X-ray irradiation-induced reversible wettability modification of titanium NRAs†
Weipeng Wanga,
Zheng Xieab,
Zhengcao Lia and
Zhengjun Zhang*c
aState Key Laboratory of New Ceramics and Fine Processing, School of Materials Science and Engineering, Tsinghua University, Beijing 100084, P.R. China
bHigh-Tech Institute of Xi'an, Shaanxi 710025, P.R. China
cKey Laboratory of Advanced Materials (MOE), School of Materials Science and Engineering, Tsinghua University, Beijing 100084, P.R. China. E-mail: zjzhang@tsinghua.edu.cn
First published on 25th November 2014
Abstract
The reversible transition between hydrophilicity and hydrophobicity of Ti NRAs induced by alternating X-ray irradiation and ethanol immersion has been revealed. The wettability modification is attributed to the chemisorption of alkyl groups on TNRA surfaces from airborne molecules.
Introduction
Smart surfaces with reversible wettability transitions under external stimuli have recently generated extensive interest due to their potential applications in various fields.1,2 Different approaches have been put forward to achieve novel wetting behaviours (e.g., superhydrophilicity, superhydrophobicity and superoleophilicity), by focusing on the two dominant factors affecting the wettability of surfaces: surface roughness and chemical state.3 Among these studies, bio-inspired nanostructured materials have been the state-of-the-art, since rough nanoscale surfaces are more sensitive to environmental stimuli.4–6 Nanostructured surfaces within different material systems have been successfully fabricated by glancing angle deposition (GLAD) and oblique angle deposition (OAD) methods, due to their advantages including large area of fabrication, substrate insensitivity and wide suitability.7,8 Because of their rough and porous characteristics, efficient wettability modifications of such surfaces have been realised, especially modifications induced by the chemical environment.9The control of wettability by ultraviolet (UV) and visible light has been extensively studied in the literature,10–13 while the use of X-ray illumination is far less investigated. X-rays, which have smaller wavelengths than UV/vis light, are reported to be efficient stimuli to generate chain scission in organic materials.14 Moreover, piezoelectric materials will be surface-charged by positively-charged holes (h+), as X-rays could generate electron–hole separation and X-ray photoelectron emission. Thus, along with their greater penetration, X-rays are expected to have different effects on materials, compared to UV/vis light. Among the studies on X-ray-generated wettability manipulation, Kwon et al. focused on the photoelectric effect,15 while Weon et al. emphasised chain scission generation.16 Although both studies obtained remarkable wettability modification upon X-ray stimuli, no reversible transition without artificial chemi-introduction was realised.
Transition metals such as Ni, Pd, Pt, Fe, Co, Rh, Cu and Ti have been widely used as catalytic media in organic industry and scientific research.17,18 Benefitting from the porous character and larger specific surface area, nanostructured catalysts have presented better efficiencies. Titanium alloys, on the other hand, have drawn great attention in the last few decades because of their unique biocompatibilities.19 As well-developed biocompatible implants, the realisation of reversible wettability modification of titanium under external stimuli is of great importance for the potential applications of titanium alloys in drug delivery and payload release.20
In this communication, inspired by the considerations above, we deposited titanium nanorod arrays (Ti NRAs) using oblique angle deposition (OAD), and remarkable wettability modification of the films was achieved upon X-ray irradiation.
Experimental
Ti NRAs of 300 nm in length were deposited onto n-Si (100) wafers and quartz substrates by OAD using an e-beam evaporation instrument. Both substrates were ultrasonically cleaned using ethanol/deionised water and dried by flowing nitrogen. For OAD, a large substrate tilting angle of 85° was set to generate a glancing incidence flux of titanium vapour. Evaporations were performed at a base pressure of 10−8 Torr, and the deposition rate was controlled at 0.5 nm s−1 with an e-beam power supply of ∼1 kW.The structure and morphology of the as-received Ti NRAs films were characterised ex situ by X-ray diffraction (XRD; Rigaku Dmax 2500 V), field emission scanning electron microscopy (FE-SEM; JSM-7001F) and high resolution transmission electron microscopy (HRTEM; JEOL 2011). SEM observations of the film cross sections were performed at a 45° tilting angle. X-ray irradiations were performed with a PHI-5400 using an Mg source under a vacuum of 10−9 Torr while collecting the XPS signals at the same time. The typical duration of X-ray illumination was 10 min with an X-ray power supply of 200 W. Contact angle (CA) measurements were performed by the sessile drop method21 on an OCA 15pro system with a tilting stage. The “as-placed” contact angles were first measured by deploying a drop and recording in real time until the droplet stabilised. The advancing and receding angles were then image-recorded and measured by tilting the test stage until the droplet started rolling. The contact angle value was obtained as an average of five measurements recorded at different surface locations for each sample. The deionised water volume was 3 μL. Each CA-measured sample was cleaned with ethanol and dried with flowing nitrogen before the X-ray irradiation process to exclude the influence of water during the XPS measurements.
Results and discussion
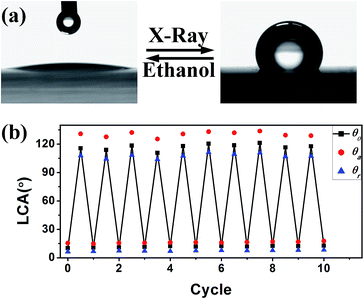
The crystal structure and morphology of the as-received Ti NRA films were studied by XRD, FE-SEM and HRTEM. Fig. 1(a) shows that the film is poorly crystallized, as all the reflection peaks have weak intensities. All the peaks can be assigned to the hexagonal α phase of titanium (JCPDS 44-1294). FE-SEM observation revealed that the nanorod arrays had a well-separated structure. A single nanorod is 300 nm in length and 30–50 nm in diameter. TEM observation confirmed the separation, while HRTEM revealed nano-crystalline Ti nanorods with the lattice fringe corresponding to the (0002) plane detected. The as-received Ti NRAs on both substrates are hydrophilic, and the hydrophilicity is well preserved for at least 10 days (Fig. S1†). Prolonged storage in ambient atmosphere will lead to a monotonic increase in the contact angles (both advancing and receding), due to the continuous absorption of carbon-based molecules.22Fig. 2 illustrates the wettability transition of Ti NRAs on Si and quartz substrates upon X-ray irradiation. For Ti NRAs on Si, the contact angle increased from 8.2° to 51.2°, while those on quartz exhibited a hydrophilic–hydrophobic transition upon X-ray irradiation; the “as-placed” contact angle increased from 12.6° to 115.5°. For X-ray irradiated Ti NRAs on quartz, the corresponding advancing and receding angles evolved to values of 130° and 107°, respectively. It is noteworthy that by adding an extra quartz beneath the silicon substrate during X-ray irradiation, the wettability of Ti NRAs on silicon showed the same transition as Ti NRAs on quartz. After hydrophobicity is achieved upon irradiation, the contact angles (the as-placed, advancing and receding angles) of the Ti NRAs can be retained for more than 30 days. The oleo-wettabilities of the films were also characterised and are shown in Fig. 3(e). All samples with or without irradiation were superoleophilic, as the CA of oil was less than 10°. Fig. 2(e) clearly illustrates that for the X-ray irradiated Ti RNAs, hydrophobicity and oleophilicity can be achieved at the same time. Various X-ray powers of 50 W, 100 W, 200 W and 260 W were also applied; the wettability of the irradiated sample was power-independent in this range as the contact angle fluctuated between 112° and 117°. The transition was also found to be independent of X-ray duration between 5 min and 60 min. Upon X-ray irradiation, the wettability of blank silicon did not change, while that of blank silicon exhibited a distinct change (Fig. S2†). Room temperature in situ oxidation of Ti NRAs for 30 min was performed by introducing floating oxygen into the deposition chamber. The oxidation process also introduced certain modifications to the contact angle upon irradiation (Fig. S3†). As the morphologies of the NRAs were unchanged by the oxidization process, the capillary effects of the Ti NRAs were also unchanged, which highlights the chemical nature of the modification of the Ti NRAs by irradiation.
As highlighted in Fig. 3, the wettability modification showed a good reversible transition that can be easily realised by immersing the irradiated sample in ethanol. The immersion into ethanol has no influence on the nanostructure, although it may dissolve the attached molecules. Subsequently, the film returns to being hydrophilic, like the as-received film. By cycling the irradiation, the process illustrated in Fig. 2 generated and re-introduced chemical modification on the Ti NRA surfaces, and the film then changed from superhydrophilic to hydrophobic.
Comprehensive investigations of the as-received films suggested that X-ray irradiation resulted in the wettability evolution of the Ti NRA films by surface chemical modification on the insulating quartz substrate.
Meanwhile, the Ti NRAs on conductive Si substrates show no remarkable changes. Similar wettability deterioration was observed by Aronov et al. in their work on low energy electron-irradiated thermo-oxide silicon under vacuum.23 Although the mechanism is complicated, they attributed the final wettability changes to alkyl group formation on the top surface. Interestingly, alkyl groups are concurrently both strongly hydrophobic and oleophilic, which is in accordance with our results in Fig. 2(e). Also, the good solubility of alkyl groups in organic solvents can explain the hydrophobic–hydrophilic transition upon ethanol immersion during the cycle transition in Fig. 3. Considering that alkyl groups have a static contact angle of about 110°,24,25 we then cautiously conclude that upon X-ray irradiation, alkyl groups were introduced to the surface of Ti NRAs and are responsible for the wettability manipulation.
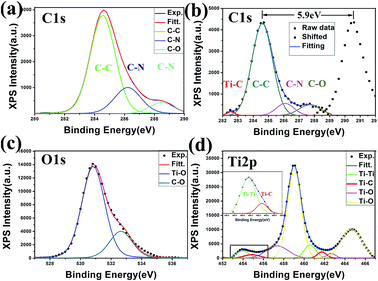
In his review paper, F. Zaera reported that alkyl halides chemisorb molecularly on transition metal surfaces, and the scission of carbon–halogen bonds can be easily activated photolytically to yield adsorbed alkyl moieties.26 Other studies have focused on the organometallic approach and reported that C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C/C–H chain scissions can be generated by X-ray illumination and catalysed by transition metals to form alkyl moieties; the as-generated moieties would then be trapped by positively charged holes.27 As definite concentrations of carbon were found on all samples studied herein during X-ray irradiation, the alkyl group formation possibly occurred during the X-ray irradiation. The direct observation of XPS peaks assigned to Ti–C bond during irradiation in Fig. 4(b) and (d) strongly supports this hypothesis of alkyl formation.
C/C–H chain scissions can be generated by X-ray illumination and catalysed by transition metals to form alkyl moieties; the as-generated moieties would then be trapped by positively charged holes.27 As definite concentrations of carbon were found on all samples studied herein during X-ray irradiation, the alkyl group formation possibly occurred during the X-ray irradiation. The direct observation of XPS peaks assigned to Ti–C bond during irradiation in Fig. 4(b) and (d) strongly supports this hypothesis of alkyl formation.
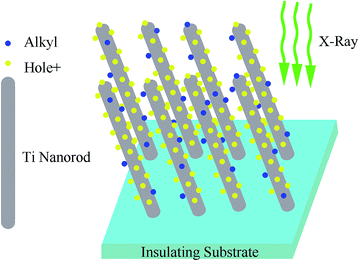
A possible mechanism for alkyl group formation on Ti NRAs upon X-ray irradiation is proposed and illustrated in Fig. 5. Four indispensable factors contributing to the modification are highlighted in Fig. 5: X-ray irradiation, Ti NRAs, substrate conductivity and ultrahigh vacuum (UHV).
Here, the X-rays are the dominating factor, as they generate the electron–hole separation. With the emission of photoelectrons, variations in the charged behaviour occurs depending on the conductivity of substrates, i.e., positively charged holes (h+) are neutralised on the conducting substrate (n-Si), while they accumulate on the insulating surface. From the XPS signals collected during irradiation, a significant peak shift to higher binding energy (284.6 eV to 290.5 eV for C1s) was observed for the insulating quartz substrates (Fig. 4(b)), while no changes were detected for the n-silicon wafer substrates (Fig. 4(a)). The continuous X-ray illumination and h+ can generate chain scission of surface-absorbed molecules on the insulating substrate, while only X-ray generation can be realised for the silicon substrate.28 Moreover, as suggested by the variation between blank silicon and quartz upon irradiation (Fig. S2†), the h+ are a key factor in the acquisition of the as-produced moieties. Positive charged holes can trap the anion alkyl moiedties with unpaired electron and promote the chemical adsorption of alkyl group onto Ti NRAs surface.
Ti NRAs play three different roles, and all the factors were correlated with the rough structure. First and conceivably, the rough surface favoured the surface absorption of organic molecules. Second, as implied by the difference between the as-received and the in situ oxidized film upon irradiation, elemental titanium plays the dominant role in the transition. Since transition metals have been efficient catalysts in the reaction in the chemical industry, titanium catalyzed the X-ray/h+-generated chain scission of absorbed organic molecules to form alkyl moieties. The rough nature would also benefit the catalytic process by increasing the specific surface area. Lastly, once the alkyl groups formed on the Ti NRAs, single nanorods became hydrophobic. According to the Wenzel model, the rough morphology of a hydrophobic surface will enlarge the contact angles.29
The proposed mechanism can then fit all the results and variations quite well. Ti NRAs on quartz showed larger modifications upon X-ray irradiation than those on silicon (Fig. 2), as h+ contributed to the generation of chain scission and the acquisition of alkyl moieties. For the blank silicon and quartz substrates, the X-ray induced modification degree (Fig. S2†) is much smaller than those with Ti NRAs (Fig. 2). This emphasized the dominating role of Ti NRAs on wettability transition. The catalytic effects of Ti upon transition were highlighted by the smaller response of the in situ oxidized film (Fig. S3†), although the response itself confirmed the contribution of h+.
Another important factor that is often ignored is the ultrahigh vacuum (UHV). From Fig. 4(c), one sees that the O1s spectrum can be well fitted to O–Ti (top surface) and O–C (absorbed molecules) bonds without other chemical states. On one hand, UHV can exclude the existence of H2O and O2, which will greatly hinder the chain scission. On the other hand, UHV limits the amount of absorbed molecules on the Ti NRAs. Furthermore, the diminishment of H2O due to the elimination of hydroxyl formation by UHV will contribute to the wetting behaviour, leading to a decrease in contact angle.
Conclusions
In summary, the reversible transition between hydrophilicity and hydrophobicity has been achieved on Ti NRAs films on insulating quartz substrates upon X-ray irradiation and ethanol cleaning. The combination of X-ray illumination, Ti nanostructure catalysis, an insulating substrate and UHV contribute to the transition by promoting alkyl formation on the film surface by molecular absorption.Acknowledgements
We thank Prof. Joseph Chiang for help in manuscript preparation. The authors are very grateful for the financial support by the National Basic Research Program of China (973 program, grant no. 2010CB731601), the National Natural Science Foundation of China (Grant no. 51228101), the Research Project of Chinese Ministry of Education (grant no. 113007A), and the Tsinghua University Initiative Scientific Research Program.Notes and references
- B. W. Xin and J. C. Hao, Reversibly Switchable Wettability, Chem. Soc. Rev., 2010, 39, 769–782 RSC.
- J. Drelich, E. Chibowski, D. D. Meng and K. Terpilowski, Hydrophilic and superhydrophilic surfaces and materials, Soft Matter, 2011, 7(21), 9804–9828 RSC.
- X. J. Feng and L. Jiang, Design and Creation of Superwetting/Antiwetting Surfaces, Adv. Mater., 2006, 18, 3063–3078 CrossRef CAS.
- X. Yao, Y. L. Song and L. Jiang, Applications of Bio-Inspired Special Wettable Surfaces, Adv. Mater., 2011, 23, 719–734 CrossRef CAS PubMed.
- J. X. Wang, Y. Z. Zhang, S. T. Wang, Y. L. Song and L. Jiang, Bioinspired Colloidal Photonic Crystals with Controllable Wettability, Acc. Chem. Res., 2011, 44, 405–415 CrossRef CAS PubMed.
- J. Drelich and A. Marmur, Physics and applications of superhydrophobic and superhydrophilic surfaces and coatings, Surf. Innovations, 2014, 2(4), 211–227 CrossRef.
- Q. Zhou, Y. He, J. Abell, Z. Zhang and Y. Zhao, Optical Properties and Surface Enhanced Raman Scattering of L-Shaped Silver Nanorod Arrays, J. Phys. Chem. C, 2011, 115, 14131–14140 CAS.
- J. Ni, Y. Zhu, S. H. Wang, Z. C. Li, Z. J. Zhang and B. Q. Wei, Nanostructuring HfO2 Thin Films as Antireflection Coatings, J. Am. Chem. Soc., 2009, 92, 3077–3080 CAS.
- S. F. Tsoi, E. Fok, J. C. Sit and J. Veinot, Surface Functionalization of Porous Nanostructured Metal Oxide Thin Films Fabricated by Glancing Angle Deposition, Chem. Mater., 2006, 18, 5260–5266 CrossRef CAS.
- R. Wang, K. Hashimoto, A. Fujishima, M. Chikuni, E. Kojima, A. Kitamura, M. Shimohigoshi and T. Watanabe, Light-induced Amphiphilic Surfaces, Nature, 1997, 388, 431–432 CrossRef CAS PubMed.
- X. J. Feng, J. Zhai and L. Jiang, The Fabrication and Switchable Superhydrophobicity of TiO2 Nanorod Films, Angew. Chem., Int. Ed., 2005, 44, 5115–5118 CrossRef CAS PubMed.
- G. Caputo, C. Nobile, T. Kipp, L. Blasi, V. Grillo, E. Carlino, L. Manna, R. Cingolani, P. D. Cozzoli and A. Athanassiou, Reversible Wettability Changes in Colloidal TiO2 Nanorod Thin-film Coatings under Selective UV Laser Irradiation, J. Phys. Chem. C, 2008, 112, 701–714 CAS.
- B. Yan, J. G. Tao, C. Pang, Z. Zheng, Z. X. Shen, C. Huan and T. Yu, Reversible UV-light-Induced Ultrahydrophobic-to-Ultrahydrophilic Transition in an alpha-Fe2O3 Nanoflakes film, Langmuir, 2008, 24, 10569–10571 CrossRef CAS PubMed.
- K. Heister, M. Zharnikov, M. Grunze, L. S. O. Johanssom and A. Ulman, Characterization of X-ray Induced Damage in Alkanethiolate Monolayers by High-Resolution Photoelectron Spectroscopy, Langmuir, 2001, 17, 8–11 CrossRef CAS.
- Y. B. Kwon, B. M. Weon, K. H. Won, J. H. Je, Y. Hwu and G. Margaritondo, X-ray-Induced Changes in Wettability, Langmuir, 2009, 25, 1927–1929 CrossRef CAS PubMed.
- B. M. Weon, Y. B. Kwon, K. H. Won, J. Yoo, J. H. Je, M. Li and J. H. Hahn, Ablation and Deposition of Poly(dimethylsiloxane) with X-Rays, ChemPhysChem, 2010, 11, 115–118 CrossRef CAS PubMed.
- A. C. Bissember, R. J. Lundgren, S. E. Creutz, J. C. Peters and G. C. Fu, Transition-Metal-Catalyzed Alkylations of Amines with Alkyl Halides: Photoinduced, Copper-Catalyzed Couplings of Carbazoles, Angew. Chem., Int. Ed., 2013, 52, 5129–5133 CrossRef CAS PubMed.
- I. Vidal, S. Melchor, I. Alkorta, J. Elguero, M. R. Sundberg and J. A. Dobado, On the Existence of Alpha-Agostic Bonds: Bonding Analyses of Titanium Alkyl Complexes, Organometallics, 2006, 25, 5638–5647 CrossRef CAS.
- X. Y. Liu, P. K. Chu and C. X. Ding, Surface Nano-Functionalization of Biomaterials, Mater. Sci. Eng. R Rep., 2010, 70, 275–302 CrossRef PubMed.
- Y. Y. Song, P. Roy, I. Paramasivam and P. Schmuki, Voltage-Induced Payload Release and Wettability Control on TiO2 and TiO2 Nanotubes, Angew. Chem., Int. Ed., 2010, 49, 351–354 CrossRef CAS PubMed.
- J. Drelich, Guidelines to measurements of reproducible contact angles using a sessile-drop technique, Surf. Innovations, 2013, 1(4), 248–254 CrossRef CAS.
- L. Boinovich, A. Emelyanenko, A. Pashinin, C. H. Lee, J. Drelich and Y. K. Yap, Origins of Thermodynamically stable superhydrophobicity of Boron Nitride nanotubes coatings, Langmuir, 2012, 28, 1206–1216 CrossRef CAS PubMed.
- D. Aronov, M. Molotskii and G. Rosenman, Charge-Induced Wettability Modification, Appl. Phys. Lett., 2007, 90, 10410410 CrossRef PubMed.
- S. L. Peczonczyk, J. Mukherjee, A. I. Carim and S. Maldonado, Wet Chemical Functionalization of III-V Semiconductor Surfaces: Alkylation of Gallium Arsenide and Gallium Nitride by a Grignard Reaction Sequence, Langmuir, 2012, 28, 4672–4682 CrossRef CAS PubMed.
- K. L. Cho, A. H. F. Wu, I. I. Liaw, D. Cookson and R. N. Lamb, Wetting Transitions on Hierarchical Surfaces, J. Phys. Chem. C, 2012, 116, 26810–26815 CAS.
- F. Zaera, Preparation and Reactivity of Alkyl-Groups Adsorbed on Metal-Surfaces, Acc. Chem. Res., 1992, 25, 260–265 CrossRef CAS.
- F. Zaera, An Organometallic Guide To The Chemistry of Hydrocarbon Moieties on Transition Metal Surfaces, Chem. Rev., 1995, 95, 2651–2693 CrossRef CAS.
- P. Roy, S. Berger and P. Schmuki, TiO2 Nanotubes: Synthesis and Applications, Angew. Chem., Int. Ed., 2011, 50, 2904–2939 CrossRef CAS PubMed.
- R. N. Wenzel, Resistance of Solid Surfaces to Wetting by Water, Ind. Eng. Chem., 1936, 28, 988–994 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ra13093f |
| This journal is © The Royal Society of Chemistry 2015 |