Modification of the emission colour and quantum efficiency for oxazoline- and thiazoline-containing iridium complexes via different N^O ligands†
Yanling Sia,
Shuai Zhanga,
Godefroid Gahungud,
Jinghai Yang*b and
Zhijian Wu*c
aCollege of Resource and Environmental Science, Jilin Agricultural University, Changchun 130118, P. R. China
bInstitute of Condensed State Physics, Jilin Normal University, Siping 136000, P. R. China. E-mail: jhyang1@jlnu.edu.cn
cState Key Laboratory of Rare Earth Resource Utilization, Changchun Institute of Applied Chemistry, Chinese Academy of Sciences, Changchun 130022, P. R. China. E-mail: zjwu@ciac.ac.cn
dUniversité du Burundi, Faculté des Sciences, Département de Chimie, Bujumbura, BP.2700, Burundi
First published on 6th February 2015
Abstract
A DFT/TDDFT method was applied to explore the geometrical, electronic and photophysical properties of the recently reported oxazoline- and thiazoline-containing iridium(III) complexes [(ppy)2Ir(oz)] (1) and [(ppy)2Ir(thoz)] (2). The calculated absorption and emission wavelengths are in agreement with experimental data. Based on complexes 1 and 2, a series of Ir(III) complexes 3, 4, 5 and 6 with different N^O ligands have been designed. The calculated results reveal that the different ancillary ligands not only affect the absorption spectra properties but also tune the emission colour. Compared with 2, the higher quantum yield of the experimental observation for 1 was explained by its larger MLCT contribution and smaller singlet–triplet splitting energy (ΔES1–T1). From this point of view, the designed complexes 3, 5 and 6 are expected to be potential phosphorescence emitters in OLEDs with high quantum efficiency.
1. Introduction
Transition metal iridium complexes with phosphorescent properties have increasingly attracted a lot of attention, especially in the application of organic light-emitting diodes (OLEDs).1–4 Transition metal atoms have strong spin–orbit coupling effects, which can enhance the intersystem crossing from the singlet to the triplet excited state, leading to a high phosphorescence efficiency.5,6 Thus, numerous attempts have been made to explore transition metal complexes as phosphorescent emitters. Moreover, Ir(III) complexes also have high thermal stability, high photo luminescence efficiency, a short excited-state lifetime, and good emission wavelength tunability.7–10For realizing the full-color displays, many interesting Ir(III) complexes emitting green (such as Ir(ppy)3 and Ir(dfppy)2(acac), ppy = 2-phenylpyridine, dfppy = 4,6-difluoro-phenylpyridine, acac = acetylacetonate), red (such as Ir(piq)3, piq = 1-phenylisoquinolato), blue (such as Ir(dfppy)2(pic), pic = picolate), and other colors have been developed with good device performances.11–18 Recently, Chao et al. reported two new oxazoline- and thiazoline-containing Ir(III) complexes [(ppy)2Ir(oz)] (1) and [(ppy)2Ir(thoz)] (2)19 (ppy = 2-phenylpyridine), oz = (2-(2′-hydroxyphenyl)-2-oxazoline) and thoz = (2-(2′-hydroxyphenyl)-2-thiazoline) with high efficient electroluminescence (EL) performance and emission colors of green and yellow. They also performed a time-dependent density functional theory (TDDFT) study on the absorption spectra of the synthesized complexes. The reliability of TDDFT method was demonstrated from the good agreement between the calculated and experimental absorption spectra.19 Encouraged by the work by Chao et al., we designed a series of Ir(III) complexes 3, 4, 5 and 6 (Fig. 1) on the basis of the experimental structures 1 and 2. Compared with red and green emitters, blue-emitting materials with high efficiency and good stability remain a great challenge. Previous studies suggested that the substitution with electron-withdrawing nitrogen (N) atom plays an important role in realizing blue-emitting properties.20,21 Assuming that this strategy can also hold for the complexes 1 and 2, some blue emitting analogues can be expected. Exploring how the strategy can affect the photophysical properties of the complexes was the main objective of this work. In particular, the influence of the different N^O ligands on the quantum efficiency was investigated, which will be of great benefit for designing novel, high efficient blue-emitting Ir(III) complexes.
2. Computational methodology
The ground state and the lowest-lying triplet excited state geometries for each complex were optimized by using the density functional theory (DFT)22 with the hybrid-type Perdew–Burke–Ernzerhof exchange correlation functional (PBE0) and the unrestricted PBE0 (UPBE0), respectively,23–25 which have been proved to be efficient and accurate for the calculation of transition metal complexes.26 Herein, the ground state geometries of open-shell singlet with UPBE0 functional for complexes 1 and 2 were also optimized. The results show that the singlet calculations with UPBE0 functional give the same energies and geometries as the close-shell PBE0 calculations, confirming that PBE0 functional is enough to describe the ground state singlet. To confirm that each configuration was a minimum on the potential energy surface, vibrational frequencies were calculated at the same theoretical level.On the basis of ground geometry optimization, TDDFT27/PBE0 calculations associated with the polarized continuum model (PCM)28 in dichloromethane (CH2Cl2) media were applied to investigate the absorption properties. With respect to the emission properties, we performed a series of computations on the complexes 1 and 2 with five different functionals (PBE0, M062X,29 M052X,30 B3LYP31 and Cam-B3LYP32). Our results indicated that M052X is more accurate in reproducing the experimental data (Table S1, ESI†). Therefore, M052X was selected in the calculation of emission spectra.
The“double-ξ”quality basis set LANL2DZ associated with the pseudopotential was employed on atom Ir and the 6-31G(d)33 basis set was used for non-metal atoms in the gradient optimizations. All calculations were performed with the Gaussian 09 software package.34 The Molekel 4.3.2 user interface35 was used to manipulate the structures and orbitals.
3. Result and discussion
3.1 Molecular geometries in ground and excited states
In the present work, we mainly focus on the structures of the singlet ground state (S0) and the lowest triplet excited state (T1) since they are involved in the phosphorescence process. The schematic structures of the studied complexes are presented in Fig. 1, and the optimized geometrical structures for complexes 1 and 2 in the ground states are shown in Fig. 2, along with the numbering of some key atoms. PBE0, B3LYP and M052X optimized bond lengths for 1 in S0 state together with the X-ray crystal diffraction data19 are listed in Table 1. The corresponding data for 2 are listed in Table S2 of ESI.† The percentage error (δ) indicates that the PBE0 functional provides a better accuracy (small δ) than M052X functional and the widely used hybrid B3LYP in the prediction of bond lengths.| PBE0 | B3LYP | M052X | |||||
|---|---|---|---|---|---|---|---|
| Exp.a | Calc.b | δc | Calc.b | δc | Calc.b | δc | |
a Crystal data from ref. 19.b Calculated.c  |
|||||||
| Ir–N1 | 2.124 | 2.155 | 1.46 | 2.196 | 3.39 | 2.175 | 2.40 |
| Ir–N2 | 2.035 | 2.036 | 0.05 | 2.060 | 1.23 | 2.050 | 0.74 |
| Ir–N3 | 2.046 | 2.047 | 0.05 | 2.072 | 1.27 | 2.065 | 0.93 |
| Ir–O1 | 2.146 | 2.156 | 0.47 | 2.182 | 1.68 | 2.172 | 1.21 |
| Ir–C1 | 1.988 | 1.993 | 0.25 | 2.014 | 1.31 | 1.989 | 0.05 |
| Ir–C2 | 2.006 | 2.002 | −0.20 | 2.019 | 0.65 | 1.997 | −0.45 |
Table 2 gives the Ir-related bond lengths for complexes 1–6 using the PBE0/LANL2DZ-6-31G(d) method. From Table 2, one can find that the Ir–N1 and Ir–N2 bond lengths of complex 3 are slightly decreased compared with those of 1 and 2. Table 2 also shows that Ir–N1 and Ir–O1 bond lengths of complexes 4–6, where C–C bond is replaced with N–N bond, are longer than those for 1–3. This change is more obvious for 6, due to the enhanced inter-molecular π-conjugation of tetrazole ring in ancillary ligand.
| 1 | 2 | 3 | ||||
|---|---|---|---|---|---|---|
| S0 | T1 | S0 | T1 | S0 | T1 | |
| Ir–N1 | 2.155 | 2.166 | 2.169 | 2.098 | 2.148 | 2.153 |
| Ir–N2 | 2.036 | 2.018 | 2.050 | 2.062 | 2.033 | 2.018 |
| Ir–N3 | 2.047 | 2.071 | 2.036 | 2.029 | 2.046 | 2.074 |
| Ir–O1 | 2.156 | 2.143 | 2.144 | 2.125 | 2.162 | 2.147 |
| Ir–C1 | 1.993 | 1.966 | 2.001 | 2.022 | 1.993 | 1.967 |
| Ir–C2 | 2.002 | 1.997 | 1.995 | 1.996 | 2.004 | 1.999 |
| 4 | 5 | 6 | ||||
|---|---|---|---|---|---|---|
| S0 | T1 | S0 | T1 | S0 | T1 | |
| Ir–N1 | 2.161 | 2.140 | 2.175 | 2.134 | 2.159 | 2.142 |
| Ir–N2 | 2.039 | 2.041 | 2.049 | 2.056 | 2.036 | 2.039 |
| Ir–N3 | 2.050 | 2.051 | 2.038 | 2.035 | 2.049 | 2.060 |
| Ir–O1 | 2.179 | 2.163 | 2.168 | 2.132 | 2.182 | 2.111 |
| Ir–C1 | 1.993 | 1.991 | 1.996 | 2.010 | 1.992 | 1.987 |
| Ir–C2 | 1.995 | 2.005 | 1.994 | 1.992 | 1.996 | 1.996 |
The geometrical parameters of the studied complexes in the lowest triplet excited states are also listed in Table 2 to show the geometry changes upon excitation. Compared with S0 states, most of the bond distances of 1–6 are shortened in T1 states. Especially, the Ir–O1 bond length of 6 is obviously decreased in T1 states. These changes indicate that the interaction between the metal and ancillary ligands will be strengthened in T1 states. Furthermore, the difference in both ground and excited states will make a great effect on the frontier molecular orbitals, resulting in different electron transition characters.
3.2 Frontier molecular orbital (FMO) analysis
The character of the FMO and the HOMO (highest occupied molecular orbital)–LUMO (lowest unoccupied molecular orbital) energy gap are known to have influence on the properties of the complexes. Therefore, we will discuss the ground state electronic structure with special emphasis on the HOMO and LUMO. The contour plots of HOMO and LUMO of complexes 1, 3, 4 and 6 are presented in Fig. 3, while those for complexes 2 and 5 can be found in Fig. S1.† | ||
| Fig. 3 HOMO and LUMO surfaces for complexes 1, 3, 4 and 6 obtained from DFT calculations at their S0 optimized geometries. | ||
The HOMO of complex 1 is mainly localized on the d(Ir) and π(oz), while the composition of LUMO is predominantly contributed by the π*(ppy).19 Compared with complex 1, the HOMO and LUMO of complex 4 have markedly different distribution. The HOMO of the complex 4 is mainly contributed by d(Ir) and π(ppy), while the LUMO is exclusively localized on the N^O ligand, implying that the composition of FMOs is drastically influenced by different N^O ligands The same changes also occur from 2–5 (Fig. S1†). For complexes 3 and 6, the LUMOs are mainly localized on the ppy ligands, but with the different site (ppy1 for 3 and ppy2 for 5). In comparison with being localized on the N^O ligand and the Ir d orbitals in the complex 3, a significant difference is, however, observed for the HOMO of complex 6, which is attributed to the ppy ligands with a mixture of Ir(d) character.
On the other hand, the different N^O ancillary ligands also affect the FMO energy levels. The calculated HOMO–LUMO energy gaps of complexes of 3–6 are 3.35 eV, 3.39 eV, 3.00 eV and 3.54 eV, respectively. Compared with the previous theoretical energy gaps for 1 and 2 (3.22 eV for 1 and 2.83 eV for 2),19 complexes 4 and 6 have larger HOMO–LUMO energy gaps. It is worth noting that complex 6 has the largest HOMO–LUMO energy gap among the studied complexes, which will affect the absorption and emission wavelengths.
3.3 Absorption spectra
TDDFT calculations are widely used to calculate the absorption properties of Ir(III) complexes on the basis of the optimized geometries.36 Here, five different functionals (PBE0, M062X, M052X, B3LYP and Cam-B3LYP) were used to assess the effect of different DFT functionals on the absorption spectra of the complexes 1 and 2 (Table S3†). It was found that the absorption wavelengths (nm) of the complexes 1 and 2 from PBE0 functional are more close to the experimental data.19 At PBE0, the adsorption spectra at gas phase are less competitive (inferior) compared with those in solution (Table S4†). In addition, based on the optimized geometries from PBE0, B3LYP and M052X, it is seen that PBE0 also gives better performance in reproducing the experimental data, although the data from the optimized geometries of the other two methods are also satisfactory (Table S5†).Table 3 lists the most leading singlet excited states (with larger CI coefficients) and the triplet state associated with their oscillator strengths, dominant orbital excitations and their assignments, along with the corresponding experimental data from PBE0 method. It is seen that the calculated maximum absorption peaks of the complexes 1 and 2 at 250 and 248 nm are comparable to the experimental values19 249 and 243 nm, respectively. This indicated that our calculated absorption spectra reproduce the experimental data very well. For complex 1, the lowest singlet–singlet transition primarily comes from HOMO → LUMO and HOMO−1 → LUMO with the absorption band at 412 nm. The HOMO of complex 1 extends to Ir, ppy and oz ligands, whereas the LUMO is mainly localized on ppy ligands. Thus, the lowest-lying absorption can be described as metal to ligand charge transfer (MLCT)/intraligand charge transfer (ILCT)/ligand to ligand charge transfer (LLCT). The lowest-lying absorption band of 2 is similar to 1 with the character of MLCT/ILCT/LLCT. Compared with 1 and 2, the lowest-lying absorption of 3 is slightly red-shifted. The results in Table 3 also show that the lowest-lying absorption of 4 is almost identical to that of the complex 1. However, the lowest-lying absorption of 5 and 6 changed significantly compared with that of 2 and 3. It was found that the wavelength of 5 is red-shifted by 65 nm compared with that of 2. On the other hand, the wavelength of 6 is blue-shifted by 21 nm as compared to that of 3. This suggests that the lowest-lying singlet–singlet absorptions are influenced by N^O ancillary ligands, which is consistent with the trend of the HOMO–LUMO energy gaps.
| State | λcal | f | Configuration | Character | Exp.19 | |
|---|---|---|---|---|---|---|
| 1 | S1 | 412 | 0.0370 | H−1 → LUMO (16%) | MLCT/ILCT/LLCT | |
| HOMO → LUMO (77%) | MLCT/LLCT/ILCT | |||||
| S33 | 250 | 0.2973 | H−3 → L + 4 (21%) | MLCT/ILCT/LLCT | 249 | |
| HOMO → L + 5 (34%) | MLCT/LLCT/ILCT | |||||
| T1 | 467 | 0.0000 | H−1 → LUMO (25%) | MLCT/ILCT/LLCT | ||
| HOMO → LUMO (36%) | MLCT/LLCT/ILCT | |||||
| 2 | S1 | 412 | 0.0402 | H−1 → LUMO (12%) | MLCT/ILCT/LLCT | |
| HOMO → LUMO (84%) | MLCT/LLCT/ILCT | |||||
| S39 | 248 | 0.1534 | H−1 → L + 5 (15%) | MLCT/LLCT/ILCT | 243 | |
| HOMO → L + 5 (11%) | MLCT/ILCT/LLCT | |||||
| HOMO → L + 6 (34%) | MLCT/LLCT/ILCT | |||||
| T1 | 498 | 0.0000 | H−1 → L + 2 (18%) | MLCT/LLCT/ILCT | ||
| HOMO → L + 2 (66%) | MLCT/ILCT/LLCT | |||||
| 3 | S1 | 421 | 0.0344 | HOMO → LUMO (85%) | MLCT/LLCT/ILCT | |
| S40 | 242 | 0.1956 | H−5 → L + 3 (11%) | MLCT/LLCT/ILCT | ||
| HOMO → L + 7 (37%) | MLCT/LLCT/ILCT | |||||
| T1 | 472 | 0.0000 | H−1 → LUMO (17%) | MLCT/LLCT/ILCT | ||
| HOMO → LUMO (46%) | MLCT/LLCT/ILCT | |||||
| 4 | S1 | 413 | 0.0058 | HOMO → LUMO (98%) | MLCT/LLCT/ILCT | |
| S32 | 256 | 0.2100 | H−8 → LUMO (13%) | ILCT | ||
| H−7 → LUMO (48%) | ILCT/LLCT | |||||
| T1 | 521 | 0.0000 | H−1 → LUMO (88%) | MLCT/ILCT | ||
| 5 | S1 | 477 | 0.0118 | HOMO → LUMO (99%) | MLCT/LLCT/ILCT | |
| S38 | 251 | 0.2055 | H−2 → L + 4 (28%) | ILCT | ||
| HOMO → L + 5 (31%) | MLCT/LLCT/ILCT | |||||
| T1 | 593 | 0.0000 | H−1 → LUMO (64%) | MLCT/ILCT/LLCT | ||
| HOMO → LUMO (29%) | MLCT/LLCT/ILCT | |||||
| 6 | S1 | 400 | 0.0666 | H−1 → LUMO (11%) | MLCT/ILCT/LLCT | |
| HOMO → LUMO (77%) | MLCT/LLCT/ILCT | |||||
| S34 | 251 | 0.2273 | H−8 → LUMO (26%) | LLCT | ||
| H−4 → L + 4 (34%) | MLCT/ILCT/LLCT | |||||
| T1 | 461 | 0.0000 | H−2 → L + 1 (17%) | ILCT | ||
| H−1 → LUMO (14%) | MLCT/LLCT/ILCT | |||||
| HOMO → LUMO (27%) | MLCT/ILCT/LLCT | |||||
| HOMO → L + 1 (19%) | MLCT/ILCT/LLCT |
Meanwhile, the calculated absorption peaks with the largest oscillator strengths in 1 is localized at 250 nm. The transition configuration of HOMO−3 → LUMO+4 and HOMO → LUMO+5 contributes to the 250 nm absorption described as MLCT/ILCT/LLCT. Compared with 1, the transitions with the largest oscillator strengths in 3 is slightly blue-shifted, while that of 4 is slightly red-shifted (Table 3). For 2, 5 and 6, the maximum absorption peaks are similar to that of 1. In the transition S0 → T1, the absorption band of 1 located at 467 nm is contributed by HOMO → LUMO and HOMO−1 → LUMO transitions with the character of MLCT/ILCT/LLCT. The calculated lowest triplet absorptions of 2 is at 498 nm, with the main transitions being found to be HOMO → LUMO+2 and HOMO−1 → LUMO+2. The calculated lowest triplet absorptions for 3–6 are at 472, 521, 593 and 461 nm, respectively. The configurations of 3 and 5 are the same with 1, which occur from HOMO → LUMO and HOMO−1 → LUMO, with mixed nature of MLCT/ILCT/LLCT. Different from 1 and 2, the transition characters of 4 and 6 are MLCT/ILCT, MLCT/LLCT/ILCT, respectively. The non-negligible MLCT is beneficial for efficient intersystem crossing, and singlet–triplet transitions are expected to enhance the quantum efficiency for these Ir(III) complexes.
3.4 Phosphorescence properties
As mentioned in the computational methodology, TDDFT/M052X (in CH2Cl2 media) was selected to study the phosphorescent emissions of all the studied complexes since it could reproduce the experimental phosphorescent spectra reasonably. The calculated phosphorescent emissions for 1–6 are shown in Table 4, together with the electron transition assignments, and the experimental data.| λcal | Configuration | Character | Exp.19 | |
|---|---|---|---|---|
| 1 | 498 | H−2 → LUMO (28%) | MLCT/ILCT/LLCT | 527 |
| H−1 → LUMO (14%) | MLCT/ILCT/LLCT | |||
| HOMO → LUMO (43%) | MLCT/LLCT/ILCT | |||
| 2 | 595 | H−1 → LUMO (12%) | MLCT/ILCT/LLCT | 542 |
| HOMO → LUMO (74%) | MLCT/LLCT/ILCT | |||
| 3 | 494 | H−2 → LUMO (27%) | MLCT/LLCT/ILCT | |
| H−1 → LUMO (14%) | MLCT/LLCT/ILCT | |||
| HOMO → LUMO (44%) | MLCT/LLCT/ILCT | |||
| 4 | 546 | H−1 → LUMO (15%) | MLCT/ILCT | |
| HOMO → LUMO (77%) | MLCT/LLCT | |||
| 5 | 619 | H−1 → LUMO (33%) | MLCT/ILCT | |
| HOMO → LUMO (60%) | MLCT/LLCT | |||
| 6 | 439 | H−1 → LUMO (14%) | MLCT/LLCT/ILCT | |
| H−1 → L + 1 (31%) | MLCT/LLCT/ILCT | |||
| HOMO → LUMO (18%) | MLCT/ILCT/LLCT | |||
| HOMO → L + 1 (15%) | MLCT/ILCT/LLCT |
The calculated lowest-energy emissions of the complexes 1 and 2 are localized at 498 and 595 nm. Upon the substitution of C atom with N atom on N^O ligands, the emission wavelength of 4 and 5 (546 and 619 nm) are red-shifted. However, the emission wavelengths of 3 (494 nm) in which N atom substitutes for O/S atom on N^O ligand and 6 (439 nm) are blue-shifted compared to complex 1, in which the wavelength 439 nm lies in the deep blue region. This revealed that the N^O ligands in the studied complexes could modify the emission color from green and yellow to deep blue. The calculated lowest emission energy for 1 corresponds to the HOMO−2 → LUMO, HOMO−1 → LUMO and HOMO → LUMO transitions with mixed nature of MLCT/ILCT/LLCT. The configuration of 3 is the same as that of 1. For 4 and 5, there are two transitions contributing to calculated lowest emission, which are HOMO → LUMO and HOMO−1 → LUMO transitions, respectively. The emission can be assigned as MLCT/ILCT/LLCT. For 6, the emission is from HOMO−1 → LUMO+1, HOMO → LUMO, HOMO → LUMO+1 and HOMO−1 → LUMO, and is assigned as MLCT/LLCT/ILCT. The larger participation of Ir(III) center in the emissive state is expected to result in a high quantum efficiency.
3.5 Quantum efficiency
The emission quantum yield (ΦPL) is linked to the radiative (kr) and nonradiative (knr) rate constants, which can be expressed by eqn (1), where τem is the emission decay time:
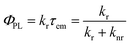 | (1) |
Accordingly, a large kr and a small knr are required for a high quantum yield. Theoretically, kr is related to the mixing between S1 and T1, which is proportional to the spin–orbit coupling (SOC) and inversely proportional to the energy difference between the two states expressed by eqn (2):37,38
 | (2) |
| Parameter | 1 | 2 | 3 | 4 | 5 | 6 |
|---|---|---|---|---|---|---|
| MLCT | 31.84 | 20.67 | 32.62 | 34.90 | 31.32 | 23.77 |
| μS1 | 0.5023 | 0.5455 | 0.4765 | 0.0791 | 0.1851 | 0.8776 |
| ΔES1–T1 | 0.7497 | 0.9698 | 0.7084 | 1.0103 | 0.8919 | 0.7314 |
| ΦPL19 | 0.55 | 0.27 |
On the other hand, the SOC effects also can be elucidated by the singlet–triplet splitting energy (ΔES1–T1).44–47 The S1 → T1 intersystem crossing induced by SOC plays an important role in phosphorescent process. Eqn (2) shows that the decreasing ΔES1–T1 leads to higher radiative rates, while a decreasing μS1 lowers kr. Using eqn (2), we recently provided a valuable theoretical analysis of the evolution of ΦPL within a series of complexes48 with the aid of computed ΔES1–T1 and μS1. The calculated ΔES1–T1 and μS1 values are also shown in Table 5. The results indicated that the μS1 value of 2 is slightly larger than that of 1, which can not explain the high ΦPL reported for 1.19 The much smaller ΔES1–T1 value for 1 accounts for the higher ΦPL compared with 2. The ΔES1–T1 for 3, 5 and 6 were found to be relatively smaller than that of 2, indicating that the N substitution in different N^O ligands is beneficial to enhance the ISC rate, and thus might increase ΦPL. Of course, we should remember that besides the factors discussed above, other factors may also affect ΦPL which are not taken into account in our discussion.
4. Conclusion
DFT/TDDFT calculations have been performed to investigate the influence on electronic structure, absorption spectra, emission colour and quantum efficiency of the studied complexes induced by the introduction of different N^O ligands. In this study, the following conclusions can be drawn: (1) the composition of the LUMO is significantly affected, when the C–C bond is substituted with N–N bond. The existence of tetrazole ring of 6 leads to a large HOMO–LUMO energy gap. (2) Compared with 1 and 2, the emission wavelengths for 4 and 5 are red-shifted, while they are blue-shifted significantly for 3 and 6. Especially, the emission colour of the complex 6 is in deep blue region, which is promising as a blue-emitting material. (3) The high quantum yield of 1 compared with 2 was found to be closely related to the large MLCT% and small ΔES1–T1. The results showed that the different ancillary ligands in 3, 5 and 6 leads to a relatively large MLCT% and small ΔES1–T1 and thus a high quantum yield. We hope that our results could provide constructive information for experimentalists in synthesizing high efficient phosphorescence materials.Acknowledgements
The authors thank the financial support from the National Natural Science Foundation of China (Grant no. 21273219, 21221061, 21203174), Natural Science Foundation of Jilin Province (20140101045JC) and computing time from the High Performance Computing Center of Jilin University, China.Notes and references
- S. Reineke, F. Lindner, G. Schwartz, N. Seidler, K. Walzer, B. Lussem and K. Leo, Nature, 2009, 459, 234 CrossRef CAS PubMed.
- Y. C. Chiu, J. Y. Hung, Y. Chi, C. C. Chen, C. H. Chang, C. C. Wu, Y. M. Cheng, Y. C. Yu, G. H. Lee and P. T. Chou, Adv. Mater., 2009, 21, 2221 CrossRef CAS.
- M. Ikai, S. Tokito, Y. Sakamoto, T. Suzuki and Y. Taga, Appl. Phys. Lett., 2001, 79, 156 CrossRef CAS PubMed.
- C. Adachi, M. A. Baldo, M. E. Thompson and S. R. Forrest, J. Appl. Phys., 2001, 90, 5048 CrossRef CAS PubMed.
- Y. Chi and P. T. Chou, Chem. Soc. Rev., 2010, 39, 638 RSC; P. T. Chou and Y. Chi, Chem.–Eur. J., 2007, 13, 380 CrossRef CAS PubMed.
- (a) Y. You, J. Seo, S. H. Kim, K. S. Kim, T. K. Ahn, D. Kim and S. Y. Park, Inorg. Chem., 2008, 47, 1476 CrossRef CAS PubMed; (b) S. Tokito, T. Iijima, Y. Suzuri, H. Kita, T. Tsuzuki and F. Sato, Appl. Phys. Lett., 2003, 83, 569 CrossRef CAS PubMed; (c) W. Y. Wong and C. L. Ho, Coord. Chem. Rev., 2009, 253, 1709 CrossRef CAS PubMed.
- (a) W. Y. Wong, G. J. Zhou, X. M. Yu, H. S. Kwok and B. Z. Tang, Adv. Funct. Mater., 2006, 16, 838 CrossRef CAS; (b) Z. Q. Chen, Z. Q. Bian and C. H. Huang, Adv. Mater., 2010, 22, 1534 CrossRef CAS PubMed; (c) C. Ulbricht, B. Beyer, C. Friebe, A. Winter and U. S. Schubert, Adv. Mater., 2009, 21, 4418 CrossRef CAS; (d) Y. You and S. Y. Park, Dalton Trans., 2009, 1267 RSC; (e) X. Li, Q. Zhang, Y. Q. Tu, H. Agren and H. Tian, Phys. Chem. Chem. Phys., 2010, 12, 13730 RSC.
- X. S. Zeng, M. Tavasli, I. F. Perepichka, A. S. Batsanov, M. R. Bryce, C. J. Chiang, C. Rothe and A. P. Monkman, Chem.–Eur. J., 2008, 14, 933 CrossRef CAS PubMed.
- L. He, L. Duan, J. Qiao, R. J. Wang, P. Wei, L. D. Wang and Y. Qiu, Adv. Funct. Mater., 2008, 18, 2123 CrossRef CAS.
- (a) Q. Zhao, S. J. Liu, M. Shi, C. M. Wang, M. X. Yu, L. Li, F. Y. Li, T. Yi and C. H. Huang, Inorg. Chem., 2006, 45, 6152 CrossRef CAS PubMed; (b) S. J. Liu, Q. Zhao, R. F. Chen, Y. Deng, Q. L. Fan, F. Y. Li, L. H. Wang, C. H. Huang and W. Huang, Chem.–Eur. J., 2006, 12, 4351 CrossRef CAS PubMed; (c) Q. Zhao, L. Li, F. Y. Li, M. X. Yu, Z. P. Liu, T. Yi and C. H. Huang, Chem. Commun., 2008, 685 RSC; (d) S. J. Liu, Q. Zhao, B. X. Mi and W. Huang, Adv. Polym. Sci., 2008, 212, 125 CAS; (e) Q. Zhao, S. J. Liu and W. Huang, Macromol. Rapid Commun., 2010, 31, 794 CrossRef CAS PubMed; (f) H. F. Shi, Y. Nakai, S. J. Liu, Q. Zhao, Z. F. An, T. Tsuboi and W. Huang, J. Phys. Chem. C, 2011, 115, 11749 CrossRef CAS.
- M. A. Baldo, S. Lamansky, P. E. Burrows, M. E. Thompson and S. R. Forrest, Appl. Phys. Lett., 1999, 75, 4 CrossRef CAS PubMed.
- R. J. Holmes, S. R. Forrest, Y. J. Tung, R. C. Kwong, J. J. Brown, S. Garon and M. E. Thompson, Appl. Phys. Lett., 2003, 82, 2422 CrossRef CAS PubMed.
- J. Li, P. I. Djurovich, B. D. Alleyne, M. Yousufuddin, N. N. Ho, J. C. Thomas, J. C. Peters, R. Bau and M. E. Thompson, Inorg. Chem., 2005, 44, 1713 CrossRef CAS PubMed.
- S. Okada, K. Okinaka, H. Iwawaki, M. Furugori, M. Hashimoto, T. Mukaide, J. Kamatani, S. Igawa, A. Tsuboyama, T. Takiguchi and K. Ueno, Dalton Trans., 2005, 1583 RSC.
- Y. Y. Lyu, Y. Byun, O. Kwon, E. Han, W. S. Jeon, R. R. Das and K. Char, J. Phys. Chem. B, 2006, 110, 10303 CrossRef CAS PubMed.
- C. H. Yang, Y. M. Cheng, Y. Chi, C. J. Hsu, F. C. Fang, K. T. Wong, P. T. Chou, C. H. Chang, M. H. Tsai and C. C. Wu, Angew. Chem., Int. Ed., 2007, 46, 2418 CrossRef CAS PubMed.
- G. J. Zhou, W. Y. Wong, B. Yao, Z. Y. Xie and L. X. Wang, Angew. Chem., Int. Ed., 2007, 46, 1149 CrossRef CAS PubMed.
- C. L. Ho, W. Y. Wong, Q. Wang, D. G. Ma, L. X. Wang and Z. Y. Lin, Adv. Funct. Mater., 2008, 18, 928 CrossRef CAS.
- K. Chao, K. Z. Shao, T. Peng, D. X. Zhu, Y. Wang, Y. Liu, Z. M. Su and M. R. Bryce, J. Mater. Chem. C, 2013, 1, 6800 RSC.
- C. F. Chang, Y. M. Cheng, Y. Chi, Y. C. Chiu, C. C. Lin, G. H. Lee, P. T. Chou, C. C. Chen, C. H. Chang and C. C. Wu, Angew. Chem., Int. Ed., 2008, 47, 4542 CrossRef CAS PubMed.
- S. J. Yeh, M. F. Wu, C. T. Chen, Y. H. Song, Y. Chi, M. H. Ho, S. F. Hsu and C. H. Chen, Adv. Mater., 2005, 17, 285 CrossRef CAS.
- E. Runge and E. K. U. Gross, Phys. Rev. Lett., 1984, 52, 997 CrossRef CAS.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865 CrossRef CAS.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1997, 78, 1396 CrossRef CAS.
- C. Adamo and V. Barone, J. Chem. Phys., 1999, 110, 6158 CrossRef CAS PubMed.
- (a) T. Liu, B. H. Xia, Q. C. Zheng, X. Zhou, Q. J. Pan and H. X. Zhang, J. Comput. Chem., 2010, 31, 628 CAS; (b) I. Ciofini, P. P. Laine, F. Bedioui and C. Adamo, J. Am. Chem. Soc., 2004, 126, 10763 CrossRef CAS PubMed; (c) I. Ciofini, P. P. Laine, F. Bedioui, C. A. Daul and C. Adamo, C. R. Chim., 2006, 9, 226 CrossRef CAS PubMed; (d) D. Jacquemin, E. A. Perpete, G. Scalmani, M. J. Frisch, I. Ciofini and C. Adamo, Chem. Phys. Lett., 2006, 421, 272 CrossRef CAS PubMed; (e) I. Ciofini, P. P. Laine, M. Zamboni, C. A. Daul, V. Marvaud and C. Adamo, Chem.–Eur. J., 2007, 13, 5360 CrossRef CAS PubMed; (f) Y. L. Si, Y. Q. Liu, X. C. Qu, Y. Wang and Z. J. Wu, Dalton Trans., 2013, 42, 14149 RSC.
- (a) J. Autschbach, T. Ziegler, S. J. A. Gisbergen and E. J. Baerends, J. Chem. Phys., 2002, 116, 6930 CrossRef CAS PubMed; (b) T. Helgaker and P. Jørgemsen, J. Chem. Phys., 1991, 95, 2595 CrossRef CAS PubMed; (c) K. L. Bak, P. Jørgemsen, T. Helgaker, K. Rund and H. J. A. Jensen, J. Chem. Phys., 1993, 98, 8873 CrossRef CAS PubMed.
- (a) E. Cancès, B. Mennucci and J. Tomasi, J. Chem. Phys., 1997, 107, 3032 CrossRef PubMed; (b) M. Cossi, V. Barone, B. Mennucci and J. Tomasi, Chem. Phys. Lett., 1998, 286, 253 CrossRef CAS; (c) B. Mennucci and J. Tomasi, J. Chem. Phys., 1997, 106, 5151 CrossRef CAS PubMed.
- Y. Zhao and D. G. Truhlar, J. Phys. Chem. A, 2006, 110, 5121 CrossRef CAS PubMed.
- Y. Zhao, N. E. Schultz and D. G. Truhlar, J. Chem. Theory Comput., 2006, 2, 364 CrossRef.
- A. D. Becke, J. Chem. Phys., 1993, 98, 5648 CrossRef CAS PubMed; C. T. Lee, W. Yang and R. G. Parr, Phys. Rev. B: Condens. Matter Mater. Phys., 1988, 37, 785 CrossRef.
- T. Yanai, D. Tew and N. Handy, Chem. Phys. Lett., 2004, 393, 51 CrossRef CAS PubMed.
- P. C. Hariharan and J. A. Pople, Mol. Phys., 1974, 27, 209 CrossRef CAS.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery Jr J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, T. Keith, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski and D. Fox, J. Gaussian 09, Revision B.01, Gaussian, Inc., Wallingford, CT, 2010 Search PubMed.
- U. Varetto, MOLEKEL Version, Swiss National Supercomputing Centre, Lugano, Switzerland Search PubMed.
- (a) H. R. Tsai, K. Y. Lu, S. H. Lai, C. H. Fan, C. H. Cheng and I. C. Chen, J. Phys. Chem. C, 2011, 115, 17163 CrossRef CAS; (b) P. Jeffrey Hay, J. Phys. Chem. A, 2002, 106, 1634 CrossRef; (c) A. R. G. Smith, M. J. Riley, S. C. Lo, P. L. Burn, I. R. Gentle and B. J. Powell, Phys. Rev. B: Condens. Matter Mater. Phys., 2011, 83, 041105 CrossRef; (d) Y. L. Si, Y. Q. Liu, G. Gahungu, X. C. Qu and Z. J. Wu, Mol. Phys., 2013, 111, 3716 CrossRef CAS.
- S. Haneder, E. Da Como, J. Feldmann, J. M. Lupton, C. Lennartz, P. Erk, E. Fuchs, O. Molt, I. Munster, C. Schildknecht and G. Wagenblast, Adv. Mater., 2008, 20, 3325 CrossRef CAS.
- N. Turro, Modern Molecular Photochemistry, University Science Books, Palo Alto, CA, USA, 1991 Search PubMed.
- C. H. Yang, Y. M. Cheng, Y. Chi, C. J. Hsu, F. C. Fang, K. T. Wong, P. T. Chou, C. H. Chang, M. H. Tsai and C. C. Wu, Angew. Chem., Int. Ed., 2007, 46, 2418 CrossRef CAS PubMed.
- J. D. Chai and M. Head-Gordon, Phys. Chem. Chem. Phys., 2008, 10, 6615 RSC.
- S. Sprouse, K. A. King, P. J. Spellane and R. J. Watts, J. Am. Chem. Soc., 1984, 106, 6647 CrossRef CAS.
- M. G. Colombo, T. C. Brunold, T. Riedener, H. U. Güdel, M. Förtsch and H. B. Bürgi, Inorg. Chem., 1994, 33, 545 CrossRef CAS.
- C. H. Lin, Y. Y. Chang, J. Y. Hung, C. Y. Lin, Y. Chi, M. W. Chung, C. L. Lin, P. T. Chou, G. H. Lee, C. H. Chang and W. C. Lin, Angew. Chem., Int. Ed., 2011, 50, 3182 CrossRef CAS PubMed.
- Z. F. Xie, F. Q. Bai, J. Wang and H. X. Zhang, Mol. Phys., 2011, 109, 1657 CrossRef CAS.
- P. J. Hay, J. Phys. Chem. A, 2002, 106, 1634 CrossRef CAS.
- A. R. G. Smith, M. J. Riley, S. C. Lo, P. L. Burn, I. R. Gentle and B. J. Powell, Phys. Rev. B: Condens. Matter, 2011, 83(1), 041105 CrossRef.
- D. N. Kozhevnikov, V. N. Kozhevnikov, M. Z. Shafikov, A. M. Prokhorov, D. W. Bruce and J. A. G. Williams, Inorg. Chem., 2011, 50, 3804 CrossRef CAS PubMed.
- S. Zhang, Y. L. Si and Z. J. Wu, RSC Adv., 2014, 4, 15849 RSC.
Footnote |
| † Electronic supplementary information (ESI) available: Fig. S1: contour plots of the HOMO and LUMO for complexes 2 and 5. Table S1: calculated phosphorescent emission wavelength (nm)/energies (eV) of the complexes 1 and 2 in CH2Cl2 media with the TDDFT method at the PBE0, M062X, M052X, B3LYP and Cam-B3LYP level, respectively, together with the experimental values. Table S2: selected optimized parameters for complex 2 in the ground state from different density functionals. Table S3: calculated absorption wavelength with the largest oscillator strengths for complexes 1 and 2 in CH2Cl2 media with the TDDFT method at the PBE0, B3LYP, M052X M062X and Cam-B3LYP level, respectively, together with the experimental data. Table S4: calculated absorption wavelength (nm) and oscillator strength of the complexes 1 and 2 with the TDDFT method at the PBE0 level in gas phase and CH2Cl2 media, together with the experimental data. Table S5: calculated absorption wavelength and oscillator strength of the complexes 1 and 2 with the TDDFT method at the PBE0 level using the PBE0, B3LYP and M052X optimized the ground state geometries, together with the experimental data. Tables S6–S11: Carrtesian coordinates for the optimized structures of 1–6 in the S0 states. See DOI: 10.1039/c4ra16269b |
| This journal is © The Royal Society of Chemistry 2015 |


