 22.80% Si, 14.94% SiO2, and 62.26% C. Here, the SiO2 content was the sum of SiO2 coating layer on the fiber surface and the oxidized SiO2 on Si nanoparticles. With increase in coating time, the SiO2 content increased. The Si@C–SiO2 nanofiber composite with 1 h coating time was composed of 22.40% Si, 16.45% SiO2, and 61.15% C, and the Si@C–SiO2 nanofiber composite with 2 h coating time contained 21.98% Si, 17.97% SiO2, and 60.05% C.The morphology of Si@C nanofibers and Si@C–SiO2 nanofibers with different coating times was examined by using SEM, as shown in Fig. 2. Continuous nanofibers were observed from the SEM image of Si@C nanofibers (Fig. 2A). From Fig. 2B–D, it is seen that the surface morphologies of Si@C–SiO2 nanofibers were similar to that of Si@C nanofibers. Si nanoparticles were dispersed throughout the carbon nanofiber matrices with some particles agglomerated on the nanofiber surfaces. From the SEM images of Si@C–SiO2 nanofibers, the presence of SiO2 coating layers was not apparent due to limited image resolution. To clearly observe the thin SiO2 layers, TEM observation was carried out, as discussed below.
 | ||
| Fig. 2 SEM images of Si@C nanofiber composite (A) and Si@C–SiO2 nanofiber composites with different coating times: (B) 0.5 h, (C) 1 h, and (D) 2 h. | ||
Fig. 3 shows TEM images of Si/C and Si@C–SiO2 nanofibers. TEM images show that for all four nanofiber composites, the Si nanoparticles were not completely confined in the nanofiber matrices and some nanoparticles were agglomerated and located on nanofiber surfaces, which agreed with the SEM observation in Fig. 2. For the pristine Si@C nanofibers, no SiO2 coating was observed on the exposed Si nanoparticles (Fig. 3A). On the other hand, Si@C–SiO2 nanofibers prepared with 0.5 h, 1 h, and 2 h SiO2 coating times exhibited amorphous SiO2 coatings on the Si nanoparticle surfaces (Fig. 3B–D).
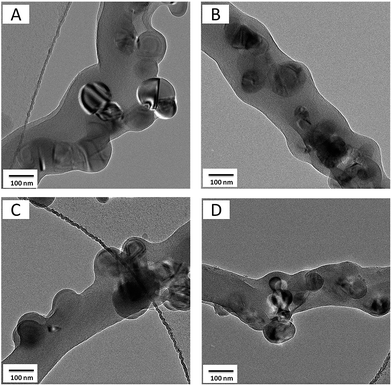 | ||
| Fig. 3 TEM images of Si@C nanofiber composite (A) and Si@C–SiO2 nanofiber composites with different coating times: (B) 0.5 h, (C) 1 h, and (D) 2 h. | ||
High-magnification TEM images of nanofiber composites are presented in Fig. 4 to further illustrate the amorphous SiO2 coating layers on the Si@C–SiO2 nanofiber composites. For the pristine Si@C nanofiber composite, no SiO2 coating was observed on the nanofiber surface (Fig. 4A). However, nanoscale amorphous SiO2 layers were clearly shown on the surfaces of Si@C–SiO2 nanofiber composites. The thickness of the SiO2 coating layers increased, on average, from 7 nm, 10 nm, to 13 nm, respectively, when the SiO2 coating time increased from 0.5 h, 1 h, to 2 h. These nanoscale SiO2 layers were important for stable SEI formation on the active materials during the repetitive electrode reactions, especially for the Si nanoparticles exposed on the carbon nanofiber surfaces.
 | ||
| Fig. 4 High-magnification TEM image of Si@C nanofiber composite (A) and Si@C–SiO2 nanofiber composites with different coating times: (B) 0.5 h, (C) 1 h, and (D) 2 h. | ||
Fig. 5 shows the FTIR spectra of Si@C and Si@C–SiO2 nanofiber composites. For all nanofiber composites, two characteristic peaks were indexed at around 1280 cm−1 and 1590 cm−1, which were attributed to the C–C and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretch bonds, respectively.20 FTIR spectra of Si@C and Si@C–SiO2 nanofiber composites also showed a characteristic SiO2 side peak at around 1100 cm−1, corresponding to the symmetric stretching, asymmetrical stretching, and bending vibrations in Si–O–Si bonds.21 Since the pristine Si@C nanofiber composite do not have SiO2 coating, the observed side peak at around 1100 cm−1 was weaker than those of Si@C–SiO2 nanofiber composites. The presence of side peak at around 1100 cm−1 for Si@C nanofibers may be ascribed to the partial oxidation of Si nanoparticles.
C stretch bonds, respectively.20 FTIR spectra of Si@C and Si@C–SiO2 nanofiber composites also showed a characteristic SiO2 side peak at around 1100 cm−1, corresponding to the symmetric stretching, asymmetrical stretching, and bending vibrations in Si–O–Si bonds.21 Since the pristine Si@C nanofiber composite do not have SiO2 coating, the observed side peak at around 1100 cm−1 was weaker than those of Si@C–SiO2 nanofiber composites. The presence of side peak at around 1100 cm−1 for Si@C nanofibers may be ascribed to the partial oxidation of Si nanoparticles.
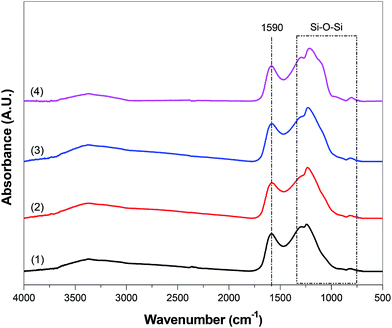 | ||
| Fig. 5 FTIR spectra of Si@C nanofiber composite (1) and Si@C–SiO2 nanofiber composites with different coating times: (2) 0.5 h, (3) 1 h, and (4) 2 h. | ||
The X-ray diffraction (XRD) patterns of Si@C and Si@C–SiO2 nanofiber composites are shown in Fig. 6. For all nanofiber composites, similar diffraction peaks were indexed. Diffraction peaks at 2θ of about 29°, 47°, 56°, 69°, 76°, and 88° represented the (111), (220), (311), (400), (331), and (422) planes of Si crystals in carbon nanofiber composites, respectively.22,23 In addition, as shown in Fig. 6, all nanofiber composites showed a broad and weak diffraction peak at around 2θ = 25°, which could be indexed as the (002) planes of disordered carbon structure, indicating the amorphous nature of the carbon nanofiber matrix.15,16
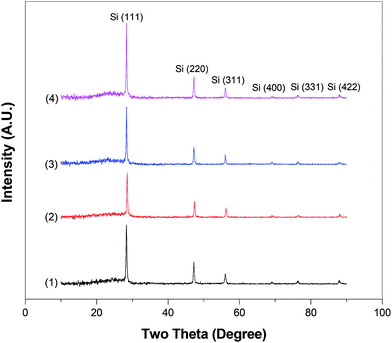 | ||
| Fig. 6 WAXD patterns of Si@C nanofiber composite (1) and Si@C–SiO2 nanofiber composites with different coating times: (2) 0.5 h, (3) 1 h, and (4) 2 h. | ||
The structural evolution of the nanofiber composites was also investigated by Raman spectroscopy. Fig. 7 shows the Raman spectra of Si@C and Si@C–SiO2 nanofiber composites. For all nanofiber composites, the characteristic carbon peaks of D-band (1360 cm−1), indicating the disordered sp2 phase, and G-band (1582 cm−1), representing the in-plane stretching vibration mode of E2g graphite, were indexed.24,25 From Fig. 7, it was also seen that due to the presence of Si nanoparticles, these nanofiber composites exhibited a Si peak at around 522 cm−1, which is a position for the transverse optical phonon.26 In addition, Si@C–SiO2 nanofiber composites with different SiO2 coating times had a broad and weak peak at around 900 cm−1, representing the SiO2 component of the Si@C–SiO2 nanofiber composites.27
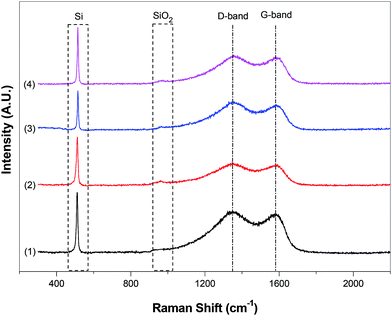 | ||
| Fig. 7 Raman spectra of Si@C nanofiber composite (1) and Si@C–SiO2 nanofiber composites with different coating times: (2) 0.5 h, (3) 1 h, and (4) 2 h. | ||
3.2. Electrochemical performance
Galvanostatic charge–discharge tests were conducted between 0.01 and 2.0 V at a constant current density of 100 mA g−1 to evaluate the electrochemical performance of the nanofiber composite anodes. The specific capacities of all nanofiber composite electrodes were calculated based on the total mass of nanofiber composites by including Si, C, and SiO2 coating if any. Fig. 8 represents the first, second and tenth cycle of the galvanostatic charge–discharge profiles of Si@C and Si@C–SiO2 nanofiber composites. The first-cycle discharge capacities were 1506, 1570, 1567, and 1481 mA h g−1, respectively, for Si@C nanofiber composite and Si@C–SiO2 nanofiber composites with different coating times: 0.5 h, 1 h, and 2 h. For the Si@C nanofiber composite, the initial discharge and charge capacities reached 1506 and 1138 mA h g−1, respectively, with a coulombic efficiency of 75.6%. At the 10th cycle, the discharge and charge capacities changed to 1193 mA h g−1 and 1168 mA h g−1, with a coulombic efficiency of 97.9% (Fig. 8A). On the other hand, the initial discharge and charge capacities of Si@C–SiO2 nanofiber composite with 0.5 h coating time were 1570 and 1222 mA h g−1, respectively, with a coulombic efficiency of 77.8%. At the 10th cycle, the discharge and charge capacities of Si@C–SiO2 nanofiber composite with 0.5 h coating time reduced to 1181 mA h g−1 and 1157 mA h g−1, with a coulombic efficiency of 98.0% (Fig. 8B). Initial charge and discharge capacities of Si@C–SiO2 nanofiber composite with 1 h coating time were 1567 and 1215 mA h g−1, respectively, with a coulombic efficiency of 77.5%. At the 10th cycle, the discharge and charge capacities of Si@C–SiO2 nanofiber composite with 1 h coating time reduced to 1078 mA h g−1 and 1058 mA h g−1, with a coulombic efficiency of 98.2% (Fig. 8C). For the Si@C–SiO2 nanofiber composite with 2 h coating time, the initial discharge and charge capacities reached 1481 and 1183 mA h g−1, respectively, with a coulombic efficiency of 79.8%. At the 10th cycle, the discharge and charge capacities reduced to 993 mA h g−1 and 957 mA h g−1, with a coulombic efficiency of 96.3% (Fig. 8D). These results show that for both first and tenth cycles, the coulombic efficiencies of Si@C–SiO2 nanofiber composites with 0.5 and 1 h coating times were better than those of Si@C nanofiber composite. From Fig. 8, it is also seen that there are two voltage plateaus, indexed at 0.7 V and 0.2 V, respectively, in the first-cycle discharge curve of Si@C–SiO2 nanofiber composites. The plateau at 0.7 V was associated with the electrolyte decomposition and SEI formation while the plateau at 0.2 V was ascribed to electrochemical reactions between lithium ions and the SiO2 ingredient of the composite nanofibers. During the initial lithiation process, SiO2 might be reduced to electrochemically inactive Li2O and Li4SiO4.28,29 On the other hand, without the presence of SiO2, the Si@C nanofiber composite did not exhibit the plateau at 0.2 V. When comparing the charge–discharge profiles of Si@C–SiO2 composites with different coating times, it can be found that the voltage plateau under the curve of the electrolyte reduction/degradation stage at 0.7 V was shortened with increasing SiO2 coating time, indicating that less electrolyte participated in the formation of SEI due to the presence of SiO2 coating on the nanofiber surface.15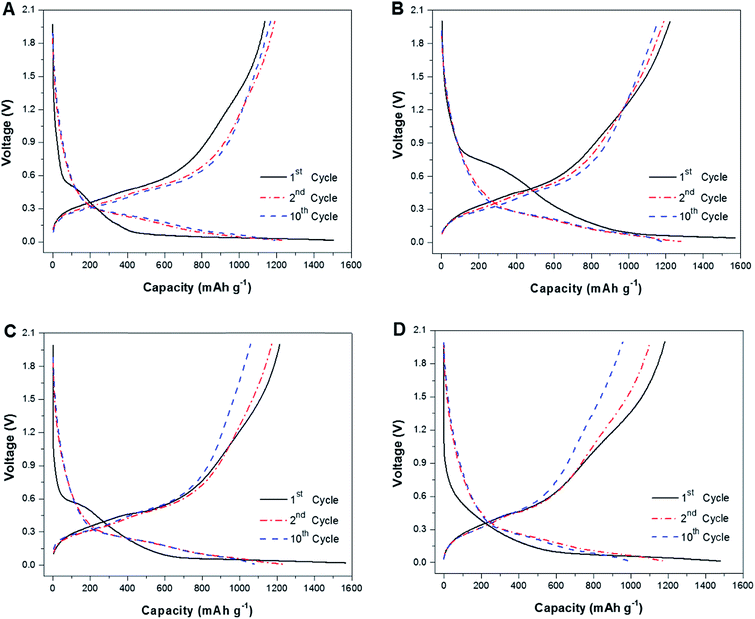 | ||
| Fig. 8 Galvanostatic charge–discharge profiles of Si@C nanofiber composite (A) and Si@C–SiO2 nanofiber composites with different coating times: (B) 0.5 h, (C) 1 h, and (D) 2 h. | ||
The cycling performance of Si@C and Si@C–SiO2 nanofiber composites was compared in Fig. 9. Capacity retention and coulombic efficiency of the Si/C nanofiber composite at the 50th cycle were around 42.5% and 95.9%, respectively, indicating unstable cycling behavior. On the other hand, relatively stable cycling performance was achieved for the Si@C–SiO2 nanofiber composite with 0.5 h coating time. At the 50th cycle, the capacity retention and coulombic efficiency of Si@C–SiO2 nanofiber composite with 0.5 h coating time were around 89.8% and 97.2%, respectively. Compared to Si@C nanofiber composite, Si@C–SiO2 nanofiber composite with 0.5 h coating time showed much slower capacity decay in 50 cycles. This result demonstrated that the nanoscale SiO2 coating enhanced the confinement of Si nanoparticles on nanofiber surfaces and helped maintain the structural integrity of the electrode during the cycling process. Furthermore, during the initial lithiation process, the amorphous SiO2 structure might be reduced to Li2O and Li4SiO4, which could help accommodate the volume expansion caused by the alloying of Si nanoparticles with Li, and thereby increased the cycling stability by minimizing the pulverization of the active Si material and the loss of electrical contact between Si nanoparticles and the carbon nanofiber matrix.28,29 SiO2 coating layer also supported more stable SEI formation by preventing direct contact of the electrolyte with the active Si material. Similar effects of nanoscale SiO2 coating have been reported by Wu, et al., who obtained improved cycling stability for ion-permeable nanoscale SiO2 surrounded silicon nanotube anodes.30 From Fig. 9, it is also seen that the cycling performance of Si@C–SiO2 nanofiber composites was worsened while increasing SiO2 coating time to 1 h or 2 h. The capacity retention and coulombic efficiency of Si@C–SiO2 nanofiber composite with 1 h coating time was around 75.0% and 97.8%, respectively, at the 50th cycle. For the Si@C–SiO2 nanofiber composite with 2 h coating time, the capacity retention and coulombic efficiency were only around 64.5% and 96.2%, respectively, at the 50th cycle. Therefore, to achieve the enhanced cycling performance, it is very crucial to select an appropriate SiO2 coating thickness for Si@C–SiO2 nanofiber composites. The cycling performance was affected by the easiness of Li-ion diffusion through the SiO2 and SEI layer formed on the nanofiber surface. The fast capacity fading of Si@C–SiO2 nanofiber composites with 1 and 2 h coating times might be caused by the hindered Li-ion insertion/deinsertion during cycling due to their relatively thick SiO2 coating layers. Similar results were reported for nano Si powder electrodes with different oxide layer thicknesses by Yu, et al.31 In many reported studies, Si based anodes were evaluated with a cutoff voltage window of 0.01 and 1.0 V. To put our results into perspective, we tested our electrodes between 0.01 and 1.0 V at a constant current density of 100 mA g−1, and the measured coulombic efficiencies are shown in Fig. 10. Results revealed that the coulombic efficiencies of the nanofiber composites in a cutoff voltage window of 0.01 and 1.0 V were quite similar to those tested with a cutoff voltage window of 0.01 and 2.0 V.
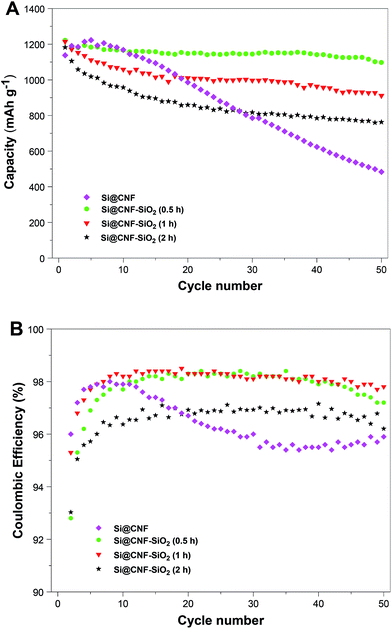 | ||
| Fig. 9 Cycling performance (A) and coulombic efficiencies (B) of Si@C nanofiber composite and Si@C–SiO2 nanofiber composites with different coating times: 0.5 h, 1 h, and 2 h. | ||
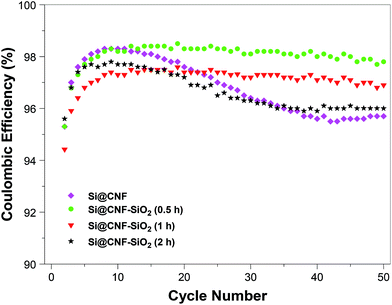 | ||
| Fig. 10 Coulombic efficiencies of Si@C nanofiber composite and Si@C–SiO2 nanofiber composites with different coating times: 0.5 h, 1 h, and 2 h with a cutoff voltage window of 0.01 and 1.0 V. | ||
The Si@C–SiO2 nanofiber composite with 0.5 h coating time showed the best cycling performance among all the studied nanofiber composites. Therefore, this composite was further investigated for its rate capability under various current densities, as shown in Fig. 11. It was observed that the charge capacities were around 1177, 1028, 873, and 683 mA h g−1, respectively, at current densities of 100, 200, 400, and 800 mA g−1. The coulombic efficiency remained relatively constant as the current density increased. In addition, the charge capacity reached 1052 mA h g−1 when the current density returned to 100 mA g−1 after ongoing cycles at higher current densities. This charge capacity (1052 mA h g−1) was very close to initial charge capacity (1177 mA h g−1), demonstrating the good rate capability of the Si@C–SiO2 nanofiber composite.
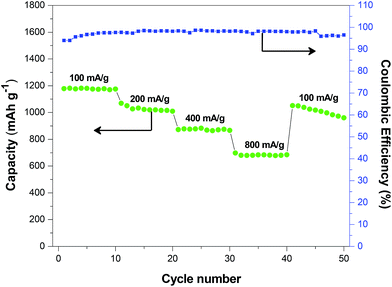 | ||
| Fig. 11 Rate capability of Si@C–SiO2 nanofiber composite with 0.5 h coating time cycled at different current densities. | ||
Electrochemically tested cells were disassembled after 50 cycles to analyze the surface morphology change of Si@C and Si@C–SiO2 nanofiber composites using TEM (Fig. 12). Before TEM observation, the cycled electrodes were washed by HCl to remove SEI. Due to the repeated large volume expansion and contraction of Si nanoparticles, the Si@C nanofiber composite suffered major structural deterioration during the cycling and Si nanoparticles fell off from the carbon matrix, causing irreversible structure damage (Fig. 12A). On the other hand, in Si@C–SiO2 nanofiber composites, most of the Si nanoparticles were maintained and encapsulated inside the nanofiber matrix after cycling, without catastrophic structural damage to the nanofiber surfaces (Fig. 12B–D). Results demonstrated that the SiO2 coating helped restrict Si nanoparticles in carbon matrix during the repetitive cycles.
 | ||
| Fig. 12 TEM images of cycled Si@C nanofiber composite (A) and Si@C–SiO2 nanofiber composites with different coating times: (B) 0.5 h, (C) 1 h, and (D) 2 h after 50 cycles. | ||
4. Conclusions
Nanoscale SiO2 coating of Si@C nanofiber composites were performed to prevent direct exposure of the Si nanoparticles with the electrolyte solution and increase the mechanical bonding strength between the Si nanoparticles and carbon nanofiber matrix. Results demonstrated that nanoscale SiO2 coating of Si@C nanofibers helped maintain the Si nanoparticles within the nanofiber structure and stabilize the SEI formation on the electrode surface, resulting in improved cycling performance. Electrochemical performance results showed that the Si@C–SiO2 nanofiber composite anode with 0.5 h coating time exhibited good capacity retention of 89.8% and high coulombic efficiency of 97.2% at the 50th cycle. It is, therefore, demonstrated that Si@C–SiO2 nanofiber composites are promising anode material candidate for next-generation high-energy lithium-ion batteries.References
- H. Wu, G. Zheng, N. Liu, T. J. Carney, Y. Yang and Y. Cui, Nano Lett., 2012, 12, 904–909 CrossRef CAS PubMed.
- X. Zhang, L. Ji, O. Toprakci, Y. Liang and M. Alcoutlabi, Polym. Rev., 2011, 51, 239–264 CrossRef CAS.
- H. Gwon, H.-S. Kim, K. U. Lee, D.-H. Seo, Y. C. Park, Y.-S. Lee, B. T. Ahn and K. Kang, Energy Environ. Sci., 2011, 4, 1277–1283 CAS.
- J.-Z. Wang, C. Zhong, S.-L. Chou and H.-K. Liu, Electrochem. Commun., 2010, 12, 1467–1470 CrossRef CAS PubMed.
- M. Dirican, M. Yanilmaz, K. Fu, Y. Lu, H. Kizil and X. Zhang, J. Power Sources, 2014, 264, 240–247 CrossRef CAS PubMed.
- B. S. Lee, S. B. Son, J. H. Seo, K. M. Park, G. Lee, S. H. Lee, K. H. Oh, J. P. Ahn and W. R. Yu, Nanoscale, 2013, 5, 4790–4796 RSC.
- L. Ji, Z. Lin, R. Zhou, Q. Shi, O. Toprakci, A. J. Medford, C. R. Millns and X. Zhang, Electrochim. Acta, 2010, 55, 1605–1611 CrossRef CAS PubMed.
- T. H. Hwang, Y. M. Lee, B. S. Kong, J. S. Seo and J. W. Choi, Nano Lett., 2012, 12, 802–807 CrossRef CAS PubMed.
- Q. Zhang, W. Zhang, W. Wan, Y. Cui and E. Wang, Nano Lett., 2010, 10, 3243–3249 CrossRef CAS PubMed.
- M. Pharr, K. Zhao, X. Wang, Z. Suo and J. J. Vlassak, Nano Lett., 2012, 12, 5039–5047 CrossRef CAS PubMed.
- K. Fu, O. Yildiz, H. Bhanushali, Y. Wang, K. Stano, L. Xue, X. Zhang and P. D. Bradford, Adv. Mater., 2013, 25, 5109–5114 CrossRef CAS PubMed.
- K. Fu, Y. Lu, M. Dirican, C. Chen, M. Yanilmaz, Q. Shi, P. D. Bradford and X. Zhang, Nanoscale, 2014, 6, 7489–7495 RSC.
- B. Wang, X. Li, X. Zhang, B. Luo, M. Jin, M. Liang, S. A. Dayeh, S. Picraux and L. Zhi, ACS Nano, 2013, 7, 1437–1445 CrossRef CAS PubMed.
- L.-F. Cui, Y. Yang, C.-M. Hsu and Y. Cui, Nano Lett., 2009, 9, 3370–3374 CrossRef CAS PubMed.
- K. Fu, L. Xue, O. Yildiz, S. Li, H. Lee, Y. Li, G. Xu, L. Zhou, P. D. Bradford and X. Zhang, Nano Energy, 2013, 2, 976–986 CrossRef CAS PubMed.
- L. Ji and X. Zhang, Carbon, 2009, 47, 3219–3226 CrossRef CAS PubMed.
- Y. Li, B. Guo, L. Ji, Z. Lin, G. Xu, Y. Liang, S. Zhang, O. Toprakci, Y. Hu, M. Alcoutlabi and X. Zhang, Carbon, 2013, 51, 185–194 CrossRef CAS PubMed.
- M. Dirican, M. Yanilmaz, K. Fu, O. Yildiz, H. Kizil, Y. Hu and X. Zhang, J. Electrochem. Soc., 2014, 161, A2197–A2203 CrossRef CAS PubMed.
- J. Xie, X. Yang, S. Zhou and D. Wang, ACS Nano, 2011, 5, 9225–9231 CrossRef CAS PubMed.
- Z. Lin, L. Ji and X. Zhang, Mater. Lett., 2009, 63, 2115–2118 CrossRef CAS PubMed.
- T. Pirzada, S. A. Arvidson, C. D. Saquing, S. S. Shah and S. A. Khan, Langmuir, 2012, 28, 5834–5844 CrossRef CAS PubMed.
- H. S. Choi, J. G. Lee, H. Y. Lee, S. W. Kim and C. R. Park, Electrochim. Acta, 2010, 56, 790–796 CrossRef CAS PubMed.
- J. Xiao, W. Xu, D. Wang, D. Choi, W. Wang, X. Li, G. L. Graff, J. Liu and J.-G. Zhang, J. Electrochem. Soc., 2010, 157, A1047 CrossRef CAS PubMed.
- Y.-S. Ding, W.-N. Li, S. Iaconetti, X.-F. Shen, J. DiCarlo, F. S. Galasso and S. L. Suib, Surf. Coat. Technol., 2006, 200, 3041–3048 CrossRef CAS PubMed.
- L. Su, Y. Jing and Z. Zhou, Nanoscale, 2011, 3, 3967–3983 RSC.
- C. Meier, S. Lüttjohann, V. G. Kravets, H. Nienhaus, A. Lorke and H. Wiggers, Phys. E, 2006, 32, 155–158 CrossRef CAS PubMed.
- Z. Li, W. Gao, A. Meng, Z. Geng and L. Gao, J. Phys. Chem. C, 2008, 113, 91–96 Search PubMed.
- Y. Yao, J. Zhang, L. Xue, T. Huang and A. Yu, J. Power Sources, 2011, 196, 10240–10243 CrossRef CAS PubMed.
- J. Wang, H. Zhao, J. He, C. Wang and J. Wang, J. Power Sources, 2011, 196, 4811–4815 CrossRef CAS PubMed.
- H. Wu, G. Chan, J. W. Choi, Y. Yao, M. T. McDowell, S. W. Lee, A. Jackson, Y. Yang, L. Hu and Y. Cui, Nat. Nanotechnol., 2012, 7, 310–315 CrossRef CAS PubMed.
- B.-C. Yu, Y. Hwa, C.-M. Park, J.-H. Kim and H.-J. Sohn, RSC Adv., 2013, 3, 9408–9413 RSC.
| This journal is © The Royal Society of Chemistry 2015 |
