Novel pH sensitive N-doped carbon dots with both long fluorescence lifetime and high quantum yield†
Cui Zheng,
Xueqin An* and
Jun Gong
School of Chemistry and Molecular Engineering, East China University of Science and Technology, Shanghai, 200237, China. E-mail: anxueqin@ecust.edu.cn; Fax: +86-021-64250804; Tel: +86-021-64250804
First published on 31st March 2015
Abstract
Novel N-doped carbon dots (CDs) were synthesized. The fluorescent quantum yield of the CDs is up to 93.3%, and the fluorescence lifetime of the CDs is up to 19.50 ns. The optical properties of the CDs in various pH solutions were probed in different ways. The fluorescence characteristics of the CDs depended on change of surface structure, which was associated with pH value.
In recent years, carbon nanomaterials such as graphene oxide,1 nanodiamond,2 graphene3 and carbon dots4–7 have drawn much attention and have been widely studied. Particularly, as a newly emerged fluorescent material, CDs have been applied in the fields of bioimaging, sensors, photocatalysis, optoelectronic devices and nano-medicine based on their excellent properties such as unique fluorescence (FL), biocompatibility, low cytotoxicity and so on.8–10 The fluorescent quantum yield (QY) is a key parameter to evaluate the quality of FL materials. CDs commonly suffered from a low QY, therefore, their application was limited. Reduction11–13 and heteroatom doping6,14,15 are two widely used approaches to improve the QY of CDs. Enhancing the CDs QY (from 2% to 24%) by a reduction pathway has been reported.13 CDs doped with N using urea as nitrogen source can give high QY of 78%.6 A series of N-doped graphene quantum dots with high QY has been reported through hydrothermal routes using citric acid and multiple amine as precursor.16 N-doped CDs were synthesized by thermal pyrolysis route and the QY was up to 88.6%.17 But these synthesis methods for high QY are time-consuming16,18,19 or they need complex post treatment process.20 Thus, simple approach is still very necessary for preparation of CDs with high QY. On the other hand, FL lifetime is an intrinsic property of fluorescent material. The CDs with long FL lifetime maybe have a potential application on fluorescence lifetime imaging microscopy (FLIM) for obtaining high signal to noise images.21 But the FL lifetime of CDs is only a couple of nanoseconds.6,22 At present, the fluorescent material for FLIM application are concentrated on quantum dot (QD),21,23 protein24 and organic dye.25,26 Nevertheless, the toxicity of QDs and the short FL lifetime of protein and organic dye restrict their performances. Besides, the CDs with long FL lifetime can contribute to their application in solar cell.27 To expand the CDs application, it is still necessary to exploit a simple approach to synthesize CDs with long FL lifetime and high QY for broad application.
In this communication, novel N-doped CDs were synthesized by using citric acid (CA) as the carbon source and tris(hydroxymethyl)aminomethane (Tris) as the nitrogen source. The CDs not only possessed high QY and long FL lifetime, but also showed fantastic pH dependent FL. The optical properties of the N-doped CDs were probed by UV spectrophotometer, the steady-state FL spectrometer and time-resolved FL spectrometer. The pH dependent optical properties were discussed in different ways.
The N-doped CDs were synthesized through a one-step microwave digestion treatment in microwave digestion container.28 CA and Tris were dissolved in water and were placed in the microwave digestion container at 160 °C for 10 min. The whole process can be controlled under designated temperature, pressure and time (experiment details see ESI†). The aqueous solution was clear and transparent after completion of the reaction. The residual CA and Tris in the CDs solution were removed by dialysis process over 12 h. The CDs solution can remain stable over half a year at room temperature.
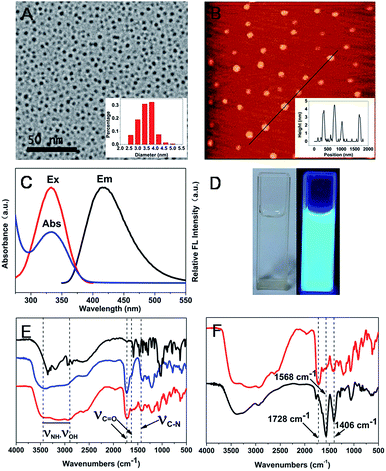
Transmission electron microscopy (TEM) image of the CDs and the size distribution are shown Fig. 1A and inset of Fig. 1A, respectively. The CDs are spherical and well disperse in a narrow size distribution. The diameter of CDs is approximately 3.5 ± 0.4 nm by calculating the average size of 250 nanoparticles (more TEM images see the Fig. S1†). The atomic force microscopy (AFM) image of the CDs is shown in Fig. 1B and the particle sizes distribute from 2.5 nm to 4.5 nm (Fig. 1B inset). The results accord with values from TEM in the experimental errors. The monodispersity of CDs in this work is much better than that prepared by microwave assisted approach.29
According to the element analysis to CDs, the CDs contained C 46.83%, H 5.14% and N 3.77%, suggesting the nitrogen doped in CDs. The QY of CDs was determined by using quinine sulphate as FL standards, and it was 93.3%(the detailed measurement process see ESI†). The QY of the N-doped CDs is much higher than that (about 15%) of CDs without N-doping.28 It is likely to be because N doping promote radiative recombination and lower down non-radiative recombination.16
The UV-visible absorption, excitation and emission spectra of CDs are presented in Fig. 1C. There is an absorption peak at 333 nm in the UV-visible absorption of CDs, which is consistent with the excitation wavelength. The FL spectra display an emission maximum at 417 nm for excitation at 333 nm. The as-prepared CDs solution exhibited distinct blue luminescence under UV irradiation (Fig. 1D).
The surface structure of CDs was investigated using Fourier transform infrared (FTIR). Compared with the FTIR of CA, new absorptions of CDs are emerged at 1630 cm−1 and 1412 cm−1 which are ascribed to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibration and C–N stretching of amide bond, respectively (Fig. 1E). The results also prove that nitrogen has been doped in the CDs, which is consistent with that from the element analysis. For CDs in acid solution, the FTIR absorption band at 2933–3400 cm−1 can be assigned to the –OH and –NH stretching vibration (Fig. 1F). The peak at 1728 cm−1 is related to the C
O stretching vibration and C–N stretching of amide bond, respectively (Fig. 1E). The results also prove that nitrogen has been doped in the CDs, which is consistent with that from the element analysis. For CDs in acid solution, the FTIR absorption band at 2933–3400 cm−1 can be assigned to the –OH and –NH stretching vibration (Fig. 1F). The peak at 1728 cm−1 is related to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibration of carboxylic acid. For CDs in non-acid solution, two major characteristic peaks for carboxylates, at around 1568 cm−1 and 1406 cm−1 instead of the C
O stretching vibration of carboxylic acid. For CDs in non-acid solution, two major characteristic peaks for carboxylates, at around 1568 cm−1 and 1406 cm−1 instead of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibration of CDs carboxylic acid (1728 cm−1), are observed as shown in Fig. 1F. Moreover, in comparison with CDs in acid solution, a weaker and narrower absorption of –OH and –NH stretching vibration was observed because lack of –OH absorption stretching vibration of carboxylic acid. The differences indicate that the surface of CDs have been changed with pH because of the deprotonation of carboxylic group. In order to make the following discussion convenient, the CDs with different surface structure are referred to as CDs–H and CDs–OH for protonation and deprotonation of carboxylic acid on the surface of CDs, respectively. The schema of CDs–H and CDs–OH are shown in Fig. 2A.
O stretching vibration of CDs carboxylic acid (1728 cm−1), are observed as shown in Fig. 1F. Moreover, in comparison with CDs in acid solution, a weaker and narrower absorption of –OH and –NH stretching vibration was observed because lack of –OH absorption stretching vibration of carboxylic acid. The differences indicate that the surface of CDs have been changed with pH because of the deprotonation of carboxylic group. In order to make the following discussion convenient, the CDs with different surface structure are referred to as CDs–H and CDs–OH for protonation and deprotonation of carboxylic acid on the surface of CDs, respectively. The schema of CDs–H and CDs–OH are shown in Fig. 2A.
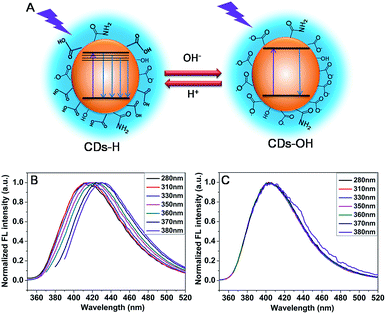 | ||
| Fig. 2 (A) The schema of CDs–H and CDs–OH. (B) The normalized FL spectra of CDs–H at pH of 3.4 and (C) CDs–OH at pH of 11.0 with various excitation wavelengths. | ||
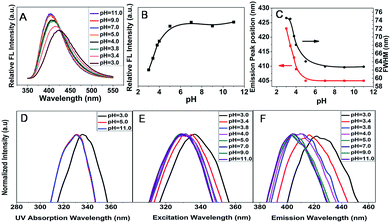
It is noteworthy to mention that the most interesting part in this communication is the pH dependent optical properties of CDs. The emission peaks of CDs–H depend on excitation wavelength when the pH value is in the range of 3.0–5.0. To illustrate the detailed changes of the emission wavelength at various excitation wavelengths for CDs–H, as an example, normalized FL spectra at pH of 3.4 is presented in Fig. 2B. The emission peaks shift from 417 to 433 nm when the excitation wavelength change from 280 to 380 nm. More information about the emission wavelength changing with excitation wavelength at various pH is provided in Fig. S3.† These properties of CDs–H may be caused by the complex surface states which could be induced by the deprotonation of carboxylic groups on CDs–H surface. The results are similar to that of common CDs, which emission wavelength depends on excitation wavelength.30–33 When the pH value is larger than 5.0, the emission peaks position of CDs–OH is unchanged with the variation of excitation wavelength. As an example of CDs–OH, normalized FL spectra of CDs–OH at pH of 11.0 is shown in Fig. 2C. The emission wavelength is 405 nm in the excitation wavelength region from 280 nm to 380 nm. The surface state of CDs–OH would be more uniform than CDs–H, because of the complete deprotonation and well formed “protective shell” with negative charge on the surface. It suggests that the surface structural change have been driven by H+ and OH− in the solution, and the different surface state of CDs may affect the band edge. It is similar to QDs whose band edge position can be controlled by pH.34
To further investigate the effect of pH on optical properties of CDs, the FL emission spectra of CDs at different pH were measured at the excitation wavelength of 330 nm (Fig. 3A). As shown in Fig. 3B, the FL intensity of CDs–H increases with pH when it is below 5.0, and FL intensity of CDs–OH almost maintain unchanged in various pH in pH region of 5.0–11.0. The phenomenon may also result from the different surface structures. With the pH increase, deprotonation of carboxylic groups is occurred on the surface of the CDs–H and a “protective shell” with negative charge is formed gradually on the surface of CDs–H with lower non-radiative recombination rate.35 This figure goes some way to explaining the changes of FL intensity. However, when pH is greater than 5.0, the deprotonation of carboxylic groups on the surface of CDs–OH has been done, and a uniform “protective shell” has been formed. The CDs–OH possess stable low non-radiative recombination rate, and the FL intensity maintain a constant as shown in Fig. 3B. In addition, it was also found out that the full width at half maximum (FWHM) of the FL CDs–H narrowed down with increase of pH as shown in Fig. 3C (the black line). Above results also demonstrate the pH value of solution would influence the surface state of CDs and alter the band edge. Moreover, the UV-visible absorption wavelength (Fig. 3D), excitation wavelength (Fig. 3E) and the emission peaks position (Fig. 3F and the red line of Fig. 3C) display a blue-shift with increase of pH for CDs–H. The inner charge-transfer and the energy bands are affected by the complex surface states of CDs–H,36 which result in narrowing of the FWHM (Fig. 3C black line) and the blue-shift (Fig. 3D–F) of above wavelength with pH increase. The blue-shift of FL emission wavelength with increase of pH also was observed in modified QDs and Au cluster.23,37 It means that the different optical properties of fluorophores could be caused by the different surface structure, no matter the surface structural change is due to surface modification or due to the medium environment. A comprehensive comparison of the range of emission wavelength, FL intensity and the FWHM between CDs–H and CDs–OH is also compared by three-dimensional map of emission–excitation-intensity (Fig. S4†).
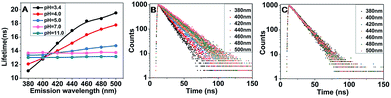
FL lifetime is an intrinsic characteristic of fluorescent reporter, which is usually independent of signal amplitude that is a function of many factors such as reporter concentration, light attenuation, excitation laser power and so on.38 However, occasionally, the FL lifetime of CDs previously reported depend on excitation wavelength or emission wavelength, which could be attributed to the effect of surface state.39,40 We were eager to know whether FL lifetime of CDs is affected by pH value, so the FL decays of CDs at different pH were measured at fixed excitation wavelength (330 nm) and at various emission wavelengths. The fluorescent decay curves could be described with a mono-exponential function, indicating that the CDs have one single FL origin, which is different with the most reported CDs with two or three FL times. The single FL origin maybe results from the uniform size distribution and single emission trap.6,17,41 Surprisingly, the FL lifetime of CDs–H increase with the emission wavelength, and the changes of FL lifetime decrease gradually with the increase of pH in the pH region of 3.0 to 5.0 (Fig. 4A). All results of FL lifetime are summarized in Table S1.† As an example, the decay curves of CDs–H at pH of 3.4 are shown in Fig. 4B. The fitted FL lifetime expand from 11.08 ns to 19.50 ns with the emission wavelength in the region of 380–500 nm. The complicated surface state of CDs–H would experience more complex de-excitation pathways to contribute to the diverse FL lifetimes.39–41 To the best of our knowledge, the long FL lifetime is up to 19.50 ns, which is the longest lifetime of CDs to date. For CDs–OH, the FL lifetime is independent of the emission wavelength (Fig. 4A pink and green lines) because the CDs–OH possess uniform surface state. The FL decays of CDs–OH at pH of 11.0, as an example, are shown in Fig. 4C. Nevertheless, more detailed study of the decay dynamics is necessary to better understand the mechanisms involved.
Conclusions
Novel pH sensitive CDs with high QY and long FL lifetime have been synthesized. The QY of N-doped CDs is up to 93.3%. The FL lifetime of CDs is up to 19.50 ns, which is the longest FL lifetime for reported CDs so far. The fluorescence lifetime of CDs is longer than that of most organic dyes (1–5 ns) and cell auto fluorescence (2–3 ns). Exploring the CDs application on FLIM is what we think should be done. The CDs optical properties closely depend on pH value of environment. For CDs–H, the emission wavelength depends on excitation wavelength and the FL lifetime is related to the emission wavelength. With increase of pH, the UV-visible absorption wavelength, excitation wavelength and emission peaks show a blue-shift, the FWHM narrow down, and the FL intensity increase. Compared with CDs–H, CDs–OH possesses more stable optical properties. The emission wavelength remains unchanged with various excitation wavelengths and the FL lifetime is not insensitive to emission wavelength. These different behaviors between CDs–H and CDs–OH could be come from the different surface states which associate to the energy gap. CDs–OH possesses stable optical properties and maybe has much potential application.Acknowledgements
This research was supported by the National Natural Science Foundation of China (21473055, 21273073 and 21073063), the National High-Tech R&D (863) Program of China (2011AA06A107). The authors also thank Prof. Jun Hu of East China University of Science and Technology for providing the microwave digestion apparatus.Notes and references
- S. K. Cushing, M. Li, F. Huang and N. Wu, ACS Nano, 2014, 8, 1002–1013 CrossRef CAS PubMed.
- U. Maitra, A. Jain, S. J. George and C. N. R. Rao, Nanoscale, 2011, 3, 3192–3197 RSC.
- J. Zhu, D. Yang, Z. Yin, Q. Yan and H. Zhang, Small, 2014, 10, 3480–3498 CrossRef CAS PubMed.
- S. N. Baker and G. A. Baker, Angew. Chem., Int. Ed., 2010, 49, 6726–6744 CrossRef CAS PubMed.
- Z. Xu, L. Yang, X. Fan, J. Jin, J. Mei, W. Peng, F. Jiang, Q. Xiao and Y. Liu, Carbon, 2014, 66, 351–360 CrossRef CAS PubMed.
- D. Qu, M. Zheng, P. Du, Y. Zhou, L. Zhang, D. Li, H. Tan, Z. Zhao, Z. Xie and Z. Sun, Nanoscale, 2013, 5, 12272–12277 RSC.
- A. B. Bourlinos, A. Stassinopoulos, D. Anglos, R. Zboril, M. Karakassides and E. P. Giannelis, Small, 2008, 4, 455–458 CrossRef CAS PubMed.
- L. Li, G. Wu, G. Yang, J. Peng, J. Zhao and J. Zhu, Nanoscale, 2013, 5, 4015–4039 RSC.
- Y. Wang and A. Hu, J. Mater. Chem. C, 2014, 2, 6921–6939 RSC.
- P. Wu and X. Yan, Chem. Soc. Rev., 2013, 42, 5489–5521 RSC.
- Y. Feng, J. Zhao, X. Yan, F. Tang and Q. Xue, Carbon, 2014, 66, 334–339 CrossRef CAS PubMed.
- H. Sun, L. Wu, N. Gao, J. Ren and X. Qu, ACS Appl. Mater. Interfaces, 2013, 5, 1174–1179 CAS.
- H. Zheng, Q. Wang, Y. Long, H. Zhang, X. Huang and R. Zhu, Chem. Commun., 2011, 47, 10650–10652 RSC.
- X. Dong, Y. Su, H. Geng, Z. Li, C. Yang, X. Li and Y. Zhang, J. Mater. Chem. C, 2014, 2, 7477–7481 RSC.
- Z. Yang, M. Xu, Y. Liu, F. He, F. Gao, Y. Su, H. Wei and Y. Zhang, Nanoscale, 2014, 6, 1890–1895 RSC.
- D. Qu, M. Zheng, L. Zhang, H. Zhao, Z. Xie, X. Jing, R. E. Haddad, H. Fan and Z. Sun, Sci. Rep., 2014, 4, 5294 CAS.
- M. Zheng, Z. Xie, D. Qu, D. Li, P. Du, X. Jing and Z. Sun, ACS Appl. Mater. Interfaces, 2013, 5, 13242–13247 CAS.
- Y. Dong, H. Pang, H. B. Yang, C. Guo, J. Shao, Y. Chi, C. M. Li and T. Yu, Angew. Chem., Int. Ed., 2013, 52, 7800–7804 CrossRef CAS PubMed.
- J. Liu, X. Liu, H. Luo and Y. Gao, RSC Adv., 2014, 4, 7648–7654 RSC.
- X. Wang, L. Cao, S. T. Yang, F. Lu, M. J. Meziani, L. Tian, K. W. Sun, M. A. Bloodgood and Y. Sun, Angew. Chem., Int. Ed., 2010, 49, 5310–5314 CrossRef CAS PubMed.
- A. Orte, J. M. Alvarez-Pez and M. J. Ruedas-Rama, ACS Nano, 2013, 7, 6387–6395 CrossRef CAS PubMed.
- S. Mondal, M. Chatti, A. Mallick and P. Purkayastha, Chem. Commun., 2014, 50, 6890–6893 RSC.
- M. J. Ruedas-Rama, A. Orte, E. A. H. Hall, J. M. Alvarez-Pez and E. M. Talavera, Chem. Commun., 2011, 47, 2898–2900 RSC.
- M. Tantama, Y. P. Hung and G. Yellen, J. Am. Chem. Soc., 2011, 133, 10034–10037 CrossRef CAS PubMed.
- K. Hoffmann, T. Behnke, D. Drescher, J. Kneipp and U. R. Genger, acs Nano, 2013, 7, 6674–6684 CrossRef CAS PubMed.
- M. K. Kuimova, G. Yahioglu, J. A. Levitt and K. Suhling, J. Am. Chem. Soc., 2010, 130, 6672–6673 CrossRef PubMed.
- R. Narayanan, M. Deepa and A. K. Srivastava, J. Mater. Chem. A, 2013, 1, 3907–3918 CAS.
- J. Gong, X. An and X. Yan, New J. Chem., 2014, 38, 1376–1379 RSC.
- W. Wang, Y. Li, L. Cheng, Z. Cao and W. Liu, J. Mater. Chem. B, 2014, 2, 46–48 RSC.
- X. Jia, J. Li and E. Wang, Nanoscale, 2012, 4, 5572–5575 RSC.
- S. Qu, X. Wang, Q. Lu, X. Liu and L. Wang, Angew. Chem., Int. Ed., 2012, 51, 12215–12218 CrossRef CAS PubMed.
- S. Sahu, B. Behera, T. K. Maiti and S. Mohapatra, Chem. Commun., 2012, 48, 8835–8837 RSC.
- Y. Wang, S. Kalytchuk, Y. Zhang, H. Shi, S. V. Kershaw and A. L. Rogach, J. Phys. Chem. Lett., 2014, 5, 1412–1420 CrossRef CAS.
- L. Vayssieres, Prepr. Symp.-Am. Chem. Soc., Div. Fuel Chem., 2011, 56, 135–136 CAS.
- W. Kong, H. Wu, Z. Ye, R. Li, T. Xu and B. Zhang, J. Lumin., 2014, 148, 238–242 CrossRef CAS PubMed.
- B. Valeur, Molecular Fluorescence: Principles and Applications, Wiley-VCH, Verlag Gmbh, New York, 2001, ch. 3, p. 58 Search PubMed.
- X. Wen, P. Yu, Y. Toh, X. Ma, S. Huang and J. Tang, Nanoscale, 2013, 5, 10251–10257 RSC.
- G. Ma, ACS Appl. Mater. Interfaces, 2013, 5, 2835–2844 CAS.
- K. Hola, A. B. Bourlinos, O. Kozak, K. Berka, K. M. Siskova, M. Havrdova, J. Tucek, K. Safarova, M. Otyepkaa, E. P. Giannelis and R. Zboril, Carbon, 2014, 70, 279–286 CrossRef CAS PubMed.
- Y. Long, C. Zhou, Z. Zhang, Z. Tian, L. Bao, Y. Lin and D. Pang, J. Mater. Chem., 2012, 22, 5917–5920 RSC.
- Y. Liu, C. Liu and Z. Zhang, J. Mater. Chem. C, 2013, 1, 4902–4907 RSC.
Footnote |
| † Electronic supplementary information (ESI) available: Detailed experiment process, measurement of quantum yield and the specific fluorescence lifetime of CDs. See DOI: 10.1039/c5ra01986a |
| This journal is © The Royal Society of Chemistry 2015 |