Development of a novel and green microwave-assisted hydrogen peroxide digestion method for total sulphur quantitative extraction in coal samples prior to inductively coupled plasma-optical emission spectroscopy and ion-chromatography determination
Nomvano Mketo,
Philiswa N. Nomngongo and
J. Catherine Ngila*
Department of Applied Chemistry, University of Johannesburg, PO Box 17011, Doornfontein 2028, Johannesburg, South Africa. E-mail: jcngila@uj.ac.za; Fax: +27 11 5596425; Tel: +27 11 5596196
First published on 21st April 2015
Abstract
A green, novel, fast and cost effective procedure has been developed for determination of total sulphur in coal samples based on the use of diluted hydrogen peroxide and microwave irradiation. The resulting digests were then analysed by using inductively coupled plasma-optical emission spectroscopy (ICP-OES) and ion-chromatography (IC). The investigated factors include hydrogen peroxide concentration, microwave temperature, extraction time and coal amount. A certified reference material (SARM 20) was used for the optimization of the influential experimental parameter. It was observed that quantitative recoveries of total sulphur in coal could be attained when microwave temperature, extraction time, [H2O2], and coal amount are 150 °C, 5 min, 3 mol L−1 and 0.05 g, respectively. In addition, the 24 factorial design results demonstrated that the interactions between coal amount and time (BD) as well as coal amount and temperature (BC) were significant at a 95% confidence level. Under optimum conditions, the accuracy of the proposed microwave-assisted hydrogen peroxide digestion (MW-AHPD) method was examined by analysing three coal certified reference materials (SARM 18, 19 and 20) and quantitative recoveries (89–101%) were achieved. Additionally, the precision of the proposed method, expressed in terms of relative standard deviation (n = 15), was satisfactory (≤1.5%). The method detection limit (0.014 μg g−1) and method quantification limit (0.047 μg g−1) for sulphur analysis were relatively good compared to those reported in the literature. The proposed digestion method converted all the sulphur species into sulphates as confirmed by the IC results and it can be routinely applied to replace the standard Eschka method. The P-XRD results confirmed the undigested kaolinite and quartz minerals.
1. Introduction
Coal's total sulphur content fluctuates from 0.2% up to 11% depending on the location of the coal's origin and the maturity.1,2 The most dominating sulphur forms in coal are the inorganic species such as sulphate and sulphide. However, some of the sulphur is organic and it forms thiolic (R–H–S), sulphidic (R–S–R′), disulphidic (R–S–S–R), sulphoxide (R–S–O–R), and thiophenic (heterocyclic) structures within the coal.3–6 Analysis of total sulphur content in coal is very crucial as it determines the quality and the value of coal. Coal with high quantities of sulphur is known to be less expensive as it releases lots of sulphur dioxide to the atmosphere during its combustion and that causes severe environmental problems.7 The growth of coal utilization is limited by the liberation of sulphur emission from coal thermal processing plants. For example, during coal combustion processes, sulphur participate in several reactions and is emitted from the coal framework mainly as SO2 and SO3 gases, while, coal gasification processes liberate sulphur as H2S, CS2 and COS gases.8–10 The emission of S from anthropogenic sources has been related to the depletion of the Earth's ozone layer, acid rain incidence and chronic respiratory diseases.7,11 It has to be noted that, the sulphur existence in coal cannot be controlled or prevented as it occurs naturally during coal genesis.Recently, the world is moving towards the implementation of renewable energy technologies (biomass, solar, wind and geothermal). However, there is no evidence of commercially-viable alternatives that have developed to significantly replace fossil fuels such as coal.12,13 Hence, coal still stands as the most trustworthy and sustainable energy with cost effective electricity generation. Therefore, the development of more efficient, cost-effective, environmentally friendly, rapid and green methods for total sulphur determination in coal is essential. Knowing the amount of total sulphur in coal will assist in determination of coal value and future designation of possible coal clean-up technologies.
Eschka method (ASTM D 3177) is a well-known classic method for total sulphur determination in coal and coal related solid matrices. This method was first developed Adalbent Eschka and it converts (using furnace temperatures and MgO & Na2CO3 mixture) all the sulphur species to sulphate ions before gravimetric or ion chromatographic analysis.14 However, Eschka method is tedious and not precise when determining low sulphur concentration levels (≤5%). In recent years, chromatographic,15 mass spectrometric,16 molecular absorption17 and optical emission18–20 related techniques have been applied for the determination of sulphur and other elements in different solid matrices after numerous sample preparation procedures. The latter include hot plate acid digestion,21 acid block digestion,22 microwave acid digestion,19,20,23,24 ultrasonic bath acid digestion,26,27 dry-ashing15 and slurry formation.17 It should be noted that, hot plate, digestion block, ultrasonic bath and microwave make use of concentrated inorganic acids (HNO3, HCl, HF, H2SO4 & HClO4) which release carcinogenic gases. In addition, these acids also cause ICP-OES spectral interferences due to undigested carbon and high concentration of the acids.16–20 Dry-ashing15 on the other hand causes loss of volatile analyte and it is tedious, whereas slurry17 gives rise to poor reproducibility and high detection limits. However, there is a lot of research that has been conducted based on the use of ultrasonic bath and microwave assist extraction of multi-elements in solid matrices using partial dilute acids.23–28 The latter has been used in order to reduce acid waste, sample preparation cost, ICP-OES spectral interferences and emitted carcinogenic gasses. Recently, more cost-effective and green approaches have been implemented to achieve effective digestion methods for multi-element quantitative extraction in different solid matrices and quantitative recoveries were obtained.29–32 The present investigation explored feasibility of using diluted hydrogen peroxide and microwave radiation for quantitative extraction of total sulphur in coal samples. The resulted digests were subsequently analysed by using ICP-OES and IC techniques for total sulphur quantification. To the best of our knowledge, there are no studies reported in literature on MW-AHPD for quantitative extraction of total sulphur in coal samples. The proposed procedure is advantageous as it uses microwave radiation (to speed up extraction) and diluted hydrogen peroxide (to reduce sample preparation cost, ICP-OES spectral interferences and laboratory acid waste). The validation of the current method was successfully achieved by using three coal certified reference materials (SARM 18, 19 and 20).
2. Experimental procedure
2.1. Materials, samples, reagents and methods
All the chemical reagents used in the current study were of analytical grade and were utilized without any additional purification. Hydrogen peroxide solution (H2O2, 30%) of Suprapur grade was purchased from Sigma-Aldrich (St. Loius, MO, USA). Coal certified reference materials (SARM 18, 19 and 20) with particle size of ≤106 μm were obtained from Mintek (RSA). SARM 20 (sampled from SASOLBURG) with sulphur content of 0.51% (w/w) was utilised in the optimization of the proposed MW-AHPD method. Therefore, the other two CRMs (SARM 18, sampled from Witbank) and (SARM 19, sampled from Orange Free State) were used for method validation with sulphur contents of 0.56% (w/w) and 1.49% (w/w), respectively. The three coal samples with unknown sulphur concentration content were received from one of the South African coal mines and were further milled and sieved to match the similar particle size (≤106 μm) of the CRMs. In order to minimize the risk of cross-contamination, all vessel materials were socked in hydrogen peroxide solution (5 M) for a minimum of 24 hours, thoroughly rinsed with Milli-Q water and then dried in the oven (Scientific series 2000, Lasec RSA) before use. The same oven was used to eliminate moisture in unknown coal samples, the coal samples were heated at 50 °C until constant mass was obtained. Coal samples together with CRMs were stored in a desiccator to prevent hygroscope.The external calibration standards for both ICP-OES and IC analysis contained a known concentration of the target analyte (S and SO4−2, respectively). For ICP-OES analysis, calibration standards were prepared by diluting a certified stock elemental solution (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 031 mg L−1 total sulphur) purchased from Merck (Darmstadt, Germany) to get total sulphur concentration levels of 1, 3, 9, 12, 15, and 20 mg L−1 in 1% H2O2. The external calibration standards for IC analysis were made from anhydrous Na2SO4 (99.8%) which was purchased from Sigma-Aldrich (St. Loius, MO, USA). The concentration of the sulphate ions in the calibration standards included 5, 10, 20, 30, 50, and 70 mg L−1 in 1% H2O2. In both techniques (ICP-OES or IC), the blank calibration standards were hydrogen peroxide solution only (1% H2O2). Mobile phase for IC analysis was the combination of 3.5 mM Na2CO3 (99.8%) and 1 mM NaHCO3 (99.8%) solution and the mixture was filtered through nylon membrane filters (0.45 μm) before use. It should to be noted that both carbonate salts were purchase from Sigma-Aldrich (St. Loius, MO, USA). Ultra-pure water (18 MΩ cm) from a Milli-Q water purification system (Milli-Q SP ICP-MS, Millipore, USA) was utilized throughout the calibration standard preparation, sample preparation, and for final rinsing of the peroxide-socked vessel materials.
031 mg L−1 total sulphur) purchased from Merck (Darmstadt, Germany) to get total sulphur concentration levels of 1, 3, 9, 12, 15, and 20 mg L−1 in 1% H2O2. The external calibration standards for IC analysis were made from anhydrous Na2SO4 (99.8%) which was purchased from Sigma-Aldrich (St. Loius, MO, USA). The concentration of the sulphate ions in the calibration standards included 5, 10, 20, 30, 50, and 70 mg L−1 in 1% H2O2. In both techniques (ICP-OES or IC), the blank calibration standards were hydrogen peroxide solution only (1% H2O2). Mobile phase for IC analysis was the combination of 3.5 mM Na2CO3 (99.8%) and 1 mM NaHCO3 (99.8%) solution and the mixture was filtered through nylon membrane filters (0.45 μm) before use. It should to be noted that both carbonate salts were purchase from Sigma-Aldrich (St. Loius, MO, USA). Ultra-pure water (18 MΩ cm) from a Milli-Q water purification system (Milli-Q SP ICP-MS, Millipore, USA) was utilized throughout the calibration standard preparation, sample preparation, and for final rinsing of the peroxide-socked vessel materials.
2.2. Instrumentation
All the MW-AHPD processes were conducted by using Mars 6 One Touch Technology Microwave lab station (CEM Microwave Technology Ltd, North Caroline, USA). The microwave system was fitted with 24 × 50 mL PTFE vessels (any of them can be chosen as a reference) and each vessel assembly consisted of a vessel body, safety relief valve, cap and venting hole. The MARS CEM was equipped with an advanced reaction sensor for temperature (maximum: 200 °C) control. After each digestion, undigested white solid residues were separated from the clear colourless supernatant solution by using centrifuging machine (REMI motors, Model 400053, Mumbai, India). The total sulphur measurement was performed using Spectro ARCOS ICP-OES (Cleve, Germany) with radial plasma equipped with Cetac ASX-520 auto-sampler, cross-flow nebulizer and double pass Scott type spray chamber. The optimal operating conditions used for ICP-OES spectrometer were as follows: radio frequency generator power (1400 W), pump speed (30 rpm), plasma gas flow rate (12 L min−1), auxiliary flow rate (2 L min−1), nebulizer flow rate (0.95 L min−1) and analyte emission line (182.034 (nm)). Sulphate ions on the other hand were quantified by using DX 120 IC system (USA) fitted with pulsation damper, ASRS suppressor and a conductivity detector (DS 4). It is worth to mention that the conductivity meter had full-scale sensitivity settings in the range of 0–30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 μs cm−1. Sample introduction was performed by using a six-port injection valve equipped with 25 μL sample injection loop. Injection volume was 25 μL with a flow rate of 1.2 mL min−1. The retention time for sulphate ions was observed to be at 9.12 min. Powder X-ray diffraction (P-XRD) measurements of the white solid residue and raw coal (SARM 20) were conducted on a PANayltical X'Pert Pro powder diffractometer. The P-XRD instrument was equipped with X'Celerator detector and programmable divergence slit. The measurements were conducted in the 2θ range from 5 to 90° with Cu Kα radiation (λ = 0.15405 nm) at 40 kV and 40 mA working conditions. The raw P-XRD data was interpreted by using High Score Plus software.
000 μs cm−1. Sample introduction was performed by using a six-port injection valve equipped with 25 μL sample injection loop. Injection volume was 25 μL with a flow rate of 1.2 mL min−1. The retention time for sulphate ions was observed to be at 9.12 min. Powder X-ray diffraction (P-XRD) measurements of the white solid residue and raw coal (SARM 20) were conducted on a PANayltical X'Pert Pro powder diffractometer. The P-XRD instrument was equipped with X'Celerator detector and programmable divergence slit. The measurements were conducted in the 2θ range from 5 to 90° with Cu Kα radiation (λ = 0.15405 nm) at 40 kV and 40 mA working conditions. The raw P-XRD data was interpreted by using High Score Plus software.
2.3. Microwave-assisted hydrogen peroxide digestion (MW-AHPD) procedure
The experimental method for the proposed digestion procedure was adapted from the previous reported study.23 To describe the procedure briefly, appropriate coal amount (CRMs or samples) was weighed and transferred into 50 mL PTFE bomb vessel, followed by addition of the digestion reagent (different H2O2 concentration). It should be noted that blank samples contained 12 mL of hydrogen peroxide only. The PTFE bomb vessels were then sealed tightly and placed inside the microwave system. Microwave power was in W and it was directly proportional to the microwave temperature applied per run. Ramping (from room temperature to 150 °C) and cooling (from 150 to 50 °C) times for microwave temperature were 20 and 15 minutes, respectively. After the completion of the MW-AHPD process, the PTFE bomb vessels were taken out of the microwave system and were allowed to cool down to room temperature. Then, the digests inside the vessels were quantitatively transferred into 50 mL polyethylene centrifuge vials and the vessels were rinsed with 10 mL of 1 mol L−1 H2O2. After that, resulted digests were centrifuged at 3000 rpm for 15 min and the supernatant solutions were then decanted into cleaned 250 mL volumetric flasks. The white solid residues left in the 50 mL polyethylene centrifuge vials were then washed with 10 mL of 1 mol L−1 H2O2, then centrifuged (3000 rpm) for another 15 min. The resulted liquid phases were then transferred to the same 250 mL volumetric flasks and the flasks were filled up to the mark with 1 mol L−1 of H2O2. Afterwards, the solutions (samples) were passed through 0.45 μm filter, transferred into 15 mL ICP tubes and were ready for ICP-OES and IC analysis. It is worth to mention that, each sample was processed in four replicates (n = 4). The white solid residues were characterized by P-XRD to investigate mineralogical composition. It has to be noted that, the proposed method solves the problems associated with the environmental issues related to microwave acid digestion and matrix effects caused by inorganic acids. In most analytical sample preparation procedure, the main influence is the chemical reagent used and conditions applied. For instant, the nitric acid matrix was not compatible for analysis of the total sulphur by the ion-chromatograph in the previous reported study.23 While the use of hydrogen peroxide in the current study made it possible to validate the method using a well-known technique like ion chromatograph. Therefore, the idea of using diluted hydrogen peroxide instead of inorganic acids is unique and original.2.4. Evaluation of MW-AHPD efficiency
Digestion efficiency was examined by calculating quantitative recoveries of total sulphur in CRMs and four experimental parameters were evaluated. The investigated parameters involved hydrogen peroxide concentration (1–3 mol L−1), microwave temperature (100–150 °C), coal amount (0.025–0.5 g) and microwave hold digestion time (5–30 min). The final volume of the extracting solution was kept constant at 12 mL throughout the optimization process as explained in the previous report.23 The effect of changing a parameter from a low to a high level value was evaluated by monitoring the percentage recovery of total sulphur after each and every MW-AHPD experiment.3. Results and discussion
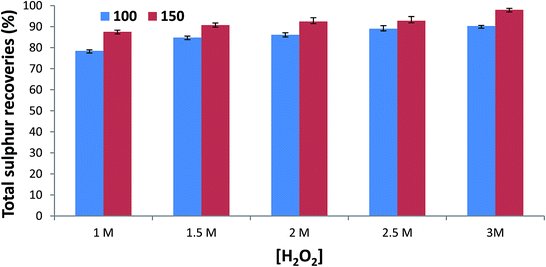
3.1. Influence of different temperatures and [H2O2] on MW-AHPD of coal
From the previous studies it was observed that temperature23,29 and concentration of the extracting/digestion reagent23,32 are the most significant parameters that affect microwave digestion/extraction processes. Therefore, the proposed MW-AHPD was optimized by first evaluating the effect of the concentration of extracting reagent (H2O2) and microwave temperature simultaneously. Only two temperature levels (100 and 150 °C) that were examined in this study, while five levels (1, 1.5, 2, 2.5 & 3 mol L−1) we evaluated for [H2O2]. The outcomes for the investigated factors are illustrated in Fig. 1. From this figure it can be observed that an increase in temperature (150 °C) improved the digestion of the coal, thereby increased the total sulphur quantitative extraction efficiency (89–96%) in all five [H2O2]. All the experiments carried out under low temperatures (100 °C) resulted in partial digestion of coal and that resulted in low total sulphur quantitative extraction efficiency (79–89%) in all the evaluated hydrogen peroxide concentrations (1–3 mol L−1). Additionally, an increase in hydrogen peroxide concentration improved the extraction efficiency from 80% up to 96%. Therefore, the maximum temperature (150 °C) and hydrogen peroxide concentration (3 mol L−1) were selected as the optimum conditions. This is because they resulted into extraction recoveries greater that 96%. These two optimum conditions were then applied for further optimization.3.2. Influence of different coal amount on the MW-AHPD of coal
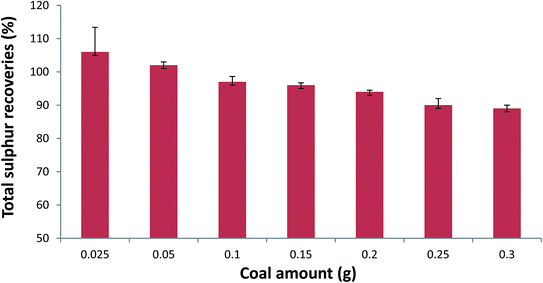
As mentioned earlier on, the crucial step in sample preparation is the extraction of the target analyte from the complexed matrix to a simple aqueous matrix that is compatible for the analytical technique proposed using an optimized procedure. Additionally, it is known that sample amount is one of the significant factors that play a role in microwave-assisted sample preparation methods.23,29–32 Hence, the influence of sample amount on MW-AHPD for coal digestion prior to quantitative extraction of total sulphur was investigated. Different coal amounts (0.025, 0.05, 0.1, 0.15, 0.2, 0.25 and 0.3 g) were investigated as shown in Fig. 2. In all the conducted experiments, the colourless clear digest were analysed for total sulphur content using optimized conditions of the ICP-OES. The results show that coal amount greater than 0.05 g (0.1, 0.15, 0.2, 0.25 and 0.3 g) resulted in poor extraction efficiency (percentage recoveries <95%). Alternatively, samples less than and equal to 0.05 g (0.025 and 0.05 g) showed quantitative recoveries (>95%). It must be noted that 0.025 g resulted in high percentage recovery (106%) but with very poor precision (% RSD = 7), hence it was omitted from the examination. Sample weight of 0.05 g was then selected as the optimum level as it showed high quantitative extraction efficiency (102%) of total sulphur with excellent precision (% RSD ≤ 1). The optimum condition of the sample amount (0.05 g) was then used in all the subsequent experiments.3.3. Influence of different digestion hold time on the MW-AHPD of coal
One of the advantages of microwave-assisted closed vessel sample preparation methods is time reduction. Consequently, the impact of irradiation time (5, 10, 15, 20, 25 and 30 min) was examined for digestion of coal. It was observed that, quantitative extraction efficiency of total sulphur from coal is not much affected by irradiation time as constant quantitative recoveries were obtained (99–100%). Since constant recoveries were achieved, there was no need to show a graph or table. However, the results obtained in the current study correspond well with the outcomes of the previous report.23 Additionally, the results showed that hold time of 5 minutes is effective for coal digestion and quantitatively extraction of the total sulphur as higher extraction efficiency (99%) were obtained with good precision (% RSD = 0.6). Based on these results, a microwave irradiation time of 5 minutes was found to be sufficient for the quantitative extraction of total sulphur. Such short digestion time add an advantage on the proposed MW-AHPD method as it improves the sample throughput and hence selected as the ideal level for further investigations. Once the examination of all the MW-AHPD parameters was completed, the optimal conditions were utilized in all subsequent experiments in order to complete validation (accuracy, precision, MDL and MQL) of the proposed method. Therefore, all the four ideal conditions were applied in three coal CRMs (SARM 18, 19 and 20) for validation purposes.3.4. Study of parameter interactions during MW-AHPD of coal
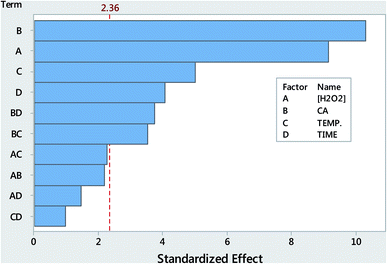
The interactions between the main effects (variables) such as hydrogen peroxide concentration (A), coal amount (B), temperature (C) and time (D) were evaluated using 24 factorial designs. The estimated main effects and their interactions can be seen in the Pareto charts presented in Fig. 3. The bars that exceed a vertical reference line (95% confidence interval) are significant values with respect to the response. According to the Pareto chart (Fig. 3), all factors were statistically significant at the 95% confidence level. However, B and A were significantly higher than the other effects. In addition, two interactions, BD and BC were also statistically significant. The results indicated that these variables have a synergistic effect on percentage recovery. This means that, the use of CA at higher levels and time as well as temperature at the lower levels and their combination also at same levels may result to a better analytical response. In addition, the influence of the other three interactions (AC, AB, AD & CD) was not statistically significant. The influence of the investigated main effects (A, B, C & D) was similar to the outcomes of the univariate optimization approach discussed above (Section 3.1–3.3).3.5. Analytical response characteristics of the MW-AHPD method
When developing an analytical method, it is very crucial to determine analytical merits such as sensitivity, precision, reproducibility, linearity, correlation coefficient, sample throughput, MDL and MQL. This is conducted in order to assess the excellency of the proposed method compared to the literature reported studies and also to ensure that the analytical technique is performing at its best. In the current study, optimized experimental conditions were utilized to evaluate the analytical features of the proposed method.Under optimum conditions, calibration function with five points was constructed for total sulphur extraction ranging from 0 (i.e., analytical method blank) to 25 mg L−1 in 50 mL. These five solutions were obtained by subjecting five different SARM 20 amounts (0.025, 0.05, 0.1, 0.15 and 0.2 g) into the optimized MW-AHPD method and the final digests were analysed by ICP-OES with aqueous external calibration standards. The concentration levels selected for sulphur showed good linearity (0.047–5100 μg g−1) with satisfactory correlation coefficient (R2 > 0.9995) and sensitivity (5479.3 cps L mg −1) as shown in Table 1. These analytical features clearly indicated that the absence of acid and the complete digestion of the coal (less carbon content residue) eliminated the matrix interferences which are due to complete solubility of the total sulphur. This phenomenon enabled the use of aqueous external calibration standard solutions to calibrate the ICP-OES system for total sulphur quantification.
| Analytical features | Specifications |
|---|---|
| Sensitivity (cps μg L−1) | 5479.3 |
| Correlation coefficient (R2) | 0.9995 |
| MDL (μg g−1) | 0.014 |
| MQL (μg g−1) | 0.047 |
| Linearity | 0.047–5100 |
| Precision (% RSD) | 1.5 |
| Reproducibility (% RSD) | 4.5 |
| Sample throughput (sample h−1) | 24 |
The method detection limit (MDL) and method quantification limit (MQL) were calculated using the IUPAC recommendation. The MDLs are defined as the lowest concentration of the analyte giving signal equal to the three times the standard deviation (SD) of the blank signals multiplied by dilution factor. Furthermore, MQLs are expressed as the lowest concentration of the analyte giving signal equal to ten times the SD of the blank signal multiplied by dilution factor. The MDL and MQL obtained for the proposed MW-AHPD method were 0.014 and 0.047 μg g−1, respectively and are presented in Table 1.
Jenner et al.16 stated that, the agreement of a data set is ‘excellent’ when % RSD is ≤3 and is ‘satisfactory’ when % RSD is between 3 and 7. When assessing the precision (repeatability) of the proposed digestion method, % RSD of 15 replicates (n = 15) was calculated and it was excellent (1.5%). That means 15 SARM 20 sample amounts of 0.05 g were subjected into MW-AHPD procedure simultaneously prior to ICP-OES analysis of the final digests samples to attain total sulphur extraction efficiency in 50 mL. Conversely, reproducibility of the proposed method was observed to be 3.9% (n = 6) and it was calculated from 6 replicates (n = 6). This implies that, six MW-AHPD procedures were conducted separately in six different days prior to ICP-OES quantification of total sulphur. It is worth to mention that, total time required for the complete digestion of the coal samples using the proposed digestion method is 40 min (20 min of microwave ramping temperature, 5 min of digestion and 15 min of digests cooling) and 24 samples can be digested simultaneously. Therefore, the sample throughput can be determined to be roughly 24 samples h−1. All the discussed analytical figures are presented in Table 1.
3.6. Validation of the MW-AHPD method
After the optimization step, the optimum conditions of the proposed digestion procedure were utilized for validation purposes. Accuracy (validation) of the proposed digestion method was performed in order to ensure that in future routine analysis, the concentration of the target analyte is close enough to the true value of the unknown sample.33 To examine validation of the proposed MW-AHPD procedure, three coal CRMs (SARM 18, 19 & 20) were subjected to the optimum conditions and the resulted digests were analysed by ICP-OES (total sulphur quantification) and IC (total sulphate quantification). No significant differences were obtained between mean (four replicates) values of total sulphur found and its certified values in the tested CRMs. Quantitative recoveries (89–102%) obtained for both ICP-OES and IC analysis (Table 2) showed satisfactory accuracy of the proposed method. The agreement between ICP-OES and IC measurements demonstrates that under the optimum conditions, all the sulphur species in coal were converted into sulphate ions (Table 2). Therefore, the proposed digestion procedure followed by IC and ICP-OES analysis is an outstanding substitute for tedious Eschka method14 routinely applied for total sulphur determination in coal samples.| CRMs | Certified value (μg g−1) | Found value (μg g−1) | Recoveries (%) | ||
|---|---|---|---|---|---|
| ICP-OES | IC | ICP-OES | IC | ||
| a Figures in parentheses are percentage relative standard deviation values (% RSD). | |||||
| SARM 18 | 5600 (16) | 5416.42 (0.8) | 5417.33 (2.1) | 97 | 97 |
| SARM 19 | 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900 (9) 900 (9) |
13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 226.50 (04) 226.50 (04) |
13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 447.50 (3.5) 447.50 (3.5) |
89 | 90 |
| SARM 20 | 5100 (10) | 5036.04 (0.7) | 5127.73 (4.1) | 99 | 101 |
3.7. Comparison of MW-AHPD method with other reported method
To investigate the effectiveness of the proposed procedure, MDL and precision of the present study were compared to those of the literature reported methods as shown in Table 3. From this table it can be observed that the proposed method showed relatively low MDL and high precision as compared to the other methods reported in literature.17,23,34–38 The use of dilute hydrogen peroxide resulted in low blank values for the proposed MW-AHPD method, thereby; improved MDL and relatively standard deviation obtained. Similar observations were reported by Bizzi and co-workers.28| Ref. | Matrix | Method | Reagents | Mass (mg) | Detection | MDL | Precision |
|---|---|---|---|---|---|---|---|
| a Microwave-assisted acid digestion.b Ultrasound-assisted acid digestion.c Microwave-assisted acid extraction.d Not determined. | |||||||
| 17 | Coal | Slurry | Triton X-100/HNO3 | 0.02 | HR-CS-ET-MAS | 100 | 4–8 |
| 23 | Coal | MW-AAEc | HNO3/H2O2 | 50 | ICP-OES | 0.03 | 3 |
| 34 | Coal | Slurry | Water | 0.8 | GF-MAS | 200 | 5–12 |
| 35 | Coal | Solid | N/A | 150 | HR-CS-MAS | 0.08 μg | 10 |
| 36 | Coal | MW-AADa | HNO3 | 200 | HR-CS-FAAS | 100 | N.Dd |
| 37 | Tailings | US-AADb | HNO3/HCl | 100 | ICP-OES | N.Dd | 3–6 |
| 38 | Peat | MW-AAD | HNO3 | 250 | ICP-OES | 5 | 17 |
| This work | Coal | MW-AHPD | H2O2 | 50 | ICP-OES | 0.01 | 1.5 |
3.8. Application of the developed MW-AHPD method in real coal samples
The optimized and validated MW-AHPD method was applied for quantitative extraction of total sulphur in three coal samples obtained in one of the South African coal mines. The coal samples were abbreviated as coal sample A, B and C (CSA, CSB and CSC respectively). The resulted digests were then analysed by both ICP-OES (for total sulphur quantification) and IC (for total sulphate quantification). The results for this investigation are presented in Table 4 and they show a good agreement between ICP-OES and IC measurements. From the IC results it can be concluded that the proposed MW-AHPD method was effective enough to convert all the sulphur species in coal to sulphate ions. Therefore the proposed procedure can be a good substitute for Eschka method routinely applied for sulphur determination in coal related samples. The total sulphur concentration obtained in three coal samples resembles the results that were reported in the previous study.23 The ascending total sulphur trend among the three coal samples was as follows: CSA > CSC > CSB. Therefore coal samples A had the highest total sulphur concentration and coal sample B was the least sample.| Samples | MW-AHPD | |
|---|---|---|
| ICP-OES | IC | |
| CSA | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 916.80 (1.4) 916.80 (1.4) |
13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 032.11 (4.0) 032.11 (4.0) |
| CSB | 3389.61 (0.6) | 3400.76 (3.5) |
| CSC | 7001.99 (0.1) | 7354.76 (5) |
3.9. P-XRD analysis of white solid residue and raw coal (SARM 20)
Powder X-ray diffraction was performed in order to investigate the mineral changes that occurred after MW-AHPD method and the results are illustrated in Fig. 4. The diffraction patterns of the white soli residue resulted from MW-AHPD and raw coal were quite similar except for the peak around 29.8 2θ. This peak is attributed to the presents of calcium in raw coal sample. However, the calcium peak was not observed for the microwave-assisted hydrogen peroxide digested coal, meaning that the proposed method removed the calcium atoms. The most dominating minerals in both samples were observed to be kaolinite [Al2Si2O5(OH)4] followed by quartz (SiO2). Similar observations were also reported by Cheng et al.39 and Zhao et al.40 From the two diffraction profiles, it can be perceived that, the intensity of all the minerals increased when coal was treated with MW-AHPD process. That is due to the white solid residue formed after MW-AHPD method being more crystalline as compared to raw coal. The latter is amorphous due to the carbon atoms present. Additionally, the coal before MW-AHPD process was black (which showed presents of carbon atoms), after the digestion procedure, white solid residues (which shows complete digestion of carbon) were obtained with clear colourless digests. These observations show that carbon content of the coal was completely digested. Kaolinite is known to be a clay mineral and the common name for quartz is silica, therefore, the white solid residue generated from the MW-AHPD procedure composed of silica and clay mixture. It is known that, kaolinite and quartz minerals can only be digested effectively by the use of hydrofluoric acid (HF). The aim of the developed method was to digest coal for quantitative extraction of total sulphur followed by ICP-OES and IC determination. The ICP-OES and IC results showed good agreement with the certified values of the CRMs; therefore, there was no need to use toxic HF in the proposed digestion procedure.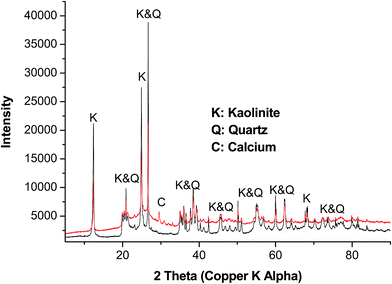 | ||
| Fig. 4 Powder X-ray diffraction patterns of raw coal (in red) and microwave-assisted hydrogen peroxide digested coal (in black). | ||
4. Conclusion
The current study describes the development of environmentally friendly, fast, novel, sensitive and precise MW-AHPD method for quantitative extraction of total sulphur in coal samples prior to ICP-OES and IC determination. The proposed procedure was successfully optimized and validated using three coal CRMs. The developed method showed good precisions (≤1.5%), accuracy (89–101%) and low MDL (0.014 μg g−1) as compared to the literature reported methods. Hydrogen peroxide converted all the sulphur species into sulphate ions as shown by the IC results. The P-XRD results showed that, the solid white residue observed after the MW-AHPD procedure are the undigested kaolinite and quartz minerals. Therefore, the developed MW-AHPD method is an alternative method for the tedious Eschka method that is routinely applied for total sulphur determination in coal related samples.Acknowledgements
The authors are grateful to NRF (scarce skills) and Eskom (TESP programme) for their financial support. University of Johannesburg, Analytical Facility (Spectrau) and chemistry department are acknowledged for providing ICP-OES, IC, XRD and other laboratory facilities.References
- G. Gryglewicz and S. Jasieko, Fuel, 1992, 71, 1225 CrossRef CAS.
- J. V. Ibarra, A. J. Bonet and R. Moliner, Fuel, 1994, 73, 933 CrossRef CAS.
- D. Zhang and S. Yani, Proc. Combust. Inst., 2011, 33, 1747 CrossRef CAS PubMed.
- E. de Visser, C. Hendriks, M. Barrio, M. J. Mlnvik, G. de Koeijer, S. Liljemark and Y. L. Gallo, Int. J. Greenhouse Gas Control, 2008, 2, 478 CrossRef CAS PubMed.
- T. Xu, J. A. Apps, K. Pruess and H. Yamamoto, Chem. Geol., 2007, 242, 319 CrossRef CAS PubMed.
- J. S. Mesroghli, E. Yperman, R. Jorjani and C. M. Noaparast, Fuel Process. Technol., 2015, 131, 193 CrossRef PubMed.
- G. S. Downward, W. Hu, D. Large, H. Veld, J. Xu, B. Reiss, G. Wuf, F. Wei, R. S. Chapmang, N. Rothmanb, L. Qingb and R. Vermeulena, Environ. Int., 2014, 68, 94 CrossRef CAS PubMed.
- B. Özum, G. Kovacik and A. Chambers, Int. J. Hydrogen Energy, 1993, 18, 847 CrossRef.
- A. Ocampo, E. Arenas, F. Chejne, J. Espinel, C. Londõno, J. Aguirre and J. D. Perez, Fuel, 2003, 82, 161 CrossRef CAS.
- L. Chen and S. Bhattacharya, Environ. Sci. Technol., 2013, 47, 1729 CAS.
- M. Masiol and R. M. Harrison, Atmos. Environ., 2014, 95, 409 CrossRef CAS PubMed.
- M. Bläsing and M. Müller, Energy Fuels, 2012, 26, 6311 CrossRef.
- R. Stanger and T. Wall, Prog. Energy Combust. Sci., 2011, 37, 69 CrossRef CAS PubMed.
- International Standard, ISO 334, Solid Mineral Fuels, Determination of Total Sulphur, Eschka Method, 1992, 1.
- W. Geng, T. Nakajima, H. Takanashi and A. Ohki, Fuel, 2008, 87, 559 CrossRef CAS PubMed.
- G. A. Jenner, H. P. Longerich, S. E. Jackson and B. J. Fryer, Chem. Geol., 1990, 83, 133 CrossRef CAS.
- F. V. Nakadi, L. R. Rosa and M. A. M. S. da Veiga, Spectrochim. Acta, Part B, 2013, 88, 80 CrossRef CAS PubMed.
- N. J. Wagner and B. Hlatshwayo, Coal Geol. Bull., 2005, 63, 228 CrossRef CAS PubMed.
- J. Wang, T. Nakazato, K. Sakanishi, O. Yamada, H. Tao and I. Saito, Anal. Chim. Acta, 2004, 514, 115 CrossRef CAS PubMed.
- J. Wang, T. Nakazato, K. Sakanishi, O. Yamada, H. Tao and I. Saito, Talanta, 2006, 68, 1584 CrossRef CAS PubMed.
- K. M. Dimpe, J. C. Ngila, N. Mabuba and P. N. Nomngongo, J. Phys., Chem. Earth Sci., 2015 DOI:10.1016/j.pce.2014.11.006.
- R. Fernández-Martínez, I. Rucandio, I. Gómez-Pinilla, F. Borlaf, F. García and M. T. Larrea, J. Food Compos. Anal., 2015, 38, 7 CrossRef PubMed.
- N. Mketo, P. N. Nomngongo and J. C. Ngila, Anal. Methods, 2014, 6, 8505 RSC.
- C. Y. Zhou, M. K. Wong and L. L. Koh, J. Anal. At. Spectrom., 1996, 11, 585 RSC.
- N. Manutsewee, W. Aeungmaitrepirom, P. Varanusupakul and A. Imyim, Food Chem., 2007, 101, 817 CrossRef CAS PubMed.
- B. L. Batista, J. L. Rodrigues, V. C. Souza and F. Barbosa, Forensic Sci. Int., 2009, 192, 88 CrossRef PubMed.
- P. Bermejo-Barrera, O. Muñiz-Naveiro, A. Moreda-Piñeiro and A. Bermejo-Barrera, Forensic Sci. Int., 2000, 107, 105 CrossRef CAS.
- C. A. Bizzi, E. L. M. Flores, J. A. Nóbrega, J. S. S. Oliveira, L. Schmidta and S. R. Mortarid, J. Anal. At. Spectrom., 2014, 29, 332 RSC.
- E. Habibi, K. Ghanemia, M. Fallah-Mehrjardi and A. Dadolahi-Sohrab, Anal. Chim. Acta, 2013, 762, 61 CrossRef CAS PubMed.
- H. Matusiewicz and M. Ślachciński, Microchem. J., 2014, 115, 6 CrossRef CAS PubMed.
- M. V. Balarama Krishna, S. V. Rao, Y. Balaji Rao, N. S. Shenoy and D. Karunasagar, Anal. Methods, 2014, 6, 261 RSC.
- K. Ghanemi, M.-A. Navidi, M. Fallah-Mehrjardi and A. Dadolahi-Sohra, Anal. Methods, 2014, 6, 1774 RSC.
- M. Rambla-Alegre, J. Esteve-Romero and S. Carda-Broch, J. Chromatogr. A, 2012, 1232, 101 CrossRef CAS PubMed.
- G. Jima, D. Katskova and P. Tittarelli, Talanta, 2011, 83, 1687 CrossRef PubMed.
- R. Mior, S. Morés, B. Welz, E. Carasek and J. B. de Andrade, Talanta, 2013, 106, 368 CrossRef CAS PubMed.
- A. Baysal and S. Akman, Talanta, 2011, 85, 2662 CrossRef CAS PubMed.
- A. H. Khan, J. Q. Shang and R. Alam, J. Hazard. Mater., 2012, 235–236, 376 CrossRef CAS PubMed.
- C. Yafa and J. G. Farmer, Anal. Chim. Acta, 2006, 557, 296 CrossRef CAS PubMed.
- J. Cheng, J. Zhou, J. Liu, X. Cao and K. Cen, Energy Fuels, 2009, 23, 2506 CrossRef CAS.
- H. Zhao, L. Wang, D. Jia, W. Xia, J. Li and Z. Guo, J. Mater. Chem. A, 2014, 2, 9338 CAS.
| This journal is © The Royal Society of Chemistry 2015 |