Experimental and computational exploration of the UV-visible properties of hexaniobate and hexatantalate ions†
Gauthier J.-P. Deblonde*ab,
Aurélien Moncomblec,
Gérard Cotea,
Sarah Bélairb and
Alexandre Chagnesa
aPSL Research University, Chimie ParisTech – CNRS, Institut de Recherche de Chimie Paris, 11 rue Pierre et Marie Curie, 75005 Paris, France. E-mail: gauthier.deblonde@chimie-paris.org
bEramet Research, Hydrometallurgy Department, F-78193 Trappes, France
cLASIR (UMR CNRS 8516) – Université de Lille – Sciences et Technologies, 59655 Villeneuve d'Ascq, France
First published on 19th December 2014
Abstract
The UV-visible properties of hexaniobate (HxNb6O19x−8) and hexatantalate (HxTa6O19x−8) ions were investigated experimentally and by DFT calculations. Due to the huge discrepancies among the reported values found in prior studies, the extinction coefficients of hexaniobate ions were determined from multiple samples in various media. A simple and low-cost method was then developed for the determination of the niobium content of both synthetic and industrial samples. Taking advantage of the UV spectra of the hexaniobate ions, the first protonation constant of Nb6O198− could be determined experimentally (pKa = 13.4 ± 0.1 at 25 °C, ΔrH = −95 kJ mol−1 in 3 M KOH/KCl) and is in accordance with the reported values previously extrapolated from potentiometric measurements (pKa = 13.3 ± 0.6). UV batch titrations performed at different ionic strengths suggest that the equilibrium between the monoprotonated and the deprotonated forms of the Lindqvist ion is accompanied by an exchange of potassium ions and that the cluster is easier to deprotonate in Na+ media. This study highlights the importance of the ion-pairing on the chemistry of these polyoxometalates. The tremendous difference between the UV spectrum of Nb6O198− and Ta6O198− was investigated by DFT computations. The UV spectra were reproduced and show a good agreement with the experimental data. Moreover this study revealed some insights into the evolution of the spectra together with the pH of the medium by studying the nature of the transitions involved in this spectral domain.
Introduction
Niobium (Z = 41) and tantalum (Z = 73) are attracting increasing attention because they play a critical role in alloys and electronic industries. However, the development of efficient, economic and environmentally friendly processes to recover them from ores is still a challenge. Although classical routes to recover and purify niobium and tantalum require highly acidic and fluoride containing solutions,1 the chemistry of both valuable metals has been extensively studied in alkaline media over the past decades. Indeed, high solubility of Nb(V) and Ta(V) can be reached in alkaline solutions along with separation from impurities such as Fe, Mn and Ti.2 The chemistry of Nb(V) and Ta(V) at basic pH is dominated by the Lindqvist type ions HxM6O19x−8 (M = Nb, Ta; 0 ≤ x ≤ 3).3 The structure of the cluster M6O19 has been widely investigated and consists of a super octahedron of six edge-sharing octahedra MO6 yielding three different types of oxygen atoms: one central atom μ6–O, six terminal oxygens η–O and twelve bridging oxygens μ2–O (Fig. 1).Contrasting with the renewed interest in alkaline solutions of Nb and Ta, methods to determine niobium and tantalum content in alkaline samples are still limited to Inductively Coupled Plasma spectrometry. In this regard, the UV-visible properties of the hexaniobate and hexatantalate ions could help to implement new and cheap methods for the determination of Nb and Ta content in samples. UV-based methods could also be developed to probe the solution thermodynamics of HxM6O19x−8 ions, including pKa determination and stability of these polyoxometalates. Surprisingly, the studies published on the absorbance properties of hexaniobate and hexatantalate ions are scarce and took place in the early development of polyoxoniobates and polyoxotantalates chemistry when the existence of the hexameric species was still controversial. In the early 60's, Jander and Ertel4 first performed a spectrophotometric study on hexaniobate ions showing a strong absorbance band below 300 nm with a maximum lying between 250 and 230 nm depending on the pH. The only hexatantalate UV spectrum was reported three years later by Nelson and Tobias5 highlighting the low absorbance properties of HxTa6O19x−8 ions compared to their niobium counterparts. Knowing that Nb(V) and Ta(V) have identical ionic radii6 and very similar electronic configurations, resulting in quasi-identical geometry for Nb6O19 and Ta6O19 clusters,7 the striking difference between their UV properties challenges our understanding and needed to be investigated.
The present study revisits the UV properties of hexaniobate and hexatantalate ions. The absorbance properties of hexaniobate ions allowed us to implement a fast and convenient method for the determination of Nb concentration in various samples including those found in industrial processes used for Nb and Ta recovery. Taking advantage of the UV absorbance of hexaniobate ions, the first protonation constant and its reaction enthalpy have been determined experimentally for the first time. Finally, the tremendous difference between the UV properties of Nb and Ta Lindqvist ions has been investigated by DFT calculations and enriches our understanding on Nb and Ta chemistry in alkaline media.
Experimental section and computational details
Regents
All stock solutions were prepared with deionized water (R > 18.2 × 106 Ω) degassed by boiling for 1 h while being purged under N2. All stock solutions were kept in a glove box under N2 atmosphere. Nb2O5 99.9% (Sigma Aldrich), Nb(HC2O4)5, nH2O (Alfa Aesar, 19.2 wt% Nb) and NbCl5 99% (Alfa Aesar) were used without further purification. Industrial alkaline solutions of Nb(V) were provided by Eramet Research company (France). These samples were slightly contaminated by Ta(V); Nb/Ta = 80 mol mol−1. Stock solutions of NaCl (VWR), KCl (VWR), CsOH, H2O (Alfa Aesar) and potassium hydrogen phthalate (99.98%, Fluka) were obtained by direct dissolution of weighed samples in CO2-free deionized water. The KOH stock solution was purchased and opened under N2 atmosphere (Alfa Aesar, Selectipur®, 30 wt% KOH. An analytical certificate provided by the manufacturer gives a CO2 contamination of 0.3 wt% K2CO3). The concentrated KOH stock solution was standardized by titrating against potassium hydrogen phthalate under positive N2 pressure. The titration was repeated four times and yielded a KOH concentration of 6.761 ± 0.020 mol L−1. The NaOH stock solution was prepared by dissolving NaOH pellets in CO2-free deionized water. The NaOH solution was further purged with N2 for 1 h and titrated against potassium hydrogen phthalate ([NaOH] = 4.866 ± 0.020 mol L−1). For the synthesis of K8Nb6O19, 16H2O, NbCl5 was hydrolyzed at pH 4 to produce Nb2O5, nH2O which was then used to produce the potassium hexaniobate salt as reported by Nyman et al.8 K8Nb6O19, 16H2O was identified by powder XRD (ICDD PDF 01-077-4938), Raman spectroscopy and thermogravimetric analysis (TGA). Raman (cm−1): 875; 827; 745; 529; 460; 376; 288; 217. TGA: %H2O found = 19.6 wt% (theoretical: 19.7). Na7HNb6O19, 15H2O was synthesized by alkaline fusion (5 h at 450 °C, NaOH/Nb2O5 = 10 mol mol−1), washed and recrystallized in water. The product was also identified by powder XRD (ICDD PDF 04-016-4410), Raman spectroscopy and TGA. Raman (cm−1): 904; 837; 769; 524; 485; 458; 380; 278; 215; 172. TGA: %H2O found = 21.5 wt% (theoretical: 20.9).Materials
A Vista Pro spectrometer (Varian) was used for ICP-AES titrations. Quantitative analyses were performed at 295.088 nm spectral emission line for Nb. All UV spectra were measured on a Cary 100-Scan spectrometer used in double beam mode. Each spectrum was acquired in Quartz Suprasil® cuvette (path length of 10.00 mm) and recorded against the corresponding blank sample. The cuvette lixiviation in high alkaline media was checked to be very limited in order to avoid silicate contamination (provider's data: dissolution of 0.27 μg cm−2 h−1 with KOH 30 wt%). The alkaline samples were hold in the cuvettes less than 5 min and then discarded.UV batch titrations
Each acid–base titration contained, at least, 8 samples prepared under N2 atmosphere by diluting the stock solutions of KCl, KOH and K8Nb6O19, 16H2O or NaCl, NaOH and Na7HNb6O19, 15H2O with CO2-free deionized water. Polyethylene vessels were used to avoid contamination by silicates. The hydroxide concentration was varied from 0.01 mol L−1 to the maximum value (which equals the ionic strength of the series) while the niobium concentration and the ionic strength were kept constant. The total niobium concentration was typically 2.4 × 10−4 mol L−1 i.e. ∑[HxNb6O19x−8] = 4 × 10−5 mol L−1. The corresponding blank samples were prepared similarly without niobium. After being thermostated under N2 atmosphere for 24 h, the UV spectrum (220–350 nm – 260 data points) of each sample was recorded against its corresponding blank.Data treatment
For each batch titration, the UV spectra, the extinction coefficient of the deprotonated species, the total hexaniobate concentration and the estimated pH were imported into the refinement program HypSpec9 and analyzed by nonlinear least-squares refinement. The determination of equilibrium constants by this software has proven its efficiency elsewhere.10–12 The protonation constant given is this work refers to the following equation:| Nb6O19(aq)8− + H3O+ = HNb6O19(aq)7− + H2O | (1) |
Due to the low concentration of hexaniobate compared to hydroxide ions in the samples ([HNb6O197−] + [Nb6O198−] = 4 × 10−5 mol L−1 compared to 0.01 ≤ [HO−] ≤ 3.00 mol L−1), the hydroxide concentration in each sample was evaluated from the volume of standardized stock solutions. The corresponding pH was calculated using eqn (2).
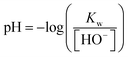 | (2) |
The Kw values in KCl/KOH media at the different ionic strengths and temperatures were taken from Sweeton et al.13 The Kw values in NaCl/NaOH media were taken from Kron et al.14 The whole procedure (samples preparation, UV batch titration and data treatment) was duplicated for 3 different ionic strengths and 2 different temperatures so that the uncertainty of the method was determined to be ±0.1 log unit.
Computational details
All calculations were carried out using DFT-based methods, and more specifically the PBE0 global hybrid functional.15,16 The Gaussian 09 software was used throughout.17 For optimizations, oxygen was represented by the Dunning basis set18 while Nb and Ta were described by the Stuttgart–Dresden ECP and its associated basis set.19 For thermodynamics and UV-vis properties, single points were processed on the optimized structured using the larger Def2-TZVP basis set for the three elements.20 Optimizations were carried out without any symmetry constraint using standard algorithms and the nature of the minima was checked by vibrational analysis. The solvent was taken into account using an implicit model, namely the Polarizable Continuum Model (PCM)21 as implemented in Gaussian 09.Electronic transitions were computed in the time-dependent variant of DFT (TD-DFT) within the adiabatic approximation taking into account the 250 lowest-lying excited states. To produce spectra, each transition was convoluted using a Gaussian-centered function with a 3000 cm−1 FWHM and all components were added.
Results
UV properties of hexaniobate ions
The UV spectrum of hexaniobate solutions displays a large absorbance band below 300 nm with a maximum lying between 250 and 240 nm depending on the hydroxide concentration (Fig. 2). Upon increasing of the pH, the absorbance wavelength of the charge transfer band shifts from 241.5 nm in presence of 0.01 M of KOH to 247.5 nm in 3.00 M KOH. Measurements performed with a KOH concentration ranging from 3 M to 9 M did not show further variations of the UV spectra (Fig. 3 and S1†). The red shift observed in the UV spectrum is characteristic of the deprotonation of the hexaniobate ion HNb6O197− and suggests that a hydroxide concentration higher than 3 M is required to fully deprotonate the hexaniobate cluster. The isosbestic point observed at 240 nm indicates the presence of an equilibrium between the deprotonated and the monoprotonated clusters. Taking into account these observations, the extinction coefficient of the fully deprotonated cluster Nb6O198− was determined experimentally.Due to huge discrepancy in the literature data,4,22,23 special care was taken in order to determine the molar absorptivity of Nb6O198−. More than thirty independent samples with various media (Fig. S2†) were diluted in 4 M KOH, their UV spectrum was recorded and their niobium content was determined by conventional Inductively Coupled Plasma spectrometric titration (ICP-AES). The extinction coefficient εNb6O19 = 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900 ± 600 L mol−1 cm−1 was determined for Nb6O198−. In order to validate this value, samples of niobium oxalate were prepared by dissolution of commercial Nb(HC2O4)5, nH2O in water, the samples were then diluted in 4 M KOH and their UV spectra were recorded after 10 min equilibration time. The same procedure was implemented for solutions of commercial NbCl5 dissolved in HCl. The Nb concentration of each sample was calculated using Beer–Lambert's law and the extinction coefficient value previously determined.
900 ± 600 L mol−1 cm−1 was determined for Nb6O198−. In order to validate this value, samples of niobium oxalate were prepared by dissolution of commercial Nb(HC2O4)5, nH2O in water, the samples were then diluted in 4 M KOH and their UV spectra were recorded after 10 min equilibration time. The same procedure was implemented for solutions of commercial NbCl5 dissolved in HCl. The Nb concentration of each sample was calculated using Beer–Lambert's law and the extinction coefficient value previously determined.
The theoretical Nb concentrations and those obtained by the UV titration are in good agreement for both niobium chloride and niobium oxalate samples as displayed in Fig. S3.† The UV-based method was also used to determine the Nb content of 17 independent industrial solutions of hexaniobate. The concentrations measured by UV titration and by ICP-AES are in good agreement (Fig. S4†) confirming the value of 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900 L mol−1 cm−1 εNb6O19. The linear range of the method was found to be 5 × 10−6 to 10−4 M of Nb6O198−. The influence of the ionic media on the UV spectrum of Nb6O198− was checked by diluting hexaniobate samples in 4 M NaOH, KOH and CsOH. The nature of the alkali ion does not influence the shape of the charge transfer band but the wavelength at maximum absorbance (λmax) is slightly shifted from 246.5 in NaOH to 247.5 in KOH and 249 nm in CsOH (Fig. 4).
900 L mol−1 cm−1 εNb6O19. The linear range of the method was found to be 5 × 10−6 to 10−4 M of Nb6O198−. The influence of the ionic media on the UV spectrum of Nb6O198− was checked by diluting hexaniobate samples in 4 M NaOH, KOH and CsOH. The nature of the alkali ion does not influence the shape of the charge transfer band but the wavelength at maximum absorbance (λmax) is slightly shifted from 246.5 in NaOH to 247.5 in KOH and 249 nm in CsOH (Fig. 4).
 | ||
| Fig. 4 Absorbance spectra of Nb6O198− measured in 4 M NaOH (orange), 4 M KOH (black) and 4 M CsOH (blue). | ||
UV properties of hexatantalate ions
Compared to their niobium counterparts, solutions of hexatantalate ions HxTa6O19x−8 (x ≤ 3) exhibit a relatively low absorbance in the UV region. Variation of the hydroxide concentration from 0.01 M to 9 M has no influence on the UV spectra suggesting that the hexatantalate ion is fully deprotonated at pH ≥ 12. At 247.5 nm, which is the wavelength of maximum absorbance for Nb6O198−, the molar absorptivity of Ta6O198− is 80 times lower than that of its niobium equivalent (Fig. 5). At the isosbestic point of 240 nm observed for hexaniobate, the ratio εNb6O19/εTa6O19 is still 20. | ||
| Fig. 5 Molar absorptivity of Ta6O198− (orange) and Nb6O198− (blue) determined experimentally and molar absorptivity of HNb6O197− (purple) calculated from UV batch titrations. | ||
pKa determination
The variation observed on the hexaniobate UV spectrum while varying the hydroxide concentration was large enough to accurately determine equilibrium constant. Nonlinear least-squares fits of the spectra included the fully deprotonated species Nb6O198− and the monoprotonated species HNb6O197− (Fig. 5). The refinement process also enabled us to determine the extinction coefficient of the monoprotonated species HNb6O197−.Spectrophotometric batch titrations were performed in KOH/KCl and NaOH/NaCl media at different ionic strengths and temperatures. The equilibrium constant decreases when the ionic strength increases. At a given ionic strength and temperature the pKa HNb6O197−/Nb6O198− was found to be lower in NaCl/NaOH media than in KCl/KOH media. The results are summarized in Fig. 6.
 | ||
| Fig. 6 Protonation constant of Nb6O198− measured at 25 °C in KCl/KOH media (square) and NaCl/NaOH media (circle) and values extrapolated by Neumann24 (triangle), Spinner et al.25 (crosses) and Etxebarria et al.26 (lozenge). The error bars correspond to ±0.1 log unit. | ||
For an ionic strength of 3 M (KOH/KCl), the equilibrium constant was determined at different temperatures (Fig. 7). The refinement of the titration data showed that the reaction is exothermic. Using Van't Hoff's equation, the reaction enthalpy was determined and yielded a ΔrH value of −95.1 kJ per mole of hexaniobate.
 | ||
| Fig. 7 Effect of temperature on the protonation constant of Nb6O198−. I = 3 M (KCl/KOH). The error bars correspond to ±0.1 log unit. | ||
Discussion
Even though hexaniobate and hexatantalate ions have been known for a long time,4,5 their UV-vis properties are still poorly investigated. To our knowledge, the only published data mentioning the UV properties of Nb and Ta Lindqvist ions are graphical.5,22–24,27 Extrapolation of the molar absorption coefficient from these pioneer studies revealed a huge discrepancy as shown in Table 1. Thus, a precise determination of the extinction coefficients was needed in order to develop UV-based methods for probing the hexaniobate and hexatantalate solution thermodynamics. The strong discrepancy between literature data is surprising but might be imputable to the titration methods used for niobium at the time like ignition and weighting of the sample as Nb2O5 which can be easily biased in presence of alkali ions (A) due to the formation of ANbO3.| pH of measurement | Species | λmax (nm) | εa (L mol−1 cm−1) | Reference |
|---|---|---|---|---|
| a Value calculated from the metal concentration, path length and absorbance published in the original articles. Absorbance values and wavelengths were determined with PlotDigitizer software. | ||||
| 9.9–11.2 | — | ∼233 | ∼25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
4 |
| 13 | — | ∼243 | ∼24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
4 |
| 14 | — | ∼248 | ∼29![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
4 |
| 13.5 | — | 242 | ∼10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900 900 |
22 |
| 10.2–11.5 | H3Nb6O195− | ∼235 | 3200 | 23 |
| 10.2–11.5 | H2Nb6O196− | ∼232 | 3000 | 23 |
| 10.2–11.5 | HNb6O197− | ∼235 | 2400 | 23 |
| Nb6O198− | ∼248 | 3000 | 23 | |
| 8.5 | — | 230 | ∼12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
27 |
| 12–14.6 | HNb6O197− | 240.5 | 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 ± 400 300 ± 400 |
This work |
| 12–14.6 | Nb6O198− | 247.5 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900 ± 600 900 ± 600 |
This work |
| 12.2 | Ta6O198− | — | 3600 at 230 nm | 5 |
| 12–15 | Ta6O198− | — | 4000 ± 150 at 230 nm | This work |
The strong absorbance band observed for HxNb6O19x−8 ions varies with the pH due to the protonation or deprotonation. The strongest absorbance is observed at hydroxide concentration higher than 3 M where the cluster is fully deprotonated. Therefore, the isolation of Nb6O198− under high alkaline conditions allows the direct determination of its molar absorptivity. In a prior study, Spinner and co-workers23 reported that after 3 days and at pH ≥ 14, hexaniobate ions are transformed into the tetrameric species Nb4O12(OH)48− and Nb4O1612−. This assumption was based on spectrophotometric measurements. We did not observe significant variations on the UV spectra over time, even after several days in solutions containing up to 9 M KOH (Fig. S1†). It should be noted that hexaniobate salts can be synthesized in very basic conditions such as alkaline fusion so it would be very unlikely that the Lindqvist ion is unstable at high pH. The same group also reported extinction coefficient values for HxNb6O19x−8 ions largely underestimated as highlighted in Table 1. In one hand, the formation of tetrameric species in solution was never confirmed by another group but Spinner's study was mentioned in few literature reviews.26,28,29 In the other hand, Nb6O198− was observed in solutions at pH higher than 14 by multiple techniques (Raman, SAXS, NMR).30–32 Consequently, the existence of tetrameric niobates seems rather unlikely.
The strong absorbance that exhibits Nb6O198− offers an easy and cheap way of titrating niobium solutions. Several UV-based methods have been developed previously for Nb(V) containing samples but they all require expensive or toxic complexing agents and none of them were applied to alkaline samples.33–36 Moreover, these methods often contain a liquid–liquid extraction step prior to the spectrophotometric measurement. After determining precisely the extinction coefficient of Nb6O198− ion, acidic samples of niobium oxalate and niobium chloride could be titrated by their dilution in 4 M KOH. In chloride media, Nb(V) forms chloro–oxo ions [NbOm(OH)nCll]5−2m−n−l whereas solutions of commercial Nb(HC2O4)5, nH2O contains both [NbO(C2O4)3]3− and [NbO(H2O)(C2O4)3]−.30,37 [NbOm(OH)nCll]5−2m−n−l, [NbO(C2O4)3]3− and [NbO(H2O)(C2O4)2]− ions are readily transformed into hexaniobate ions since (i) the strong absorbance band at 247.5 nm is observed after dilution of niobium chloride or niobium oxalate solutions in alkaline media, and (ii) the Nb contents determined by spectrophotometric titration match those determined by ICP-AES. This could open ways for new synthetic methods of hexaniobate salts under mild aqueous conditions and using commercially available materials since it is not the case for Nb2O5, nH2O used in many synthesis. The method developed (dilution in 4 M KOH and UV titration) could also be useful for the determination of Nb content in hydrous niobium oxide Nb2O5, nH2O which is known to be soluble in alkaline solutions8 and used in several niobium studies.38 Our UV-based method was also applied to industrial samples of hexaniobates since alkaline media are catching growing attention in Nb and Ta recovery processes. The Nb samples were slightly contaminated by tantalum (Nb/Ta = 80 mol mol−1) but due to the low absorbance properties of Ta6O198− compared to Nb6O198− (εNb6O19/εTa6O19 = 80 at 247.5 nm), the niobium concentration can still be determined by UV spectrophotometry. Thus, UV titrations of niobium can be helpful in the development of hydrometallurgical processes that aim at recovering or purifying niobium from ores.
Previous studies revealed that Nb6O198− and Ta6O198− clusters can be protonated. The protonation constants reported for these ions are given in Table 2. All reported data on hexaniobate ions are derived from potentiometric titrations, and due to the high value of the first protonation constant (>12.5), the authors had to extrapolate it. At an ionic strength of 3 M KCl/KOH, the value determined in the present study is in the range of the previously extrapolated values. It should be underlined that the pKa reported by Spinner25 is about 1 log unit lower than those reported by Neumann, Etxebarria et al. and this work.
| i | log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) βi for reaction Nb6O198− + iH+ = HiNb6O19i−8 βi for reaction Nb6O198− + iH+ = HiNb6O19i−8 |
|||
|---|---|---|---|---|
| a Potentiometric measurements. | ||||
| 1 | 13.8 ± 0.2 | 12.6 ± 0.1 | 13.63 ± 0.04 | 13.4 ± 0.1 |
| 2 | 24.68 ± 0.05 | 23.46 ± 0.15 | 23.55 ± 0.04 | — |
| 3 | — | 32.85 ± 0.17 | 32.90 ± 0.07 | — |
| 3 M KCl, T = ? | 3 M KCl | 3 M KCl | 3 M KCl | |
| 10.7 ≤ pH ≤ 12.4 | 25 °C | 25 °C | 25 °C | |
| 8 ≤ pH ≤ 12.1 | 9.7 ≤ pH ≤ 13.1 | 12 ≤ pH ≤ 14.5 | ||
| Pot.a | Pot.a | Pot.a | UV-vis | |
| Ref. | 24 | 25 | 26 | This work |
The protonation constant measured at different ionic strengths in K+ containing media shows a decrease of this constant when the potassium concentration increases (Fig. 6). The trend is opposed to what was observed by Spinner et al. but, as explained above, their pioneer studies seem doubtful. Interactions between hexaniobate and K+, Rb+ or Cs+ ions at very high pH values have been highlighted by the Nyman's group based on small-angle X-ray scattering experiments.31,39 The authors reported the formation of the solvent-shared ion-pairs K8Nb6O19 and [K10Nb6O19]2+ in 3 M potassium hydroxide solutions. In the solid state, the eight K+ ions are associated by the cluster faces and edges through the bridging oxygens (Fig. 1).8 It has also been proved by multinuclear NMR studies that the protonation of Nb6O198−, in both the solid state and solution, occurs at the bridging oxygens and not at the terminal oxygens.40 This was also checked by our DFT computations: while the addition of one proton on a terminal oxygen leads to structures that are very difficult to stabilize, the binding of a proton on a bridging oxygen is easily realized and leads to a structure consistent with expectations. Therefore, high alkali concentration should facilitate the formation of non-protonated Nb6O198− yielding a lower protonation constant.
Due to low solubility of sodium hexaniobate salts and the alkaline error in Na+ media, the previous potentiometric studies were not performed in such media (Table 2). The low Nb concentration required by UV titration ([Nb6O19] = 1 to 5 × 10−5 M) allowed us to investigate the protonation of Nb6O198− in Na+ media. The measurements in NaCl/NaOH media yielded lower protonation constants than in KOH/KCl media (Fig. 6). It is well-known that ion-pairing between alkali and hexaniobate ions follows the trend Cs > Rb > K > Na > Li.3,39 Contrary to Li+ and Na+ media, the strong ion-pairing effect in K+, Rb+ and Cs+ media allows the stabilization of protonated clusters as it is visible in the solid state. Indeed, di-protonated and mono-protonated hexaniobate salts can be synthesized with potassium, rubidium and cesium8,41 whereas di-protonated sodium hexaniobate and protonated lithium hexaniobate have never been isolated. Thus, HxNb6O19x−8 ions are easier to deprotonate in Li+ and Na+ than in K+, Rb+ and Cs+ media. This implies that proton equilibria for hexaniobate clusters should be seen as an exchange between alkali ions and protons rather than a simple proton release. Nonetheless, the stoichiometry and formation constants of the ion-pairs formed between K+ and HNb6O197− are still unknown so that the current acid–base model cannot take into account the ion-pairing effect.
For HxTa6O19x−8, no changes where observed on the UV spectra at pH ≥ 12 indicating that the tantalum cluster is fully deprotonated under these conditions. The published values on the first protonation constant of Ta6O198− are largely dispersed compared to those for Nb6O198− (Table 2). Our observations are in agreement with the most recent study performed by Balogh et al.7 Knowing that a protonated hexatantalate salt has never been isolated by aqueous synthesis, the pKa values of 12.68 and 13.89, extrapolated from potentiometric measurements by Arana et al.42 and Spinner and Kheddar,43 respectively, seem overestimated. To support this conclusion, the proton transfer was studied by DFT computations. More precisely, the thermodynamic quantities of reaction (3) were computed.
| HNb6O19(aq)7− + Ta6O19(aq)8− = Nb6O19(aq)8− + HTa6O19(aq)7− | (3) |
Interestingly, ΔrGo = 7.1 kJ mol−1 is obtained (as explained in computational details, the energetic contribution to this quantity is computed at the Def2-TZVP level while the entropic contribution is computed with a smaller model). For the sake of clarity, ΔrGo should be expressed in terms of pKa difference under the form pKa(HNb6O197−/Nb6O198−) − pKa(HTa6O197−/Ta6O198−) = 1.3. Considering the precision of the methods used, this value is in good agreement with the pKa's determined experimentally in this work and by Balogh et al. whose value is 1.9 (Table 2), thus, confirming the more difficult protonation of the hexatantalate versus the hexaniobate. These computational results are also consistent with those obtained in previous works.7
The effect of temperature on the protonation of Nb6O198− was evaluated (Fig. 7). The protonation reaction was found exothermic with a reaction enthalpy of ΔrH = −95.1 kJ per mole of hexaniobate. Consequently, the non-protonated species is favored when the temperature increases. The protonation constant was shifted by almost 1 log unit between 20 °C and 40 °C highlighting the importance of this parameter during the synthesis of hexaniobate salts and for separation processes. The temperature effect could also explain the slightly higher value reported in Neumann's study which was performed at room temperature (Table 2).
The tremendous difference between the UV spectra of the isostructural ions Nb6O198− and Ta6O198− was investigated by DFT calculations. The UV spectra were computed for each species and are depicted in Fig. 8. A good agreement is obtained between the computed and experimental spectra as shown by the comparison between Fig. 5 and 8 (the wavelengths domain is more extended on the computed spectra than on the experimental one). Especially, the computed absorption is much lower in the case of hexatantalate from 230 nm. It should be noticed than the convoluted bands originates from several transitions; to give more insights on this aspect, the nature of the main transitions are reported in Table 3.
 | ||
| Fig. 8 Computed transitions (orange) and convoluted spectra (blue) for Nb6O198− (a) and Ta6O198− (b) ions. The scale is the same for both spectra. | ||
| Species | Transition wavelength (nm) | Oscillator strength | Nature of the transition |
|---|---|---|---|
| a Several transitions are computed around 199 nm that justifies the general features of the convoluted spectrum for hexatantalate. | |||
| Nb6O198− | 248.3 | 0.135 | n(μ2O) → π*(Nb–μ2O) |
| 248.1 | 0.136 | n(μ2O) → π*(Nb–μ2O) | |
| 248.1 | 0.136 | n(μ2O) → π*(Nb–μ2O) | |
| 222.8 | 0.140 | n(O (all)) → n(μ2O) | |
| 222.7 | 0.136 | n(O (all)) → n(μ2O) | |
| 222.7 | 0.136 | n(O (all)) → n(μ2O) | |
| Ta6O198−a | 215.4 | 0.118 | n(μ2O) → π*(Ta–μ2O) |
| 215.3 | 0.118 | n(μ2O) → π*(Ta–μ2O) | |
| 215.2 | 0.115 | n(μ2O) → π*(Ta–μ2O) | |
The precise investigation of the transitions explains two important aspects of the measured spectra. First, the apparent lack of absorption of the hexatantalate is simply explained by a blue-shift of the transitions whose lowest absorption bands emerge from. Indeed, the transitions displace electron density from bridging oxygens to π* orbitals mainly localized on the metal atom. The energetic level being higher in Ta(V) than in Nb(V) (the latter is above than the former in the periodic table), the transition in consistently blue-shifted in Ta vs. Nb. Second, the effect of the protonation of hexaniobate (reported in Fig. 5) consists mainly in a blue-shift of the measured band. Once again it should be explained while considering the nature of the transition: the protonation of μ2O (justified above) leads to a lower possibility for the electron density around those atoms to be implied in transitions, then to a blue-shift of the absorption band.
Conclusions
The hexaniobate ions, HNb6O197− and Nb6O198−, exhibit a strong absorbance band in the UV region 230–250 nm. Taking advantage of the different UV-vis properties of Nb6O198− and Ta6O198−, a cheap and fast method was developed to determine the Nb content of both synthetic and industrial samples. For the first time, the protonation constant of Nb6O198− has been determined experimentally. The nature of the electrolyte and the temperature were found to play a major role on the protonation of Nb6O198− highlighting the need for further investigations on the ion-pairing between alkali ions and polyoxometalates. Due to the large uncertainties on previously reported studies, the absorbance properties and the protonation of the isostructural ions Ta6O198− and Nb6O198− were investigated by quantum-chemistry-based methods and corroborate the experimental findings. The UV properties of the hexaniobate ions could help to develop UV-based methods for probing the solution thermodynamics of Nb(V) in alkaline media. We are currently exploiting the spectral properties of HxNb6O19x−8 and HxTa6O19x−8 to study alkaline solutions of Nb(V) and Ta(V) by capillary electrophoresis.Acknowledgements
This work was granted access to the HPC resources of CINES under the allocation 2014086933 made by GENCI (Grand Equipement National de Calcul Intensif). We also thank the CRI (Centre de Ressources Informatiques) of the Université de Lille for providing computing time for part of the theoretical calculations. The experimental part of this work was financed by Eramet Research (France).Notes and references
- Z. Zhu and C. Y. Cheng, Hydrometallurgy, 2011, 107, 1–12 CrossRef CAS PubMed.
- X. Wang, S. Zheng, H. Xu and Y. Zhang, Hydrometallurgy, 2009, 98, 219–223 CrossRef CAS PubMed.
- M. Nyman, Dalton Trans., 2011, 40, 8049 RSC.
- G. Jander and D. Ertel, J. Inorg. Nucl. Chem., 1960, 14, 71–76 CrossRef CAS.
- H. W. Nelson and S. Tobias, Inorg. Chem., 1963, 2, 985–992 CrossRef.
- R. t. Shannon, Acta Crystallogr., Sect. A: Cryst. Phys., Diffr., Theor. Gen. Crystallogr., 1976, 32, 751–767 CrossRef.
- E. Balogh, T. M. Anderson, J. R. Rustad, M. Nyman and W. H. Casey, Inorg. Chem., 2007, 46, 7032–7039 CrossRef CAS PubMed.
- M. Nyman, T. M. Alam, F. Bonhomme, M. A. Rodriguez, C. S. Frazer and M. E. Welk, J. Cluster Sci., 2006, 17, 197–219 CrossRef CAS PubMed.
- P. Gans, A. Sabatini and A. Vacca, Talanta, 1996, 1739–1753 CrossRef CAS.
- M. Sturzbecher-Hoehne, C. Ng Pak Leung, A. D'Aléo, B. Kullgren, A.-L. Prigent, D. K. Shuh, K. N. Raymond and R. J. Abergel, Dalton Trans., 2011, 40, 8340 RSC.
- G. J.-P. Deblonde, M. Sturzbecher-Hoehne and R. J. Abergel, Inorg. Chem., 2013, 52, 8805–8811 CrossRef CAS PubMed.
- M. Sturzbecher-Hoehne, G. J.-P. Deblonde and R. J. Abergel, Radiochim. Acta, 2013, 101, 359–366 CrossRef CAS.
- F. H. Sweeton, R. E. Mesmer and C. F. Baes Jr, J. Solution Chem., 1974, 3, 191–214 CrossRef CAS.
- I. Kron, S. L. Marshall, P. M. May, G. Hefter and E. Königsberger, Monatshefte für Chemie, 1995, 126, 819–837 CrossRef CAS.
- J. P. Perdew, M. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865–3868 CrossRef CAS.
- C. Adamo and V. Barone, J. Chem. Phys., 1999, 110, 6158 CrossRef CAS PubMed.
- M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery Jr, J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, M. J. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, Ö. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski and D. J. Fox, Gaussian 09, Revision D.01, Gaussian, Inc., Wallingford CT, 2009 Search PubMed.
- T. H. Dunning Jr and P. J. Hay, in Modern Theoritical Chemistry, ed. H. F. Schafer, New-York, 1977, vol. 3, pp. 1–28 Search PubMed.
- M. Dolg, H. Stoll, H. Preuss and R. M. Pitzer, J. Chem. Phys., 1993, 97, 5852 CrossRef CAS.
- F. Weigend and R. Ahlrichs, Phys. Chem. Chem. Phys., 2005, 7, 3297–3305 RSC.
- J. Tomasi, B. Mennucci and R. Cammi, Chem. Rev., 2005, 105, 2999–3094 CrossRef CAS PubMed.
- G. I. Gridchina, Russ. J. Inorg. Chem., 1963, 8, 322–326 Search PubMed.
- A. Goiffon, R. Granger, C. Bockel and B. Spinner, Rev. Chim. Miner., 1973, 10, 487 CAS.
- G. Neumann, Acta Chem. Scand., 1964, 18, 278–280 CrossRef CAS PubMed.
- B. Spinner, Rev. Chim. Miner., 1968, 839–868 CAS.
- N. Etxebarria, L. A. Fernandez and J. M. Madariaga, J. Chem. Soc., Dalton Trans., 1994, 3055 RSC.
- J. R. Black, M. Nyman and W. H. Casey, J. Am. Chem. Soc., 2006, 128, 14712–14720 CrossRef CAS PubMed.
- F. Sahureka, R. C. Burns and E. I. von Nagy-Felsobuki, Inorg. Chim. Acta, 2003, 351, 69–78 CrossRef CAS.
- G. M. Rozantsev, O. I. Dotsenko and G. V. Taradina, Russ. J. Coord. Chem., 2000, 26, 264–270 Search PubMed.
- J.-M. Jehng and I. E. Wachs, J. Raman Spectrosc., 1991, 22, 83–89 CrossRef CAS.
- M. R. Antonio, M. Nyman and T. M. Anderson, Angew. Chem., Int. Ed., 2009, 48, 6136–6140 CrossRef CAS PubMed.
- G. K. Goh, F. F. Lange, S. M. Haile and C. G. Levi, J. Mater. Res., 2003, 18, 338–345 CrossRef CAS.
- K. Patel, S. K. Menon and Y. K. Agrawal, Microchem. J., 1996, 53, 158–163 CrossRef CAS.
- A. K. Chhakkar and L. R. Kakkar, Fresenius. J. Anal. Chem., 1995, 351, 720–723 CrossRef CAS.
- N. Agnihotri, R. Kamal and J. R. Mehta, Ann. Chim., 2006, 96, 479–485 CrossRef CAS.
- N. Agnihotri and J. R. Mehta, Chem. Anal., 2005, 50, 429 CAS.
- V. Pershina, Radiochim. Acta, 1998, 80, 75–84 CAS.
- K. Nakajima, Y. Baba, R. Noma, M. Kitano, J. N. Kondo, S. Hayashi and M. Hara, J. Am. Chem. Soc., 2011, 133, 4224–4227 CrossRef CAS PubMed.
- M. K. Kinnan, W. R. Creasy, L. B. Fullmer, H. L. Schreuder-Gibson and M. Nyman, Eur. J. Inorg. Chem., 2014, 2014, 2361–2367 CrossRef CAS.
- T. M. Alam, M. Nyman, B. R. Cherry, J. M. Segall and L. E. Lybarger, J. Am. Chem. Soc., 2004, 126, 5610–5620 CrossRef CAS PubMed.
- I. Santos, L. H. Loureiro, M. F. P. Silva and A. Cavaleiro, Polyhedron, 2002, 21, 2009–2015 CrossRef CAS.
- G. Arana, N. Etxebarria, L. A. Fernandez and J. M. Madariaga, J. Solution Chem., 1995, 24, 611–622 CrossRef CAS.
- B. Spinner and N. Kheddar, C. R. Seances Acad. Sci., Ser. C, 1969, t. 269, 1108–1111 Search PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra14866e |
| This journal is © The Royal Society of Chemistry 2015 |



