Evaluation and modeling of a newly designed impinging stream photoreactor equipped with a TiO2 coated fiberglass cloth
Morteza Jafarikojoura,
Mohammad Moein Mohammadib,
Morteza Sohrabi*ac and
Sayed Javid Royaeed
aChemical Engineering Department, Amirkabir University of Technology, Tehran, Iran. E-mail: Sohrabi@aut.ac.ir
bDepartment of Chemical and Petroleum Engineering, Sharif University of Technology, Tehran, Iran
cIran Academy of Sciences, Tehran, Iran
dPetroleum Refining Technology Development Division, Research Institute of Petroleum Industry, Tehran, Iran
First published on 23rd December 2014
Abstract
The photocatalytic degradation of p-nitrophenol (PNP) using TiO2 particles immobilized on a fiberglass cloth was investigated in a novel design of a photo-impinging stream reactor. A spray painted method has been used for the coating process. The structural properties of the immobilized sample were examined using X-ray diffraction and a scanning electron microscope (SEM). The photocatalytic degradation results showed the good performance of the reactor. The flow regime within the reactor was characterized and modeled by applying a liquid residence time distribution. A compartment model consisting of four continuous stirred regions was assigned to describe the flow pattern in the reactor. A Langmuir–Hinshelwood kinetic scheme has been used to describe the degradation of p-nitrophenol and to examine the behavior of the reaction system. A comparison between the sum of the square errors of the experimental results and those predicted by the model for PNP degradation revealed that a good agreement exists between the two sets of data.
1. Introduction
P-Nitrophenol (PNP), having a high stability and solubility in water is one of the more hazardous organic substances present in industrial wastewater. This compound is involved in the synthesis of a wide range of products such as pesticides, pharmaceuticals, plastics, azo dyes, explosives and solvents.1 PNP is known as a priority toxic pollutant by the United States Environmental Protection Agency (EPA) as it may cause blood disorders.2,3 Hence, the removal of PNP from industrial effluents is an essential practical problem.Among all wastewater treatment processes, heterogeneous photocatalytic oxidation with TiO2 as a photocatalyst has drawn much attention in elimination of toxic organic compounds.4 The advantages of photocatalysis include low operation temperature, low cost and relatively low energy consumption. These parameters have led the relevant application of photocatalysis to the stage of commercialization.5
However, two major issues in photocatalytic processes are mass and photon transfer limitations.6 To overcome photon transfer limitations, various photoreactors, such as bubble column reactors, spinning disk reactors, rotating disk reactors, monolith reactors, and fluidized bed reactors have been applied.7–11 An ideally intensified reactor, however, should be able to integrate both maximized light efficiency and minimized mass transfer resistance.
For this purpose, photo-impinging streams reactor configurations have attracted a number of studies in both academic and industrial areas.6,12,13 In such configurations, normally oppositely directed fluid streams are made to impinge with in a relatively narrow zone with a high turbulence intensity and as such excellent conditions for enhancing interphase mass-transfer rates are provided.6 The essential phenomenon in solid–fluid systems is the penetration of particles into the opposite streams, creating a longer mean residence time and higher relative velocity. Consequently, the solid particles' flow pattern, mixing intensity, photon adsorption, and transport characteristics may be significantly improved within such reactors.14 Some of the practical difficulties in the use of suspended solid–fluid photocatalytic processes in impinging streams systems are limited penetration of the radiation in the suspension, fouling of UV source due to the deposition of catalyst particles and separation of the fine solid particles from liquid. Immobilization of the catalyst on an inert support could be an effective practical solution.15 Consequently, a number of research efforts have been dedicated to the development of immobilized systems following various synthesis routes and support materials.16 So far all investigations on photocatalytic processes carried out in impinging streams reactors available in the literature have been deal with slurry systems with catalyst particles in suspension.12,14 Several materials as support for TiO2 immobilization, including glass beads,17 glass tubes,18 fiberglass,19 quartz,20 silica,21 activated carbon,22 aluminium,23 pumice stone,24 perlite granules,25 and stainless steel26 have been applied. As a promising support among these materials, fiberglass cloth (FGC) has become popular for its special characteristics such as lightweight, low cost, transparency for visible light and high stability against ultraviolet rays.27 In addition, fiberglass cloth can be cut into different sizes, and folded into many shapes.
Photocatalytic reactors are often fundamentally different from conventional reactors.28 Hence, determination of the residence time distributions (RTD) of materials and flow regimes within the reactor are some key information required for successful design, modelling and scale-up of the latter devices.29,30 On the basis of RTD data, it may be possible to simulate non-ideal systems by a configuration of ideal regions such as ideal perfect mixed and/or ideal plug flow reactors.14,28
Reactor modelling is sometimes performed based on certain flow regimes such as continuous stirred tank reactors in series.31 Evaluating reactor performance under conditions that mass and photon transfer limitations are not predominant, is an important requirement for the determination of intrinsic kinetic parameters independent from the reactor configuration.32 In order to provide a correlation between the predictions and the experimental data, an error function is required to enable the optimization procedure. The residual or sum of square error (SSE) of the estimate can be used to determine the goodness-of-fit.33
In this study, a new photo-impinging streams reactor using TiO2 coated fiberglass cloth was designed, constructed and modelled. The capability of the latter to degrade PNP in the liquid phase was assessed. X-ray diffraction (XRD) and scanning electron microscope (SEM) were used to examine the success of the procedure of the catalyst immobilization. In order to obtain a comprehensive insight into the reactor performance, effective control of the process and determination of the intrinsic kinetic coefficients independent from the reactor configuration, flow regime in photoreactor was characterized. In the first step, TiO2 was coated on fiberglass cloth using spray painted method and the sample was placed in the impingement zone of the reactor. Effects of PNP initial concentration and liquid flow rate on the reactor's performance were examined. Residence time distribution (RTD) of the fluid in the reactor was determined to characterize the photoreactor hydrodynamic behaviour. The reactor was then modelled, applying a cascade of continuous stirred tank reactors (CSTRs). The data predicted from the proposed model were used to calculate the kinetic coefficients for degradation reaction.
2. Experimental
2.1. Materials
Aeroxide® P25-TiO2 Powder (Evonik; BET surface area of 53.9 m2 g−1, pore volume of 0.172 m3 g−1, 70% anatase/30% rutile) was purchased from Evonik (Germany). Diethanolamine, Ethanol and p-nitrophenol were obtained from Merck Co. and used without further purification. Fiberglass cloth (FGC) was delivered by Kripa Co., India, and used as a support for the photocatalyst with the single fiber diameter of about 15 μm.2.2. Preparation of the immobilized TiO2
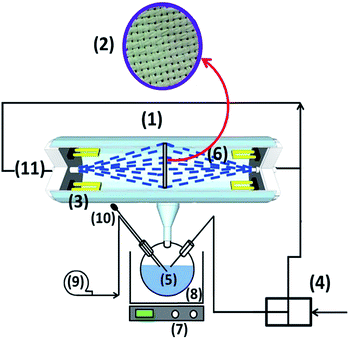
The procedure for spray painting of TiO2 has been described elsewhere.34 5 g of P25 was added to 25 mL of ethanol as the base medium of the slurry in which the titania powder can be properly dispersed. Subsequently, 5 mL of diethanolamine was added as a stabilizing agent to the slurry.35 This emulsion was sprayed onto the fiberglass cloth with a spray gun after the air pressure, slurry flow rate and distance of the fiber and gun were fixed at 4 bar, 10 μL and 10 cm, respectively. Such a technique, known as spray painted method, has been used before with minor changes and is normally applied as a standard method of coating.36,37 In order to achieve a controllable uniform immobilization, FGC was being moved in front of the spray gun by a linear step motor at the fixed speed of 1 cm s−1. A picture of the coating process is shown in Fig. 1. The substrate was then dried using a blow drier. Finally, the as-obtained samples were calcined at 400 °C for 1 h to enhance the adhesion strength of the films.2.3. Photoreactor design and photocatalytic degradation conditions
A schematic diagram of the photo-impinging streams reactor used in this study is shown in Fig. 2. The apparatus consisted of a cylindrical vessel made of pyrex glass and equipped with six low-pressure mercury vapor lamps, with a dominant emission line at 253.7 nm (TUV 5W from Philips Co.) as irradiation sources. Two pressure nozzles were mounted on the same axis in front of each other, spraying two jets of wastewater. FGC (exposed area of 87.5 cm2) was positioned in the middle of the reactor (the impingement zone). To start the process a gear pump was first switched on and the PNP solution (pH = 5.1), after passing through the two nozzles, was irradiated with UV light and collided in the impingement zone on TiO2 coated FGC. Before switching on the lamp, the process was being performed at dark for 30 min in order to reach adsorption/desorption equilibrium.PNP degradation was followed using Perkin-Elmer Lambda2S spectrophotometer. The concentration of PNP was determined by measuring the absorbance at 410 nm wavelength. These samples were prepared by mixing of 1 mL effluent with 5 mL of 0.1 mol L−1 NaOH and kept for 5 min.38 The PNP degradation ratio was evaluated from the following equation:
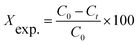 | (1) |
2.4. Characterization of TiO2 films
X-ray diffraction (XRD) analysis (INEL X-ray diffractometer, model EQuinox 3000, France) was used to detect and analyze the crystalline phases of the prepared samples using CuKα radiation in the 2θ range of 10° to 80°. The crystal size of TiO2 was calculated from the Scherrer relation.39 Scanning electron microscopy (SEM) measurements were performed with an ESEM XL30 Philips micro-scope, operating at 25 kV.2.5. RTD measurement
To characterize the liquid flow pattern in the photoreactor, the liquid residence time distribution curve was determined at the flow rate of 550 mL min−1. To generate a pulse function (Dirac delta function), a tracer (1 mL of 400 mg L−1 Reactive Black 8 dye) at time t = 0 was injected instantly into the inflow stream, using a syringe. Samples were collected at the outlet of the photoreactor at regular time intervals (Fig. 3). The collected samples were analyzed by a UV-visible spectrophotometer to determine the concentration of dye. The RTD or exit age distribution function (E) is defined as follows:40,41
 | (2) |
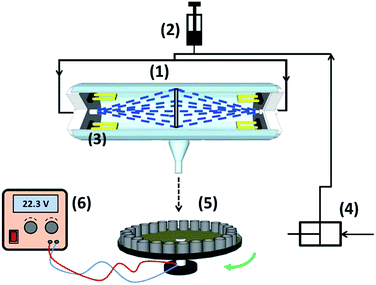 | ||
| Fig. 3 Schematic diagram of the photoreactor in the case of RTD. Study: (1) reaction vessel, (2) dye injection port, (3) UV lamps, (4) gear pump, (5) rotating disk for sampling, (6) power supplier. | ||
3. Results and discussion
3.1. Characterization of the immobilized TiO2 film on fiberglass
As shown in Fig. 4a, the FGC is made of woven fiberglass ribbons with of 15–20 μm width. The surface of the nude fiberglass is smooth. Fig. 4b–d displays the SEM images of the TiO2 films on FGC calcined at 400 °C. After coating, the surface of the fiberglass becomes coarse, indicating that the TiO2 has been successfully coated on the fiberglass cloth. It may be observed from images that the TiO2 film with a uniform surface has been successfully coated on fiberglass by the spray painted method and that TiO2 transformation into particles as a result of immobilization has been negligible. | ||
| Fig. 4 SEM images of nude fiberglass cloth – 500× (a) and TiO2 film coated on fiberglass cloth by spray painted method: (b) 500×, (c) 40×, (c) 5000× magnification. | ||
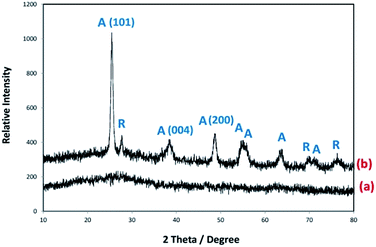
X-ray diffraction was performed in order to examine the crystalline phases of the coated samples. Fig. 5 shows the XRD patterns of P25 coated onto FGC and calcined at 400 °C. The results showed the diffraction peaks of anatase and rutile phases. Three distinctive diffraction peaks of anatase in TiO2 crystal structure are found mainly at 25.34°, 37.88°, and 48.10° corresponding to anatase (1 0 1), (0 0 4), and (2 0 0) crystal planes, respectively. The average crystallite size of TiO2 particles was calculated according to Scherrer's equation from (1 0 1) diffraction peak of anatase and was ca. 23 nm at 400 °C.
3.2. Photocatalytic degradation of p-nitrophenol
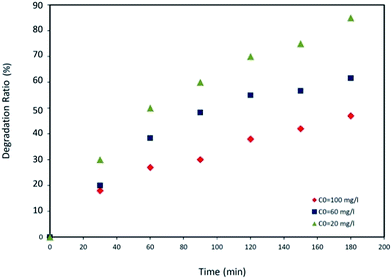
Three possible reasons can be provided to explain such an observation: First, at a high initial PNP concentration most active sites of the catalyst are occupied and fewer photons may reach the photocatalyst surface. In addition, a competition would be occurred between the contaminant and the rest molecules for adsorption at the active sites.43 Second, while initial PNP concentration is increased, the reactive species (˙OH) required for the degradation are also increased. Hence, the number of OH radicals necessary for degradation would be inadequate at higher concentrations of PNP. Third, an increase in PNP inlet concentration may enhance generation of intermediates which could be adsorbed at the surface of the catalyst. This phenomenon may lead to deactivation of active sites.44–47 Subsequently, PNP degradation rate is dropped as the inlet concentration of PNP is increased.
The absorption spectra obtained during the photocatalytic degradation of PNP (initial concentration of 20 mg L−1) is shown in Fig. 8. This figure shows that the characteristic absorption of PNP disappeared rapidly and implies that PNP has been effectively removed from the reaction solution. In photocatalytic degradation mechanisms of PNP the following intermediates, namely 4-nitrocatechol, hydroquinone and benzoquinone have been observed in previous studies.48,49 However, the absorbance peaks of these compounds have not been appeared in the absorption spectra. This could be related to the extremely low concentration of such intermediates.
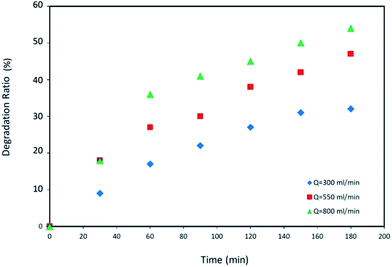
In addition, at each reaction cycle, PNP degradation was reported for three distinct flow rates. As it is observed, by increasing the number of reaction cycles, PNP degradation decreases under the same conditions. This effect may be attributed to the catalyst falling off at each cycle and hence reduction of active sites during the reaction time.
3.3. Photoreactor modeling
The RTD experiment was performed by measuring the concentration of tracer at one second time intervals using a flow rate of 550 mL min−1. The RTD curve was plotted applying eqn (1) (Fig. 11). Since RTD data are normally known at a number of discrete time intervals, the mean residence time (![[t with combining macron]](https://www.rsc.org/images/entities/i_char_0074_0304.gif) m), the variance of the residence time data (σ2) and the dimensionless variance (σθ2) are all expressed by the following relations:28
m), the variance of the residence time data (σ2) and the dimensionless variance (σθ2) are all expressed by the following relations:28
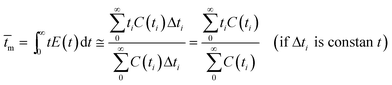 | (3) |
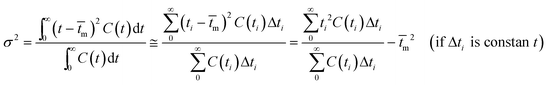 | (4) |
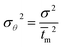 | (5) |
From eqn (3) and (5), mean residence time (![[t with combining macron]](https://www.rsc.org/images/entities/i_char_0074_0304.gif) ) and dimensionless variance (σθ2) were determined as 8.01 s and 0.264, respectively.
) and dimensionless variance (σθ2) were determined as 8.01 s and 0.264, respectively.
CSTRs in series model has been applied previously to simulate the non-ideal behavior of liquid streams in impinging steam reactors.31,41 In such a model, the actual volume of a photoreactor is divided by N equal sized ideal CSTRs (Fig. 10). The number of tanks is calculated from eqn (6).31
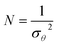 | (6) |
The number of CSTRs (N) representing the flow (550 mL min−1 flow rate) within the photoreactor in each cycle was obtained as 3.78 (≅4).
The expression for the theoretical RTD of this model can be predicted using Martin method via Gamma distribution function:50
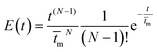 | (7) |
![[t with combining macron]](https://www.rsc.org/images/entities/i_char_0074_0304.gif) m is the mean residence time and N is total number of CSTRs. The predicted E(t) curve from the CSTRs in series model for a cascade of four tanks has been plotted in Fig. 11 and compared with the experimental data.
m is the mean residence time and N is total number of CSTRs. The predicted E(t) curve from the CSTRs in series model for a cascade of four tanks has been plotted in Fig. 11 and compared with the experimental data.
 | ||
| Fig. 11 Comparison between the residence time distribution E(t) of the photo-impinging streams reactor using fiberglass cloth and that predicted from tank in series model with N = 4. | ||
The RTD is one of the major informative characteristics of the flow pattern in a chemical reactor. It provides information on the duration of stay of various elements within a system and allows comparison between systems having different configuration of reactors. It is evident that, the key phenomenon in impinging streams with fluid-solid flows is the penetration of particles into the opposite stream, creating a longer mean residence time. In Fig. 12, the experimental RTD data obtained for the impinging stream reactor with FGC placed at the impinging zone is compared with the experimental results determined without presence of FGC. As it may be observed from this figure, there is a notable difference between the two sets of RTD data. A comparison between the two mean residence times revealed that the reactor equipped with a fiberglass cloth has a longer mean residence time under identical operating conditions (flow rate of 550 mL min−1). This may be due to the wetting properties of fiberglass warp and woofs leading to a longer traversal path and lower flow rates of liquid droplets. It may be concluded therefore, that presence of fiberglass clothes at the impinging zone of the reactors can increase the contact time of reactants with catalyst surface during each cycle. This advantage would somehow compensate the lower activity of immobilized catalysts in comparison with suspended systems.
3.4. Degradation kinetic model
Kinetic experiments have been performed applying a liquid flow rate of 550 mL min−1, pH value of 5.1 and initial PNP concentrations of 20, 60 and 100 mg L−1. To describe the PNP degradation rate Langmuir–Hinshelwood (L–H) model has been applied. Langmuir–Hinshelwood model has been widely used for liquid and gas-phase photocatalysis.51–53 This model explains the kinetic of reactions that occur between two adsorbed species (a free radical and an adsorbed substrate, or a surface-bound radical and a free substrate).54 By application of L–H expression, the photocatalytic degradation rate (r) of PNP becomes:
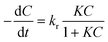 | (8) |
It may be assumed that the impingement zone acts as a cascade of four ideal CSTRs in series (Fig. 10). Evaluation of PNP concentrations in a system consisting of N CSTRs in series, is defined by a set of N mass balance expressions:40
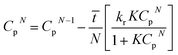 | (9) |
| Cp0,n = Cpr,in, Cp4,n = Cpr,out |
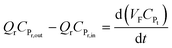 | (10) |
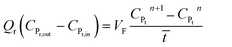 | (11) |
![[t with combining macron]](https://www.rsc.org/images/entities/i_char_0074_0304.gif) is the mean residence time and
is the mean residence time and 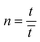 .
.
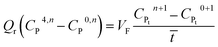 | (12) |
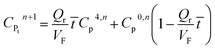 | (13) |
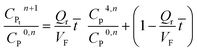 | (14) |
To determine the kinetic coefficients, an optimization program was applied by which the sum of the square errors (SSE) for experimental and predicted degradation ratios is minimized:
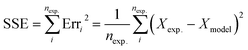 | (15) |
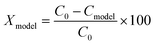 | (16) |
The kinetic parameters, experimental and predicted degradation ratios and SSEs for each run are presented in Table 1.
| Time (min) | C0 = 100 mg L−1 | C0 = 60 mg L−1 | C0 = 20 mg L−1 | |||
|---|---|---|---|---|---|---|
| Xexp. | Xmodel | Xexp. | Xmodel | Xexp. | Xmodel | |
| 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 10 | 3.8 | 4.6 | 6.0 | 7.0 | 9.6 | 10.5 |
| 20 | 8.7 | 9.1 | 13.9 | 14.1 | 18.9 | 20.0 |
| 30 | 13.2 | 13.5 | 20.9 | 20.5 | 28.4 | 28.6 |
| 40 | 17.2 | 17.8 | 26.2 | 26.6 | 36.7 | 36.3 |
| 50 | 20.9 | 21.9 | 31.7 | 32.4 | 42.5 | 43.2 |
| 60 | 27.7 | 25.9 | 38.7 | 37.7 | 50.6 | 49.4 |
| kr (mg min−1 L−1) | 0.033 | 0.058 | 0.123 | |||
| K (L mg−1) | 0.0099 | 0.0063 | 0.0034 | |||
| SSE | 0.00051 | 0.00031 | 0.00040 | |||
As it may be observed from Table 1, SSEs of the experimental PNP conversions are close to the predicted values from CSTRs in series model. This could indicate that a satisfactory agreement exists between the model predictions and the experimental results.
4. Conclusion
Photocatalytic decomposition of p-nitrophenol in liquid phase was investigated in a new designed photo-impinging streams reactor using a fiberglass cloth coated with TiO2 catalyst. The PNP initial concentration range was 20 to 100 mg L−1 and the range of liquid flow rates was 300 to 800 mL min−1. With lower PNP concentrations, higher degradation of the latter was observed. Increasing the liquid flow rate promotes the diffusion of PNP molecules within the catalyst leading to a decrease in PNP degradation ratio. RTD of fluid in photoreactor was obtained applying the impulse tracer method. The RTD data revealed that CSTRs in series model with a cascade of four ideal tanks was well correlated with the reactor behavior. The residence time of fluid in the reactor equipped with a fiberglass cloth was higher than that with no FGC operating under identical conditions. This could lead to higher degradation efficiency of the former configuration. The CSTRs in series model combined with PNP photo-degradation rate equation was used to predict the kinetic parameters. A good correlation observed between the predicted values from CSTRs in series model and those determined experimentally, indicating the validity of the selected model.Nomenclature
| E | Residence time distribution |
![[t with combining macron]](https://www.rsc.org/images/entities/i_char_0074_0304.gif) m m | Mean liquid residence time (min) |
| σ2 | Variance of the residence time |
| σθ2 | Dimensionless variance |
| N | Number of equal sized ideal stirred tank reactors |
| K | Adsorption equilibrium constant (L mg−1) |
| C | PNP concentration (mg L−1) |
| C0 | PNP initial concentration (mg L−1) |
| kr | Apparent reaction rate coefficient (mg min−1 L−1) |
| SSE | Sum of the square errors |
| nexp. | Total number of experiments |
| Xexp. | Experimental degradation ratio (%) |
| Xmodel | Model degradation ratio (%) |
| CN−1 | Inlet PNP concentrations from the Nth reactor (mg L−1) |
| CN | Outlet PNP concentrations from the Nth reactor (mg L−1) |
Acknowledgements
The authors would like to thank Eng. M. Rezaei for his helpful comments and Evonik GmbH for kindly supplying Nano-sized TiO2.References
- K. P. Mishra and P. R. Gogate, Ultrason. Sonochem., 2011, 18, 739–744 CrossRef CAS PubMed.
- Z. Wu, Y. Cong, M. Zhou and T. e. Tan, Chem. Eng. J., 2005, 106, 83–90 CrossRef CAS.
- S.-Z. Wu, C.-H. Chen and W.-D. Zhang, Chin. Chem. Lett., 2014, 25, 1247–1251 CrossRef CAS.
- M. M. Mohammadi, M. Vossoughi, M. Feilizadeh, D. Rashtchian, S. Moradi and I. Alemzadeh, Colloids Surf., A, 2014, 452, 1–8 CrossRef CAS.
- C. M. Ling, A. R. Mohamed and S. Bhatia, Chemosphere, 2004, 57, 547–554 CrossRef CAS PubMed.
- S. J. Royaee and M. Sohrabi, Desalination, 2010, 253, 57–61 CrossRef CAS.
- M. Tasbihi, U. Lavrencic Štangar, U. Cernigoj, J. Jirkovsky, S. Bakardjieva and N. Novak Tusar, Catal. Today, 2011, 161, 181–188 CrossRef CAS.
- A. K. Ray, in Advances in Chemical Engineering, ed. I. d. L. Hugo and R. Benito Serrano, Academic Press, 2009, vol. 36, pp. 145–184 Search PubMed.
- P. Du, J. T. Carneiro, J. A. Moulijn and G. Mul, Appl. Catal., A, 2008, 334, 119–128 CrossRef CAS.
- J. H. Lee, W. Nam, M. Kang, G. Y. Han, K. J. Yoon, M.-S. Kim, K. Ogino, S. Miyata and S.-J. Choung, Appl. Catal., A, 2003, 244, 49–57 CrossRef CAS.
- D. D. Dionysiou, G. Balasubramanian, M. T. Suidan, A. P. Khodadoust, I. Baudin and J.-M. Laîné, Water Res., 2000, 34, 2927–2940 CrossRef CAS.
- S. J. Royaee, M. Sohrabi and F. Soleymani, J. Chem. Technol. Biotechnol., 2011, 86, 205–212 CrossRef CAS.
- S. J. Royaee, M. Sohrabi and P. Jabari Barjesteh, Chem. Eng. Res. Des., 2012, 90, 1923–1929 CrossRef CAS.
- S. J. Royaee and M. Sohrabi, Ind. Eng. Chem. Res., 2012, 51, 4152–4160 CrossRef CAS.
- Z. M. Shaykhi and A. A. L. Zinatizadeh, J. Taiwan Inst. Chem. Eng., 2014, 45, 1717–1726 CrossRef CAS.
- J.-R. Gurr, A. S. S. Wang, C.-H. Chen and K.-Y. Jan, Toxicology, 2005, 213, 66–73 CrossRef CAS PubMed.
- M. Karches, M. Morstein, P. Rudolf von Rohr, R. L. Pozzo, J. L. Giombi and M. A. Baltanás, Catal. Today, 2002, 72, 267–279 CrossRef CAS.
- J.-C. Lee, M.-S. Kim and B.-W. Kim, Water Res., 2002, 36, 1776–1782 CrossRef CAS PubMed.
- S. Horikoshi, N. Watanabe, H. Onishi, H. Hidaka and N. Serpone, Appl. Catal., B, 2002, 37, 117–129 CrossRef CAS.
- I. N. Martyanov and K. J. Klabunde, J. Catal., 2004, 225, 408–416 CrossRef CAS.
- M. S. Vohra and K. Tanaka, Water Res., 2003, 37, 3992–3996 CrossRef CAS PubMed.
- J. Matos, J. Laine, J.-M. Herrmann, D. Uzcategui and J. Brito, Appl. Catal., B, 2007, 70, 461–469 CrossRef CAS.
- H. Chen, S. W. Lee, T. H. Kim and B. Y. Hur, J. Eur. Ceram. Soc., 2006, 26, 2231–2239 CrossRef CAS.
- K. Venkata Subba Rao, A. Rachel, M. Subrahmanyam and P. Boule, Appl. Catal., B, 2003, 46, 77–85 CrossRef CAS.
- S. Hosseini, S. Borghei, M. Vossoughi and N. Taghavinia, Appl. Catal., B, 2007, 74, 53–62 CrossRef CAS.
- M. Jafarikojour, M. Sohrabi, S. J. Royaee and A. Hassanvand, Clean: Soil, Air, Water, 2014 DOI:10.1002/clen.201300985.
- Z. Liu, P. Fang, S. Wang, Y. Gao, F. Chen, F. Zheng, Y. Liu and Y. Dai, J. Mol. Catal. A: Chem., 2012, 363–364, 159–165 CrossRef CAS.
- M. Fathinia and A. R. Khataee, J. Ind. Eng. Chem., 2013, 19, 1525–1534 CrossRef CAS.
- W. Wibel, A. Wenka, J. J. Brandner and R. Dittmeyer, Chem. Eng. J., 2013, 227, 203–214 CrossRef CAS.
- S. S. Waje, A. K. Patel, B. N. Thorat and A. S. Mujumdar, Drying Technol., 2007, 25, 249–259 CrossRef.
- S. Royaee, M. Sohrabi and M. Jafarikojour, Res. Chem. Intermed., 2014, 1–23 Search PubMed.
- G. Charles, T. Roques-Carmes, N. Becheikh, L. Falk, J.-M. Commenge and S. Corbel, J. Photochem. Photobiol., A, 2011, 223, 202–211 CrossRef CAS.
- S. Basha, D. Keane, A. Morrissey, K. Nolan, M. Oelgemöller and J. Tobin, Ind. Eng. Chem. Res., 2010, 49, 11302–11309 CrossRef CAS.
- S. Kozerski, F.-L. Toma, L. Pawlowski, B. Leupolt, L. Latka and L.-M. Berger, Surf. Coat. Technol., 2010, 205, 980–986 CrossRef CAS.
- A. Verma, M. Kar and S. A. Agnihotry, Sol. Energy Mater. Sol. Cells, 2007, 91, 1305–1312 CrossRef CAS.
- J. Colmenares-Angulo, S. Zhao, C. Young and A. Orlov, Surf. Coat. Technol., 2009, 204, 423–427 CrossRef CAS.
- I. Burlacov, J. Jirkovský, L. Kavan, R. Ballhorn and R. B. Heimann, J. Photochem. Photobiol., A, 2007, 187, 285–292 CrossRef CAS.
- M. Motamedi, A. Habibi, M. Maleki and F. Vahabzadeh, Clean: Soil, Air, Water, 2014 DOI:10.1002/clen.201300635.
- S. M. B. S. N. Hosseini, M. Vossoughi and N. Taghavinia, Appl. Catal., B, 2007, 74, 53–62 CrossRef CAS.
- G. Vincent, A. Queffeulou, P. M. Marquaire and O. Zahraa, J. Photochem. Photobiol., A, 2007, 191, 42–50 CrossRef CAS.
- M. Jafarikojour, M. Sohrabi, S. J. Royaee and M. Rezaei, RSC Adv., 2014, 4, 53097–53104 RSC.
- Y. Wu, Impinging Streams: Fundamentals, Properties and Applications, Elsevier Science, 2007 Search PubMed.
- T. N. Obee and R. T. Brown, Environ. Sci. Technol., 1995, 29, 1223–1231 CrossRef CAS PubMed.
- M. Antonopoulou, V. Papadopoulos and I. Konstantinou, J. Chem. Technol. Biotechnol., 2012, 87, 1385–1395 CrossRef CAS.
- C. F. Daniel Vildozo, M. Sleiman and J.-M. Chovelon, Appl. Catal., B, 2010, 94, 303–310 CrossRef.
- O. Carp, C. L. Huisman and A. Reller, Prog. Solid State Chem., 2004, 32, 33–177 CrossRef CAS.
- J. Lea and A. A. Adesina, J. Chem. Technol. Biotechnol., 2001, 76, 803–810 CrossRef CAS.
- M. S. Dieckmann and K. A. Gray, Water Res., 1996, 30, 1169–1183 CrossRef CAS.
- W. Zhang, X. Xiao, T. An, Z. Song, J. Fu, G. Sheng and M. Cui, J. Chem. Technol. Biotechnol., 2003, 78, 788–794 CrossRef CAS.
- Y. Wang, S. Liu, M. Brannock and G. Leslie, Desalination, 2009, 236, 120–126 CrossRef CAS.
- R. Vargas and O. Núñez, Sol. Energy, 2010, 84, 345–351 CrossRef CAS.
- G. Pardo, R. Vargas and O. Núñez, J. Phys. Org. Chem., 2008, 21, 1072–1078 CrossRef CAS.
- T. A. Egerton, P. A. Christensen, R. W. Harrison and J. W. Wang, J. Appl. Electrochem., 2005, 35, 799–813 CrossRef CAS.
- V. Augugliaro and R. S. o. Chemistry, Clean by Light Irradiation: Practical Applications of Supported TiO2, Royal Society of Chemistry, 2010 Search PubMed.
| This journal is © The Royal Society of Chemistry 2015 |