An imidazo-phenanthroline scaffold enables both chromogenic Fe(ii) and fluorogenic Zn(ii) detection†
Abstract
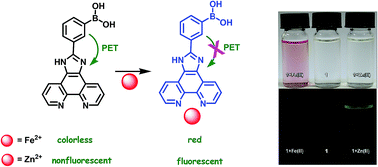
A novel, simple and efficient dual channel probe built on an imidazo-phenanthroline scaffold with a boronic acid unit, viz. 3-(1H-imidazo[4,5-f][1,10]phenanthrolin-2-yl)phenylboronic acid, is disclosed. It is found that this novel probe induces chromogenic and fluorogenic responses to Fe(II) and Zn(II) ions, respectively. To our best knowledge, this is one of the rare examples of dual channel responsive probes that can be used for visual detection of Fe(II) and turn-on fluorogenic detection of Zn(II) ions, simultaneously.

 Please wait while we load your content...
Please wait while we load your content...