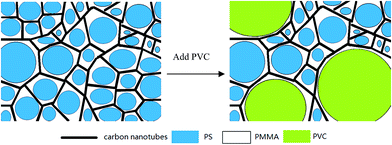
Self-assembly of miscible homopolymer/quasi-block copolymer blends/MWNT composites: a strategy to obtain ultralow electrical percolation threshold and mechanism†
Ri Xu and
Xuecheng Xu*
Department of Physics, East China Normal University, 500 DongChuan Road, Shanghai, China. E-mail: xcxu@phy.ecnu.edu.cn
First published on 26th January 2015
Abstract
Herein we developed a strategy to lower the electrical percolation threshold by the self-assembly of miscible polymer blends of homopolymer/quasi-block copolymer blends/CNT composites. Orderly formation of CNT networks and p–π interaction was observed. Further explorations of the variations of miscibility and segmental motion are required.
Electrically conductive polymer/MWNTs composites have received constant and extensive attention due to their applications in a wide range of electronic devices, with the main purpose of lowering the percolation threshold and achieving higher maximum conductivity.1
Until recently, ultralow percolation threshold has been achieved by selective localization of carbon nanotubes, which can be divided into 2 sets: one is the well-known double percolation concept based on melt-mixed immiscible polymer blends,2–13 the other is to fabricate the sample by separating processing time points of compounding of partial polymer/CNTs and addition of pure polymer beads deliberately before compression molding.14,15
Moreover, some efforts have been devoted to improving compatibilities of polymer blends and a number of methods has been developed.16–18 While conducting polymer/nanoparticles composites focused on miscible polymers are barely explored, Taegyun Kwon and co-workers19 adopted CuPt nanorods as both conductive fillers and compatibilizers to modify the percolating morphology and lower the percolation threshold lately. And in our recent work, quasi-block copolymer/MWNTs composites gained better conductivity than immiscible blends under solution-based condition.20 In this work, we developed a solution-based self-assembling strategy to obtain miscible polymer blends of PVC (poly(vinylene chloride))/PMMA-qb-PS (poly(methyl methacrylate)-quasi-block-polystyrene)/MWNTs (PVC/qb/CNTs) composites with an ultralow percolation threshold of 0.071 wt%. It is concluded by Jamieson and coworkers21 that PMMA-b-PS and PVC are miscible polymers, which is also attested by our DSC curves (ESI Fig. 1†).
The conductive mechanism for quasi-block copolymer/CNTs composites based on double-percolation threshold was proposed by our latest work.20 In this sample design, a miscible polymer is added to form micro-phase separation in order to further self-assemble to promote the formation of CNTs networks, as depicted in Fig. 1. Besides, the possible electronic p–π interaction could be introduced by PVC,22 which would make contribution to the carrier mobility. The preparation of the composites is described in ESI.†
Miscibility could be affected by molecular weight of both qb and PVC. Table 1 shows the GPC results of the two polymers. According to the analysis of Jamieson and coworkers,21 this system should be miscible because the weight-average molecular weights are rather small.
| Name | ![[M with combining macron]](https://www.rsc.org/images/entities/i_char_004d_0304.gif) n n |
Mw max | Mw min | ![[M with combining macron]](https://www.rsc.org/images/entities/i_char_004d_0304.gif) w w |
PDI |
|---|---|---|---|---|---|
| Qb | 28![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 357 357 |
148![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 228 228 |
3446 | 46![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 786 786 |
1.65 |
| PVC | 66![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 970 970 |
359![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 758 758 |
9674 | 107![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 688 688 |
1.61 |
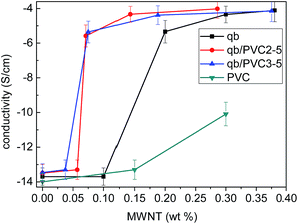
Fig. 2 shows PVC/qb/MWNTs, qb/MWNTs, PVC/MWNTs composites' conductivity variations with the MWNTs content. The minimum percolation threshold of approximately 0.071% in PVC/qb/MWNTs composites appears at the PVC/qb mass ratio of 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, 5
2, 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, while qb/MWNTs composite has percolation threshold of 0.20% and PVC/MWNTs of more than 0.30%.
3, while qb/MWNTs composite has percolation threshold of 0.20% and PVC/MWNTs of more than 0.30%.
Fig. 3(a and b) shows the cross-section of 0.071% qb/PVC(2–5) and (c and d) that of 0.148% qb/PVC(2–5). Since PVC and qb are miscible, etching cannot be used in this kind of composite. In 0.071% qb/PVC(2–5) (Fig. 3(a and b)), the CNTs disperse in the framed network in the continuous phase qb as reported,20 with 300–500 nm radii PVC “archipelagos” surrounded by its miscible PMMA-segment.21 The “archipelagos” of PVC transform into big ellipsoid “isolated islands” with average radius of 1.5 μm as CNTs content grows to 0.148% (Fig. 3(c and d)). The CNTs content in this cross-section seems to be superfluous. The interfacial width rises dramatically, illustrating that the compatibility between miscible polymer blends lessens as CNTs are added, which should be considered to be ascribable to diminishment of the segmental motion of PMMA when adding CNTs, while the DSC results reveal that the glass transition temperature of the composites decreased by 55 °C from non-CNTs-blends to 0.148% PVC/qb/CNTs (ESI Fig. 1†), which means the segmental motion was improved by CNTs in this composite. Therefore, this phenomenon awaits to be further explored.
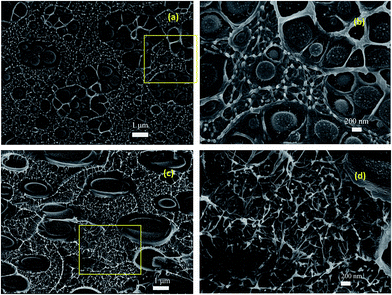 | ||
| Fig. 3 SEM image of cross-section of (a and b) 0.071% qb/PVC(2–5); (c and d) 0.148% qb/PVC(2–5). (b and d) are magnified images of (a and c). | ||
Table 2 shows parameters of the XPS spectra and peak fitting of qb/PVC(2–5)/MWNTs composites at MWNTs content of 0.057% (before percolation), 0.071% (during percolation), 0.286% (after percolation) (ESI Fig. S2†). C 1s spectra are on the left side, Cl 2p3 spectra are on the right side, corresponding to XPS parameters. Before the formation of CNTs networks, by comparison of ESI Fig. S2(a–f),† C 1s and Cl 2p3 peaks have only 2 sub-peaks, that is, C–Cl + C–O sub-peak and C–C chain sub-peak. C–Cl + C–O peak represents the situation of interaction between PVC and PMMA-segments because C–Cl and C–O bonds share the same peak.23 Cl 2p3 peak can be sub-peaked into the one interacting with PMMA-segments (peak 1) and the one doesn't (peak 2). After the CNTs network formation (ESI Fig. S2(c–f)†), a new sub-peak appears in both C 1s and Cl 2p3 spectra, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O peak in C 1s spectrum and Cl⋯qb/CNTs interaction peak in Cl 2p3 (peak 3).
O peak in C 1s spectrum and Cl⋯qb/CNTs interaction peak in Cl 2p3 (peak 3).
| Peak | Binding energy (eV) | FWHM (eV) | Atomic% | Peak | Binding Energy (eV) | FWHM (eV) | Atomic% | |
|---|---|---|---|---|---|---|---|---|
| 0.057% | C–C | 285.5 | 2.14 | 88.4% | C–Cl(2) | 201.2 | 2.27 | 81.8% |
| C–Cl + C–O | 287.6 | 1.70 | 11.6% | Cl⋯PMMA(1) | 202.8 | 1.71 | 18.2% | |
| 0.071% | C–C | 286.4 | 2.93 | 89.2% | C–Cl(2) | 201.5 | 2.13 | 80.9% |
| C–Cl + C–O | 288.5 | 1.49 | 4.9% | Cl⋯PMMA(1) | 202.9 | 1.02 | 6.4% | |
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O O |
290.5 | 1.97 | 5.9% | Cl⋯qb/CNT(3) | 203.5 | 1.44 | 12.6% | |
| 0.286% | C–C | 285.5 | 2.13 | 86.5% | C–Cl(2) | 200.5 | 2.05 | 71.9% |
| C–Cl + C–O | 287.3 | 2.00 | 10.9% | Cl⋯PMMA(1) | 202.0 | 1.64 | 24.2% | |
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O O |
289.0 | 1.35 | 2.6% | Cl⋯qb/CNT(3) | 203.2 | 1.12 | 3.9% |
After the formation of CNTs networks, as the CNTs content grows, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O peak and peak 3 diminishes, suggesting that CNTs' more doping effect to PMMA-segments20 and interaction between PVC and qb/CNTs are weakened; peak intensity of C–Cl + C–O rarely changes but the peak is widen and the area increases, implying that the compatibility between PVC and PMMA-segments decreases with more potent doping of CNTs (ESI Fig. S2(c and e)†), which is in accordance with the PVC's islands' transformation. Peak 1 enlarges, which indicates that PVC has more electronic interaction with PMMA-segments at the interface, corresponding to the broadening of the interfacial width between PVC and qb (Fig. 3).
O peak and peak 3 diminishes, suggesting that CNTs' more doping effect to PMMA-segments20 and interaction between PVC and qb/CNTs are weakened; peak intensity of C–Cl + C–O rarely changes but the peak is widen and the area increases, implying that the compatibility between PVC and PMMA-segments decreases with more potent doping of CNTs (ESI Fig. S2(c and e)†), which is in accordance with the PVC's islands' transformation. Peak 1 enlarges, which indicates that PVC has more electronic interaction with PMMA-segments at the interface, corresponding to the broadening of the interfacial width between PVC and qb (Fig. 3).
C–C peak position increases to 286.4 eV from 285.5 eV (ESI Fig. S2(a and c)†), as network formation is essential to the enhancement of carrier concentration. The C–C peak from C 1s and C–Cl (peak 2) from Cl 2p3 peak both witness a drop by 1 eV, which means after the CNTs network formation the lone pair electrons from Cl atoms on the interface between PVC and qb begin to interact with affluent π-electrons in CNTs as the CNTs content grows, that is, p–π interaction. This effect induces enhancement of carrier mobility, which improves the electrical conductivity further.
Conclusions
In this paper, we added 5/7 wt% of miscible homopolymer into quasi-block-copolymer/MWNTs and obtained ultralow electrical percolation threshold of 0.071 wt%. Self-assembly of the blocks and miscible polymers transpired in NMP solution and further improvement of selective localization of CNTs orderly network frame in the composites was achieved. Carrier concentration enhanced dramatically at the point of CNTs network formation and carrier mobility increased as the CNTs content continues to grow due to p–π interaction between electron lone-pairs around Cl atoms and CNTs π-electrons at the interface. Since conductive miscible polymer blends/CNTs composites were scarcely explored, and miscible polymers are found to be easily mixed in ambient condition by solvents, this strategy is worth proposed.On the other hand, we found that the miscibility of PVC/PMMA-qb-PS blends is slightly reduced as the CNTs content grows, which is against the conclusion with immiscible polymer blends/nanoparticles that nanoparticle fillers can be used as compatibilizers. Moreover, glass transition temperature decreases dramatically as CNTs content grows, which means segmental motion may be stronger with more CNTs. Therefore, further investigation in relevant experiment and theory could be ameliorated due to the conclusion in this article.
Notes and references
- W. Bauhofer and J. Z. Kovacs, Compos. Sci. Technol., 2009, 69, 1486–1498 CrossRef CAS PubMed.
- Z. Xu, Y. Zhang, Z. Wang, N. Sun and H. Li, ACS Appl. Mater. Interfaces, 2011, 3, 4858–4864 CAS.
- R. Jung, W.-I. Park, S.-M. Kwon, H.-S. Kim and H.-J. Jin, Polymer, 2008, 49, 2071–2076 CrossRef CAS PubMed.
- J.-F. Gao, D.-X. Yan, B. Yuan, H.-D. Huang and Z.-M. Li, Compos. Sci. Technol., 2010, 70, 1973–1979 CrossRef CAS PubMed.
- P. Pötschke, A. R. Bhattacharyya and A. Janke, Polymer, 2003, 44, 8061–8069 CrossRef PubMed.
- C. Su, L. Xu, C. Zhang and J. Zhu, Compos. Sci. Technol., 2011, 71, 1016–1021 CrossRef CAS PubMed.
- D. Yan and G. Yang, Mater. Lett., 2009, 63, 1900–1903 CrossRef CAS PubMed.
- C. Zhang, H. Han and X. Yi, Compos. Interfaces, 1999, 6, 227–236 CAS.
- E. Cohen, L. Zonder, A. Ophir, S. Kenig, S. McCarthy, C. Barry and J. Mead, Macromolecules, 2013, 46, 1851–1859 CrossRef CAS.
- L. Zhang, C. Wan and Y. Zhang, Compos. Sci. Technol., 2009, 69, 2212–2217 CrossRef CAS PubMed.
- S. Bose, A. R. Bhattacharyya, R. A. Khare, S. S. Kamath and A. R. Kulkarni, Polym. Eng. Sci., 2011, 51, 1987–2000 CAS.
- C. Mao, Y. Zhu and W. Jiang, ACS Appl. Mater. Interfaces, 2012, 4, 5281–5286 CAS.
- L. Zonder, A. Ophir, S. Kenig and S. McCarthy, Polymer, 2011, 52, 5085–5091 CrossRef CAS PubMed.
- S. Maiti, S. Suin, N. K. Shrivastava and B. B. Khatua, RSC Adv., 2014, 4, 7979–7990 RSC.
- N. Shrivastava, S. Suin, S. Maiti and K. Bhanu, Ind. Eng. Chem. Res., 2013, 52, 2858–2868 CrossRef CAS.
- N. P. Panapitiya, S. N. Wijenayake, Y. Huang, D. Bushdiecker, D. Nguyen, C. Ratanawanate, G. J. Kalaw, C. J. Gilpin, I. H. Musselman, K. J. Balkus and J. P. Ferraris, Polymer, 2014, 55, 2028–2034 CrossRef CAS PubMed.
- A. Taguet, P. Cassagnau and J.-M. Lopez-Cuesta, Prog. Polym. Sci., 2014, 39, 1526–1563 CrossRef CAS PubMed.
- T. Kwon, T. Kim, F. binti Ali, D. J. Kang, M. Yoo, J. Bang, W. Lee and B. J. Kim, Macromolecules, 2011, 44, 9852–9862 CrossRef CAS.
- T. Kwon, K. H. Ku, D. J. Kang, W. B. Lee and B. J. Kim, ACS Macro Lett., 2014, 3, 398–404 CrossRef CAS.
- R. Xu and X. Xu, RSC Adv., 2014, 4, 42226–42233 RSC.
- C. Prahsarn and A. M. Jamieson, Polymer, 1997, 38, 1273–1283 CrossRef CAS.
- B. Yu and X. Xu, RSC Adv., 2014, 4, 3966 RSC.
- K. Artyushkova and J. E. Fulghum, Surf. Interface Anal., 2001, 31, 352–361 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra13689f |
| This journal is © The Royal Society of Chemistry 2015 |