Open Access Article
Open Access ArticleTwo equivalent methyl internal rotations in 2,5-dimethylthiophene investigated by microwave spectroscopy†
Vinh
Van
*a,
Wolfgang
Stahl
a and
Ha Vinh Lam
Nguyen
b
aInstitute of Physical Chemistry, RWTH Aachen University, Landoltweg 2, D-52074 Aachen, Germany. E-mail: vinh.van@rwth-aachen.de
bLaboratoire Interuniversitaire des Systèmes Atmosphériques (LISA), UMR 7583 (CNRS/Univ. Paris Est & Paris Diderot), Université Paris Est, 61 avenue du Général de Gaulle, F-94010 Créteil cedex, France
First published on 29th September 2015
Abstract
The microwave spectrum of 2,5-dimethylthiophene, a sulfur-containing five-membered ring with two conjugated double bonds, was recorded in the frequency range from 2 to 40 GHz using molecular beam Fourier transform technique. Highly accurate molecular parameters were determined. A labeling scheme for the group G36 written as the semi-direct product (CI3 × CI3) ⋊ C2v was introduced.
Introduction
Monomethyl derivatives of unsaturated heterocyclic five-membered rings have been widely studied by microwave spectroscopy. The barriers to internal rotation of the methyl group cover a wide range from low barriers such as in 2-methylthiazole (1),1N-methylpyrrole (2),2 and 4-methylisothiazole (3)3 to intermediate barriers like in 4-methyloxazole (4),4 2-methylfuran (5),5 and 3-methylfuran (6),6 as well as 2-methylthiophene (7)7 and 3-methylthiophene (8),8 as summarized in Fig. 1.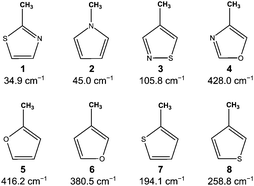 | ||
| Fig. 1 Barriers to internal rotation of some monomethyl unsaturated heterocyclic five-membered rings. | ||
To the best of our knowledge, no microwave investigations on the rotational spectra of dimethyl substituted unsaturated heterocyclic five-membered rings are available. Here, we present our studies on 2,5-dimethylthiophene (DMTP) aimed at determining the torsional barriers of two equivalent methyl tops. A comparison of the experimental results with state of the art quantum chemical calculations (QCCs) will also be discussed.
Quantum chemical calculations
In DMTP, two conjugated double bonds force the thiophene frame to planarity, and thus only one conformer is possible.9 However, the orientations of the two methyl groups in the equilibrium, which determine the molecular symmetry (C2v, C2, or Cs), remain questionable. In a previous electron diffraction study,10 one of the C–H bonds of the methyl groups is cis with respect to the S–C bond, which is in contrast to QCCs at the HF/3-21G(d) level. In our work, much higher levels of theory are applied to study this observation.Potential energy curves were obtained by varying the dihedral angle φ1 = ∠(S15,C12,C10,H4) in a step width of 1°, while all other geometry parameters were optimized (for atom numbers see Fig. 2). Calculations were performed using the GAUSSIAN 09 package11 with the MP2, B3LYP, and M06-2X methods and various basis sets. Three potential energy curves calculated with the cc-pVTZ basis set are shown in Fig. S-F1 in the ESI.† The V3 potentials possess negligible contributions of higher order terms, which can be clearly recognized through the Fourier coefficients indicated in Table S1 also in the ESI.† The energy minima show a C2v symmetry, where a hydrogen atom of each methyl group is antiperiplanar to the sulfur atom. The two methyl groups are thus equivalent. Full geometry optimizations and harmonic frequency calculations yielded similar results (see Fig. 2), which are also in agreement with the calculations given in ref. 10. The Cartesian coordinates are available in Table S2 (ESI†).
To study the coupling between the two methyl tops, a potential energy surface (PES) was calculated at the MP2/6-311++G(d,p) level of theory by varying the dihedral angles φ1 and φ2 = ∠(S15,C11,C9,H1) in a step width of 10°, as given in Fig. S-F2 (ESI†). The corresponding energies were parameterized with a two-dimensional Fourier expansion based on terms representing the correct symmetry; the Fourier coefficients are listed in Table S3 (ESI†). No significant coupling between the two tops could be observed. We found a similar situation for the PES calculated at the B3LYP/6-311++G(d,p) level (visualized in Fig. S-F3, ESI†). However, the energy minima are broader; the contribution of cross terms is smaller, as can also be recognized in Table S3 (ESI†).
Geometry optimizations to a first order transition state of one methyl group using the Berny algorithm12 at various levels of theory were used to calculate the barriers to internal rotation. The calculated rotational constants, angles between an internal rotor axis and the principal axes, as well as the V3 potentials are summarized in Table S4 in the ESI.† The angle ∠(i2,a) for the second rotor is π − ∠(i1,a), whereas ∠(i1,n) = ∠(i2,n) for n = b, c. We noticed that the geometry optimizations and harmonic frequency calculations at the MP2/6-311++G(d,p) level yielded one imaginary vibrational mode, which is a bending of the planar thiophene frame. Stating stable planar ring systems as unstable is a well-known behavior found at this level of theory, which has been reported for benzene and arenes.13
Group theory
Symmetry labels
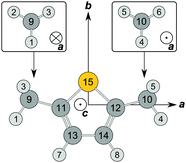
DMTP has a C2v frame symmetry and two equivalent methyl rotors. It is well known that the molecular symmetry group of such a system is G36 with the character table given in Table 1. There are different symmetry labels in use which arise from different product decompositions of G36. Probably, the earliest method is based on the direct product C−3v × C+3v, and the symmetry species of G36 are labeled with the labels of both subgroups C−3v and C+3v14 as given in column S3 of Table 1. Using the permutation-inversion group formalism, Bunker and Jensen15 derived a character table of acetone, where the different species are denoted by their degeneracy letter A, E, G and a running index (shown in column S2 of Table 1).| G36a | E | a −1 | ab | ab −1 | P | bP | Q | aQ | R | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Equiv. rot.b | R 0 | R 0 | R 0 | R 0 | R π b | R π b | R π a | R π a | R π c | |||
| S1c | S2d | S3e | 1 | 4 | 2 | 2 | 3 | 6 | 3 | 6 | 9 | wtg |
| a a = (1 2 3), b = (4 5 6), P = (1 4)(2 5)(3 6)(7 8)(9 10)(11 12)(13 14), Q = (1 4)(2 6)(3 5)(7 8)(9 10)(11 12)(13 14)*, R = (2 3)(5 6)*, for atom numbers see Fig. 2. b Equivalent rotations of the four-group. c Symmetry labels based on the semi-direct product (CI3 × CI3) ⋊ C2v, see ref. 16. d Symmetry labels according to ref. 15. e Symmetry labels based on the direct product C−3v × C+3v, see ref. 14. f Row between S3 and wt: number of elements in the respective class. g Spin statistical weight. | ||||||||||||
| (00)·A1 | A1 | A1A1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 36 |
| (00)·B1 | A2 | A2A1 | 1 | 1 | 1 | 1 | −1 | −1 | 1 | 1 | −1 | 28 |
| (00)·A2 | A3 | A1A2 | 1 | 1 | 1 | 1 | 1 | 1 | −1 | −1 | −1 | 36 |
| (00)·B2 | A4 | A2A2 | 1 | 1 | 1 | 1 | −1 | −1 | −1 | −1 | 1 | 28 |
| (12)·A′ | E1 | E A1 | 2 | −1 | 2 | −1 | 0 | 0 | 2 | −1 | 0 | 16 |
| (12)·A′′ | E2 | E A2 | 2 | −1 | 2 | −1 | 0 | 0 | −2 | 1 | 0 | 16 |
| (11)·A | E3 | A1E | 2 | −1 | −1 | 2 | 2 | −1 | 0 | 0 | 0 | 20 |
| (11)·B | E4 | A2E | 2 | −1 | −1 | 2 | −2 | 1 | 0 | 0 | 0 | 12 |
| (01)·A | G | EE | 4 | 1 | −2 | −2 | 0 | 0 | 0 | 0 | 0 | 64 |
We prefer a third labeling scheme (column S1), for which G36 is written as the semi-direct product (CI3 × CI3) ⋊ C2v, as reported by Ezra.16 The theoretical background was described in detail also by Altmann.17 The direct product CI3 × CI3 of the two intrinsic (superscript I) C3 groups of the internal rotors, which is an invariant subgroup of G36, decomposes into four orbits under C2v (Table 2). One representative of each orbit forms the first part of the symmetry label, e.g. (01). The numbers σ = 0, 1, 2 represent the three symmetry species A, Ea, Eb, respectively, of the group C3 and correspond to the transformation properties of the C3-adapted planar rotor wave functions ei(3k+σ)φ with 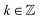 and the torsional angle φ. Each orbit is associated with its little co-set. They form subgroups of C2v. The symmetry species of the little co-sets provide the second part of our symmetry labels.
and the torsional angle φ. Each orbit is associated with its little co-set. They form subgroups of C2v. The symmetry species of the little co-sets provide the second part of our symmetry labels.
| Orbit (σ1, σ2) | Little co-set |
|---|---|
| (00) | C 2v |
| (11), (22) | C 2 |
| (12), (21) | C s |
| (01), (10), (02), (20) | C 1 |
Nuclear spin statistics
The total wave function ψtot = ψel·ψvib·ψrotors·ψns must obey the Pauli principle, i.e. it changes sign under an odd number of fermion permutations (here protons) and retains sign under an even number of permutations. It belongs thus either to the species (00)·A1 or (00)·A2. There are 256 nuclear spin functions arising from 8 protons in DMTP with the representation Γns = 36 (00)·A1 + 28 (00)·B2 + 10 (12)·A′ + 6 (12)·A′′ + 10 (11)·A + 6 (11)·B + 32 (01)·A.We determined the spin weights (column wt in Table 1) by counting the number of allowed total wave functions for Γel = Γvib = (00)·A1.
Selection rules
The dipole moment vector in space fixed coordinates is invariant under pure permutation operations but changes sign under inversion. Therefore, it transforms as Γμ = (00)·A2. In the vibronic ground state, transitions between two rotorsional levels i and j are allowed, if Γi × Γμ × Γj ⊃ (00)·A1. The resulting four torsional components of two different b-type transitions, namely KaKc: ee ↔ oo and eo ↔ oe (e = even, o = odd) along with their spin statistical weights are given in Table 3. They are determined by correlation with the molecular symmetry group C2v(M) of the hypothetical rigid DMTP. If the quantum numbers Ka and Kc are known, the torsional state can be labeled conveniently by the first part (σ1,σ2) of the full symmetry label. We will use this abbreviated notation throughout this paper.| ee ↔ oo transitions | wt | eo ↔ oe transitions | wt |
|---|---|---|---|
| (00)·A1 ↔ (00)·A2 | 36 | (00)·B1 ↔ (00)·B2 | 28 |
| (12)·A′ ↔ (12)·A′′ | 16 | (12)·A′ ↔ (12)·A′′ | 16 |
| (11)·A ↔ (11)·A | 20 | (11)·B ↔ (11)·B | 12 |
| (01)·A ↔ (01)·A | 64 | (01)·A ↔ (01)·A | 64 |
Microwave spectroscopy
Experimental setup
The rotational spectra were measured with two molecular beam Fourier transform microwave spectrometers covering the frequency ranges from 2 to 26.5 GHz18 and from 26.5 to 40 GHz.19 DMTP was purchased from Alfa Aesar GmbH & Co KG, Karlsruhe, Germany with a stated purity of over 98%. Some drops of the liquid substance were put on a 5 cm piece of a pipe cleaner, which was placed inside a steel tube close to the nozzle. A helium stream at a pressure of approximately 200 kPa flowed over the substance and the helium–substance mixture was expanded into the cavity.Spectral analysis
The QCC results have shown that DMTP does not possess dipole moment components in a- and c-direction. Only b-type transitions are thus expected, which is in agreement with the group theoretical results (see section Selection rules). In the first step, the b-type rigid-rotor spectrum was predicted using the rotational constants calculated at the MP2/cc-pVTZ level. Based on this prediction, a broadband scan in the frequency range from 8.5 to 12.0 GHz was measured, where overlapping spectra in a step width of 0.25 MHz were automatically recorded. In the next step, all lines were remeasured at higher resolution.At the beginning, the transitions 220 ← 211, 321 ← 312, and 422 ← 413 as well as 221 ← 212 and 322 ← 313 were considered. They were not the most intensive transitions in the scan but their positions could be predicted quite precisely. After these transitions had been assigned and fitted, further lines in the scan were added to the fit. Afterwards, transitions outside the scan region were also included. The results are given as Fit I in Table 4. A prediction considering the internal rotation of both methyl groups was then carried out using the program XIAM.20 The angles ∠(i1,n) and ∠(i2,n), n = a, b, c, as well as the initial V3 potential were taken from the values calculated at the MP2/cc-pVTZ level. Supported by the intensities from the spin statistical weights, the (01), (11), and (12) species in the scan were successfully assigned. The splittings were up to 0.5 GHz. Finally, further torsional transitions up to J = 7 and Ka = 5 were measured to increase the data set.
| Par.a | Unit | Fit I | Fit II | Calc.b |
|---|---|---|---|---|
| a All parameters refer to the principal axis system. Watson’s A reduction and Ir representation were used. A comparison with the fits using Watson’s S reduction is given in Table S6 in the ESI. b At MP2/6-311++G(d,p) level. c The angle ∠(i2,a) for the second rotor is derived from the relation π − ∠(i1,a). d Standard deviation of the fit. e Number of lines. | ||||
| A | GHz | 4.97224360(42) | 4.96500697(89) | 4.9414 |
| B | GHz | 1.74696506(27) | 1.74694756(13) | 1.7426 |
| C | GHz | 1.31337981(20) | 1.313336623(81) | 1.3094 |
| ΔJ | kHz | 0.0684(28) | 0.0672(11) | 0.0630 |
| ΔJK | kHz | 0.2071(93) | 0.1579(37) | 0.171 |
| ΔK | kHz | 3.2158(98) | 0.9161(43) | 0.803 |
| δ J | kHz | 0.0196(14) | 0.01839(51) | 0.0171 |
| δ K | kHz | 0.136(23) | 0.1031(89) | 0.0390 |
| V 3 | cm−1 | 247.95594(30) | 241.2 | |
| I α | uÅ2 | 3.16442(39) | ||
| ∠(i,a) | ° | 14.5931(78)c | 14.90 | |
| ∠(i,b) | ° | 75.4069(78) | 75.10 | |
| ∠(i,c) | ° | 90.00(fixed) | 89.96 | |
| F 12 | GHz | −7.320(12) | ||
| D pi2J | kHz | −2.78(25) | ||
| D pi2K | kHz | −98.9(42) | ||
| D pi2− | kHz | 1.97(19) | ||
| σ | kHz | 2.6 | 1.9 | |
| N | 41 | 157 | ||
The typical high resolution spectra of the four torsional components are visualized in Fig. 3. We notice that in many transitions, the (00) and the (01) species indicate additional small splittings of about 10 kHz. These splittings are probably due to spin–spin or spin–rotation coupling of the protons. However, for the (01) species, another explanation is possible. In G36 molecules, the (01) and the (10) species are degenerate. Under the influence of centrifugal distortion, this degeneracy could be lifted if rotations about axes other than a, b, and c are considered.21 Nevertheless, since the splittings are small and no theory is available for spin–spin and spin–rotation couplings of molecules with 8 protons like DMTP, nor for the effect mentioned above, no support for this argument can be given. We note that though the PES in section Quantum chemical calculations shows negligible top–top interaction, the (11)–(12) splittings are not small. This observation is mainly related to the kinetic coupling.
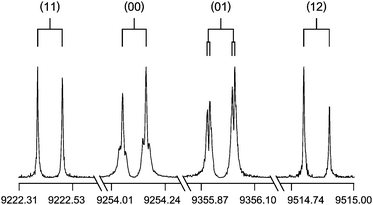 | ||
| Fig. 3 Typical spectra of the four torsional species (00), (01), (11), and (12) of the 321 ← 312 transition of DMTP (in MHz). The experimental resolution is 2 kHz, the typical line width 10 kHz (FWHH). The larger splittings indicated by brackets are due to the Doppler effect; the smaller brackets indicate splittings of unknown origin. For these spectra, 21–614 decays were co-added. The intensities are normalized. An energy level diagram to illustrate the splitting is given in Fig. S-F4 in the ESI.† | ||
Results of the fits and discussion
In total, 157 torsional components were fitted to a standard deviation of 1.9 kHz, within the measurement accuracy. The rotational and centrifugal distortion constants were well determined. The V3 potential, the moment of inertia Iα, the angles δ between the internal rotation axes and the principal a-axis, the kinetic coupling between the two rotors F12, and three higher order parameters Dpi2J, Dpi2K, and Dpi2− were also fitted as linear combinations of the two tops. The results are given as Fit II in Table 4; the frequency list is in Table S5 (ESI†).The experimental rotational constants were compared with those from QCCs. We found the best agreements with the method M06-2X, followed by MP2 and B3LYP, as shown in Table S4 (ESI†). The smallest Pople basis set shows better agreement than bigger basis sets, which is probably the result of error compensation.
The calculated barriers to internal rotation vary in a wide range between 215.5 cm−1 and 405.2 cm−1, but they are still in the same order of magnitude as the experimental V3 potential of 248.0 cm−1. The HF method overestimates the barrier by over 100 cm−1, whereas the MP2 method underestimates the barrier by up to 30 cm−1. We found that the Dunning basis sets yield better results than the Pople ones. The barrier of 248.4 cm−1 calculated at the MP2/6-311G(d,p) level matches the experimental value best.
The barriers of DMTP are closer to the value found for the mono-substituted molecule (8) rather than that of (7), which has a more similar structure (see Fig. 1). In molecules with other hetero atoms (oxygen or nitrogen) or with two hetero atoms, the barrier changes significantly. Our attempts to find out a rule to predict the methyl barriers in unsaturated heterocyclic five-membered rings failed. Unlike in alkyl acetates where the barrier remains largely invariant at approximately 100 cm−1, this value cannot be easily predicted when electronic effect exists, e.g. by the presence of double bonds. In unsaturated acetates,22,23 π-electron conjugation, extending from the double bond to the ester group, is possible, which causes significantly different barriers.
For unsaturated rings with conjugated double bonds like DMTP, it is mostly probable that this electronic system influences the barrier heights. However, more investigations have to be performed to confirm this speculation and moving towards a more precise prediction of the methyl barriers.
Conclusions
The spectrum of DMTP recorded under molecular beam conditions was successfully assigned using a combination of FT microwave spectroscopy, group theory, and quantum chemistry. The rotational constants obtained by geometry optimizations at different levels of theory are in good agreement with the experimental values. A C2v equilibrium structure was calculated, where one hydrogen atom of each methyl group is anti-periplanar to the sulfur atom, and the two methyl groups are thus equivalent. They undergo internal rotations with a barrier height of 247.95594(30) cm−1, which is in reasonable agreement with the calculated barrier.Acknowledgements
V.V. thanks the Fonds der Chemischen Industrie (FCI) for a PhD fellowship. Simulations were performed with computing resources granted by JARA-HPC from the RWTH Aachen University under the project jara0124.References
- J.-U. Grabow, H. Hartwig, N. Heineking, W. Jäger, H. Mäder, H. W. Nicolaisen and W. Stahl, J. Mol. Struct., 2002, 612, 349 CrossRef CAS.
- W. Arnold, H. Dreizler and H. D. Rudolph, Z. Naturforsch., A: Astrophys., Phys. Phys. Chem., 1968, 23, 301 CAS.
- H.-W. Nicolaisen, J.-U. Grabow, N. Heineking and W. Stahl, Z. Naturforsch., A: Phys. Sci., 1991, 46, 635 CrossRef CAS.
- E. Fliege, Z. Naturforsch., A: Phys. Sci., 1990, 45, 911 CrossRef CAS.
- W. G. Norris and L. C. Krisher, J. Chem. Phys., 1969, 51, 403 CrossRef CAS PubMed.
- T. Ogata and K. Kozima, Bull. Chem. Soc. Jpn., 1971, 44, 2344 CrossRef CAS.
- N. M. Pozdeev, L. N. Gunderova and A. A. Shapkin, Opt. Spektrosk., 1970, 28, 254 CAS.
- T. Ogata and K. Kozima, J. Mol. Spectrosc., 1972, 42, 38 CrossRef CAS.
- B. Bak, D. Christensen, L. Hansen-Nygaard and J. Rastrup-Andersen, J. Mol. Spectrosc., 1961, 7, 58 CrossRef CAS.
- M. Tanabe, N. Kuze, H. Fujiwara, H. Takeuchi and S. Konaka, J. Mol. Struct.: THEOCHEM, 1995, 372, 173 CrossRef CAS.
- M. J. Frisch, et al., Gaussian 09 (Revision A.02), Gaussian, Inc., Wallingford CT, 2009 Search PubMed.
- H. B. Schlegel, J. Comput. Chem., 1982, 3, 214 CrossRef CAS PubMed.
- D. Moran, A. C. Simmonett, F. E. Leach, W. D. Allen, P. v. R. Schleyer and H. F. Schaefer, J. Am. Chem. Soc., 2006, 128, 9342 CrossRef CAS PubMed.
- H. Dreizler, Z. Naturforsch., 1961, 16a, 1354 CAS.
- P. R. Bunker and P. Jensen, Molecular Symmetry and Spectroscopy, NRC Research Press, Ottawa, Ontario, Canada, 2006 Search PubMed.
- G. S. Ezra, Symmetry Properties of Molecules, Lecture Notes in Chemistry 28, Springer-Verlag, Berlin, Heidelberg, New York, 1982 Search PubMed.
- S. L. Altmann, Induced Representations in Crystals and Molecules, Point, space and nonrigid molecule groups, Academic Press, London, New York, San Francisco, 1977 Search PubMed.
- J.-U. Grabow, W. Stahl and H. Dreizler, Rev. Sci. Instrum., 1996, 67, 4072 CrossRef CAS PubMed.
- I. Merke, W. Stahl and H. Dreizler, Z. Naturforsch., A: Phys. Sci., 1994, 49, 490 CrossRef CAS.
- H. Hartwig and H. Dreizler, Z. Naturforsch., A: Phys. Sci., 1996, 51, 923 Search PubMed.
- P. Jensen and P. R. Bunker, J. Mol. Spectrosc., 1994, 164, 315 CrossRef.
- H. V. L. Nguyen, A. Jabri, V. Van and W. Stahl, J. Phys. Chem. A, 2014, 118, 12130 CrossRef.
- H. V. L. Nguyen and W. Stahl, J. Mol. Spectrosc., 2010, 264, 120 CrossRef.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c5cp03513a |
| This journal is © the Owner Societies 2015 |