Esterification of carboxylic acids with alkyl halides using imidazolium based dicationic ionic liquids containing bis-trifluoromethane sulfonimide anions at room temperature
Arvind H. Jadhava,
Kyuyoung Leea,
Sangho Kooab and
Jeong Gil Seo*a
aDepartment of Energy Science and Technology, Energy and Environment Fusion Technology Center, Myongji University, Nam-dong, Cheoin-gu, Yongin-si, Gyeonggi-do 449-728, Republic of Korea. E-mail: jgseo@mju.ac.kr; Fax: +82-31-336-6336
bDepartment of Chemistry, Myongji University, Nam-dong, Cheoin-gu, Yongin-si, Gyeonggi-do 449-728, Republic of Korea
First published on 4th March 2015
Abstract
Task-specific room temperature ionic liquids (RTILs) composed of symmetrical N-methylimidazolium rings linked with a short oligo (ethylene glycol) chain (cationic part) and bis-trifluoromethane sulfonimide (NTf2, anionic part) were successfully synthesized, and their physicochemical properties were determined by various modern analytical techniques. The catalytic activity of the synthesized RTILs was evaluated in the esterification reaction of acids with alkyl halides in solvent-free conditions at room temperature. From the screening test, all the synthesized RTILs showed a high yield with significant selectivity for respective esters in a very short reaction time. Especially, 0.1 equimolar of RTIL-1 ([tetraEG(mim)2][NTf2]2) was found to be, the most efficient and reusable catalyst for this reaction. As a result, 100% conversion and up to a 94% yield of the respective ester product was obtained in a 30 min reaction time. This might be due to their synergetic effect of Lewis acidity, wide liquid range, and high miscibility compared to the other homogeneous and heterogeneous catalysts. Beside this, RTIL was easily separated from the reaction mixture and reused several times without any significant loss of catalytic activity and structural property. The present dicationic ionic liquids (ILs) under a solvent-free catalytic system were found to be kinetically fast, naturally benign, and achieved good yields for esterification of carboxylic acids with alkyl halides.
1. Introduction
With the rapid improvement in the field of synthetic organic chemistry, more and more naturally benign and eco-friendly processes are coming up at tremendous rate.1 Powered by the current scenario of environmental awareness and green chemistry, it seems that the ILs are here to stay in the current pattern of scientific research.2 Especially, RTILs have attracted great attention from the field of science and engineering. RTILs were initially introduced as a green reaction media for the substitution for organic solvents because of their unique chemical and physical properties such as thermal stability, nonvolatility, and nonflammability, specific miscibility, and easy reusability.3–7 Nowadays, they have trailed far away from this edge, showing their significant role in directing the reaction as catalysts as well as solvent.8–10 The properties of ILs are strongly reliant on their cations and anions therefore, ILs can be referred to as “designer solvents or catalyst” with tailored properties either by altering their structures or by varying cations and anions.1–4,11,12Esters and their derivatives are substantial products or intermediates in the chemical and pharmaceutical industries.13 Therefore, the Fischer ester synthesis method14 and various profitable esterification approaches catalysed by ion exchange resins, heteropoly acids, Lewis acids, zeolite, Brønsted acids, etc. have been reported.15–21 Today, eco-friendly organic synthesis has become very significant and widespread, directing towards green chemistry. Especially, RTILs attracted great interest as environmentally friendly reaction media for organic synthesis.1–5 In this respect, RTILs have been successfully utilized as catalyst or solvent in number of organic reactions such as hydrogenation,22 Friedel–Crafts reaction,23 Heck, Suzuki, Sonogashira,24–26 and Diels–Alder reactions,27 oxidation,28 Michael addition,29 nucleophilic substitution such as the Williamson ether synthesis,30 and etc.31,32
Esterification of carboxylic acids with alcohol using homogenous and heterogeneous organic or inorganic acid catalysts is prominent.33 On the other hand, elimination of water formed during the reaction and use of additional amount of the reactants are generally required for reasonable conversion with sufficient yield, and a bulky volume of volatile organic solvents is regularly mandatory. Meanwhile, it is very problematic to recover the homogenous or soluble acid catalyst which should be neutralized subsequently after the reaction. In addition, large volumes of volatile organic solvents and acidic catalyst may result in contamination to the environment.34–37 It has been reported that esterification could be carried out using ILs, such as ([Hmim][HSO4]) and ([Heemim][HSO4]).38 Though, the synthesis of these Brönsted acidic ILs were not appropriate, and volatile organic solvents were inevitable for the preparation and reuse. Additionally, the yield and conversions were lower than average for most of the substrates.
Esterification of acid with alkyl halide is a simple process, nevertheless there are still many encounters to produce a simple and clean route to produce esters from organic acids and alkyl halides.39 We believe that the predominantly attractive method uses hydrophobic mild acidic RTILs to carry out the reaction.40 The homogeneous phase of RTILs formed more interaction with the reactants, apparently when hydrophobic RTILs are used as an alternative, poor solubility of the ester products in the RTILs produced phase separation, shifting the equilibrium towards the product.40 High yield could be achieved with a simple green separation method due to the hydrophobic nature, and RTILs could be reused several times beneath the protection of green chemistry. The NTf2 containing RTILs are hydrophobic and stable at atmospheric condition. They exhibit a wide liquid range, even recent study indicates that decomposition temperatures were overestimated in previous thermal studies.2–5 Moreover, the relatively low cost and their high chemical stability at atmospheric condition, the NTf2 containing RTILs are attracting more attention for their industrial use.2–5 So far hydrophilic ILs with acidic counter anions and protonated acidic ILs either solvent or catalyst have been used in esterification reactions.38
Herein, we report the synthesis of a series of hydrophobic imidazolium based RTILs functionalized with oligo ethylene glycol chain between two cations and associated with NTf2 anions. Particularly, we concentrated on imidazolium based RTILs as catalysts for biodegradability and toxicity reasons.2–5 The prepared hydrophobic RTILs were achieved fascinating physicochemical properties such as viscosity, wide liquid range, and high thermal stability. The catalytic activity was tested for the esterification of carboxylic acids with alkyl halides. The obtained results were compared with the other homogenous as well as heterogeneous catalysts. In addition, recyclability test was also performed for these RTILs.
2. Experimental
2.1. Materials and methods
N-Methylimidazole (99.0%), methanesulfonyl chloride (99.0%), triethyl amine (99.0%), tetraethylene glycol (99.0%), bis(trifluoromethane)sulphonamide lithium salt (99.0%), triethylene glycol (99.0%), anhydrous pyridine (99.8%), di-ethylene glycol (99.0%), acetonitrile (HPLC grade), ethyl acetate, anhydrous sodium sulfate, and all substrate for ester derivatives were purchased from Sigma-Aldrich and used without further purification. Ester derivatives were characterized by using TLC, different spectroscopic methods and compared with authentic samples. All solvents were obtained from commercial sources and were distilled from appropriate agents prior to use it.2.2. Characterization
All synthesized RTILs were characterized by 1H NMR and 13C NMR spectroscopy on Bruker spectrometer 400 MHz and 100 MHz respectively using CDCl3 or DMSO as a solvent. The reported chemical shifts were against TMS as reference for 1H and 13C NMR. Mass spectra were acquired using electro spray ionization on Waters Micrimass ZQ LC/MS 2000 mass spectrometer. FT-IR spectra were obtained on Varian 2000 (Scimitar series) spectrophotometer. TLC analysis was accomplished on silica-gel Poly Gram SIL G/UV 254 plates to monitor the reaction growth. All ester products were also characterized by 1H and 13C NMR, IR spectroscopy and matched with the authentic samples.Determination of acidity by pyridine adsorption investigates with prepared RTILs were accomplished using dry pyridine with RTIL by 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 weight ratio. The mixture of sample was speared on CaF2 windows and these all experiments were performed in dry condition and analysed at room temperature. Differential Scanning Calorimetry (DSC) results were obtained using aluminium sample pan of diameter 7 mm and between 10–15 mg of the RTIL was measured into the aluminium sample pan. A minor incision was made in the tops of both the reference pan and sample pan. Samples were measured at atmospheric pressure between −30 and +80 °C temperature with a heating (cooling) rate 5 °C min−1 in a nitrogen atmosphere. Thermogravimatric analyses (TGA) were performed on Scinco TGA N-100 instrument with heating rate 5 °C min−1 in a nitrogen atmosphere. The RTILs samples were measured in 30 and 800 °C temperature range. Viscosity determination experiments were performed on Brook-field DV-II+ model Programmable viscometer associated with temperature measured heating bath at atmospheric pressure.
5 weight ratio. The mixture of sample was speared on CaF2 windows and these all experiments were performed in dry condition and analysed at room temperature. Differential Scanning Calorimetry (DSC) results were obtained using aluminium sample pan of diameter 7 mm and between 10–15 mg of the RTIL was measured into the aluminium sample pan. A minor incision was made in the tops of both the reference pan and sample pan. Samples were measured at atmospheric pressure between −30 and +80 °C temperature with a heating (cooling) rate 5 °C min−1 in a nitrogen atmosphere. Thermogravimatric analyses (TGA) were performed on Scinco TGA N-100 instrument with heating rate 5 °C min−1 in a nitrogen atmosphere. The RTILs samples were measured in 30 and 800 °C temperature range. Viscosity determination experiments were performed on Brook-field DV-II+ model Programmable viscometer associated with temperature measured heating bath at atmospheric pressure.
2.3. Synthesis of dicationic room temperature ionic liquids (RTILs)
2.3.2.1. Tetraethylene glycol-bis(3-methylimidazolium)bis-(trifluoromethanesulfonyl)imide ([tetraEG(mim)2][NTf2]2) (RTIL-1). A mixture of tetraethylene glycol-bis(3-methylimidazolium)dimesylate ([tetraEG(mim)2][OMs]2) (1.0 mmol) RTIL and bis-trifluoromethane sulfonimide (LiNTf2) (2.0 mmol) in double distilled water was stirred magnetically for 2 days in a single neck round bottom flask at room temperature. After completion of reaction, the reaction mixture showed two phases in which hydrophobic IL was separated out from the aqueous layer. The aqueous layer was decanted and remaining IL was washed three times with double distilled water to remove unreacted starting materials and finally heated at 80 °C under reduced pressure on rotary evaporator and afforded oily liquid as resulting product tetraethylene glycol-bis(3-methylimidazolium)(bis-trifluoromethanesulfonyl)imide ([tetraEG(mim)2][NTf2]2) (1): yield 88%; oily liquid; 1H NMR (400 MHz, CDCl3): δ 9.41 (s, 2 × H), 7.72 (s, 2 × H), 7.14 (s, 2 × H), 4.41 (t, J = 4.8 Hz, 2 × 2H), 3.91 (s, 2 × 3H), 3.78 (t, 2 × 2H), 3.61 (t, 2 × 2H); 3.46 (s, 2 × 2H); 13C NMR (100 MHz, CDCl3): δ 149.53, 137.42, 122.89, 123.09, 70.41, 70.11, 66.94, 56.26, 37.13; FT-IR (500–4000 cm−1): 3158, 3123, 2968, 2884, 1569, 1453, 1348, 1180, 1134, 1053, 934, 838. MS-ESI: m/z [M − NTf2]+ calcd: 604.13; found: 604.11. Elem. anal. calc. (%) for C20H28F12N6O11S4: C, 27.15; H, 3.19; N, 9.50; found: C, 27.17; H, 3.12; N, 9.59.
2.3.2.2. Triethylene glycol-bis(3-methylimidazolium)(bis-trifluoromethanesulfonyl)imide ([triEG(mim)2][NTf2]2) (RTIL-2). Yield 86%; oily liquid; 1H NMR (400 MHz, CDCl3): δ 9.38 (s, 2 × H), 7.71 (s, 2 × H), 7.32 (s, 2 × H), 4.21 (t, J = 4.8 Hz, 2 × 2H), 3.87 (s, 2 × 3H), 3.73 (t, 2 × 2H), 3.58 (t, 2 × 2H); 3.42 (s, 2 × 2H); 13C NMR (100 MHz, CDCl3): δ 149.46, 137.39, 122.92, 123.16, 71.04, 71.09, 66.86, 56.39, 37.27; FT-IR (500–4000 cm−1): 3159, 3122, 2958, 2880, 1569, 1453, 1347, 1180, 1134, 1053, 929, 833. MS-ESI: m/z [M − NTf2]+ calcd: 560.11; found: 560.19. Elem. anal. calc. (%) for C18H24F12N6O10S4: C, 25.72; H, 2.88; N, 10.00; found: C, 25.77; H, 2.83; N, 10.29.
2.3.2.3. Diethylene glycol-bis(3-methylimidazolium)(bis-trifluoromethanesulfonyl)imide ([diEG(mim)2][NTf2]2) (RTIL-3). Yield 84%; oily liquid; 1H NMR (400 MHz, CDCl3): δ 9.36 (s, 2 × H), 7.68 (s, 2 × H), 7.29 (s, 2 × H), 4.19 (t, J = 4.8 Hz, 2 × 2H), 3.89 (s, 2 × 3H), 3.71 (t, 2 × 2H), 3.61 (t, 2 × 2H); 3.41 (s, 2 × 2H); 13C NMR (100 MHz, CDCl3): δ 148.91, 136.81, 123.02, 122.76, 71.00, 71.11, 67.01, 55.92, 38.01; FT-IR (500–4000 cm−1): 3148, 3121, 2968, 2884, 1556, 1456, 1342, 1182, 1133, 1057, 921, 840. MS-ESI: m/z [M − NTf2]+ calcd: 516.08; found: 516.11, Elem. anal. calc. (%) for C16H20F12N6O9S4: C, 24.12; H, 2.53; N, 10.55; found: C, 24.19; H, 2.60; N, 10.51.
2.4. Typical esterification reaction procedure in presence of RTILs
In a two necked round bottom flask containing a magnetic needle, 1.0 mmol of organic acid and 1.0 mmol of alkyl halide was added into it. 0.1 mmol of synthesized RTILs as catalyst was added and stirred at room temperature. Reaction progress was monitored by thin layer chromatography (TLC). After completion of the reaction, the mixture was washed three times by diethyl ether to extract reaction product and collected the ether layer. The synthesized RTILs are insoluble in diethyl ether. The collected ether layer was dried on anhydrous sodium sulphate and concentrated on rotary evaporator to obtained crude ester product. The residue containing RTIL was washed three times with double distilled water to remove by-product (hydrogen halide) formed in the reaction and obtained pure hydrophobic RTIL at the bottom of round bottom flask. Later on, collected RTIL dried at 80 °C in vacuum oven for 2 h and reused it for next attempt of esterification reaction.3. Results and discussion
The imidazolium based dicationic RTILs with NTf2 anions were developed using straightforward synthesis technique as presented in Scheme 1. In short, tetraethylene glycol (a) reacted with methanesulfonyl chloride utilizing triethyl amine as base in dichloromethane to obtain tetraethylene glycol dimesylate (b). In succeeding step, addition of N-methylimidazole was done in b to afford tetraethylene glycol-bis(3-methylimidazolium)dimesylate ([tetraEG(mim)2][OMs]2) (c) as a thick oily liquid. Followed by the metathesis reaction was done in double distilled water with Li(NTf2)2 to obtain ([tetraEG(mim)2][NTf2]2) (d) as thick oily liquid. All produced new dicationic RTILs catalysts after totally characterized by analytical and spectroscopic approaches later on, estimation of their physicochemical properties such as acidity, viscosity, DSC, TGA, and etc. were done.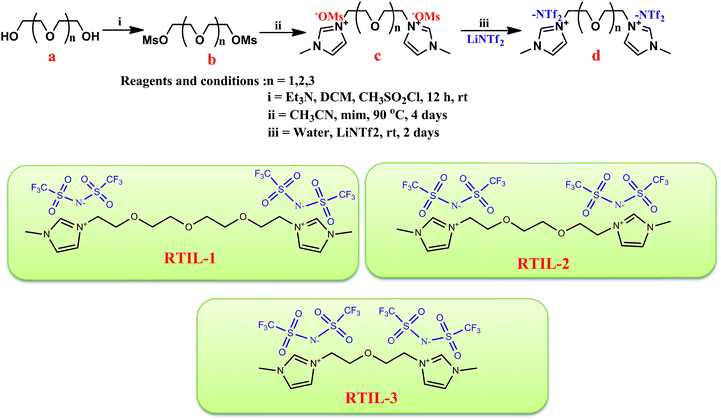 | ||
| Scheme 1 Synthesis of imidazolium based dicationic RTILs used for the esterification reaction of acids with alkyl halides. | ||
3.1. Lewis acidity determination of RTILs by FT-IR spectroscopy
The results of FT-IR spectra of pyridine adsorbed RTILs and pure pyridine are shown in Fig. 1. Generally, pyridine adsorbed complexes shows two major absorption peaks at 1438 and 1548 cm−1 analogous to Lewis and Brønsted acidity, respectively.42 Consequently, to define both of Lewis and Brønsted acidity, pyridine adsorption study by FT-IR spectroscopy is appropriate tool. It is reported, the presence of band for individual N–H bending and C–C stretching modes of protonated pyridine experience upward shifts or increased intensity of peaks upon synchronization of pyridine molecule with both acid sites.42 | ||
| Fig. 1 Acidity determination of prepared imidazolium based dicationic RTILs containing NTf2 anions by FT-IR spectroscopy. | ||
The IR peak detected at 1438 cm−1 is allocated to characteristic protonation of pyridine molecule onto Lewis acid sites and the peak at 1578 cm−1 is illustrative peak for chemisorbed pyridine onto Lewis acid sites and both of these peaks were observed in the spectra of prepared RTILs with pyridine in our study.42 When pyridine was mixed with all three types of RTILs respectively, could not find any peak at 1548 cm−1 corresponding to the Brønsted (protic) acid sites. The intense peak at 1440 cm−1 proved that Lewis acidity present in the RTIL-1.
In addition, this peak is shifted to higher wavenumber from 1438 cm−1 to 1444 cm−1 and the intensity increased compared with the pure pyridine. Subsequently, RTIL-2 and RTIL-3 also showed peaks at higher wavenumber shifted at 1440 cm−1 and 1442 cm−1, respectively. These results confirmed that all the prepared RTILs have Lewis acidity and the order is RTIL-1 > RTIL-2 > RTIL-3. The highest acidity was observed in RTIL-1 ([tetraEG(mim)2][NTf2]2), which has the tetraethylene glycol linker between the imidazolium cations. We believed that, the variation between these Lewis acidities of RTILs appeared because of their linker in between symmetrical imidazolium cations and synergetic effect of NTf2 anions present in RTILs.
3.2. Thermogravimetric analysis (TGA) of dicationic RTILs
NTf2 containing RTILs have a very wide stable liquid range for example monocationic [C4mim][NTf2] as compared to organic compounds.43 This liquid range is organized by their glass forming (Tg) or melting temperature (Tm), which are both usually below ambient temperature, moreover, leading to the new generation tailored RTILs, and often even below 0 °C. Because of their non-boiling character, the higher temperature characteristics limit is given by their thermal decomposition (Td) temperature.2–5 In order to explain the thermal performance and the temperature which permit for a perfect and harmless procedure, it is important to study the exhibition of RTILs at elevated temperatures and to study the degradation mechanisms.2,3In the present study, conducted thermal analysis of all prepared RTILs from room temperature to 800 °C in nitrogen atmosphere and gas flow rate was 5 °C min−1. Prior to thermal analysis, all RTILs were kept at 70 °C for 12 h in vacuum oven. The thermal decomposition profiles are shown in the Fig. 2. Td of all the RTILs estimated from these curves was compared with reported values of monocationic imidazolium based ILs which contains the conventional anions, as summarized in the Table 1. It was observed that the thermal stability of prepared RTILs was significantly higher than that of other conventional imidazolium based monocationic and dicationic RTILs.43 This might be due to high molecular weight of NTf2 anions conjugate with two imidazolium rings separated with oligo ethylene glycol chains and strong intermolecular interaction between them. Moreover, the length of linker chain separating two imidazolium cations was partially responsible for their high Td.3,4
 | ||
| Fig. 2 Thermogravimetric analysis (TGA) of prepared imidazolium based dicationic RTILs containing NTf2 anions. | ||
| Sr. no. | IL | Type of IL | Cations | Anions | Td (°C) | Reference |
|---|---|---|---|---|---|---|
| a TGA condition: 25–800 °C in nitrogen atmosphere and gas flow was 5 °C min−1. | ||||||
| 1 | ([C4mim][Cl]) | Monocationic | Imidazolium | Cl- | 125 | 44 |
| 2 | ([C4mim][NTf2]) | Monocationic | Imidazolium | NTf2 | 409 | 43 |
| 3 | ([N2222][Cl]) | Monocationic | Ammonium | Cl- | 264 | 45 |
| 4 | ([N2222][NTf2]) | Monocationic | Ammonium | NTf2 | 403 | 45 |
| 5 | [C9(mim)2][NTf2]2 | Dicationic | Imidazolium | NTf2 | 350 | 46 |
| 6 | ([C12(benzim)2][NTf2]2) | Dicationic | Imidazolium | NTf2 | 350 | 46 |
| 7 | ([C9(mpy)2][NTf2]2) | Dicationic | Pyrrolidinium | NTf2 | >400 | 46 |
| 8 | ([C9(bpy)2][NTf2]2) | Dicationic | Pyrrolidinium | NTf2 | 330 | 46 |
| 9a | ([TetraEG(mim)2][NTf2]2) | Dicationic | Imidazolium | NTf2 | 455 | In present study |
| 10a | ([TriEG(mim)2][NTf2]2) | Dicationic | Imidazolium | NTf2 | 442 | In present study |
| 11a | ([DiEG(mim)2][NTf2]2) | Dicationic | Imidazolium | NTf2 | 437 | In present study |
In this study, the nature of linker chains and anions are effective factors in facilitating excellent thermal property of prepared RTILs. All the prepared RTILs did not lose their weight as heated up to 435 °C. This is an outstanding thermal stability of RTILs containing the organic moieties, suggesting that prepared RTILs could be utilized in high temperature organic transformation reaction. Among the RTILs tested, RTIL-1 having tetraethylene glycol chain with NTf2 anions showed the most thermally stable (Td = 455 °C) characteristic. Thermograms of RTIL-2 and RTIL-3 showed Td at 442.92 °C and 437.42 °C, respectively and these slight reductions in Td of RTIL-2 and RTIL-3 were might be due to the association of tri and di-ethylene glycol separating two imidazolium rings and their synergetic effect on RTILs (insight Fig. 2). However, the results describes that well-constructed RTILs showed great thermal stability compared with traditional monocationic ILs composed of alkyl imidazolium cations and conventional halide anions.36
3.3. Differential scanning calorimetry (DSC) analysis of dicationic RTILs
Differential scanning calorimetry (DSC) is normally used for studies of heat capacities, phase transitions, enthalpies, and chemical reactions. This method offers significant benefits, such as the minor amount of sample needed for the experiment, suitable of manipulation, flexibility, and quick measurement with exactness of temperature control. DSC is also very suitable for the study of physicochemical progresses, such as the determination of the kinetics and thermodynamics of organic compounds due to the fact that these kinds of reactions are exothermic and are easily followed dynamically and isothermally by DSC.In the present study, analysed all the prepared RTILs by DSC analysis and heating traces results are collected in the Fig. 3. As RTILs were stable at atmospheric condition, before conducting the DSC analysis, all RTILs were kept for drying at 70 °C for 24 h in vacuum oven.
 | ||
| Fig. 3 Differential scanning calorimetry (DSC) analysis of prepared imidazolium based dicationic RTILs containing NTf2 anions. | ||
Typically, 6–15 mg of RTIL was kept in a standard 40 μL aluminium DSC pan. Air flexibility alterations were made by using known densities of resources. The DSC pan was hermetically wrapped with a cover to eliminate any evaporation of the substances. The pan was weighed before and after each experiment to detect any mass loss during the investigation. Measurements were accomplished in two cycles and temperature range was −30–80 °C with scanning rates of 5 °C min−1. From all the DSC analysis results of RTILs, we could observe that RTILs did not experience any phase change behaviour throughout the cycles down to −30 °C as well as no other transitions detected. This means that pure RTILs could not be crystallized with no glass transition temperature in the DSC pans and remain liquid even at very low temperature (−30 °C). This is likely attributed to the largely unfavourable packing of the short oligo ethylene glycol linked with imidazolium based cations associated with the bulky NTf2 anions despite repeated cycling at low temperatures. Such an analogous behaviour was earlier detected in similar mixed anion and cations ILs systems which were not able to be crystallized and did not showed any glass transition temperature.47 These extraordinary and interesting features of RTILs confirm that the use of these RTILs in extensive selection of applications, particularly liquid range at high as well as low temperature can be utilized.
3.4. Viscosity determination of dicationic RTILs
The viscosity is an important property of RTILs, because it intensely influences the diffusion of molecules dispersed or dissolved in the RTILs. In addition, it is the internal friction or resistance to flow produced by intermolecular components and is consequently very significant in all physicochemical developments. For that purpose, any scientific unit operations assigning with the transfer of fluids or energy involve knowledge of the viscosity of pure compounds and/or their mixtures. General prediction of viscosity is normally problematic, and flexible predictive models and their values will require additional experimental data in order to attain a better understanding of viscosity property. The NTf2 containing ILs exhibit a wide liquid range even if a recent study point out that, decomposition temperatures were overestimated in previous thermal studies.43 The viscosities of prepared RTILs were measured from 25 °C to 100 °C temperature and results are reported in Fig. 4. | ||
| Fig. 4 Viscosity determination at various temperature of prepared imidazolium based dicationic RTILs containing NTf2 anions. | ||
Before performing the viscosity measurement all RTILs were vacuum dried at 70 °C for 12 h. The viscosity of these neat RTILs decreases with the temperature rising from 25 to 100 °C. In this study, we found that RTIL-1 containing imidazolium rings separated by tetraethylene glycol chain showed the highest viscosity among the prepared RTILs. At 25 °C, RTIL-1 specified 330 cP viscosity and gradually it is decreased while increased the temperature. Finally, at 100 °C it displayed 160 cP viscosity, which is very high compared to other RTILs studied in this report.2–5 The higher viscosity may be attributed due to the tetraethylene glycol chain length between the imidazolium cations. The length of linker chain is sufficient to make interaction between two imidazolium cations, resulting in a formation of cage-like structure with the bulky NTf2 cations which make RTIL more viscous. On the other hand RTIL-2 showed viscosity range between 280–110 cP at 25 °C to 100 °C respectively, which is quite favourable viscosity compared to the reported NTf2 containing RTILs.48 This might be due to that triethylene glycol chain is enough long to keep both imidazolium cation rings apart from each other and make RTIL structure straight. The lowest viscosity was observed in the RTIL-3, which has the diethylene glycol linker between the two imidazolium rings and the range was 100–220 cP at 25 °C to 100 °C. In this study, consequently all RTILs exhibited high viscosity compared to their reported monocationic ILs. The higher viscosities of these prepared RTILs may be attributed to supplementary hydrogen bonds containing the ether functional groups of the cations with other cations or anions present in the RTILs. In addition, conducted few more experiments for the determination of hydrophobicity of prepared RTILs and results are displayed in a photograph of Fig. 5.
In a certain amount of water and RTILs were mixed respectively and stirred for 24 h and analysed separation layer. After 24 h results clearly showed that, there are still two layers corresponding to RTIL (bottom) and water (upper) which designate the hydrophobicity of RTILs.
3.5. Catalytic activity of dicationic RTILs in esterification reaction
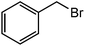
Table 2 show the results of esterification reactions of benzoic acid (1) with benzyl bromide (2) to obtain benzyl benzoate (3) as a model ester compound using various homogeneous catalysts including all the synthesized RTILs under solvent-free condition. In the beginning, performed catalyst-free and solvent-free synthesis of benzyl benzoate (3) at room temperature and progress of the reaction was monitored continuously and there was no reaction up to 24 h (entry 1).| Entry | Catalyst | Catalyst amount | Time (min) | Conversion (%) | Yieldb (%) |
|---|---|---|---|---|---|
| a All reactions were carried out on 1.0 mmol scale of substrate with catalyst and without addition of any co-catalyst and solvent.b Yield refers to the isolated product. Products were characterized by NMR and IR spectroscopy. | |||||
| 1 | — | — | 24 h | — | — |
| 2 | ([DiEG(mim)2][NTf2]2) | 0.5 equiv. | 120 | 100 | 81 |
| 3 | ([TriEG(mim)2][NTf2]2) | 0.5 equiv. | 60 | 100 | 86 |
| 4 | ([TetraEG(mim)2][NTf2]2) | 0.5 equiv. | 10 | 100 | 94 |
| 5 | ([TetraEG(mim)2][NTf2]2) | 0.3 equiv. | 20 | 100 | 93 |
| 6 | ([TetraEG(mim)2][NTf2]2) | 0.2 equiv. | 20 | 100 | 94 |
| 7 | ([TetraEG(mim)2][NTf2]2) | 0.1 equiv. | 30 | 100 | 94 |
| 8 | [BMIm][NTf2] | 0.1 equiv. | 30 | 37 | 22 |
| 9 | [BMIm][NTf2] | 0.1 equiv. | 120 | 59 | 55 |
| 10 | 1-Methyl imidazole | 0.1 equiv. | 30 | 42 | 36 |
| 11 | Tetra ethylene glycol | 0.1 equiv. | 30 | 12 | 8 |
| 12 | Li[NTf2]2 | 0.1 equiv. | 30 | 68 | 61 |
| 13 | [BMIm][Br] | 0.1 equiv. | 30 | 44 | 34 |
| 14 | [BMIm][Cl] | 0.1 equiv. | 30 | 56 | 42 |
| 15 | FeCl3 | 0.1 equiv. | 30 | 32 | 26 |
| 16 | CuCl2·2H2O | 0.1 equiv. | 30 | 28 | 23 |
Next step, conducted the same reaction of 1 (1 mmol) and 2 (1 mmol) in 0.5 equiv. of ([diEG(mim)2][NTf2]2) RTILs as catalyst and stirred at room temperature (entry 2). After the completion of reaction (120 min) the reaction mixture was extracted from RTIL using diethyl ether to get crude reaction mass and subjected to 1H NMR analysis. In 1H NMR spectrum of crude mass, it was observed that the reaction of 1 with 2 using RTILs was preceded smoothly and provide high yield (81%) of 3 within 120 min showing the significant catalytic activity of prepared ([diEG(mim)2][NTf2]2) catalyst.
In the beginning, performed catalyst-free and solvent-free synthesis of benzyl benzoate (3) at room temperature and progress of the reaction was monitored continuously and there was no reaction up to 24 h (entry 1). Next step, conducted the same reaction of 1 (1 mmol) and 2 (1 mmol) in 0.5 equiv. of ([diEG(mim)2][NTf2]2) RTILs as catalyst and stirred at room temperature (entry 2). After the completion of reaction (120 min) the reaction mixture was extracted from RTIL using diethyl ether to get crude reaction mass and subjected to 1H NMR analysis. In analysis of 1H NMR spectrum of crude mass, it was observed that the reaction of 1 with 2 using RTILs was preceded smoothly and provide high yield (81%) of 3 within 120 min showing the significant catalytic activity of prepared ([diEG(mim)2][NTf2]2) catalyst.
Later on, performed the same reaction using catalytic amount of ([triEG(mim)2][NTf2]2) and ([tetraEG(mim)2][NTf2]2), respectively (entries 3 and 4) and reactions were completed in very short reaction time with excellent yield of 3. These results reveal that all prepared RTILs are highly reactive in esterification of acids with alkyl halides. Especially, among all prepared RTILs, ([tetraEG(mim)2][NTf2]2) showed 94% yield in 10 min reaction time was found the most active RTIL. We believe that the tetraethylene glycol chain with dicationic imidazolium rings of ([tetraEG(mim)2][NTf2]2) IL produced highly active structure associated with supplementary hydrogen bonding with reactant. On the other hand, the rest of dicationic RTILs have less hydrogen bonding with the reactant and obtained less yield. Therefore ([tetraEG(mim)2][NTf2]2) dicationic RTIL is selected for the advance study in esterification reaction at ambient condition.
In subsequent step, determined the minimum amount of ([tetraEG(mim)2][NTf2]2) RTIL as catalyst required for rapid conversion of reactants and formation of ester product in efficient yield by carrying out the several number of catalytic reactions using 0.3, 0.2, 0.1 equiv. of ([tetraEG(mim)2][NTf2]2) RTIL in constant reaction condition (entries 5–7). These reactions outcome showed that all reactions with ([tetraEG(mim)2][NTf2]2) were consummate efficiently well and deliver outstanding yield of 3 within 30 min reaction time. The amount of catalyst dosage showed major influence on the reaction time, 0.5 equiv. of ([tetraEG(mim)2][NTf2]2) completed reaction within 10 min, successively when amount of catalyst decreased, reactions are sluggish and need more reaction time. 0.3 and 0.2 equiv. of catalyst completed the reaction in 20 min under same condition, respectively. 94% respective ester product yield was obtained in 30 min reaction time by using 0.1 equiv. of ([tetraEG(mim)2][NTf2]2) as catalyst. Production an allowance for the mole economy, 0.1 equiv. of catalyst was nominated for additional study with this protocol.
Our next task was to compare and determine synergetic effect of dicationic ([tetraEG(mim)2][NTf2]2) catalyst with other mono-cationic ILs as well as homogenous and heterogeneous catalyst in esterification reaction at room temperature. The conventionally popular mono-cationic IL [BMIm][NTf2] was selected for the comparison in esterification reaction at similar condition. A 22% yield of respective ester product 3 was obtained using catalytic amount of monocationic IL under same reaction condition (entry 8). In addition, increased reaction time up to 120 min with [BMIm][NTf2] was also fails to give the efficient respective ester product yield (entry 9). We also carried out reaction with 1-methyl imidazole and tetraethylene glycol as catalyst, these reactions are also unable to show the efficient yield of relevant product in short reaction period (entries 10 and 11). To rule out the potential promotional effect of the [NTf2]2 anions with prepared RTILs, we performed reaction with heterogeneous Li[NTf2]2 as catalysts. It is already reported and well known that, [NTf2]2 anions forms weak complexes with acidic moieties.4,5 Both fluoro-alkyl groups of [NTf2]2 with a large ionic size are known to increase the interaction of the ILs and acidic groups as well as least co-ordination with cations that is favourable for reaction kinetics compared to traditional anions.1–4 It was found that ester product could be generated with this catalyst but reaction was sluggish dramatically and obtained insufficient amount of respective product (entry 12). Meanwhile, synthesized RTILs were successfully achieved efficient yield of respective ester in very short reaction time. This is a strong indication that, the imidazolium cation with [NTf2]2 anions significantly influences the reaction progress of the esterification conversion. These results indicates that, there is a synergetic mutual action of short oligo ethylene glycol functionalized imidazolium cation and [NTf2]2 anions, which plays a crucial role on the improvement of conversion of acids and alkyl halides into esters. These observations underline the fact that the positive effect of ionic liquids in the esterification reaction is closely related to synergetic effect of both counter ions “anions and cations” as well as organic functional groups on the imidazolium rings, which determine the acidic property of RTILs.
Furthermore, various traditional ILs were applied in the reaction system to gain further insights into the effect of the counter ions and the substituents of RTILs components on the esterification reaction. As elucidated in Table 2, the efficiency of production of ester was modulated strongly by the different type of counter ions examined. [BMIm][Br] and [BMIm][Cl] were tested for the esterification reaction under similar reaction condition and resulted in 34% and 42% yields of corresponding product (entries 13 and 14). Additionally, to determine the catalytic activity of heterogeneous catalyst in esterification, FeCl3, and CuCl2·H2O were also tested in similar reaction condition and results provide 26 and 23% yield of 3 in 30 min reaction time (entries 15 and 16).
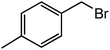
The successful development of highly active dicationic ([tetraEG(mim)2][NTf2]2) RTIL as catalyst for the esterification reaction prompted us to initiate a preliminary program to investigate its usefulness as catalyst for synthesis of various ester derivatives at room temperature. Table 3 shows the results of several prepared ester derivatives using dicationic ([tetraEG(mim)2][NTf2]2) RTIL at ambient condition. Benzoic acid and benzyl bromide were selected as initial model reactant and respective ester product as synthetic target on which to test and support our design that synthesis of ester derivative could be promoted in these prepared dicationic RTILs and this reaction showed 94% yield in 30 min reaction period. In next step, we performed substituted benzoic acid and substituted alkyl halides, these reactions were also showed promising results and reveals the catalytic activity of ([tetraEG(mim)2][NTf2]2) RTIL (entries 1–4).
| Sr. no. | Acid | R–X | Time (min) | Product | Yieldb (%) |
|---|---|---|---|---|---|
| a All reactions were carried out on 1.0 mmol scale of substrate with 0.1 equiv. of catalyst and without addition of any co-catalyst and solvent.b Yield refers to the isolated product. Products were characterized by NMR and IR spectroscopy. | |||||
| 1 | 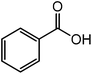 |
 |
40 | 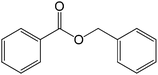 |
94 |
| 2 | 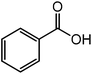 |
 |
30 | 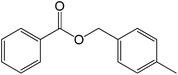 |
90 |
| 3 | 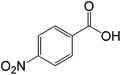 |
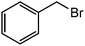 |
50 | 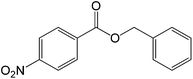 |
76 |
| 4 | 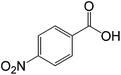 |
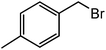 |
30 | 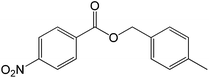 |
81 |
| 5 | 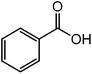 |
 |
30 | 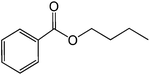 |
86 |
| 6 | 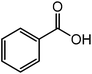 |
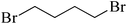 |
30 | 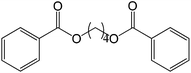 |
87 |
| 7 | 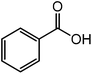 |
 |
50 | 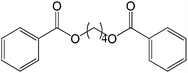 |
81 |
| 8 | 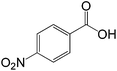 |
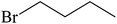 |
30 | 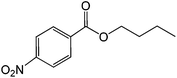 |
88 |
| 9 | 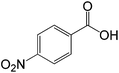 |
 |
40 | 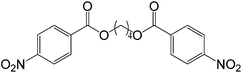 |
94 |
| 10 | 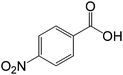 |
 |
60 | 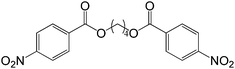 |
80 |
| 11 | 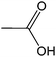 |
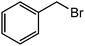 |
50 | 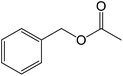 |
81 |
| 12 | 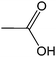 |
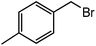 |
30 | 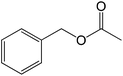 |
88 |
| 13 | 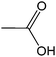 |
 |
40 | 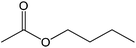 |
92 |
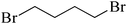
| 14 | 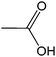 |
 |
60 | 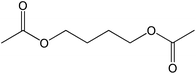 |
90 |
| 15 | 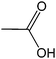 |
 |
60 | 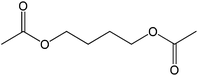 |
81 |
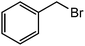
| 16 | 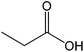 |
 |
60 | 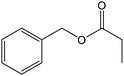 |
78 |
| 17 | 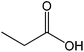 |
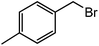 |
40 | 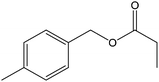 |
90 |
| 18 | 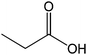 |
 |
50 | 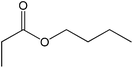 |
90 |
| 19 | 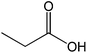 |
 |
60 | 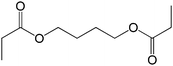 |
92 |
| 20 | 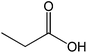 |
 |
60 | 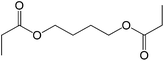 |
78 |
| 21 | 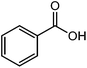 |
 |
180 | 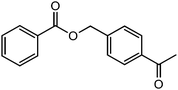 |
76 |
| 22 | 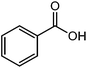 |
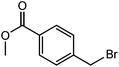 |
145 | 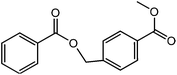 |
80 |
| 23 | 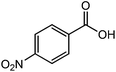 |
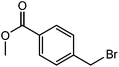 |
90 | 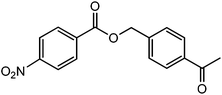 |
74 |
| 24 | 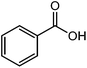 |
 |
130 |  |
79 |
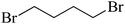
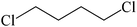
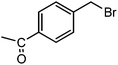
The straight chain mono and di-alkyl halides with substituted and un-substituted acid were also exhibited efficient yield of respective ester products at room temperature in short reaction time (entries 5–10). Acetic acid reacted with benzyl bromide and substituted benzyl bromide or alkyl bromide in presence of RTIL were also showed efficient yield of corresponding ester product in adequate time (entries 11 and 12). Butane-1,4-diyl di-acetate were obtained in very good yield by reacting with di-alkyl halides with acetic acid (entries 13–15). In addition, propionic acid were also showed ester products with benzyl bromide, substituted benzyl bromide, mono alkyl halide, di-alkyl halide correspondingly in efficient yield at ambient condition with ([tetraEG(mim)2][NTf2]2) RTIL as a catalyst (entries 16–20). It is noteworthy to mention here, the present catalytic system is favourable for esterification of acid in presence of other acid sensitive functional groups (entries 21–24). We consider that, the present new approach for the synthesis of esters using organic acids with alkyl halides offers numerous benefits over traditional method. Whereas, present method does not involve the use of alkali throughout the stages, it provides high yield and conversion in short reaction time and completed under mild and green condition. Currently, investigating of mechanism of this esterification reaction with these RTILs and the extension of this method to multifunctional molecules is in under way.
3.6. Reusability test of ([tetraEG(mim)2][NTf2]2) RTIL
Progressive achievement of new eco-friendly, green, mild, advance, cheap and reusable dicationic RTILs as a new catalytic coordination for the esterification reaction of acids to alkyl halides under solvent free mild reaction condition is the major ambition of this study. Meanwhile, the catalytic recyclability and their strength after utilization are very substantial factors in catalysed organic reactions and it is essential to understand the stability of catalyst. To study the reuse of the ([tetraEG(mim)2][NTf2]2) catalyst, the RTIL was collected as a thick oily liquid in round bottom flask at the end of reaction and washed three times with double distilled water and used it in next attempt. Recyclability of ([tetraEG(mim)2][NTf2]2) catalyst was studied over eight cycles for the esterification reaction. There is no significant loss in the catalytic activity and selectivity over eight cycles (Fig. 6). Additionally, conversion of reactant was 100% and the yield of product was sustained at 92% during recycling tests. In addition, performed 1H and 13C NMR analysis of reused ([tetraEG(mim)2][NTf2]2) catalyst for the resolve of structural changes in recycled catalyst. The results of NMR and IR analysis revealed that, the protection of structural integrity, no remarkable changes were found in the structure of recycled RTIL after being used eight cycles in esterification. 95% RTILs could be achieved after completion of reaction for the next cycle. | ||
| Fig. 6 Recyclability test of imidazolium based dicationic ([tetraEG(mim)2][NTf2]2) RTILs in esterification reaction. | ||
Therefore, the catalyst ([tetraEG(mim)2][NTf2]2) RTIL is capable for recycle eight times without loss in its selectivity and catalytic activity.
4. Conclusions
In conclusion, a series of symmetrical imidazolium based tailor-made dicationic RTILs were prepared successfully and screened its property and catalytic activity in esterification reaction using organic acids and alkyl halides. These RTILs possess excellent physicochemical properties compared with tradition monocationic ILs, such property can be utilized in various research applications. The present hydrophobic catalytic system showed 100% consumption of reactants and achieved 94% yield of respective ester products at room temperature in 30 min reaction time using 0.1 equiv. of RTIL. Moreover, effect of catalyst dosage and homogenous as well as heterogeneous catalyst were also determined and discussed. The present catalyst showed outstanding performance using very less amount of catalyst in very short reaction period. At elevated reaction condition, several ester derivatives were prepared and characterized. The RTIL can be separated by a simple decantation method and regenerated several times. Regenerated catalyst showed efficient stability without considerable loss of physicochemical property and catalytic activity. Such task-specific ILs may well prove to be very significant in transferring ILs from the laboratory to production scale processes in esterification reaction.Acknowledgements
This work was supported by KCRC through the NRF funded by Ministry of Science, ICT, and Future Planning (NRF-2014M1A8A1049258) and National Research Foundation of Korea (NRF) grant funded by the Ministry of Education (no. 2009-0093816).References
- M. B. Gawande, V. D. B. Bonifacio, R. Luque, P. S. Branco and R. S. Varma, Chem. Soc. Rev., 2013, 42, 5522 RSC
.
- S. Zhang, J. Sun, X. Zhang, J. Xin, Q. Miao and J. Wang, Chem. Soc. Rev., 2014, 43, 7838 RSC
.
- M. Petkovic, K. R. Seddon, L. P. N. Rebello and C. S. Pereira, Chem. Soc. Rev., 2011, 40, 1383 RSC
.
- N. V. Plechkova and K. R. Seddon, Chem. Soc. Rev., 2008, 37, 123 RSC
.
- T. Erdmenger, C. G. Sanchez, J. Vitz, R. Hoogenboom and U. S. Schubert, Chem. Soc. Rev., 2010, 39, 3317 RSC
.
- A. H. Jadhav, H. Kim and I. T. Hwang, Catal. Commun., 2012, 21, 96 CrossRef CAS PubMed
.
- A. H. Jadhav and H. Kim, Tetrahedron Lett., 2012, 53, 5338 CrossRef CAS PubMed
.
- A. C. Gujar and M. G. White, Catalysis, 2008, 21, 154 Search PubMed
.
- H. Li, P. S. Bhadury, B. Song and S. Yang, RSC Adv., 2012, 2, 12525 RSC
.
- A. H. Jadhav, A. Chinnappan, R. H. Patil and H. Kim, Chem. Eng. J., 2014, 236, 300 CrossRef CAS PubMed
.
- A. H. Jadhav and H. Kim, Bioresour. Technol., 2013, 132, 342 CrossRef CAS PubMed
.
- A. H. Jadhav and H. Kim, RSC Adv., 2013, 3, 5131 RSC
.
- S. C. Fox, B. Li, D. Xu and K. J. Edgar, Biomacromolecules, 2011, 12, 1956 CrossRef CAS PubMed
.
- E. Fischer and A. Speier, Berichte, 1895, 28, 3252 CrossRef CAS
.
- Comprehensive Organic Synthesis, ed. G. Beaz, B. M. Trost and I. Fleming, Pergamon: Oxford, 1991, vol. 6 Search PubMed
.
- H. Li, Y. Qiao, L. Hua, Z. Hou, B. Feng, Z. Pan, Y. Hu, X. Wang, X. Zhao and Y. Yu, ChemCatChem, 2010, 2, 1165 CrossRef CAS
.
- J. Otera, Chem. Rev., 1993, 93, 1449 CrossRef CAS
.
- K. Ishihara, S. Ohara and H. Yamamoto, Science, 2000, 290, 1140 CrossRef CAS
.
- K. Ishihara, M. Nakayama, S. Ohara and H. Yamamoto, Tetrahedron, 2002, 58, 8179 CrossRef CAS
.
- N. Nakayama, A. Sato, K. Ishihara and H. Yamamoto, Adv. Synth. Catal., 2004, 346, 1275 CrossRef
.
- K. Ishihara, S. Nakagawa and A. Sakakura, J. Am. Chem. Soc., 2005, 127, 4168 CrossRef CAS PubMed
.
- P. J. Dyson, D. J. Ellis, D. G. Parker and T. Welton, Chem. Commun., 1999, 25 RSC
.
- Y. R. Jorapur, C. H. Lee and D. Y. Chi, Org. Lett., 2005, 7, 1231 CrossRef CAS PubMed
.
- A. J. Carmichael, M. J. Earle, J. D. Holbrey, P. B. McCormac and K. R. Seddon, Org. Lett., 1999, 1, 997 CrossRef CAS
.
- V. Calo, A. Nacci, L. Lopez and N. Mannarini, Tetrahedron Lett., 2000, 41, 8973 CrossRef CAS
.
- C. J. Mathews, P. J. Smith and T. Welton, Chem. Commun., 2000, 1249 RSC
.
- J. G. Huddleston and R. D. Rogers, Chem. Commun., 1998, 1765 RSC
.
- J. S. Yadav, B. V. S. Reddy, A. K. Basak and A. V. Narsaiah, Tetrahedron, 2004, 60, 2131 CrossRef CAS PubMed
.
- V. Calo, A. Nacci, L. Lopez and V. L. Lerario, Tetrahedron Lett., 2000, 41, 8977 CrossRef CAS
.
- J. S. Yadav, B. V. S. Reddy, A. K. Basak and A. Venkat Narsaiah, Tetrahedron Lett., 2003, 44, 2217 CrossRef CAS
.
- A. H. Jadhav, A. Chinnappan, R. H. Patil and H. Kim, Chem. Eng. J., 2014, 240, 228 CrossRef CAS PubMed
.
- A. H. Jadhav, A. Chinnappan, R. H. Patil and H. Kim, Chem. Eng. J., 2014, 243, 82 CrossRef PubMed
.
- H. P. Zhu, F. Yang, J. Tang and M. Y. He, Green Chem., 2003, 5, 38 RSC
.
- M. Mäki-Arfela, T. Salmi, M. Sundell, K. Ekman, R. Peltonen and J. Lehtonen, Appl. Catal., A, 1999, 184, 25–32 CrossRef
.
- M. Hino and K. Arata, Appl. Catal., 1985, 18, 401 CrossRef CAS
.
- M. A. Schwegler and H. Van Bekkum, Appl. Catal., 1999, 74, 191 Search PubMed
.
- P. Wasserscheid and W. Keim, Angew. Chem., Int. Ed., 2000, 39, 3772 CrossRef CAS
.
- J. Fraga-Dubreuil, K. Bourahla and M. Rahmouni, Catal. Commun., 2002, 3, 185 CrossRef CAS
.
- J. McNulty, S. Cheekoori, J. J. Nair, V. Larichev, A. Capretta and A. J. Robertson, Tetrahedron Lett., 2005, 46, 3641 CrossRef CAS PubMed
.
- D. J. Tao, Y. T. Wu, Z. Zhou, J. Geng, X. B. Hu and Z. B. Zhang, Ind. Eng. Chem. Res., 2011, 50, 1989 CrossRef CAS
.
- A. H. Jadhav and H. Kim, Chem. Eng. J., 2012, 200–202, 264 CrossRef CAS PubMed
.
- Y. Yang and Y. Kou, Chem. Commun., 2004, 226 RSC
.
- F. Heym, B. J. M. Etzold, C. Kern and A. Jess, Green Chem., 2011, 13, 1453 RSC
.
- Z. J. Zhang and R. G. Reddy, in Fundamentals of Advanced Materials for Energy Conversion, ed. P. R. Taylor, D. Chandra and R. G. Bautista, Minerals, Metals & Materials Soc., Warrendale, 2002, p. 33 Search PubMed
.
- H. L. Ngo, K. LeCompte, L. Hargens and A. B. McEwen, Thermochim. Acta, 2000, 357, 97 CrossRef
.
- J. L. Anderson, R. Ding, A. Ellern and D. W. Armstrong, J. Am. Chem. Soc., 2005, 127, 593 CrossRef CAS PubMed
.
- M. B. Foreiter, H. Q. Nimal Gunaratne, P. Nockemann, K. R. Seddon and G. Srinivasan, Phys. Chem. Chem. Phys., 2014, 16, 1208 RSC
.
- M. T. Clough, C. R. Crick, J. Grasvik, P. A. Hunt, H. Niedermeyer, T. Welton and O. P. Whitaker, Chem. Sci., 2015, 6, 1101 RSC
.
| This journal is © The Royal Society of Chemistry 2015 |



