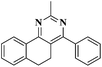
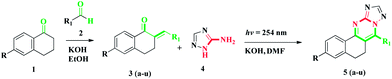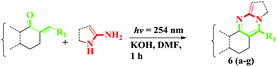UV-light induced domino type reactions: synthesis and photophysical properties of unreported nitrogen ring junction quinazolines†
Jeyakannu Palaniraja and
Selvaraj Mohana Roopan*
Chemistry of Heterocycles & Natural Product Research Laboratory, Organic Chemistry Division, School of Advanced Sciences, VIT University, Vellore, Tamil Nadu – 632014, India. Web: www.mohanaroopan.comE-mail: mohanaroopan.s@gmail.com; mohanaroopan.s@vit.ac.in; Fax: +91-416-224-3092; Tel: +91-416-220-2352
First published on 1st April 2015
Abstract
An expedient method for the synthesis of 5,6-dihydrobenzo[h][1,2,4]triazolo[5,1-b]quinazolines by UV light has been developed. Our aim was to synthesize various α, β-unsaturated carbonyl compounds and to further react them with different amines in DMF, in the presence of potassium hydroxide as base, which leads to cyclization and the aromatized products in a single step using UV irradiation at 254 nm. We have performed the reaction with various other bases and solvents that lead to the desired product with a lower yield. The synthesized ring junction compounds have been characterized by suitable spectroscopic techniques. The fluorescence emission spectra of the synthesized compounds were recorded in DMF.
Introduction
Quinazoline derivatives are nitrogen-containing heterocyclic compounds which have a universal impact due to their biological and pharmaceutical activities.1 Researchers have already determined many therapeutic activities of quinazoline derivatives, including anti-cancer,2–4 anti-inflammation,5,6 anti-bacterial,7–9 analgesia,10 anti-virus,11 anti-cytotoxin,12 anti-spasmodic,13 anti-tuberculosis,14 anti-oxidation,15 antimalarial,16 anti-hypertension,17 anti-obesity,18 anti-psychotic,19 anti-diabetes,20etc., along with the quinazoline core motif. We have also found several triazolo pyrimidine core structures with highly important medical activities for neurological disorders, anti-cancer, anti-bacterial, anti-viral cases, and also with cytotoxic activities. Over past decades, researchers have been focusing on fused heterocyclic compounds with a nitrogen ring junction due to their biologically potent properties.21–23 Unfortunately, the majority of ring junction systems do not occur naturally, but they have more importance than just a theoretical viewpoint. Replacement of a carbon atom and its attached hydrogen atoms by heteroatoms like nitrogen, sulphur or oxygen, either in five or six-membered rings, leads to a wide variety of heterocycles. Their pharmacological activities also vary hugely. Some of the ring junction nitrogen analogues have been used as dyes as well. Recent literature has been enriched with an overview on the syntheses of various heterocyclic compounds using non-conventional energy sources like microwave irradiation, ultraviolet light,24,25 ultrasonic waves, etc. To bring novelty to the existing work, our interest was mainly to focus on the synthesis of triazoloquinazoline ring junction nitrogen compounds using UV-irradiation via a domino type reaction.26 The cascade reaction or domino reaction or tandem reaction has drawn special focus in organic transformations. Despite their biological activity, there are limited reports of fluorescence studies of triazoloquinazoline fused systems. This gap was identified by our research group and so we planned the synthesis of a series of triazoloquinazolines and to subject them to fluorescence studies.Results and discussion
We have also reported a series of short reviews on arylation via transition metal free conditions.27 The background literature revealed that Scheme 1 was closely related to our present research experiments. Drizin et al. (2002), synthesized dihydropyrolopyrimidine fused rings by a one pot methodology with excellent yields.28 The reactions were carried out in the absence of a catalyst. In 2005, Shikhaliev et al. demonstrated the neat reaction to synthesise dihydrotriazolo pyrimidine derivatives with excellent yields.29In 2008, Majid et al. synthesised dihydrotriazolo pyrimidine derivatives via a one pot multi component methodology using a tungsten catalyst.30
Desenko et al., in 1991, synthesized a set of tetrahydro triazoloquinazolines from chalcones and 2H-[1,2,4]triazol-3-ylamine in DMF.31 Many literature reports record either the synthesis of ring junction nitrogen compounds without aromatization or two step procedures were necessary to get the aromatized ring junction nitrogen compound.32–34 To aromatize the compound, DDQ35/toluene, PhCl/p-chloranil36 or CAN/acetone37 were used. However, these methodologies require a longer time for the completion of the reaction and provide a lower yield. These gaps prompted us towards the synthesis of a couple of 5,6-dihydrobenzo[h][1,2,4]triazolo[5,1-b]quinazolines.
Our research methodology involves the reaction between a series of α,β-unsaturated carbonyl compounds and 2H-[1,2,4]triazol-3-ylamine in the presence of potassium hydroxide as a base in DMF under UV-irradiation (Scheme 1). The reaction mixture was irradiated for 3 h at 254 nm and led to the cyclization, followed by the aromatized products, in a single step with good to excellent yields. We have synthesised an array of 5,6-dihydrobenzo[h][1,2,4]triazolo[5,1-b]quinazoline derivatives.
The synthesized triazoloquinazolines were characterized by spectral analyses. The synthesised compounds were subjected to fluorescence studies and the quantum yields of fluorescent-active compounds were calculated. During fine tuning of the reaction conditions, no product conversion was observed with ethanol or with neat conditions (entry 1 and 2, Table 1). A moderate yield was observed when using strong bases like tBuOK, NaOH and KOH.
| Entry | Base | Solvent | Temp. (°C) | Source | Time (h) | Yieldb (%) |
|---|---|---|---|---|---|---|
| a Where Δ = conventional heating, MW = microwave, UV = ultraviolet irradiation, US = ultra sonication, NR = no reaction and RT = room temperature. The optimal conditions are shown by bold letters. b Isolated yield. | ||||||
| 1 | — | — | 100 | Δ | 16 | NR |
| 2 | — | EtOH | 80 | Δ | 16 | NR |
| 3 | Triethylamine | EtOH | 80 | Δ | 16 | NR |
| 4 | Piperidine | EtOH | 80 | Δ | 16 | NR |
| 5 | NaOMe | MeOH | 80 | Δ | 16 | Trace |
| 6 | NaOEt | EtOH | 80 | Δ | 16 | 75 |
| 7 | NaOtBu | t-BuOH | 100 | Δ | 16 | 40 |
| 8 | KOtBu | t-BuOH | 100 | Δ | 16 | 30 |
| 9 | NaOH | EtOH | 80 | Δ | 16 | 50 |
| 10 | KOH | EtOH | 80 | Δ | 16 | 56 |
| 11 | KOH | DMSO | 80 | Δ | 3 | 45 |
| 12 | KOH | DMSO | 80 | Δ | 5 | 25 |
| 13 | KOH | THF | 80 | Δ | 3 | 16 |
| 14 | KOH | ACN | 80 | Δ | 3 | 16 |
| 15 | KOH | DMF | 80 | Δ | 3 | 87 |
| 16 | KOH | DMF | 60 | Δ | 5 | 67 |
| 17 | KOH | DMF | 100 | Δ | 3 | 50 |
| 18 | KOH | DMF | 80 | MW (100 W) | 0.5 | 60 |
| 19 | KOH | DMF | RT | US | 3 | 55 |
| 20 | KOH | DMF | RT | UV (254 nm) | 1 | 92 |
| 21 | KOH | DMF | RT | UV (312 nm) | 3 | 67 |
| 22 | KOH | DMF | RT | UV (365 nm) | 3 | 55 |
After performing the reaction under different conditions, we found that the KOH/DMF combination with 1 h at room temperature in the presence of UV irradiation at 254 nm provided the best yield (entry 21). The reaction time also played an important role in increasing the yield of the product (Table 1). With optimized conditions in hand, the scope of the methodology was examined. We synthesized 22 α,β-unsaturated carbonyl compounds and known compounds were confirmed by their melting points and 1H NMR data matching with reported data. The compounds 5a and 5n formed with 92% product (isolated yield). From the above result, the substitution on the α,β-unsaturated carbonyl compounds were not playing any important role in the product formation. The synthesized α,β-unsaturated carbonyl compounds were reacted with 2H-[1,2,4]triazol-3-ylamine in the presence of KOH/DMF to afford the product with good to excellent yield (Table 2).
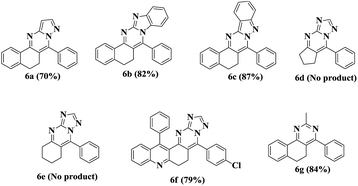
On the basis of the above fine-tuned reaction conditions, we utilized the same methodology for different amines and α,β-unsaturated carbonyls to provide compounds with fair to good yields. The synthesized compounds 2-benzylidene cyclohexanone 6e and 2-benzylidene cyclopentanone 6d failed to afford the required products under the same conditions (Table 3).
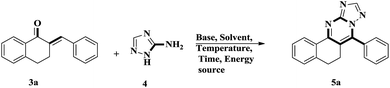
The compound 5a was also synthesized by a one pot multi component reaction. The conditions involved equimolar amounts of the three reactants with a KOH/DMF combination under UV irradiation to afford the product with an isolated yield of 20% (Scheme 2). In the multistep reaction, the compounds 3a and 5a were synthesized with 94% yield38 and 92% isolated yield, respectively. Compared with the above conditions, the Multi Component Reaction (MCR) gave 20% product (isolated yield).
 | ||
| Scheme 2 Synthesis of 7-phenyl-5,6-dihydrobenzo[h][1,2,4]triazolo[5,1-b]quinazoline via a multi component one pot methodology. | ||
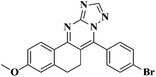
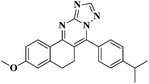
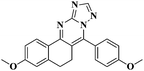
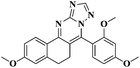
The fluorescence emission spectra of the synthesized compounds 5a–v were recorded in DMF (10−5 M). Among these compounds 5d, 5e, 5h, 5k, 5l, 5m, 5o, 5q, 5u and 5v showed fluorescence properties (Fig. 1).
We calculated the quantum yield (Φ) of the fluorescence active compounds. The Φ was calculated using the formula,
| Φ = (ΦR × IS × ODR × ηs)/(IR × ODS × ηR) |
| Entry | λ max (abs, nm) | λ max (em, nm) | Stokes shift (nm) | OD | I | Φ |
|---|---|---|---|---|---|---|
| a Sh = shoulder; abs = absorbance; em = emission; OD = excited absorbance; I = integral area; Φ = quantum yield. | ||||||
| Tryptophan39 | 280 | 355 | 75 | 0.384 | 158![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 517 517 |
0.130 |
| 5d | 271 | 392 | 121 | 0.823 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 887 887 |
0.006 |
| 253 (Sh) | 392 | 139 | 0.671 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 887 887 |
0.008 | |
| 5e | 259 | 365 | 106 | 0.948 | 9773 | 0.003 |
| 277 (Sh) | 365 | 88 | 0.547 | 9773 | 0.006 | |
| 5h | 265 | 415 | 150 | 1.137 | 9029 | 0.002 |
| 275 (Sh) | 415 | 140 | 0.712 | 9029 | 0.004 | |
| 5k | 264 (Sh) | 391 | 127 | 1.117 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 363 363 |
0.003 |
| 277 | 391 | 114 | 0.724 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 363 363 |
0.005 | |
| 5l | 276 (Sh) | 386 | 110 | 0.679 | 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 578 578 |
0.009 |
| 264 | 386 | 122 | 0.430 | 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 578 578 |
0.014 | |
| 5m | 274 (Sh) | 423 | 149 | 0.592 | 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 844 844 |
0.010 |
| 261 | 423 | 162 | 0.302 | 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 844 844 |
0.020 | |
| 5o | 266 | 430 | 164 | 0.745 | 9546 | 0.004 |
| 5q | 267 | 428 | 161 | 0.983 | 37![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 482 482 |
0.013 |
| 5u | 270 (Sh) | 450 | 180 | 0.707 | 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 747 747 |
0.008 |
| 246 | 450 | 204 | 0.638 | 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 747 747 |
0.010 | |
| 5v | 260 | 384 | 124 | 1.178 | 5181 | 0.001 |
| 251 (Sh) | 384 | 133 | 0.575 | 5181 | 0.002 | |
Conclusions
In conclusion, we have developed an efficient and easy protocol for the synthesis of triazoloquinazoline fused ring systems through a domino type transformation. Moreover, this method offers shorter reaction times, remarkable yields and transition metal free reaction conditions. Some of the derivatives showed fluorescence activity and we have calculated the quantum yield for these fluorescence active compounds. In the future, we plan to investigate the biological activities of the synthesized compounds.Experimental section
All commercially available reagents were used without further purification and the reactions were monitored by TLC. The 1H and 13C NMR spectra were obtained using a Bruker Avance 400 MHz spectrometer in CDCl3 solvent with TMS as an internal standard. Chemical shift values (δ) were expressed in parts per million (ppm). Abbreviations are as follows: s, singlet; d, doublet; t, triplet; m, multiplet. The melting points were measured on an Elchem Microprocessor based DT apparatus using open capillary tubes and are uncorrected. The mass spectra were obtained by high resolution mass spectrometry. The UV-Visible spectra were obtained on a UV-2550, Shimadzu Corporation, Kyoto, Japan. The fluorescence spectra were obtained on a Hitachi F-7000 FL spectrophotometer. Column chromatography was performed using 60–120 mesh silica gel. UV-irradiation was carried out in a Heber multi-wavelength multi-lamp photo reactor (Model HML-LP88).General procedure for the synthesis of 5,6-dihydrobenzo[h][1,2,4]triazolo[5,1-b]quinazolines [5a–i & 6a–f]
A mixture of the α,β-unsaturated carbonyl compound (1 mmol) and amine (1 mmol) were mixed in a 50 mL quartz UV reaction vial containing 10 mL of dimethylformamide and potassium hydroxide (1.2 mmol) was added at room temperature. The mixture was irradiated under UV at 254 nm (8 lamps) with constant stirring. The progress of the reaction was monitored by TLC. After the completion of the reaction, the reaction mixture was poured into crushed ice and the solid was filtered. The solid was dissolved in EtOAc and mixed with water and the organic layer was separated. The separated organic layer was dried over sodium sulphate and the solvent was evaporated. The crude product was further purified by column chromatography to afford the product as a solid.Characterization data for the compounds [5a–i & 6a–f]
Acknowledgements
SMR thanks DST-SERB (no. SB/FT/CS-126/2012), Government of India, New Delhi for providing the research grant. JP wishes to express their gratitude to DST for providing a Research Assistant Position. Furthermore, we thank the VIT management for providing the research facility, and thanks to VIT-SIF and DST-FIST for providing NMR facilities to carry out this work.Notes and references
- (a) S. M. Roopan, T. Maiyalagan and F. N. Khan, Can. J. Chem., 2008, 86, 1019–1025 CrossRef CAS; (b) A. Bharathi, S. M. Roopan, A. Kajbafvala, R. D. Padmaja, M. S. Darsana and G. Nandhini Kumari, Chin. Chem. Lett., 2014, 25, 324–326 CrossRef CAS; (c) C. Chaudhari, S. M. A. Hakim Siddiki, M. Tamura and K.-I. Shimizu, RSC Adv., 2014, 4, 53374–53379 RSC; (d) S. Mohana Roopan, Indian J. Heterocycl. Chem., 2008, 18, 183–184 Search PubMed.
- S. T. Al-Rashood, I. A. Aboldahab, M. N. Nagi, L. A. Abouzeid, A. A. Abdel-Aziz, S. G. Abdel-Hamide, K. M. Youssef, A. M. Al-Obaid and H. I. El-Subbagh, Bioorg. Med. Chem., 2006, 14, 8608–8621 CrossRef CAS PubMed.
- N. Vasdev, P. N. Dorff, A. R. Gibbs, E. Nandanan, L. M. Reid, J. P. O. Neil and H. F. VanBrocklin, J. Labelled Compd. Radiopharm., 2005, 48, 109–115 CrossRef CAS.
- A. E. Wakeling, S. P. Guy, J. R. Woodburn, S. E. Ashton, B. J. Curry, A. J. Barker and K. H. Gibson, Cancer Res., 2002, 62, 5749–5754 CAS.
- V. Alagarsamy, V. R. Solomon and K. Dhanabal, Bioorg. Med. Chem., 2007, 15, 235–241 CrossRef CAS PubMed.
- A. Baba, N. Kawamura, H. Makino, Y. Ohta, S. Taketomi and T. Sohda, J. Med. Chem., 1996, 39, 5176–5182 CrossRef CAS PubMed.
- R. Rohini, P. M. Reddy, K. Shanker, A. Hu and V. Ravinder, Eur. J. Med. Chem., 2010, 45, 1200–1205 CrossRef CAS PubMed.
- L. Antipenko, A. Karpenko, S. Kovalenko, A. Katsev, E. Komarovska-Porokhnyavets, V. Novikov and A. Chekotilo, Chem. Pharm. Bull., 2009, 57, 580–585 CrossRef CAS PubMed.
- V. Jatav, S. Kashaw and P. Mishra, Med. Chem. Res., 2008, 17, 205–211 CrossRef.
- A. A. Aly, Chin. J. Chem., 2003, 21, 339–346 CrossRef CAS.
- H. Li, R. Huang, D. Qiu, Z. Yang, X. Liu, J. Ma and Z. Ma, Prog. Nat. Sci., 1998, 8, 359–365 CAS.
- P. M. Chandrika, T. Yakaiah, B. Narsaiah, V. Sridhar, G. Venugopal, J. V. Rao, K. P. Kumar, U. S. N. Murthy and A. R. R. Rao, Indian J. Chem., 2009, 48B, 840–847 CAS.
- P. Paneersalvam, T. Raj, P. S. M. Ishar, B. Singh, V. Sharma and B. A. Rather, Indian J. Pharm. Sci., 2010, 72, 375–378 CrossRef CAS PubMed.
- P. Nandy, M. T. Vishalakshi and A. R. Bhat, Indian J. Heterocycl. Chem., 2006, 15, 293–294 CAS.
- G. Saravanan, V. Alagarsamy and C. R. Prakash, Int. J. Pharm. Pharm. Sci., 2010, 2, 83–86 CAS.
- R. Lakhan, O. P. Singh and R. L. Singh-J, J. Indian Chem. Soc., 1987, 64, 316–318 CAS.
- H. J. Hess, T. H. Cronin and A. Scriabine, J. Med. Chem., 1968, 11, 130–136 CrossRef CAS PubMed.
- S. Sasmal, G. Balaji, H. R. K. Reddy, D. Balasubrahmanyam, G. Srinivas, S. Kyasa, P. K. Sasmal, I. Khanna, R. Talwar, J. Suresh, V. P. Jadhav, S. Muzeeb, D. Shashikumar, K. H. Reddy, V. J. Sebastian, T. M. Frimurer, O. Rist, L. Elster and T. Högberg, Bioorg. Med. Chem. Lett., 2012, 22, 3157–3162 CrossRef CAS PubMed.
- M. Alvarado, M. Barceló, L. Carro, C. F. Masaguer and E. Raviña, Chem. Biodiversity, 2006, 3, 106–117 CrossRef CAS PubMed.
- M. S. Malamas and J. Millen, J. Med. Chem., 1991, 34, 1492–1503 CrossRef CAS PubMed.
- J. Palaniraja and S. M. Roopan, RSC Adv., 2015, 5, 8640–8646 RSC.
- K. S. Kumar, P. M. Kumar, V. S. Rao, A. A. Jafar, C. L. T. Meda, R. Kapavarapu, K. V. L. Parsa and M. Pal, Org. Biomol. Chem., 2012, 10, 3098–3103 RSC.
- G. R. Reddy, T. R. Reddy, S. C. Joseph, K. S. Reddy and M. Pal, RSC Adv., 2012, 2, 3387–3395 RSC.
- Y. Shiraishi, M. Ikeda, T. Daijiro, S. Tanakab and T. Hiraia, Chem. Commun., 2011, 47, 4811–4813 RSC.
- L. Zhang, Y. Deng and F. Shi, Tetrahedron Lett., 2013, 54, 5217–5219 CrossRef CAS.
- (a) J. M. Lee, Y. Na, H. Han and S. Chang, Chem. Soc. Rev., 2004, 33, 302–312 RSC; (b) H. C. Kolb, M. S. VanNieuwenhze and K. B. Sharpless, Chem. Rev., 1994, 94, 2483–2547 CrossRef CAS; (c) J.-C. Wasilke, S. J. Obrey, R. T. Baker and G. C. Bazan, Chem. Rev., 2005, 105, 1001–1020 CrossRef CAS PubMed.
- S. M. Roopan and J. Palaniraja, Res. Chem. Intermed. DOI:10.1007/s11164-014-1880-6.
- I. Drizin, M. W. Holladay, L. Yi, H. Q. Zhang, S. Gopalakrishnan, M. Gopalakrishnan, K. L. Whiteaker, S. A. Buckner, J. P. Sullivan and W. A. Carroll, Bioorg. Med. Chem. Lett., 2002, 12, 1481–1484 CrossRef CAS PubMed.
- K. S. Shikhaliev, D. V. Krylskii, A. Y. Potapov and M. Y. Krysin, Russ. Chem. Bull., 2005, 54, 2903–2904 CrossRef CAS.
- M. M. Heravi, L. Ranjbar, F. Derikvand, B. Alimadadi, H. A. Qskooie and F. F. Bamoharram, Mol. Diversity, 2008, 12, 181–185 CrossRef CAS PubMed.
- S. M. Desenko, H. Estrada, V. D. Orloy and O. A. Ponomarey, Khim. Geterotsikl. Soedin., 1991, 1, 105–108 Search PubMed.
- I. E. Varshalomidze, A. G. Golikov and A. P. Krivenko, Chem. Heterocycl. Compd., 2009, 45, 1014–1016 CrossRef CAS.
- V. V. Lipson, S. M. Desenko, M. G. Shirobokova and V. V. Borodina, Chem. Heterocycl. Compd., 2003, 39, 1213–1217 CrossRef CAS.
- B. G. Jadhav and S. D. Samant, Russ. J. Org. Chem., 2014, 50, 1301–1305 CrossRef CAS.
- A. Shaabani, E. Farhangi and S. Shaabani, Iran. J. Chem. Chem. Eng., 2013, 32, 3–10 CAS.
- A.-F. E. Mourad, A. A. Aly, H. H. Farag and E. A. Beshr, Beilstein J. Org. Chem., 2007, 3, 1–5 Search PubMed.
- M. Boutros, R.-P. Maskey, C. Koch, F. Fuchs, S. Steinbrink and D. A. Gilbert, PCT Int. Appl., 06 Oct 2011, 2011121096 Search PubMed.
- F. Yu, D. Xiao, C. Tao, C. Lili, L. Fang, J. Xu, L. Xiaomin, S. Xu, C. Kaixian, J. Hualiang, W. Hui, L. Hong and L. Dongxiang, J. Med. Chem., 2010, 53, 3465–3479 CrossRef PubMed.
- H. Adam and L. W. David, J. Phys. Chem., 1970, 74, 4473–4480 CrossRef.
- P. Grewal, M. Mallaney, K. Lau and A. Sreedhara, Mol. Pharmaceutics, 2014, 11, 1259–1272 CrossRef CAS PubMed.
- S. Sujitraj, B. Aurélie, H. Christian, V. Boris, A. Ally and L. Winfried, Photochem. Photobiol. Sci., 2013, 12, 1074–1078 CrossRef PubMed.
- L. S. Andrzej, S. Dorit and D. Wolfgang, J. Phys. Chem. A, 2009, 113, 542–550 CrossRef PubMed.
- P. L. Muiño and P. R. Callis, J. Phys. Chem. B, 2009, 113, 2572–2577 CrossRef PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5ra00229j |
| This journal is © The Royal Society of Chemistry 2015 |