A green electrochemical method for the synthesis of new N,N′-diphenylbenzene-1,4-diamine derivatives†
Davood Nematollahi*,
Saeideh Mahdinia,
Peiman Karimi,
Hamid Salehzadeh and
Sajad Kaihani
Faculty of Chemistry, Bu-Ali Sina University, Hamedan, 65178-38683, Iran. E-mail: nemat@basu.ac.ir; Fax: +98-811-8257407
First published on 18th March 2015
Abstract
A green method for the synthesis of new organosulfur derivatives of N,N′-diphenylbenzene-1,4-diamine (2a–e) based on the Michael reaction of electrochemically generated N,N′-diphenyl-p-quinonediimine with 2-mercaptopyridine, 1H-1,2,4-triazole-3-thiol, 1-phenyl-1H-tetrazole-5-thiol, 2-mercaptobenzoxazole and 2-mercaptobenzothiazole as nucleophiles in a water/ethanol (25/75, v/v) mixture is described. The thioethers (2a–e) were synthesized in high yields, without toxic reagents and solvents at a carbon electrode using an environmentally friendly method with high atom economy.
Introduction
1,4-Phenylenediamines are important compounds in the manufacture of dyes, pharmaceuticals, polymers, and other industrial products.1 They are also very susceptible to oxidative polymerization via oxidation of amino groups (one or two) to give linear azopolymers, ladder polyphenazines, poly aminoanilines, and phenazine/iminoquinonoid-unit containing polymers.2 In addition, they are extensively used in the manufacture of azo dyes.3 Furthermore, they are used as antioxidant agents in various vulcanized rubbers and they provide preferable protection.4 Among these, N,N′-substituted p-phenylenediamines, represent the most important group of antioxidants5 used in rubber industry6 and reducing NOx emissions in the diesel engines.7 It is well known that antioxidants (e.g. N,N′-substituted p-phenylenediamines) interact with free radicals in various ways, including hydrogen abstraction, addition of a radical species to the antioxidant and simple electron transfer reactions.5
 | (1) |
 | (2) |
 | (3) |
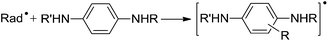
It should be noted that, in pathways eqn (1)–(3), the products are less reactive than the initial radical species. The last pathway includes electron transfer between oxidizing radical species and reducing substrate. In this context, the antioxidant activity of six N,N′-substituted p-phenylenediamines in polyisoprene rubber matrix was studied by differential scanning calorimetry and shown that antioxidant activity of these compounds is dependent on their structure.8 In addition, some other studies indicate that in the mechanism of antioxidant action of N,N′-substituted p-phenylenediamines instead of the classical reaction pathway leading to the N,N′-substituted p-quinonediimines.9 Their studies indicated that the structure of the individual N,N′-substituted p-phenylenediamines, is a major determinant in predicting antioxidant activities of these compounds.
Despite of numerous studies concerning the oxidation of N,N′-substituted p-quinonediimines, particularly N,N′-diphenylbenzene-1,4-diamine (DPD) as a model compound in the field of antioxidant activities,4–9 synthesis of organic compounds10 and synthesis of conducting polymers,11 there is no report on the electrochemical functionalization of DPD. Following our strategy for synthesis of organic compounds under green conditions,12 we wish to study the functionalization of DPD and to describe a one-pot and straightforward protocol for the synthesis of some new organosulfur derivatives of N,N′-diphenylbenzene-1,4-diamine (2a–e). This idea prompted us to investigate the electrochemical oxidation of DPD in the presence of 2-mercaptopyridine (pyridine-2-thiol) (1a), 1H-1,2,4-triazole-3-thiol (1b), 1-phenyl-1H-tetrazole-5-thiol (1c), 2-mercaptobenzoxazole (1d) and 2-mercaptobenzothiazole (1e) (Fig. 1). This method represents a facile electrochemical process for the synthesis of some thioethers (2a–e) in high yields and purities under green conditions, without toxic reagents and solvents in a divided cell using an environmentally friendly method at a carbon electrode.
Results and discussion
Cyclic voltammograms of a solution of N,N′-diphenyl-p-phenylenediamine (DPD) in water (phosphate buffer, pH = 2.0, c = 0.2 M)/ethanol (25/75, v/v) mixture, in the absence and the presence of 2-mercaptopyridine (1a) are shown in Fig. 2. Curve a which is attributed to the electrochemical oxidation of DPD shows an anodic peak (A1) at 0.24 V and corresponding cathodic peak (C1) at 0.15 V, which corresponds to the transformation of DPD to N,N′-diphenyl-p-quinonediimine (DQD) and vice versa within a quasi-reversible two-electron process.13 A measure of reversibility is provided by the ratio of the cathodic to anodic peak current (IpC1/IpA1), which is closer to 1. This confirms the stability of the electrogenerated DQD under the experimental conditions. In other words, the side reactions, such as hydroxylation,14 dimerization15 or oxidative ring cleavage16 that involve quinones, quinoneimines or quinonedidiimines, are too slow to be observed at the time scale of cyclic voltammetry.14–16 The oxidation of DPD in the presence of 1a as a nucleophile was studied in some detail (Fig. 2, curve b). As can be seen from this figure, the cathodic peak (C1) decreases and a new anodic peak (A2) appeared in more positive potentials. Under these conditions, the ratio of the cathodic to anodic peak current (IpC1/IpA1), is less than 1. It decreases with decreasing scan rate and increasing 1a concentration (Fig. 2, inset), indicating that the product of the electron transfer (DQD) is undergoing a following chemical reaction.17 In Fig. 2, curve c is related to 2-mercaptopyridine (1a) itself. The comparison of cyclic voltammogram of 1a with curve b shows that anodic peak A2 is due to the oxidation of 1a.These studies were followed by controlled potential coulometry (cpc) of DPD (0.25 mmol) in the presence of 1a (0.25 mmol) in water (phosphate buffer, pH = 2.0, c = 0.2 M)/ethanol (25/75, v/v) mixture at the potential of peak A1. The number of transferred electrons was calculated from the charge that had accumulated when the current fell below 5% of its initial value. The calculated number of transferred electrons per molecule of DPD was 2.
Diagnostic criteria of cyclic voltammetry and controlled potential coulometry accompanied by 1H NMR, 13C NMR and MS spectra of final product allow us to propose the following mechanism for the electrochemical oxidation of DPD in the presence of 1a (Scheme 1). According to our results, the Michael addition reaction of 1a with electrogenerated DQD followed by the aromatization of the resulting intermediate to give 2a as the final product. The same results were obtained for the other sulfur compounds given in Fig. 1 (data not shown).
 | ||
| Scheme 1 Proposed mechanism for the electrochemical oxidation of DPD in in the presence of thioles 1a–e. | ||
The preparative synthesis of 2a–e was performed in potentiostatic condition by oxidation of DPD in the presence of thioles 1a–e, at 0.25 V versus SCE a carbon anode in a divided cell. More detail is described in the experimental section.
In order to the investigation of the electrochemical properties of the isolated products, the cyclic voltammetric behaviour of 2b was examined (Fig. 3, curve b). As can be seen, voltammogram exhibits a quasi-reversible system with E1/2 = 0.21 V vs. SCE. Comparing the half wave potential of cyclic voltammogram of 2b with that of DPD (E1/2 = 0.24 V vs. SCE), shows that half wave potential of product (2b) is less than DPD. This may increase the antioxidant activity of 2b in comparison with DPD.
It should be noted that, different to cyclic voltammograms of DPD in pHs ≥ 2 (Fig. 2, curve a), at pH values ≤ 1, cyclic voltammogram of DPD shows more complex behavior. In this condition the cyclic voltammograms of DPD exhibits two anodic peaks AI and AII in the positive-going scan and two corresponding cathodic peaks CI and CII in the negative-going scan (Fig. 4). The anodic peaks AI and AII correspond to two successive oxidations, the first one of DPD to radical cation DPD˙+ (or DPD˙) and the second one of DPD˙+ (or DPD˙) to N,N′-diphenyl-p-quinonediimine (DQD). The cathodic peaks CI and CII correspond respectively to the one-electron reduction of DQD to DPD˙+ (or DPD˙) and to the one-electron reduction of DPD˙+ (or DPD˙) to DPD.
 | ||
| Fig. 4 Cyclic voltammogram of DPD (1.0 mM), at a glassy carbon electrode in water (HCl 0.1 M)/ethanol (25/75, v/v) mixture. Scan rate: 10 mV s−1. T = 25 ± 1 °C. | ||
From the point of view of green chemistry, the presented method has some important advantages. High atom economy (>99%), use of electricity as energy instead of oxidative reagents, clean synthesis, use of aqueous media (75% water) instead of organic solvents, work in room temperature, one-step reaction, and pressure and technical feasibility are of preeminent green advantages.
Conclusion
To the best of our knowledge, only a few reports have appeared on the functionalization of DPD.18 On the other hand, this is the first report of synthesis DPD derivatives by electrochemical method. The results of this work show that DPD is oxidized to N,N′-diphenyl-p-quinonediimine (DQD) within a quasi-reversible two-electron process.13 The formed DQD is attacked by thioles 1a–e to form thioethers 2a–e in high yields and purities. The reaction mechanism for anodic oxidation of DPD in the presence of 1a–e is presented in Scheme 1. The present method for the synthesis of thioethers 2a–e has several advantages. (a) This process is practically convenient to carry out and can be performed in aqueous solution/ethanol mixture, at room temperature and atmospheric pressure. (b) Neither catalyst nor organic/inorganic oxidizing agents are necessary and the reaction can be performed in one-pot, under green and mild conditions. (c) The synthesized compounds (2a–e), may show efficient antioxidant activity. To conclude, while the reactions are performed on a mmol scale, there is little difficulty in producing larger quantities by using larger cells.Experimental
Cyclic voltammetry, controlled-potential coulometry and preparative electrolysis were performed using an Autolab model PGSTAT 20 potentiostat/galvanostat. The working electrode used in the voltammetry experiments was a glassy carbon disc (1.8 mm2 area) and platinum wire was used as counter electrode. The working electrode used in controlled-potential coulometry and synthesis was an assembly of four carbon rods (31 cm2) and large platinum gauze constitute the counter electrode. The working electrode potentials were measured versus SCE (all electrodes from AZAR electrode). N,N′-Diphenylbenzene-1,4-diamine, 2-mercaptopyridine, 1H-1,2,4-triazole-3-thiol, 1-phenyl-1H-tetrazole-5-thiol, 2-mercaptobenzoxazole and 2-mercaptobenzothiazole and other solvents and reagents were reagent-grade materials from Aldrich. The glassy carbon electrode was polished using alumina slurry (from Iran Alumina Co.)General procedure for synthesis of 2a–e
In a typical procedure, a solution (80 mL) of water (phosphate buffer, pH = 2.0, c = 0.2 M)/ethanol mixture (25/75, v/v) containing DPD (0.25 mmol, 0.0664 g) and 0.25 mmol of thiols (1a: 0.0281 g, 1b: 0.0261 g, 1c: 0.0455 g, 1d: 0.0398 g, 1e: 0.0431) was electrolyzed in a divided cell at 0.20 V vs. SCE. The electrolysis was terminated when the decay of the current became more than 95%. The solid precipitated was collected by filtration and was washed several times with water. After drying, the residual solid dissolved in acetone and filtered. Acetone was removed under vacuum and the residual washed with diethylether and dried. The products were characterized by IR, 1H NMR, 13C NMR and MS.Acknowledgements
We acknowledge the Bu-Ali Sina University Research Council and Center of Excellence in Development of Environmentally Friendly Methods for Chemical Synthesis (CEDEFMCS) for their support of this work.References
- S. A. Lawrence, Amines: Synthesis Properties and Applications, Cambridge University Press, Cambridge, 2004, vol. 200, pp. 104–109 Search PubMed.
- G. Ćirić-Marjanović, B. Marjanović, P. Bober, Z. Rozlívková, J. Stejskal, M. Trchová and J. Prokeš, J. Polym. Sci., Part A: Polym. Chem., 2011, 49, 3387–3403 CrossRef.
- M. M. Dell'Anna, S. Intini, G. Romanazzi, A. Rizzuti, C. Leonelli, F. Piccinni and P. Mastrorilli, J. Mol. Catal. A: Chem., 2014, 395, 307–314 CrossRef PubMed.
- (a) E. Klein, Z. Cibulková and V. Lukeš, Polym. Degrad. Stab., 2005, 88, 548–554 CrossRef CAS PubMed; (b) L. Guo, H. Lei, J. Zheng, G. Huang and G. Li, Polym. Compos., 2013, 34, 1856–1862 CrossRef CAS; (c) S. S. Choi1, S. H. Im and C. Nah, J. Appl. Polym. Sci., 2012, 125, E342–E347 CrossRef.
- P. Rapta, A. Vargova, J. Polovkova, A. Gatial, L. Omelka, P. Majzlik and M. Breza, Polym. Degrad. Stab., 2009, 94, 1457–1466 CrossRef CAS PubMed.
- (a) F. Cataldo, Polym. Degrad. Stab., 2001, 72, 287–296 CrossRef CAS; (b) F. Cataldo, Eur. Polym. J., 2002, 38, 885 CrossRef CAS; (c) A. Gatial, J. Polovková and M. Breza, Acta Chim. Slov., 2008, 1, 72–84 Search PubMed; (d) Y. Fukushima, K. Mori and A. Murase, Adv. Inclusion Sci., 1984, 3, 305–315 Search PubMed.
- (a) S. M. Palash, M. A. Kalam, H. H. Masjuki, M. I. Arbab, B. M. Masum and A. Sanjid, Energy Convers. Manage., 2014, 77, 577–585 CrossRef CAS PubMed; (b) K. Varatharajan and M. Cheralathan, Fuel Process. Technol., 2013, 106, 526–532 CrossRef CAS PubMed.
- (a) Z. Cibulkova, P. Simon, P. Lehocky and J. Balko, Polym. Degrad. Stab., 2005, 87, 479–486 CrossRef CAS PubMed; (b) Z. Cibulkova, P. Simon, P. Lehocky and J. Balko, J. Therm. Anal. Calorim., 2005, 80, 357–361 CrossRef CAS.
- (a) T. Moll and J. Heinze, Synth. Met., 1993, 55, 1521–1526 CrossRef CAS; (b) A. Sakamoto, Y. Furukawa and M. Tasumi, J. Phys. Chem. B, 1997, 101, 1726–1732 CrossRef CAS; (c) M. I. Boyer, S. Quillard, E. Rebourt, G. Louarn, J. P. Buisson, A. Monkman and S. Lefrant, J. Phys. Chem. B, 1998, 102, 7382–7392 CrossRef CAS; (d) H. de Santana, S. Quillard, E. Fayad and G. Louarn, Synth. Met., 2006, 156, 81–85 CrossRef CAS PubMed.
- (a) S. Lengvinaite, J. V. Grazulevicius, S. Grigalevicius, B. Zhang, J. Yang, Z. Xie and L. Wang, Synth. Met., 2008, 158, 213–218 CrossRef CAS PubMed; (b) Z. Minliang, L. Hao, W. Liping, Y. Gui and L. Yunqi, Acta Chim. Sin., 2012, 70, 1599–1603 CrossRef; (c) D. Narayanan and K. R. Haridas, Molbank, 2013, M799, DOI:10.3390/M799.
- (a) B. Grossmann, J. Heinze, T. Moll, C. Palivan, S. Ivan and G. Gescheidt, J. Phys. Chem. B, 2004, 108, 4669–4672 CrossRef CAS; (b) T. Hagiwara, T. Demura and K. Iwata, Synth. Met., 1987, 18, 317–322 CrossRef CAS; (c) R. Moucka, M. Mrlik, M. Ilcikova, Z. Spitalsky, N. Kazantseva, P. Bober and J. Stejska, Chem. Pap., 2013, 67, 1012–1019 CAS; (d) K. G. Neoh, E. T. Kang and K. L. Tan, J. Phys. Chem., 1992, 96, 6777–6783 CrossRef CAS.
- (a) D. Nematollahi and M. Rafiee, Green Chem., 2005, 7, 638–644 RSC; (b) F. Varmaghani, D. Nematollahi, S. Mallakpour and R. Esmaili, Green Chem., 2012, 14, 963–967 RSC; (c) H. Salehzadeh, D. Nematollahi and H. Hesari, Green Chem., 2013, 15, 2441–2446 RSC.
- H. de Santana, S. Quillard, E. Fayad and G. Louarn, Synth. Met., 2006, 156, 81–85 CrossRef CAS PubMed.
- (a) T. E. Young, J. R. Griswold and M. H. Hulbert, J. Org. Chem., 1974, 39, 1980–1982 CrossRef CAS; (b) L. Papouchado, G. Petrie and R. N. Adams, J. Electroanal. Chem., 1972, 38, 389–395 CrossRef CAS.
- (a) M. D. Ryan, A. Yueh and C. Wen-Yu, J. Electrochem. Soc., 1980, 127, 1489–1495 CrossRef CAS PubMed; (b) H. Salehzadeh, D. Nematollahi and M. Rafiee, J. Electroanal. Chem., 2011, 650, 226–232 CrossRef CAS PubMed; (c) D. Nematollahi, M. Rafiee and A. Samadi-Maybodi, Electrochim. Acta, 2004, 49, 2495–2502 CrossRef CAS PubMed.
- (a) F. Varmaghani and D. Nematollahi, Electrochim. Acta, 2011, 56, 6089–6096 CrossRef CAS PubMed; (b) R. A. Izydore, H. E. Johnson and R. T. Horton, J. Org. Chem., 1985, 50, 4589–4595 CrossRef CAS.
- A. J. Bard and L. R. Faulkner, Electrochemical Methods, Wiley, New York, 2nd edn, 2001, p. 497 Search PubMed.
- (a) H. Tabata and T. Okuno, Acta Crystallogr., 2011, 67, o3169 CAS; (b) Y. Yu, H. Mao, L. Chen, X. Lu, W. Zhang and Y. Wei, Macromol. Rapid Commun., 2004, 25, 664–668 CrossRef CAS; (c) D. Narayanan and K. R. Haridas, Molbank, 2013, M799 CrossRef CAS; (d) Z. Minliang, L. Hao, W. Liping, Y. Gui and L. Yunqi, Acta Chim. Sin., 2012, 70, 1599–1603 CrossRef.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra17102k |
| This journal is © The Royal Society of Chemistry 2015 |



