Crystalline Si photovoltaic modules functionalized by a thin polyethylene film against potential and damp-heat-induced degradation
Abstract
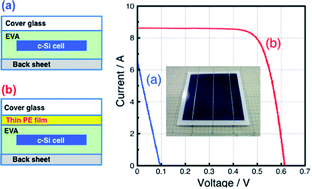
Potential-induced degradation (PID) in p-type-based multicrystalline Si photovoltaic (PV) modules was experimentally generated applying −1000 V from an Al plate, which is attached on the front cover glass of the module, to the Si cell at 85 °C for 2 h. The solar energy-to-electricity conversion efficiency (η) of the standard Si PV module significantly decreased after the PID test. In contrast, no degradation was observed in the modules, including a thin polyethylene (PE) film (30 μm thickness) with the copolymer of ethylene and vinyl acetate (EVA) as the encapsulant. It was suggested that the PE film whose volume resistivity is higher than that of EVA prevented the diffusion of Na+ from the front cover glass toward the Si cell, resulting in a suppression of PID because different degradation processes during PID were observed in the EL images for the two modules, including a half PE film. In addition, the Si PV module, including a PE film, demonstrated stable performance after a damp-heat test (85 °C/85% relative humidity) for 4000 h, although the η of the standard module significantly decreased from 16.0% to 7.6% after the test. Our results indicate an attractive and promising low-cost technique for improving the long-term stability of crystalline Si PV modules against potential and damp-heat-induced degradation.

 Please wait while we load your content...
Please wait while we load your content...