Synthesis and characterization of ferric tannate as a novel porous adsorptive-catalyst for nitrogen removal from wastewater
Ruina Zhang,
Lin Li and
Junxin Liu*
Research Center for Eco-Environmental Sciences, Chinese Academy of Sciences, 18 Shuangqing Road, Haidian District, Beijing 100085, China. E-mail: jxliu@rcees.ac.cn; Fax: +86 10 62849133; Tel: +86 10 62849133
First published on 14th April 2015
Abstract
Ferric tannate has unique adsorption capacities for NH4+-N and NO2−-N simultaneously. So far, no normative method is available for synthesizing ferric tannate. In this work, an optimized method for synthesizing ferric tannate by using tannic acid and ferric chloride is established under the conditions of room temperature and neutral pH. The optimal molar ratio of tannic acid and ferric chloride were determined to be in the range of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 to 1
20 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25 based on the yield and stability of the ferric tannate composite. Scanning electron microscopy, the Brunauer–Emmett–Teller method, X-ray diffraction, and Fourier transform infrared spectrometry were used to characterize the ferric tannate composite. The results showed that the ferric tannate has a rough and porous surface, a large surface area, and an amorphous structure. Experiments of NH4+-N and NO2−-N adsorption reflected that the ferric tannate composite has a unique capacity for adsorption-catalyzed conversion of NH4+-N and NO2−-N to N2. When the mass ratios of ferric tannate/NH4+-N and ferric tannate/NO2−-N were both 200, the removal of NH4+-N and NO2−-N was 98.1% and 96.2%, respectively, after 3.0 hours of reaction. The conversion rate of N2 increased to 87.1%. Therefore, ferric tannate can be used as a potential material for removing nitrogen from wastewater in the future.
25 based on the yield and stability of the ferric tannate composite. Scanning electron microscopy, the Brunauer–Emmett–Teller method, X-ray diffraction, and Fourier transform infrared spectrometry were used to characterize the ferric tannate composite. The results showed that the ferric tannate has a rough and porous surface, a large surface area, and an amorphous structure. Experiments of NH4+-N and NO2−-N adsorption reflected that the ferric tannate composite has a unique capacity for adsorption-catalyzed conversion of NH4+-N and NO2−-N to N2. When the mass ratios of ferric tannate/NH4+-N and ferric tannate/NO2−-N were both 200, the removal of NH4+-N and NO2−-N was 98.1% and 96.2%, respectively, after 3.0 hours of reaction. The conversion rate of N2 increased to 87.1%. Therefore, ferric tannate can be used as a potential material for removing nitrogen from wastewater in the future.
Introduction
Nitrogen in wastewater is acknowledged as one of the critical causes of severe eutrophication of receiving water bodies. In order to satisfy the strict sewage discharge standards, many technologies for wastewater nitrogen removal have been extensively developed, such as biological nitrogen removal, chemical oxidation, adsorption, and ion exchange. Currently, biological nitrogen removal through the combination of aerobic nitrification and anoxic denitrification is widely applied in wastewater treatment plants. However, this approach requires a large amount of oxygen for nitrification, and a sufficient amount of organic substrates to maintain denitrification, which results in high energy consumption.1 Another biotechnology, anaerobic ammonium oxidation (Anammox), is recognized as a promising alternative for nitrogen removal. In the Anammox process, NO2−-N is converted to nitrogen gas (N2) with NH4+-N as the electron donor; this process consumes less energy and does not require additional carbon substrate as the electron donor.2 However, Anammox bacteria exhibit an extremely slow growth rate, limiting its wide practical application.3,4The chemical approach for nitrogen removal gives a greater reaction rate and a higher removal efficiency, but this approach requires additional oxidants or reductants, thereby causing high costs and complicating operational procedures.5,6 Adsorption and ion exchange approaches, such as using activated carbon and zeolites, can quickly remove NH4+-N, NO2-N, and NO3-N from wastewater. However, the adsorbent and ion exchange resins must be regenerated after the saturation of adsorbed nitrogen, and the regenerated solution needs further disposal, leading to higher environmental and economical costs.7–11 Thus, adsorption would be an attractive alternative approach for wastewater nitrogen removal if the adsorbent resin can be regenerated in situ.
Theoretically, the redox reaction for N2 production occurs between NH4+-N and NO2-N based on their standard redox potential,12 but the reaction rate depends on the reaction conditions significantly. Chemical kinetics indicate that the reaction rate can be accelerated through increasing the concentrations of reactants.13 Adsorption can transform reactants from liquids into solids, causing the reactants to be concentrated on the surface of adsorbents.14 A very recent patent implied that the presence of ferric tannate (a kind of magnetic nanoparticle) is capable of accelerating this reaction at room temperature.15 Furthermore, a highly cross-linked network of ferric tannate was found to exhibit a rough and porous surface,16 capable of absorbing both negative and cationic ions due to its ferric ions and negative oxygen ions. In addition, N2H4 and NH2OH are intermediate products in the reaction of NO2-N and NH4+-N,17–19 while ferric ions can convert N2H4 and NH2OH to N2.20–22 Thus, ferric tannate may be used as a novel solution for nitrogen removal. Even though many previous studies have illustrated the development and applications of magnetic nanoparticles in nitrogen removal,23–25 no literature has clearly demonstrated that these magnetic nanoparticles, including ferric tannate, could efficiently eliminate NH4+-N and NO2−-N simultaneously, as well as accelerate the redox reaction between these two substances. Additionally, no approach has been developed to date for synthesizing ferric tannate that can be efficiently used in wastewater treatment.
Therefore, this study aims to develop an optimized approach for synthesizing ferric tannate, which can accelerate the reaction between NH4+-N and NO2−-N. Tannic acid and ferric chloride were used as the raw materials to synthesize ferric tannate under the combined conditions of room temperature and neutral pH. Moreover, Fourier transform infrared spectrometry (FTIR) and the Brunauer–Emmett–Teller (BET) method were used to analyze the functional groups and surface morphology of the produced ferric tannate. This study also investigated the potential nitrogen removal performance of ferric tannate in wastewater treatment. The objective of this study is to synthesize a material that can accelerate the reaction between NO2−-N and NH4+-N.
Materials and methods
Materials
Tannic acid and ferric chloride (FeCl3) were used to synthesize ferric tannate. Tannic acid (C76H52O46, molar mass: 1701.18 g mol−1) was purchased from Sigma-Aldrich Co., USA, with a pure grade of ≥99.5%. FeCl3, NH4Cl, and NaNO2 were purchased from Sinopharm Chemical Reagent Co., China, with a pure grade of ≥99%. NaHCO3 was purchased from Tianjin Jinke Fine Chemical Research Institute, China, with a pure grade of ≥99.5%. The solutions of tannic acid, FeCl3, and NaHCO3 were prepared with distilled water at concentrations of 0.1, 1, and 0.65 mol L−1, respectively.Synthesis of ferric tannate
Tannic acid is an abundant polyphenolic compound. Its ortho-phenolic hydroxyl structure can cause complexation reactions with ferrous and ferric ions, which form a hybrid with metal–organic frameworks. The chemical structures of tannic acid and ferric tannate are shown in Fig. 1.26,27 In view of the complexity of the chemical structure of tannic acid, six molar ratios of tannic acid and FeCl3 (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10, 1
10, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15, 1
15, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20, 1
20, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25, 1
25, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30, and 1
30, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40) were chosen to investigate the influence of the varying proportions on ferric tannate yield. The synthesis of ferric tannate was performed in a conical flask with a volume of 150 mL. An amount of 10 mL of 0.1 mol L−1 tannic acid solution was mixed with various volumes of 1 mol L−1 FeCl3 solution. The pH of the mixture was adjusted to 7 by using 0.65 mol L−1 NaHCO3 solution. The reaction products were separated via sedimentation, and the resultant precipitate was washed with deionized water four times, centrifuged (3000 rpm, 2 min), and dried in a freezer dryer at −50 °C.
40) were chosen to investigate the influence of the varying proportions on ferric tannate yield. The synthesis of ferric tannate was performed in a conical flask with a volume of 150 mL. An amount of 10 mL of 0.1 mol L−1 tannic acid solution was mixed with various volumes of 1 mol L−1 FeCl3 solution. The pH of the mixture was adjusted to 7 by using 0.65 mol L−1 NaHCO3 solution. The reaction products were separated via sedimentation, and the resultant precipitate was washed with deionized water four times, centrifuged (3000 rpm, 2 min), and dried in a freezer dryer at −50 °C.
Characterization of ferric tannate
The ferric tannate yield (ye) was calculated using the following equation (eqn (1)):
 | (1) |
To determine the stability of the ferric tannate yield, 0.2 g of the ferric tannate composition, which was prepared with different ratios as mentioned earlier, was placed into six shake flasks. Then, each shake flask was charged with 200 mL of deionized water. After 1 h of continuous stirring, the mixture in each shake flask was filtered through a 0.45 μm pore size filter. The filtrate was measured with a UV-vis spectrophotometer (UV-1700, Hitachi Co., Japan) at a 200 nm to 500 nm wavelength.
Field emission scanning electron microscopy (FE-SEM), which was coupled with an energy dispersive X-ray spectroscopy (EDS) detector (SU-8020, Hitachi Co., Japan), was employed to observe the surface physical morphology and major elements of ferric tannate.
An automatic micropore and mesopore analyzer (ASAP 2020 HD88, USA) was operated to analyze the specific surface area and pore size distribution of ferric tannate. Moreover, the specific surface area and pore size distribution of the ferric tannate were determined with a N2 adsorption isotherm at 77 K by using a Micromeritics ASAP 2010 accelerated surface area analyzer and the BET method.28
Fourier transform infrared spectrometry (FTIR, Tensor 27, Germany) was applied to measure the functional groups of tannic acid and ferric tannate with the potassium bromide pellet method in the range of 400 cm−1 to 4000 cm−1.29
X-ray diffraction (XRD, X’Pert PRO MPD, PANalytical, Holland) was used to analyze the purity and crystallinity of ferric tannate, ferric chloride, and tannic acid with Cu-Kα radiation (λ = 0.015418 nm) in the 2θ range of 10° to 80°. The XRD data were collected in a scan mode with a step length of 0.5° with a scanning rate of 6° min−1. The accelerating voltage and the applied current were 45 kV and 45 mA, respectively.
Determination of the nitrogen removal performance
Several batch tests were conducted in stoppered conical flasks to estimate the nitrogen removal performance of the synthesized ferric tannate. Stock solutions were prepared by dissolving NH4Cl and/or NaNO2 in deionized water. Each time, 0.1 g ferric tannate and 50 mL stock solution were mixed in a flask, which was then shaken with a shaker at 200 rpm at room temperature. In the procedure of nitrogen removal by using ferric tannate, the pH was 7, which was adjusted with HCl or NaHCO3.30In the adsorption isotherm studies, stock solutions with different initial concentrations of NH4+-N or NO2−-N were added into 100 mL conical flasks, and the equilibrium time was set to 2 h. The samples were separated via filtration.
In the experiments, the concentrations of both NH4+-N and NO2−-N in the reaction solutions were 50 mg L−1. 100 mL stock solution and 1.0 g ferric tannate were added into a 150 mL stoppered conical flask. Then, the flask was shaken at 200 rpm for 3.0 h at room temperature, and 500 μL water samples and 50 μL gas samples were collected every 20 min. In the study, the data of NH4+ and NO2− adsorption on the ferric tannate at room temperature were obtained through three groups of parallel tests.
The concentrations of NH4+-N and NO2−-N were measured according to standard methods.31 The Agilent 6890N Gas Chromatograph equipped with a TCD was used to analyse the content of N2.
The adsorption amount of NH4+-N or NO2−-N on the ferric tannate at equilibrium, qe (mgN g−1), was calculated with the following equations (eqn (2) and (3)):
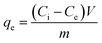 | (2) |
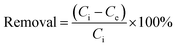 | (3) |
The Langmuir adsorption equation and the Freundlich adsorption equation can be linearised according to the equation in ref. 32.
Due to the inherent bias resulting from linearisation of the isotherm model, error analysis was employed as a criterion for the fitting quality. This statistical analysis is based on the sum of the squares of the differences between the experimental and the Langmuir model and Freundlich model calculated data. The error was calculated using the following equations (eqn (4) and (5)):
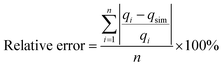 | (4) |
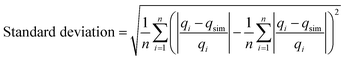 | (5) |
A low value of the relative error or standard deviation indicates that the experimental data fit better to the value from the model. In order to confirm the best-fit isotherms and kinetic models for the adsorption system, there is a need to analyse the data set using the Nash–Sutcliffe efficiency (NSE).33,34 As a dimensionless goodness-of-fit indicator, the values of the NSE could be widely used and a potentially reliable statistic for assessing the goodness-of-fit of isotherm adsorption models. NSE = 1 indicated a perfect fit, 0.75 ≤ NSE ≤ 0.9 suggested a good fit, and 0.65 ≤ NSE ≤ 0.749 suggested an acceptable fit. NSE is calculated as follows (eqn (6)):
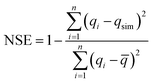 | (6) |
![[q with combining macron]](https://www.rsc.org/images/entities/i_char_0071_0304.gif) is the average adsorption capacity (mg g−1).
is the average adsorption capacity (mg g−1).
Synthesis of ferric tannate
As expected, the reaction of tannic acid with FeCl3 resulted in the formation of an abundant precipitate of ferric tannate. Table 1 presents a summary of the results of the ferric tannate yield from the six proportions of tannic acid and FeCl3, which showed an obvious response between the ratio of tannic acid and FeCl3 and the ferric tannate yield. Increasing the proportion of tannic acid to FeCl3 from 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 to 1
10 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 resulted in the gradual increase of ferric tannate yields. The highest yield, 82.6%, appeared in the case of the 1
30 resulted in the gradual increase of ferric tannate yields. The highest yield, 82.6%, appeared in the case of the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 ratio. Moreover, the ferric tannate yields are nearly equal in the cases of the 1
30 ratio. Moreover, the ferric tannate yields are nearly equal in the cases of the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20, 1
20, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25, and 1
25, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 ratios.
40 ratios.
| Ratio (tannic acid/FeCl3) | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10 |
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15 15 |
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 20 |
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25 25 |
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 30 |
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 40 |
| Yield (%) | 71.3 | 75.4 | 78.4 | 79.2 | 82.6 | 78.4 |
The stability of the ferric tannate composition in liquid will be highly important to apply it to nitrogen removal from wastewater. Therefore, the dissolution of the ferric tannate compositions in water was determined. The UV-visible absorbance spectra of the soak water of the ferric tannate compositions are shown in Fig. 2. The results showed that nothing could be dissolved in the cases of the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15, 1
15, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20, and 1
20, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25 ratios, but an absorption peak appeared at 302 nm in the case of the 1
25 ratios, but an absorption peak appeared at 302 nm in the case of the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 ratio, and at 303 nm in the cases of the 1
10 ratio, and at 303 nm in the cases of the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 and 1
30 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 ratios, which indicated that some substrates were dissolved into the water.
40 ratios, which indicated that some substrates were dissolved into the water.
The preceding results indicated that the molar ratio of tannic acid to ferric ions was responsible for the formation of the ferric tannate complex. With strong reducibility, the phenolic hydroxyl groups in tannic acid can reduce Fe3+ to Fe2+.35–37 In the case of the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 ratio, the excess tannic acid can produce a dissolved ferrous tannate, with an absorption peak appearing at 302 nm.38 In the cases of the 1
10 ratio, the excess tannic acid can produce a dissolved ferrous tannate, with an absorption peak appearing at 302 nm.38 In the cases of the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 30 and 1
30 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40 ratios, the ferric ion was in excess, which could lead to the formation of Fe(OH)3. Therefore, the absorption peak at 303 nm is probably from the ferric ion dissolution.39 Therefore, based on the yield and stability of the ferric tannate compositions, the optimal molar ratio of tannic acid and FeCl3 to synthesize ferric tannate is between 1
40 ratios, the ferric ion was in excess, which could lead to the formation of Fe(OH)3. Therefore, the absorption peak at 303 nm is probably from the ferric ion dissolution.39 Therefore, based on the yield and stability of the ferric tannate compositions, the optimal molar ratio of tannic acid and FeCl3 to synthesize ferric tannate is between 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 and 1
20 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25.
25.
Characterization of ferric tannate
According to the results of the preceding experiment, the synthesized sample in the case of the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 ratio was selected to characterize the ferric tannate composite. FE-SEM images of the ferric tannate composite are illustrated in Fig. 3. The ferric tannate composite had a rough and porous surface structure. The adsorption isotherms of N2 on the ferric tannate composite were used to deduce the surface characteristics. The results showed that the BET surface area, pore volume, and average pore diameter were 97 m2 g−1, 0.073 cm3 g−1, and 66.58 nm, respectively. In general, a large surface area and pore diameter can increase the adsorption capacity of the material.40,41
20 ratio was selected to characterize the ferric tannate composite. FE-SEM images of the ferric tannate composite are illustrated in Fig. 3. The ferric tannate composite had a rough and porous surface structure. The adsorption isotherms of N2 on the ferric tannate composite were used to deduce the surface characteristics. The results showed that the BET surface area, pore volume, and average pore diameter were 97 m2 g−1, 0.073 cm3 g−1, and 66.58 nm, respectively. In general, a large surface area and pore diameter can increase the adsorption capacity of the material.40,41
The SEM-EDS spectrum of the ferric tannate composite is shown in Fig. 4. The composite mainly consists of C, O, and Fe elements, and the percentage content of C, O, and Fe in the ferric tannate composite are 23.06%, 46.83%, and 29.91%, respectively. However, the ratio of C to O from the SEM-EDS spectrum is anomalous compared with the chemical formula of tannic acid (C76H52O46). A possible explanation is that ferric ions partially undergo a hydrolysis reaction at neutral pH, which leads to an increase of O in the ferric tannate composite.35,42,43 However, this finding should be verified with further research.
The XRD patterns of the tannic acid, FeCl3, and ferric tannate composite are shown in Fig. 5. XRD data indicate that FeCl3 has many diffraction peaks because of its crystalline structure.44 The diffraction peaks of the tannic acid and ferric tannate composite appear in the 2θ = 20° to 30° range, which demonstrates that they have an amorphous structure.
The FTIR spectra of the tannic acid and ferric tannate composite are illustrated in Fig. 6. Two broad adsorption bands in the 3600 cm−1 to 3100 cm−1 range indicating the presence of a phenolic hydroxyl group were observed, and the band at 1320 cm−1 exhibits a characteristic bending vibration of O–C in a phenolic hydroxyl group. The sharp band at 1400 cm−1 in the spectrum of the ferric tannate composite is very pronounced, which can be ascribed to the adsorption peak of a C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O vibration of benzoic acid. This phenomenon can be explained by the chelation and redox reaction of Fe3+ with the phenolic hydroxyl group, which results in the coexistence of Fe3+ and Fe2+ in ferric tannate.27,35 The interaction between Fe3+ and the phenolic hydroxyl group has an effect on the O–C bond stretching vibration, in which the band is shifted from 1320 cm−1 in the spectrum of the tannic acid to 1338 cm−1 in the spectrum of the ferric tannate composite.45–48 During the combination process of Fe3+ with the phenolic hydroxyl group, an oxygen anion was formed.
O vibration of benzoic acid. This phenomenon can be explained by the chelation and redox reaction of Fe3+ with the phenolic hydroxyl group, which results in the coexistence of Fe3+ and Fe2+ in ferric tannate.27,35 The interaction between Fe3+ and the phenolic hydroxyl group has an effect on the O–C bond stretching vibration, in which the band is shifted from 1320 cm−1 in the spectrum of the tannic acid to 1338 cm−1 in the spectrum of the ferric tannate composite.45–48 During the combination process of Fe3+ with the phenolic hydroxyl group, an oxygen anion was formed.
Evaluation of the nitrogen removal performance of the ferric tannate composite
Some batch tests for adsorbing NH4+-N and NO2−-N were conducted separately to estimate the nitrogen removal performance of the synthesized ferric tannate. The data of NH4+-N and NO2−-N adsorption on the ferric tannate composites, with only NH4+-N or NO2−-N in the solution, are shown in Fig. 7. The Langmuir and Freundlich models are usually used to describe the equilibrium isotherm data. The results from linear plots of these isotherms of NH4+-N and NO2−-N on the ferric tannate composites are listed in Table 2. Compared with the correlation coefficient value of the linear plot of the Freundlich isotherm, that of the Langmuir model was in better agreement with the observed data (R2 = 0.990 for NH4+-N adsorption and R2 = 0.957 for NO2−-N adsorption). The maximum adsorption capacities of NH4+-N and NO2−-N calculated from the Langmuir equations were 13.6 mg g−1 and 10.2 mg g−1 at room temperature, respectively. The results indicated that the adsorption capacity of ferric tannate toward NH4+-N was better than that toward NO2−-N.| Langmuir parameters | Freundlich parameters | |||||
|---|---|---|---|---|---|---|
| KL (L mg−1) | qm (mg g−1) | R2 | KF [(mg g−1)/(mg L−1)n] | n | R2 | |
| NH4+-N | 0.079 | 13.9 | 0.990 | 7.06 | 0.109 | 0.860 |
| NO2−-N | 0.009 | 10.2 | 0.957 | 2.84 | 4.53 | 0.952 |
In the study, the values of the NSE (Table 3) indicated that the Langmuir models of NH4+-N and NO2−-N can be considered to be very good (0.9 ≤ NSE ≤ 1). The Freundlich models of NH4+-N and NO2−-N can be considered to be good (0.8 ≤ NSE ≤ 0.9) and very good (0.9 ≤ NSE ≤ 1), respectively.
| Error estimation (%) | NSE | |||||||
|---|---|---|---|---|---|---|---|---|
| Langmuir | Freundlich | Langmuir | Freundlich | |||||
| NH4+ | NO2− | NH4+ | NO2− | NH4+ | NO2− | NH4+ | NO2− | |
| Test1 | 1.51 ± 1.24 | 1.46 ± 1.44 | 3.50 ± 2.28 | 2.94 ± 2.07 | 0.972 | 0.994 | 0.854 | 0.992 |
| Test2 | 1.58 ± 0.901 | 1.67 ± 1.19 | 3.51 ± 2.32 | 2.74 ± 2.29 | 0.976 | 0.996 | 0.849 | 0.991 |
| Test3 | 1.89 ± 1.19 | 2.42 ± 1.44 | 3.85 ± 2.63 | 1.60 ± 1.43 | 0.965 | 0.994 | 0.824 | 0.997 |
The adsorption performances of ferric tannate toward NH4+-N and NO2−-N were different and varied significantly with time. Fig. 8 shows the adsorptive behaviors for NH4+-N and/or NO2−-N on ferric tannate. The concentrations of NH4+-N and NO2−-N decreased with time, while the production of N2 increased over the same time (Fig. 8). When the mass ratios of ferric tannate/NH4+-N and ferric tannate/NO2−-N were both 200, the removal efficiencies of NH4+-N and NO2−-N were 98.1% and 96.2%, respectively, after 3.0 hours of reaction. The conversion rate of N2 increased to 87.1%. The results showed that after the addition of ferric tannate, the removal of NH4+-N and NO2−-N increased obviously with N2 production increasing simultaneously, which indicated that ferric tannate would be beneficial for the reaction between NH4+-N and NO2−-N, and lead to greater N2 generation. These results indicated that the ferric tannate had a unique capacity to adsorb anions and cations simultaneously, and an adsorption-catalyzed reaction occurred on the ferric tannate with the adsorption of NH4+-N and NO2−-N. The adsorption-catalyzed reaction between NH4+-N and NO2−-N could refresh the adsorption sites of ferric tannate, and the continuous adsorption of NH4+-N and NO2−-N on the ferric tannate could be promoted. Thus, the removal amount of NH4+-N and NO2−-N was enhanced. Therefore, the ferric tannate may be used as a promising material to remove nitrogen from wastewater in the future.
 | ||
| Fig. 8 The removal of NH4+-N and NO2−-N and the conversion rate of N2 with the addition of ferric tannate. | ||
For practical use, the dosage of ferric tannate will be adjusted with the concentrations of NH4+-N and NO2−-N in the wastewater, for the purpose of increasing the amounts of NH4+-N and NO2−-N converted to N2.
Conclusions
A novel method to synthesize porous ferric tannate composites was demonstrated by using tannic acid and ferric chloride under the conditions of room temperature and neutral pH. According to the yield and stability of the ferric tannate composite, the optimal molar ratio of tannic acid and FeCl3 is between 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 and 1
20 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 25. Combined with the SEM and BET characterization, the characteristics of a porous surface and a large surface area of the ferric tannate composite were confirmed. XRD analysis confirmed that the ferric tannate composite has an amorphous structure. The adsorption test results indicated that the adsorption isotherm of NH4+-N and NO2−-N on the ferric tannate composite agrees well with that obtained from the Langmuir model. Moreover, the maximum adsorption capacities of NH4+-N and NO2−-N calculated from the Langmuir equations were 13.6 mg g−1 and 10.2 mg g−1 at room temperature, respectively. Furthermore, ferric tannate has a unique capacity to adsorb NH4+-N and NO2−-N simultaneously. In addition, an adsorption-catalyzed reaction can occur on the ferric tannate with the adsorption of NH4+-N and NO2−-N, which enhances the removal efficiency of NH4+-N and NO2−-N. When the mass ratios of ferric tannate/NH4+-N and ferric tannate/NO2−-N were both 200, the removal efficiencies of NH4+-N and NO2−-N were 98.1% and 96.2%, respectively, after 3.0 hours of reaction. The conversion rate of N2 increased to 87.1%. Therefore, ferric tannate may be used as a promising material to remove nitrogen from wastewater in the future.
25. Combined with the SEM and BET characterization, the characteristics of a porous surface and a large surface area of the ferric tannate composite were confirmed. XRD analysis confirmed that the ferric tannate composite has an amorphous structure. The adsorption test results indicated that the adsorption isotherm of NH4+-N and NO2−-N on the ferric tannate composite agrees well with that obtained from the Langmuir model. Moreover, the maximum adsorption capacities of NH4+-N and NO2−-N calculated from the Langmuir equations were 13.6 mg g−1 and 10.2 mg g−1 at room temperature, respectively. Furthermore, ferric tannate has a unique capacity to adsorb NH4+-N and NO2−-N simultaneously. In addition, an adsorption-catalyzed reaction can occur on the ferric tannate with the adsorption of NH4+-N and NO2−-N, which enhances the removal efficiency of NH4+-N and NO2−-N. When the mass ratios of ferric tannate/NH4+-N and ferric tannate/NO2−-N were both 200, the removal efficiencies of NH4+-N and NO2−-N were 98.1% and 96.2%, respectively, after 3.0 hours of reaction. The conversion rate of N2 increased to 87.1%. Therefore, ferric tannate may be used as a promising material to remove nitrogen from wastewater in the future.
Acknowledgements
This study was financially supported by the National Science Foundation of China (no. 51138009 and 51208495).References
- A. Dey, Environ. Eng. Sci., 2010, 27, 757–765 CrossRef CAS.
- B. Kartal, J. G. Kuenen and M. C. M. van Loosdrecht, Science, 2010, 328, 702–703 CrossRef CAS PubMed.
- M. Strous, J. J. Heijnen, J. G. Kuenen and M. S. M. Jetten, Appl. Microbiol. Biotechnol., 1998, 50, 589–596 CrossRef CAS.
- K. Isaka, Y. Date, T. Sumino, S. Yoshie and S. Tsuneda, Appl. Microbiol. Biotechnol., 2006, 70, 47–52 CrossRef CAS PubMed.
- I. Ihara, K. Umetsu, K. Kanamura and T. Watanabe, Bioresour. Technol., 2006, 97, 1360–1364 CrossRef CAS PubMed.
- Y. H. Liou, S. L. Lo, C. J. Lin, W. H. Kuan and S. C. Weng, J. Hazard. Mater., 2005, 127, 102–110 CrossRef CAS PubMed.
- G. D. Ji, Y. Zhou and J. J. Tong, Environ. Eng. Sci., 2010, 27, 871–878 CrossRef CAS.
- P. A. Terry, Environ. Eng. Sci., 2009, 26, 691–696 CrossRef CAS.
- S. Annouar, M. Mountadar, A. Soufiane, A. Elmidaoui and M. A. M. Sahli, Desalination, 2004, 165, 437 CrossRef CAS.
- S. Leakovic, I. Mijatovic, S. Cerjan-Stefanovic and E. Hodzic, Water Res., 2000, 34, 185–190 CrossRef CAS.
- H. Zheng, L. Han, H. Ma, Y. Zheng, H. Zhang, D. Liu and S. Liang, J. Hazard. Mater., 2008, 158, 577–584 CrossRef CAS PubMed.
- J. C. Fanning, Coord. Chem. Rev., 2000, 199, 159–179 CrossRef CAS.
- S. J. Klippenstein, V. S. Pande and D. G. Truhlar, J. Am. Chem. Soc., 2014, 136, 528–546 CrossRef CAS PubMed.
- X. F. Wu, Y. L. Hu, F. Zhao, Z. Z. Huang and D. Lei, J. Environ. Sci., 2006, 18, 1167–1175 CrossRef CAS.
- Y. Yuasa, Jpn Kakai JP 197 196[87 197196] 31 Aug 1987.
- H. Wilson, C. Carr and M. Hacke, Chem. Cent. J., 2012, 6, 1–13 CrossRef PubMed.
- D. A. Nguyen, M. A. Iwaniw and H. S. Fogler, Chem. Eng. Sci., 2003, 58, 4351–4362 CrossRef CAS.
- W. R. L. van der Star, M. J. van de Graaf, B. Kartal, C. Picioreanu, M. S. M. Jetten and M. C. M. van Loosdrecht, Appl. Environ. Microbiol., 2008, 74, 4417–4426 CrossRef CAS PubMed.
- B. Kartal, W. J. Maalcke, N. M. de Almeida, I. Cirpus, J. Gloerich, W. Geerts, H. J. M. O. den Camp, H. R. Harhangi, E. M. Janssen-Megens, K. J. Francoijs, H. G. Stunnenberg, J. T. Keltjens, M. S. M. Jetten and M. Strous, Nature, 2011, 479, 127 CrossRef CAS PubMed.
- J. Mondal, K. T. Nguyen, A. Jana, K. Kurniawan, P. Borah, Y. L. Zhao and A. Bhaumik, Chem. Commun., 2014, 50, 12095–12097 RSC.
- Z. M. Peng, M. Z. Wu, Y. Xiong, J. Wang and Q. W. Chen, Chem. Lett., 2005, 34, 636–637 CrossRef CAS.
- Y. P. He, Q. L. Sheng, B. Liu and J. B. Zheng, Electrochim. Acta, 2012, 66, 82–87 CrossRef CAS PubMed.
- Y. Liu, J. Zhou, J. Gong, W. P. Wu, N. Bao, Z. Q. Pan and H. Y. Gu, Electrochim. Acta, 2013, 111, 876–887 CrossRef CAS PubMed.
- O. Gutfleisch, M. A. Willard, E. Bruck, C. H. Chen, S. G. Sankar and J. P. Liu, Adv. Mater., 2011, 23, 821–842 CrossRef CAS PubMed.
- S. Rakshit, C. J. Matocha and G. R. Haszler, J. Environ. Qual., 2005, 34, 1286–1292 CrossRef CAS PubMed.
- A. T. Iffat, Z. T. Maqsood and N. Fatima, J. Chem. Soc. Pak., 2005, 27, 174–177 CAS.
- T. K. Ross and R. A. Francis, Corros. Sci., 1978, 18, 351–361 CrossRef CAS.
- Q. L. Hongsen Zhang, J. Wang and J. Liu, RSC Adv., 2015, 5, 8 Search PubMed.
- F. Elkabbany, Y. Badr and M. Tosson, Phys. Status Solidi A, 1981, 63, 699–704 CrossRef CAS PubMed.
- A. Ritter and A. Peppas, J. Controlled Release, 1987, 37–42 Search PubMed.
- M. H. Mccrady, American Journal of Public Health and the Nations Health, 1966, 56, 684–685 CrossRef PubMed.
- V. Vimonses, S. M. Lei, B. Jin, C. W. K. Chowd and C. Saint, Chem. Eng. J., 2009, 148, 354–364 CrossRef CAS PubMed.
- A. Ritter and R. Munoz-Carpena, J. Hydrol., 2013, 480, 33–45 CrossRef PubMed.
- L. Wu, R. Munoz-Carpena, B. Gao, W. Yang and Y. A. Pachepsky, Environ. Sci. Technol., 2014, 48, 3883–3890 CrossRef CAS PubMed.
- J. A. Jaen, J. De Obaldia and M. V. Rodriguez, Hyperfine Interact., 2011, 202, 25–38 CrossRef CAS PubMed.
- R. Y. Liu and A. W. Xu, RSC Adv., 2014, 4, 40390–40395 RSC.
- G. K. B. Lopes, H. M. Schulman and M. Hermes-Lima, Biochim. Biophys. Acta, Gen. Subj., 1999, 1472, 142–152 CrossRef CAS.
- A. H. Basaran and O. H. Tuovinen, Appl. Microbiol. Biotechnol., 1986, 24, 338–341 CrossRef CAS.
- X. M. Li, T. T. Shen, D. B. Wang, X. Yue, X. Liu, Q. Yang, J. B. Cao, W. Zheng and G. M. Zeng, J. Environ. Sci., 2012, 24, 269–275 CrossRef CAS.
- C. L. Mangun, M. A. Daley, R. D. Braatz and J. Economy, Carbon, 1998, 36, 123–129 CrossRef CAS.
- L. B. Kong, H. Li, J. Zhang, Y. C. Luo and L. Kang, Appl. Surf. Sci., 2010, 256, 6688–6693 CrossRef CAS PubMed.
- J. A. Jaen and C. Navarro, Hyperfine Interact., 2009, 192, 61–67 CrossRef CAS.
- R. S. Peres, E. Cassel, C. A. Ferreira and D. S. Azambuja, Ind. Eng. Chem. Res., 2014, 53, 2706–2712 CrossRef CAS.
- Y. Zhang, Z. X. Zhang, T. B. Li, X. G. Liu and B. S. Xu, Spectrochim. Acta, Part A, 2008, 70, 1060–1064 CrossRef PubMed.
- E. G. I. d. S. N. J. A. Jáen and C. Herńandez, Hyperfine Interact., 1999, 122, 8 CrossRef.
- A. T. Iffat, Z. T. Maqsood, K. Ali and S. Nisar, J. Chem. Soc. Pak., 2004, 26, 151–156 CAS.
- J. Iglesias, E. G. De Saldana and J. A. Jaen, Hyperfine Interact., 2001, 134, 109–114 CrossRef CAS.
- R. G. Andrade, J. S. Ginani, G. K. B. Lopes, F. Dutra, A. Alonso and M. Hermes-Lima, Biochimie, 2006, 88, 1287–1296 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2015 |







