Catalyst free, multicomponent-tandem synthesis of spirooxindole-indazolones and spirooxindole-pyrazolines: a glycerol mediated green approach†
Swastika Singha,
Mohammad Saquiba,
Shyam Babu Singhb,
Mandavi Singha and
Jagdamba Singh*a
aEnvironmentally Benign Synthesis Lab, Department of Chemistry, University of Allahabad, Allahabad-211002, India. E-mail: dr.jdsau@gmail.com; Tel: +91 9415218507
bDepartment of Chemistry, Government College, Ramchandrapur, Chhattisgarh-497220, India
First published on 8th May 2015
Abstract
The development of a versatile new one pot, catalyst free, multicomponent-tandem strategy for assembly of spirooxindole-indazolones and spirooxindole-pyrazolines is described. The reported protocol includes several advantages like environmental friendliness, cost effectiveness, high atom economy, short reaction times and high yields. The reported method is the first green synthesis of spirooxindole-indazolones and spirooxindole-pyrazolines.
Introduction
Multicomponent reactions (MCRs) have emerged as a powerful new strategy in synthetic organic chemistry and drug discovery. MCRs are a precise synthetic tool to access products with structural complexity and diversity in a single operation with improved atom and time economy, simplicity, synthetic efficiency and a lower number of reactions and purification steps.1Spirooxindole is an important heterocyclic moiety, present as a key structural motif in a variety of alkaloids and biologically active molecules.2,3
Though a large number of methods for the synthesis of spirooxindoles have been reported till date, yet the development of more efficient routes to access existing as well as novel spirooxindole derivatives remains an important goal for organic chemists.2a,4,5
Indazolones are biologically important class of molecules.6 Thus the construction of hybrid molecules containing both the spirooxindole and indazolone motifs integrated unto one platform is highly desirable from medicinal point of view. Similarly in the last few years, the synthesis of spirooxindoles-pyrazolines has received increasing attention, due to the presence of the oxindole and pyrazoline skeletons in several bioactive molecules.7 Recently, spirooxindoles-pyrazoline derivatives have been reported as exhibiting very good anti-cancer activity (Fig. 1).8
However to the best of our knowledge, till date not a single method for the synthesis of spirooxindole-indazolones has been reported while only a few methods for the synthesis of spirooxindoles-pyrazolines are known. Therefore, the development of new routes for construction of spirooxindole-indazolones remains a synthetic challenge. Likewise newer methods for the synthesis of spirooxindoles-pyrazolines are also urgently needed.
In the last few decades the use of environment friendly green techniques in organic syntheses has seen a rapid growth due to the growing awareness for the need to reduce harm to the environment as well as to reduce economic costs.9
A major thrust area in this regard has been to replace conventional harmful petroleum based organic solvents by safe, cheap and environmentally benign solvents obtained from bio-renewable feedstocks.10 Consequently biomass-derived reaction media such as lactic acid, γ-valerolactone, 2-methyl-THF and glycerol etc. are fast emerging as promising substitutes to conventional solvents.11
Among them, glycerol, a sustainable biodegradable compound obtained as an organic waste in the fast developing biodiesel industry and new processes for the conversion of cellulose and lignocelluloses into value-added chemicals, is attracting special attention. It combines the benefits of water (low toxicity, easy availability, cheap price, renewability) and ionic liquids (high boiling point, relatively low vapor pressure, reusability, ability to dissolve a wide range of organic and inorganic compounds).12 Another area that has attracted much attention in green chemistry is the development of catalyst free reactions.13
In the background of the above discussions we herein report a simple and efficient multicomponent-tandem one pot catalyst free green approach to spirooxindole-indazolones and spirooxindole-pyrazolines from easily available starting materials in glycerol–water solvent system. The present report is part of our ongoing research program on the development of novel green synthetic routes to important heterocyclic molecules14 and to the best of our knowledge is the first synthesis of spirooxindole-indazolones and first green synthesis of spirooxindole-pyrazolines.
Results and discussion
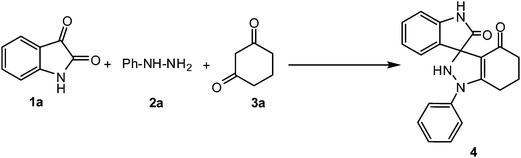
To initiate this work we conducted a model reaction wherein isatin (1a, 1 mmol) and phenyl hydrazine (2a, 1 mmol) were reacted at room temperature (RT) in water. However no hydrazone formation was observed even after 12 h of stirring. We conducted the same reaction at reflux. Hydrazone formation was observed in this case after 5 h of stirring. However formation of only a trace amount of a new product was observed after addition of dimedone (3a, 1 mmol). The experiment was now put up in the presence of CTAB at RT but it was unsuccessful. However, when the reaction was repeated at reflux, encouraging result was obtained. Immediately after the addition of isatin (1a) and phenyl hydrazine (2a) a new spot just above the spot of isatin began to form on the TLC, which we assumed to be of the expected hydrazone. In about 2 h the isatin spot disappeared completely. At this juncture we added dimedone and allowed the reaction to stir at reflux till the complete disappearance of the hydrazone spot (TLC). The product was isolated and identified as spirooxindole-indazolone 4. Yield of the reaction was found to be 41%. The experiments were repeated using SDS, with almost similar results. The same reaction was now attempted using β-CD in water under reflux leading to the formation of the desired product in 64% yield. Now in order to improve the yield of the product we tried ethanol as a solvent instead of water. When the reaction was conducted under reflux the reaction proceeded smoothly leading to the formation of the desired product in 5 h in much better yield (80%). The reaction was also conducted at a lower temperature (75 °C) but with the same outcome. Use of glycerol in place of ethanol led to an almost similar result. However when the reaction was conducted in a mixture of glycerol–water (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) at 100 °C, a dramatic enhancement in yield (91%) and reduction of the reaction time (3 h) was observed. When the experiment was conducted at a lower temperature (75 °C) no change was observed in yield and reaction time. However when the reaction temperature was lowered further (50 °C) there was a marked reduction in yield and increase in reaction time. Conducting the same experiment at RT led to further reduction in yield and increase in reaction time. Very interestingly increasing the percentage of water in glycerol–water solvent system (1
1) at 100 °C, a dramatic enhancement in yield (91%) and reduction of the reaction time (3 h) was observed. When the experiment was conducted at a lower temperature (75 °C) no change was observed in yield and reaction time. However when the reaction temperature was lowered further (50 °C) there was a marked reduction in yield and increase in reaction time. Conducting the same experiment at RT led to further reduction in yield and increase in reaction time. Very interestingly increasing the percentage of water in glycerol–water solvent system (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) led to a significant reduction in yield. The use of PEG-400 as solvent was also experimented with but good results were not obtained.
1) led to a significant reduction in yield. The use of PEG-400 as solvent was also experimented with but good results were not obtained.
From the above sets of experiment it was evident that the best condition for conducting the above reaction was at 75 °C using glycerol–water mixture (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as a reaction medium under catalyst free conditions leading to the formation of the desired spirooxindole-indazolone 4 in 91% yield in 3 h (Table 1, entry 12).
1) as a reaction medium under catalyst free conditions leading to the formation of the desired spirooxindole-indazolone 4 in 91% yield in 3 h (Table 1, entry 12).
| Entry | Solvent | Additive | Temperature | Time | Yield of 4b % |
|---|---|---|---|---|---|
| a All reactions were carried out with 1a (1 mmol), 2a (1 mmol), 3a (1 mmol) in 5 mL solvent under air.b Isolated yields. | |||||
| 1 | Water | None | RT | 12 h | No reaction |
| 2 | Water | None | Reflux | 12 h | Trace amount |
| 3 | Water | CTAB | RT | 10 h | Trace amount |
| 3 | Water | CTAB | Reflux | 8 h | 41 |
| 4 | Water | SDS | RT | 11 h | Trace amount |
| 5 | Water | SDS | Reflux | 8.5 h | 36 |
| 6 | Water | β-CD | Reflux | 7 h | 64 |
| 7 | Ethanol | None | Reflux | 5 h | 80 |
| 8 | Ethanol | None | 75 °C | 5 h | 80 |
| 9 | Glycerol | None | 100 °C | 6 h | 78 |
| 10 | Glycerol | None | 75 °C | 6 h | 78 |
| 11 | Glycerol | None | RT | 11 h | 48 |
| 12 | Glycerol–water/4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
None | 100 °C | 3 h | 91 |
| 13 | Glycerol–water/4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
None | 75 °C | 3 h | 91 |
| 14 | Glycerol–water/4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
None | 50 °C | 5 h | 80 |
| 15 | Glycerol–water/4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
None | RT | 8 h | 69 |
| 16 | Glycerol–water/1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
None | 75 °C | 5 h | 75 |
| 17 | Glycerol–water/1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
None | 100 °C | 5 h | 75 |
| 18 | PEG-400 | None | 75 °C | 7 h | 40 |
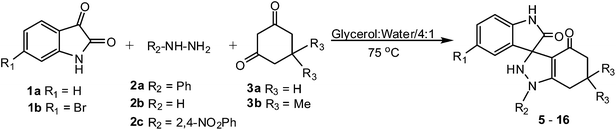
Once ideal conditions for conducting this reaction have been identified, the scope and efficiency of the developed synthetic protocol was explored under the optimized reaction conditions with isatins, hydrazines and dimedones having different substituents, to furnish the corresponding spirooxindole-indazolones. In all the cases the desired product were obtained in high yields and short reaction times (Table 2).
It was observed that use of simple hydrazine gave the best result. In case of phenyl hydrazine there was a small decrease in yield and slight increase in reaction time. However, when 2,4-dinitrophenyl hydrazine was used there was a noticeable decrease in yield and increase in reaction time due to the presence of the strongly electron-withdrawing nitro group on the phenyl ring. However the substituents on dimedone and isatin had negligible effect on the yield or time of the reaction.
The plausible mechanism for synthesis of spirooxindole-indazolones is shown below in Fig. 2. The reaction proceeds through the glycerol assisted formation of the anion I which then attacks the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N bond of the hydrazone, followed by the attack of the inner nitrogen lone pair of the hydrazone on the dimedone carbonyl resulting in cyclization, leading to the formation of the indazolones or pyrazolines as the case may be.
N bond of the hydrazone, followed by the attack of the inner nitrogen lone pair of the hydrazone on the dimedone carbonyl resulting in cyclization, leading to the formation of the indazolones or pyrazolines as the case may be.
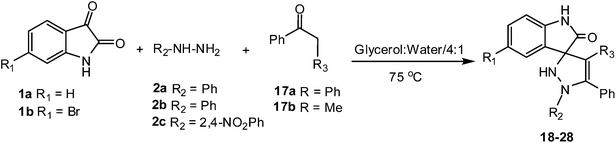
During the course of carrying out the above reaction we figured out that if an open chain carbonyl compound having an active methylene group like 2-phenyl acetophenone or propiophenone is used in place of dimedone we could obtain highly functionalized spirooxindole-pyrazolines as the product instead of spirooxindole-indazolones. So once the methodology for the synthesis of spirooxindole-indazolones was established, we focused our attention on exploiting the synthetic protocol for the construction of the targeted spirooxindole-pyrazolines.
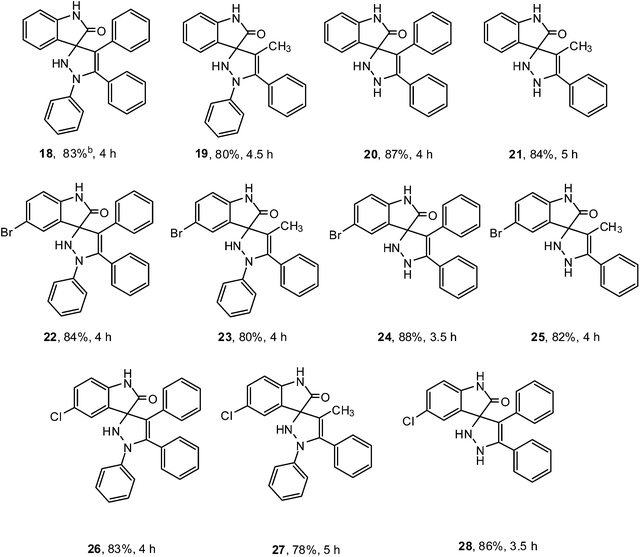
Consequently when isatin derivatives 1 were reacted with substituted hydrazines 2 and carbonyl compounds 17 under optimized reaction conditions, spirooxindole-pyrazolines 18–28 were obtained in good to very good yields (Table 3).
Conclusion
In summary, we have developed a facile and versatile, one pot, multi-component-tandem green methodology to access spirooxindole-indazolones and highly functionalized spirooxindole-pyrazolines. To the best of our knowledge this is the first green synthesis of spirooxindole-indazolones and spirooxindole-pyrazolines. The use of a green solvent system, catalyst free mild reaction conditions, high yields, short reaction times, high atom economy and easy workup procedure are the key features of this method. The tricyclic spirooxindole-indazolones and spirooxindole-pyrazoline frameworks constructed in this study could serve as important scaffolds for drug discovery. The biological screening of the title compounds, are presently underway and would be reported later on in a separate paper.Experimental
General remarks
All chemicals used were purchased from commercial suppliers such as Aldrich, Alfa Aesar, Merck, Spectrochem, Qualigens etc. and were used without purification. The reactions were monitored using pre-coated TLC plates of silica gel G/UV-254 of 0.25 mm thickness (Merck 60 F-254). Column chromatography was performed using silica gel (60–120) and (100–200). NMR spectra were recorded on a Bruker Avance-II 400FT spectrometer at 400 MHz (1H) and 100 MHz (13C) in DMSO using TMS as an internal reference. Mass Spectra (ESIMS) were obtained on Micromass quadro II spectrometer. Elemental analyses were carried out in a Thermo Scientific (FLASH 2000) CHN Elemental Analyser.General procedure for the synthesis of spirooxindole-indazolones (4–16)
To a stirred solution of glycerol–water/4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 solvent system (5 mL) at 75 °C, isatin (1.0 mmol) and hydrazine (1.0 mmol) were added and the mixture was stirred for 15–30 min. To this, 1,3-diketone (1.0 mmol) was added and the resulting reaction mixture was stirred till completion of the reaction (TLC). The reaction mixture was quenched with water and extracted with ethyl acetate (4 × 5 mL). The combined organic layers were washed with brine solution, dried over anhydrous Na2SO4 and evaporated under reduced pressure. The crude product was purified by column chromatography using hexane/ethyl acetate to obtain the pure spirooxindole-indazolone derivatives.
1 solvent system (5 mL) at 75 °C, isatin (1.0 mmol) and hydrazine (1.0 mmol) were added and the mixture was stirred for 15–30 min. To this, 1,3-diketone (1.0 mmol) was added and the resulting reaction mixture was stirred till completion of the reaction (TLC). The reaction mixture was quenched with water and extracted with ethyl acetate (4 × 5 mL). The combined organic layers were washed with brine solution, dried over anhydrous Na2SO4 and evaporated under reduced pressure. The crude product was purified by column chromatography using hexane/ethyl acetate to obtain the pure spirooxindole-indazolone derivatives.
General procedure for the synthesis of spirooxindole-pyrazolines (18–28)
To a stirred solution of solvent system glycerol–water/4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 solvent system (5 mL) at 75 °C, isatin (1.0 mmol) and hydrazine were added and the mixture was stirred for 15–30 min. To this, 1,2-diphenylethanone (1.0 mmol) was added and the resulting reaction mixture was stirred till completion of the reaction (TLC). The reaction mixture was quenched with water and extracted with ethyl acetate (4 × 5 mL). The combined organic layers were washed with brine solution, dried over anhydrous Na2SO4 and evaporated under reduced pressure. The crude product was purified by column chromatography using hexane/ethyl acetate to obtain the pure spirooxindole-pyrazoline derivatives.
1 solvent system (5 mL) at 75 °C, isatin (1.0 mmol) and hydrazine were added and the mixture was stirred for 15–30 min. To this, 1,2-diphenylethanone (1.0 mmol) was added and the resulting reaction mixture was stirred till completion of the reaction (TLC). The reaction mixture was quenched with water and extracted with ethyl acetate (4 × 5 mL). The combined organic layers were washed with brine solution, dried over anhydrous Na2SO4 and evaporated under reduced pressure. The crude product was purified by column chromatography using hexane/ethyl acetate to obtain the pure spirooxindole-pyrazoline derivatives.
Acknowledgements
The authors are thankful to SAIF, Punjab University, Chandigarh, India and SAIF, CDRI, Lucknow, India for providing spectral data. The authors also acknowledge financial support from UGC, New Delhi, India in form of fellowships for Swastika Singh and Mandavi Singh and for the Major Research Project (Project no. 42-263/2013 (SR)). Dr Mohammad Saquib specifically thanks UGC, New Delhi for Dr D. S. Kothari Postdoctoral Fellowship.Notes and references
- X. Guo and W. Hu, Acc. Chem. Res., 2013, 46, 2427–2440 CrossRef CAS PubMed.
- (a) P. Khanna, S. S. Panda, L. Khanna and S. C. Jain, Mini-Rev. Org. Chem., 2014, 11, 73–86 CrossRef CAS; (b) J. Day, M. Uroos, R. A. Castledine, W. Lewis, B. McKeever-Abbas and J. Dowden, Org. Biomol. Chem., 2013, 11, 6502–6509 RSC; (c) T. Honda, Pure Appl. Chem., 2010, 82, 1773–1783 CrossRef CAS; (d) C. V. Galliford and K. A. Scheidt, Angew. Chem., Int. Ed., 2007, 46, 8748–8758 CrossRef CAS PubMed.
- (a) D. B. Ramachary, C. Venkaiah and R. Madhavachary, Org. Lett., 2013, 15, 3042–3045 CrossRef CAS PubMed and references cited therein; (b) G. S. Singh and Z. Y. Desta, Chem. Rev., 2012, 112, 6104–6155 CrossRef CAS PubMed.
- (a) A. Bazgir, G. Hosseini and R. Ghahremanzadeh, ACS Comb. Sci., 2013, 15, 530–534 CrossRef CAS PubMed; (b) A. P. Antonchick, C. Gerding-Reimers, M. Catarinella, M. Schürmann, H. Preut, S. Ziegler, D. Rauh and H. Waldmann, Nat. Chem., 2010, 2, 735–740 CrossRef CAS PubMed.
- N. R. Ball-Jones, J. J. Badillo and A. K. Franz, Org. Biomol. Chem., 2012, 10, 5165–5181 CAS.
- (a) W. E. Conrad, K. X. Rodriguez, H. H. Nguyen, J. C. Fettinger, M. J. Haddadin and M. J. Kurth, Org. Lett., 2012, 14, 3870–3873 CrossRef CAS PubMed and references cited therein; (b) C. Li, T. Zhang, Z. Zeng, X. Liu, Y. Zhao, B. Zhang and Y. Feng, Org. Lett., 2012, 14, 479–481 CrossRef CAS PubMed; (c) A. Correa, I. Tellitu, E. Domınguez and R. SanMartin, J. Org. Chem., 2006, 71, 3501–3505 CrossRef CAS PubMed; (d) K. A. M. Abouzid and H. S. El-Abhar, Arch. Pharmacal Res., 2003, 26, 1–8 CrossRef CAS.
- (a) M. M. M. Santos, Tetrahedron, 2014, 70, 9735–9757 CrossRef CAS PubMed and references cited therein; (b) M. N. Ibrahim, M. F. El-Messmary and M. G. A. Elarfi, Eur. J. Chem., 2010, 7, 55–58 CAS.
- (a) B. Yu, D.-Q. Yu and H.-M. Liu, Eur. J. Med. Chem., 2014 DOI:10.1016/j.ejmech.2014.06.056 and references cited therein; (b) A. Monteiro, L. M. Gonçalves and M. M. M. Santos, Eur. J. Med. Chem., 2014, 79, 266–272 CrossRef CAS PubMed.
- (a) M. B. Gawande, V. D. B. Bonifacio, R. Luque, S. Paula, P. S. Brancoa and R. S. Varma, Chem. Soc. Rev., 2013, 42, 5522–5551 RSC.
- (a) C. Vidal and J. García-Álvarez, Green Chem., 2014, 16, 3515–3521 RSC; (b) Y. Gu and F. Jerome, Chem. Soc. Rev., 2013, 42, 9550–9570 RSC; (c) T. Laird, Org. Process Res. Dev., 2012, 16, 1–2 CrossRef CAS; (d) P. J. Dunn, Chem. Soc. Rev., 2012, 41, 1452–1461 RSC; (e) P. G. Jessop, Green Chem., 2011, 13, 1391–1398 RSC.
- (a) A. E. Díaz-Álvarez, J. Francos, P. Crochet and V. Cadierno, Curr. Green Chem., 2014, 1, 51–65 CrossRef; (b) D. M. Alonso, S. G. Wettstein and J. A. Dumesic, Green Chem., 2013, 15, 584–585 RSC; (c) J. Yang, J.-N. Tan and Y. Gu, Green Chem., 2012, 14, 3304–3317 RSC; (d) V. Pace, P. Hoyos, L. Castoldi, P. Domínguez de María and A. R. Alcántara, ChemSusChem, 2012, 5, 1369–1379 CrossRef CAS PubMed.
- (a) J. I. García, H. García-Marín and E. Pires, Green Chem., 2014, 16, 1007–1033 RSC; (b) Y. Gu and F. Jerome, Green Chem., 2010, 12, 1127–1138 RSC.
- (a) F. Nemati, M. M. Hosseini and H. Kiani, J. Saudi Chem. Soc., 2013 DOI:10.1016/j.jscs.2013.02.004; (b) H. R. Safaei, M. Shekouhy, S. Rahmanpur and A. Shirinfeshan, Green Chem., 2012, 14, 1696–1704 RSC; (c) G. Choudhary and R. K. Peddinti, Green Chem., 2011, 13, 276–282 RSC; (d) P. T. Anastas and T. C. Williamson, Green Chemistry: Designing Chemistry for the Environment, ACS Symposium Series, Washington DC, 1996 Search PubMed.
- (a) M. Singh, M. Saquib, M. Singh, S. B. Singh, P. Ankit, S. Fatma and J. Singh, Tetrahedron Lett., 2014, 55, 6175–6179 CrossRef CAS PubMed; (b) M. Singh, S. B. Singh, S. Fatma, P. Ankit and J. Singh, New J. Chem., 2014, 38, 2756–2759 RSC; (c) S. B. Singh, P. K. Verma, K. Tiwari, M. Srivastava, J. Singh and K. P. Tiwari, Supramol. Chem., 2013, 25, 255–262 CrossRef CAS PubMed; (d) M. Srivastava, J. Singh, S. B. Singh, K. Tiwari, V. Pathak and J. Singh, Green Chem., 2012, 14, 901–905 RSC.
Footnote |
| † Electronic supplementary information (ESI) available: Characterization data of all new compounds. See DOI: 10.1039/c5ra02794b |
| This journal is © The Royal Society of Chemistry 2015 |