Potato starch: binder and pore former in nanoframes of nanolayered oxides for Pb2+ and Ni2+ as pollutants in water and industrial sludge applications
M. A. Aguilar González*a,
A. A. Zaldívar Cadenab,
C. N. Aguilarc,
E. M. Múzquizd and
F. Equihuae
aCenter for Research and Advanced Studies of the National Polytechnic Institute (CINVESTAV-IPN), Avenida Industria Metalúrgica # 1062 Parque Industrial Saltillo-Ramos Arizpe, Ramos Arizpe, Coahuila, Mexico C.P. 25900. E-mail: mgzlz@hotmail.com; miguel.aguilar@cinvestav.edu.mx
bInstitute of Civil Engineering, Universidad Autónoma de Nuevo León, Av. Fidel Velázquez y Av. Universidad S/N Cd. Universitaria, San Nicolás de los Garza, Nuevo León 66451, Mexico
cDepartment of Food Science and Technology, School of Chemistry, Universidad Autónoma de Coahuila, Unidad Saltillo, 25 000, Saltillo, Coahuila, Mexico
dDepartment of Ceramic Materials, School of Chemistry, Universidad Autónoma de Coahuila, Unidad Saltillo, 25 280, Saltillo, Coahuila, Mexico
eFaculty of Mechanical and Electronic Engineering, Universidad Autónoma de Coahuila, Av. Barranquilla S/N, Colonia Guadalupe., Monclova, Coahuila, Mexico C.P. 25750
First published on 13th March 2015
Abstract
This article deals with the importance of potato starch in two different applications, as a pore former and also as a binder in microporous/mesoporous ceramic adsorbents for the removal of toxic metal pollutants in water. These porous ceramic adsorbents have unique and high adsorption and ion exchange properties in addition to an intricate structure, and were obtained by mixing 50% potassium polytitanates (PPTs) and 50% potato starch (PS). Cylindrical pieces were obtained through a typical extrusion process. Additionally, a heat treatment in different stages was applied to these cylinders and later they were crushed to 2–5 mm size. The physicochemical behavior of the adsorbents was assessed before and after the sintering process using different techniques: thermal analysis, chemical analysis, optical microscopy, SEM, TEM, XRD, BET, mercury porosimetry, determination of mechanical properties and physicochemical methods. Amounts of PS of 5–50 weight percent were applied with PPT in order to produce adsorbents with a properly configured structure. The mechanical properties and results of adsorption tests showed that the adsorbents removed cations of lead and nickel from the solution excellently. PPTs have technological and academic importance attributed to the fact that they have not yet been studied in linked form. It is possible to reduce diseases in people across the five continents by treatment of polluted water using these materials, and they also provide an opportunity for usage of potato waste.
1. Introduction
Potato starch has been well recognized as a binder and pore forming agent in ceramic microstructures.1 Some studies have established typical and modern processing methods for ceramic materials bonded by starch.2 Ceramic porous materials play a very important role in the field of new advanced materials with functional properties. The application of PS (Potato Starch) in industrial processes is very extensive and diverse, including food,3 paper,4 diesel particle filters,5 porous piezoelectric ceramics,6 hydrogel composites,7 anodes in fuel cells,8 and clinical applications.9Porous ceramic bodies formed with advanced nanomaterials of titanates have great potential for many applications; however, they have not been utilized either in the immobilization of toxic pollutants in water nor in metal coating sludge. In both cases, it seems as if having a porous support with an activated surface for increased adsorption can enhance the efficiency of the applied material.
New potassium polytitanate (PPT) based ceramic oxides of potassium have shown major and essential importance in the field of research due to their unique structures and properties, such as their thermal, chemical, catalytic, mechanical, non-frictional, refractory, optical, adsorbent, biomimetic and ion exchange properties.
In the last decade, several routes have been proposed for the synthesis of potassium titanates, among which can be found chemical, thermal and hydrothermal syntheses, and modification of titanium precursors by reactions in the solid or liquid state.10
Recently, the development of a new type of ceramic nanomaterial called “potassium polytitanates” (PPTs), obtained from molten salts using a short heat treatment route, was reported.11 PPTs are inexpensive and easily handled in ceramic technology. Also, they do not pose health risks in handling due to their laminar morphology. Treatment at temperatures from 900 to 1300 °C allows the formation and recrystallization of different potassium titanates, depending on their synthesis.
Molten salt processes are regarded as highly important to chemical technology. The main applications are metallic alloy production,12 batteries,13 fuel cells,14 catalysis,15 photocatalysts16 and solar energy.17 Molten salts are studied for different purposes through many different techniques. The chemical and thermodynamic considerations of binary or ternary systems in solutions of molten salts have been very well established.18
Nowadays PPTs are considered as a kind of intelligent ceramic material19 due to their ion exchange, intercalation and adsorption properties in radionuclide and radioactive materials, preventing damage to human health and the environment. It is important to consider that there are some reports of other modified ceramic materials related to ion exchange and metal removal properties.20–24
Potassium titanates are ceramic systems with very similar chemical compositions, but their properties are very different, especially when the process of obtaining them involves heat treatments. The extrusion method was chosen for this research because it is a process that provides directionality to fine powders of laminar nature and/or irregular shape (such as PPT). Besides this, the advantages of extrusion include the ability to add sintering aids, low shrinkage and the elimination of flaws by colloidal processing of the paste. In this research we apply conventional extrusion, which is the best method for granular production, is straightforward and is inexpensive.
There is no evidence in the literature of titanates being bonded to starch to study their adsorption or catalysis applications. The difficulty in linking titanates arises mainly through the manipulation of layered powders on a micro- or nanometric scale.
The innovation of our work suggests the handling of starch granules in ceramic pastes at a strict temperature of 50 °C. This treatment gives the maximum plasticity in order to form bodies in the green state (before sintering) and saves the application of chemical or organic binders (an environmental contribution). It is well-known that the degree of swellability will depend on uniformity in porosity. There are others important methods for making porous ceramics with starch and water, such as consolidation (slip casting), gel casting or tape casting.
In this paper, we present important evidence for the application of PS as a pore former and as a binder in microporous/mesoporous ceramic adsorbents for the removal of toxic metallic pollutants in water. Porous ceramics based on PPT linked to PS, extruded conventionally and heat treated, were obtained and evaluated.
2. Experimental procedure
2.1 PPT preparation (as precursor materials)
PPTs were prepared through molten salt synthesis. Analytical reagents (wt% = 82 (KNO3), 8 (KOH) and 10 (TiO2), all with 99.7% purity) were used to synthesize quasi-crystalline PPT according to previous work.11 The obtained molten salts were heat treated at 500 °C. Afterwards, dried TiO2 powder (average particle size = 0.4 μm) was added to the salts in a stainless steel crucible and reacted for 1 hour in order to obtain PPT as a precursor material. The molten material was cooled to room temperature and then crushed. The ground material was thoroughly washed with distilled water and separated with a filter paper (no. 42). Powders were dried at 90 °C for 2 hours.2.2 Preparation of adsorbents
Commercial grade PS (amylose–27 amylopectin, 73 wt%) was obtained from Sabritas® S.A de C.V (Saltillo Coahuila, México). PS was chosen according to previous research of 5 different starches for ceramic applications25 and due to its negligible ash content after heat treatment (at least from the standpoint of materials science) compared with others organics. Potato starch was applied in natural form. The method for synthesis of the adsorbents is presented in Fig. 1. It was reported that this route can yield the best volumetric expansion of the starch.262.3 Reagents and solutions
Lead nitrate and nickel nitrate (Aldrich, 99% of purity) were used for adsorption tests in aqueous solutions at a concentration of 155 mg L−1 in distilled water and at pH values of 5.6 and 7.5 for lead and nickel, respectively. These solutions were used directly in adsorption tests. Both solutions were used to investigate the amount of Pb(II) or Ni(II) removed from solutions as industrial waste.272.4 Characterization
The density of the adsorbent materials was determined using the Archimedes principle, using toluene of 99.5% purity. Measurements were taken at 25 °C on an analytical balance (Ohaus, Explorer) according to ASTM C373-88 (2006). A BET surface area of 13.4 m2 g−1 and an average particle size of 35 microns for PPT were found for the starting materials. The determination of Brunauer–Emmet–Teller (BET) surface area and analysis of micro-pores (MP) were carried out in a Quantachrome Autosorb 1C sortometer from the adsorption/desorption of a monolayer of nitrogen. The average grain size for PS was 64 μm, determined through the Laser Coulter Technique. SEM analyses were carried out using a Philips XL30 scanning electron microscope for ESEM (Environmental Scanning Electron Microscopy), equipped with EDS (Pegasus, operated at 20 kV acceleration voltage). The identification of crystalline phases was performed by X-ray diffractometry in a Philips X’Pert X-ray diffractometer using Kα(Cu) radiation and a secondary monochromator (Ni filtering) at 40 kV and 30 mA. Thermal analysis was performed on a Perkin Elmer analyzer, model 1700. 10 mg of each sample was weighed and heated at a rate of 3 °C min−1 during the overall analysis in the thermobalance.Using two different methods, the porosity of the samples was evaluated: (a) the novel ceramographic method, using the software Image Pro Plus 5.1 (Media Cybernetics) for SEM images (BSE), and (b) the mercury intrusion porosimetry method. In the former, epoxy resin, in the presence of a vacuum, was introduced into the samples according to the ASTM E-3 standard. Then, the metallographic surfaces were prepared and coated with gold. The porosity evaluation was performed with 20 different fields. In the latter method, the samples were directly applied to the determination. The mechanical properties of the porous ceramics were measured through the compressive strength with a hydraulic machine (Controls, Sercom 7) using a cell of 15 kN and a loading rate of 100 N s−1. 6 cylindrical samples were tested in each experiment.
Fact-Sage® software was used to calculate crystalline phases and possible transformations.
2.5 Adsorption test
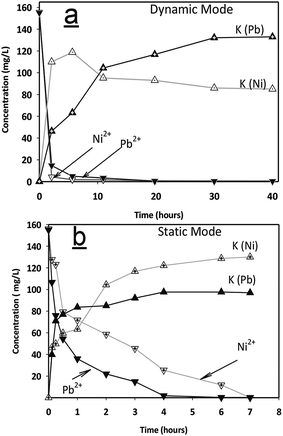
In the static experiments, 2 grams of adsorbents were placed in 200 ml solutions. Different contact times (0, 7, 15, 30, 60, 120, 180, 240, 360 and 420 min) were considered for static adsorption tests.
For the dynamic mode, a special pyrex glass device was used to feed fluid into a Pyrex® glass column (diameter = 25 mm) with the capacity of 500 cm3 at 23 ± 2 °C. Adsorbent pellets (165 g) occupying a third of the glass column were used during this experiment. The flow rate of the solution was kept constant at 140 ml h−1 for 40 hours.
The adsorption kinetics of Pb(II) and Ni(II) in their aqueous solutions were determined using an atomic emission spectrometer (ICP Thermojarrel Thermoelemental Ash, Iris Intrepid II model). The spectrometer was calibrated with NIST certified standards.
The metal concentration retained in the adsorbent phase (qe, mg g−1) was calculated as follows:
| qe = ((Co − Ce)/V)/m, | (1) |
3. Results and discussion
3.1 Thermal behavior
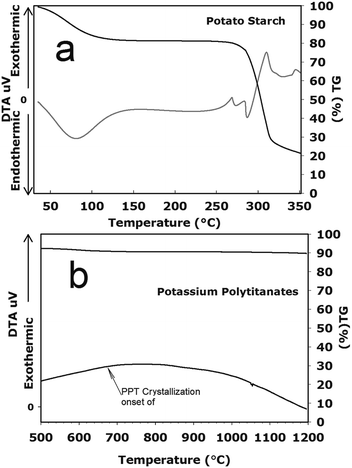
The chemical processes, properties and types of PS have been investigated in ceramic applications in order to understand the structural and crystalline arrangements of each component of PS28,29 and therefore the thermal behavior. It is well known that PS granules are insoluble in water below 50 °C; in our work, a gelling temperature range from 55 and up to 80 °C was present, as can be seen in Fig. 2(a) for the PS raw material. During the gelation stage, water absorption was limited due to the swelling of the starch particles, which increased their size several times by an irreversible process.30We found that the gelatinization temperature for the PS granules mixed with PPT and water was about 65 °C in a 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 w% relationship. While heating, the wet pastes began to swell, forming a highly viscous white paste.
50 w% relationship. While heating, the wet pastes began to swell, forming a highly viscous white paste.
In the first stage, the extruded samples were dried at 65 °C for two hours, which was enough to allow swelling up of the starch granules. In the temperature range of 25 to 140 °C, PS showed two outcomes: (1) a loss in weight related to the evaporation of adsorbed water and (2) the endothermic effect at a more intense heat of 80 °C. This process corresponds to volumetric expansion of the starch and hence results in a gelation stage.31 After that, the temperature was increased to 180 °C in order to evaporate water completely.
When the temperature was increased from 275 to 350 °C, the starch granules showed a weight loss of about 60%, which corresponds to thermal decomposition.32 Then, to totally ensure the decomposition of PS in the extruded bodies, a second stage of heat treatment at 500 °C was carried out. While burning, the starch granules were calcinated in pyrodextrinization reactions30 in order to make hollows in the ceramic bodies.
Fig. 2(a) shows the representative graphs of the transformation of the potato starch, in the process of swelling as it comes into contact with water at a temperature range between 25 and 350 °C; a similar behavior was studied by Laurentin and co-workers.30 Furthermore, in PPT, adsorbed water (removed from 350 to 550 °C) and structural water (removed from 500 to 700 °C) were detected (Fig. 2(b)). The thermogram, TG, in Fig. 2(b) shows a partial weight loss of the sample during the analysis of 9.8 (w%) at 714 °C, generated by the decomposition of Ti–OH groups,33 which corresponds to the removal of structural water. In the final stage, a small endothermic change was detected at 1050 °C, due to the recrystallization and structural transformation that occurred during the Ti phase formation.
Additionally, regarding the sintering process, one can say that the final products from starch will always be CO2 and H2O, as long as this is all performed in an oxidizing atmosphere. Of course, under reducing conditions, residual carbon may occur. The PS breakdown begins to be detected below 300 °C.30
3.2 Morphology, microstructure and phase composition
Moreover, the PPT were composed of aggregates of micrometric size, which have a composition of lamellar particles (plaques) with dimensions from 400 to 1000 nm and a plate thickness smaller than 100 nm (Fig. 3(b)).
The advantage of a granular material is that it allows for impurities or other pollutants in a fluid to be reversibly separated. The porosity was open and different morphologies were observed for PPT before and after sintering. By applying a granular adsorbent, agglomeration of the adsorbed particles at the bottom of the adsorption container was prevented; this was also reviewed through SEM.
The adsorbent material developed a network of pores (average diameter of 35 microns). The spaces left by PS in the sintered structures of PPT favor an increase in the interlaminar spacing of the crystalline structure of 2.3 nm (Fig. 5(b)) where a significant amount of ion exchange had apparently occurred. Fig. 5(a) shows agglomerates of irregular morphology, such as K2Ti4O9 (short fibers) and K2Ti6O13 (needles) in powders synthesized at 500 °C with agglomerates of different sizes and morphologies.
3.3 XRD
 | ||
| Fig. 6 XRD patterns from: (a) PPT synthesized at 500 °C, (b) potato starch, (c) extruded sample of 50 SP–50 PPT (w%) in green condition and (d) 50 SP–50 PPT (w%) at sintered stage. | ||
The estimation of the anatase content in the produced PPT was similar to those realized by means of quantitative X-ray analysis.35 The treatment time allows the anatase to fully transform into PPT. A survey of transformation of TiO2 was realized by Wallenberg and co-workers.39
With all aforementioned data, it was determined that the PS was not involved in the process of recrystallization of PPT.
3.4 Porosity
Transport pores were promoted naturally through the extrusion and their presence was confirmed at the sintering stage. This favored the open porosity.A portion of the generated porosity is considered to be a consequence of the recrystallization between acicular and tangled microstructures. When the ceramic bodies were formed, they generated spaces (internally), which allowed the growth and recrystallization of PPT and thus a structural rearrangement of potassium hexa-titanate, always in the presence of heat. Initially, they had an irregular shape (Fig. 3(b)), which was converted to a fibrous form (Fig. 7(f)). Through mercury porosimetry it was observed that the starting adsorbent contained transport pores of 0.4 microns (35% of total porosity), macropores in a range of 0.05 to 0.4 microns in diameter as well as mesopores with diameters smaller than 0.05 microns. Similar results were obtained by Ohta and Fujiki.37 This means that there is no risk for health in the manipulation of the fibers from the extrusion process (homogeneous and fluid pastes). The evaluation of total porosity in the adsorbents was 63.7% (optical) and 67% (SEM).
 | ||
| Fig. 7 SEM photomicrographs of samples of adsorbents with different PS contents: (a) 0%, (b) 10%, (c) 25%, (d) 45, (e) 50%, and (f) same as that of (e) at 2500×. | ||
Research on the factors that affect the morphology, such as the diameter, length and synthesis rate of potassium titanates, was carried out by Zaremba et al.38 However, the most important physical aspect of PPTs is that they allow the retention of pollutants by interactions in their interlayer spaces.
3.5 Specific surface area
The specific surface area values are frequently altered for various reasons, mainly due to the content of titanium.39 PPT precursors showed an average value of 13.9 m2 g−1 when analyzed through nitrogen adsorption, which coincides with the research reported by Wallenberg and co-workers.39 The BET values of the PPTs were lower than those of commercial adsorbents.Morphological characteristics, such as a non-porous particulate structure and an accordion-like shape, can affect the exposed surface area of such materials. The PPT–PS ceramic adsorbents (sintered) achieved an average value of specific surface area of 0.65 m2 g−1. The specific surface area of the sintered materials was lower than that of the synthesized ones; this can be attributed to the effects of recrystallization during heat treatment and thus presents an exchange in their morphology.39 This report allowed us to observe irregular agglomerated materials (Fig. 3(b)) such as in the raw materials, which were later converted to intricate beams with an acicular morphology as observed after the heat treatment Fig. 7(e) and (f). On the other hand, the exhausted adsorbent showed an average value of 1.2 m2 g−1.
3.6 Mechanical properties
The best mechanical properties were presented in the 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 50 PPT–PS system (3.2 of density, compared with 4.5 of PPT without starch) and were sufficient for rough handling of the materials in the green condition. PS acted as a binder and pore former in the obtained porous ceramic material, favoring a high mechanical strength in the order of 103 MPa. The mechanical strength of filters/adsorbents must have a value around 100 MPa, according to the application of each material.40
50 PPT–PS system (3.2 of density, compared with 4.5 of PPT without starch) and were sufficient for rough handling of the materials in the green condition. PS acted as a binder and pore former in the obtained porous ceramic material, favoring a high mechanical strength in the order of 103 MPa. The mechanical strength of filters/adsorbents must have a value around 100 MPa, according to the application of each material.40
Heat treatment generated an intricately interconnected structure in the adsorbent, promoted by the use of PS granules. This structure improved the mechanical properties compared to the PPT without PS (Fig. 7(a)) and also prevented the effect of clogging. The results were 14.57, 22.92, 34.62, 47.63 and 61.83 MPa for the green condition and 48.62, 59.14, 70.27, 84.99 and 103.70 MPa in the sintered samples, for 10, 20, 30 40 and 50% PS, respectively. The worst results were attributed to the system with 10% PS.
3.7 Adsorption kinetics
The initial pH of the solution for the case of nickel was set at a 7.5; this agreed with several experiments previously conducted and was also according to reports in the literature for nickel ion electrochemistry.27
The maximum amount of lead ions removed by the adsorbent was 28.8 mg g−1 at a pH value of 5.6, showing 99.94% of total effectiveness of removal. This allowed us to propose that the adsorption may have occurred by ion exchange, as explained by eqn (2) and (3).
The main reactions for metal ions may occur as follows:
| Me2+(solution) + H2O ↔ MeOH+(solution) + H+(solution) | (2) |
| MeOH+(solution) + H2O ↔ Me(OH)2(solution) + H+(solution) | (3) |
And the metal ion exchange could be as follows:
| H+(solution) + Me+(adsorbent) ↔ H+(adsorbent) + Me+(solution) | (4) |
| Me2+(solution) + K+(adsorbent) ↔ Me2+(adsorbent) + 2K+(solution) | (5) |
The adsorbents showed high effectiveness of adsorption for both Pb ions and Ni ions and after 15 minutes of contact time, concentrations were reduced to 0.1 mg dm−3 in both cases, which are allowable levels according to standard EPA 55 FR 22520, 1990, USA. During the process, potassium ions were released into the solution and simultaneously increased in accordance with the decrement of metal content. This allowed us to propose that the adsorption may have occurred by ion exchange between Me2+ (solution) and K+ cations (of the adsorbent structure).
The fibers of PPT are considered to have different and unique morphologies, since their materials underwent one crystalline transformation in a single direction.
The PPT absorbent capability for trapping hazardous cations permanently allowed us to isolate them from contaminated water. When the used adsorbents become saturated they can be confined safely without the risk of releasing the absorbed cations (through leaching effects).
The features of the adsorbents applied in this work, such as porosity, particle size, density, surface area, pore size and mechanical properties, are consistent with those in the publications of Heckmann and Wenger and Harada and co-workers.40,41 The concentration of metal ions in solution used in this research of 155 mg L−1 was selected according to previous studies for adsorption of metal ions in solution.42–44
4. Conclusions
The materials were obtained from extruded pastes, whose composition was 50% PPT, with a molar ratio of TiO2/K2O = 5.1, and 50% potato starch and distilled water (60 w% relative to the weight of solids). The materials were sintered at 1100 °C for 30 minutes and were used as ceramic adsorbents. The crystalline phases occurred in the composition of potassium titanates and had a structure similar to that of K2Ti6O13. This structure is formed during heat treatment by a chemical reaction among the PPT, favoring high mechanical strength (about 110 MPa). The adsorbent material has developed a network of transportation pores (average diameter of 35 microns). The PPTs favor increased interlayer spacing of up to 2.3 nm in the crystalline structure. As a result, the material showed a high rate of adsorption of lead and nickel through an ion exchange mechanism with the potassium incorporated into the structure of the adsorbents.Acknowledgements
The authors would like to thank the CONACyT (México) for economic support for the realization of this research and M.Sc. Enrique Diaz Barriga for the support of the TEM characterization.References
- Z. Živcová, M. Černý, W. Pabst and E. Gregorová, J. Eur. Ceram. Soc., 2009, 29, 2765–2771 CrossRef PubMed.
- M. E. Gomes, A. S. Ribeiro, P. B. Malafaya, R. L. Reis and A. Cunha, Biomaterials, 2001, 22, 883–889 CrossRef CAS.
- A. Cheyne, J. Barnes and D. I. Wilson, J. Food Eng., 2005, 66, 1–12 CrossRef PubMed.
- S. Jobling, Curr. Opin. Plant Biol., 2004, 7, 210–218 CrossRef CAS PubMed.
- T. Wolff and H. Friedrich, GE Pat. # 029481A1, 2011.
- A. N. Rybyanates, Ferroelectrics, 2011, 419, 90–96 CrossRef.
- M. R. Guilherme, R. S. Oliveira, M. R. Mauricio, T. S. Cellet, G. M. Pereira, M. H. Kunita, E. C. Muniz and A. F. Rubira, Soft Matter, 2012, 8, 6629–6637 RSC.
- A. Sanson, P. Pinasco and E. Roncari, J. Eur. Ceram. Soc., 2008, 28, 1221–1226 CrossRef CAS PubMed.
- F. Romero and M. J. Bernal, Starch/Staerke, 2011, 1–11 Search PubMed.
- T. Zaremba and D. Witkowsca, Mater. Sci.-Pol., 2010, 28, 25 CAS.
- M. A. Aguilar, A. Gorokhovsky, A. Aguilar and J. I. Escalante, Bol. Soc. Esp. Ceram. Vidrio, 2008, 47, 29–34 CrossRef.
- M. Harata, K. Yasuda, H. Yakushiji and T. H. Okabe, J. Alloys Compd., 2009, 474, 124–130 CrossRef CAS PubMed.
- A. Fukunaga, S. Sakai, C. Hiraiwa, K. Nitta, M. Majima and S. Inazawa, Molten Salt Battery, Jp. Pat. WO 2011148864 A1, December 1 2011.
- A. Hilmi, C.-Y. Yuh and M. Farooque, High performance electrolyte for molten carbonate fuel cells, US Pat. US 20120021328 A1, January 26 2012.
- Q. Wang, Z. Guo and J. S. Chung, Mater. Res. Bull., 2009, 44, 1973–1977 CrossRef CAS PubMed.
- T. Docters, J. M. Chovelon, J. M. Herrmann and J. P. Deloume, Appl. Catal., B, 2004, 50, 219–226 CrossRef CAS PubMed.
- Y. J. Park, S. E. Bae, Y.-H. Cho and K. Song, Device for On-Site Measurement of Concentration of Uranium in High Temperature Molten Salts, US Pat. US 20120026495 A1, February 2 2012.
- M. Blander, NATO Series C Mathematical and Physical Sciences, in Molten Salts Chemistry An Introduction and Selected Applications, ed. G. Mamantov and R. Marassi, D. Reidel Publishing Company, Dordrecht, Holland, 1987, vol. 1, ch. 22, pp. 17–45 Search PubMed.
- D. J. Yang, Z. F. Zheng, H. Zhu, H. W. Liu and X. P. Gao, Adv. Mater., 2008, 20, 2777–2781 CrossRef CAS PubMed.
- F. Akhtar, L. Andersson, S. Ogunwumi, N. Hedin and L. Bergström, J. Eur. Ceram. Soc., 2014, 34, 1643–1666 CrossRef CAS PubMed.
- R. Chen, Z. Zhang, Z. Lei and N. Sugiura, Desalination, 2012, 286, 56–62 CrossRef CAS PubMed.
- S. Chattopahyay, US Pat. # 0306555, 2014.
- Y. T. Fang, T. Liu, Z. C. Zhang and X. N. Gao, Renewable Energy, 2014, 63, 755–761 CrossRef CAS PubMed.
- Y. Fang, W. Yao, J. Guo and X. Gao, Int. J. Low-Carbon Technol., 2012, 1–4 Search PubMed.
- E. Gregorová, W. Pabst and I. Boacenko, J. Eur. Ceram. Soc., 2006, 26, 1301–1309 CrossRef PubMed.
- M. H. Talou, M. A. Villar and M. A. Camerucci, Ceram. Int., 2010, 36, 1017–1026 CrossRef CAS PubMed.
- J. G. Dean, F. L. Bosqui and K. H. Lanouette, Environ. Sci. Technol., 1972, 6, 518–524 CrossRef CAS.
- E. Gregorová, Z. Zivcová and W. Pabst, Starch/Staerke, 2009, 61, 495 CrossRef.
- A. Kraak, Ind. Crops Prod., 1993, 1, 107–112 CrossRef.
- A. Laurentin, M. Cardenas, J. Ruales, E. Pérez and J. Tovar, J. Agric. Food Chem., 2003, 51, 5510–5515 CrossRef CAS PubMed.
- L. Kroh, W. Jalyshko and J. Haseler, Starch/Staerke, 1996, 48, 426–433 CrossRef CAS.
- C. Mestres, F. Matencio, B. Pons, M. Yajid and G. Fliedel, Starch/Staerke, 1996, 48, 2–6 CrossRef CAS.
- A. V. Gorokhovsky, J. I. Escalante, T. Sánchez and C. A. Gutiérrez, J. Eur. Ceram. Soc., 2004, 24, 3541–3546 CrossRef CAS PubMed.
- E. Gregorová and W. Pabst, J. Eur. Ceram. Soc., 2008, 27, 669–672 CrossRef PubMed.
- T. Sanchez, A. Gorokhovsky and J. I. Escalante, J. Am. Ceram. Soc., 2008, 91, 3058–3065 CrossRef PubMed.
- Å. Rindlav-Westling, M. Stading and P. Gatenholm, Biomacromolecules, 2002, 3, 84–91 CrossRef PubMed.
- N. Ohta and Y. Fujiki, J. Ceram. Soc. Jpn., 1980, 88, 1–10 CAS.
- T. Zaremba, Mater. Sci.-Pol., 2012, 30, 180–188 CrossRef CAS PubMed.
- L. Wallenberg, M. Sanati and A. Andersson, Microsc., Microanal., Microstruct., 1990, 1, 357–364 CrossRef CAS.
- K. Heckmann and T. Wenger, Porous ceramic, US Pat. US6737376 B1, May 18 2004.
- M. Harada, T. Sasaki, Y. Ebina and M. Watanabe, J. Photochem. Photobiol., A, 2002, 148, 273–276 CrossRef CAS.
- M. Sobh, M. Moussawi, W. Rammal, A. Hijazi, H. Rammal, M. Reda, J. Toufaily and T. Hamieh, Am. J. Phytomed. Clin. Ther., 2014, 2, 1070–1080 CAS.
- T. Yin and X. Huang, Adv. Mater. Res., 2014, 955, 2747–2750 CrossRef.
- V. K. Gupta and A. Rastogi, J. Hazard. Mater., 2008, 152, 407–414 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2015 |