Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Self-assembled supramolecular cages containing ruthenium(II) polypyridyl complexes†
Jiajia
Yang
ab,
Mohan
Bhadbhade
c,
William A.
Donald
b,
Hasti
Iranmanesh
b,
Evan G.
Moore
d,
Hong
Yan
a and
Jonathon E.
Beves
*ab
aState Key Laboratory of Coordination Chemistry, School of Chemistry and Chemical Engineering, Nanjing University, Nanjing 210093, China
bSchool of Chemistry, The University of New South Wales (UNSW), Sydney, NSW 2052, Australia. E-mail: j.beves@unsw.edu.au
cMark Wainwright Analytical Centre, The University of New South Wales (UNSW), Sydney, NSW 2052, Australia
dSchool of Chemistry and Molecular Biosciences, The University of Queensland, Brisbane, QLD 4072, Australia
First published on 13th February 2015
Abstract
Substitution-inert, redox- and photo-active ruthenium(II) complexes based on 2,2′,6′,2′′-terpyridine ligands were self-assembled into discrete supramolecular cages via coordination to palladium(II) centres and characterised by NMR, ESI-MS and X-ray crystallography.
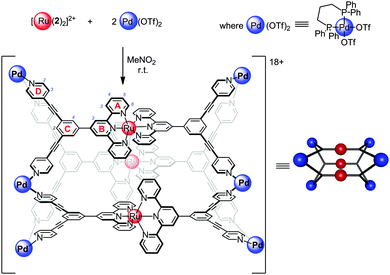
Ruthenium(II) polypyridyl complexes exhibit remarkable photophysical properties and stability,1 continuing to find new applications including water splitting,2 visible light photoredox processes3 and biological imaging.4 Many examples of ruthenium(II) polypyridines have been reported being incorporated into extended structures such as organic polymers,5 vesicles,6 coordination polymers7 and networks,8 as well as acting as photosensitizers for molecular machines9 and as components of discrete 2D macrocycles10 or related assemblies.11 However, examples of 3D molecular cages12–14 containing Ru(II) centres remain rare,12,15 in part due to the inert nature of this low-spin d6 complexes. Herein we report, to the best of our knowledge, the first structural characterisation of incorporation of the prototypical [Ru(tpy)2]2+ (tpy = 2,2′,6′,2′′-terpyridine16–18) unit into a discrete 3D molecular cage.19 Functionalised [Ru(tpy)2]2+ units are particularly appealing as only a single isomer of this complex is possible, in contrast to bidentate analogues such as Ru(bpy)32+ (bpy = 2,2′-bipyridine), which greatly simplifies the number of possible species formed when linking multiple Ru(II) centres. Complexes [Ru(1)2]2+ and [Ru(2)2]2+ feature inert [Ru(tpy)2]2+ units decorated with pendant pyridyl groups capable of binding additional metal centres (Fig. 1).20 Square planar Pd(II) complexes are well suited to binding pyridyl ligands to form metallosupramolecular structures, using approaches pioneered by Fujita21 and Stang.22 Reaction of [Ru(1)2](PF6)2 with two equivalents of Pd(dppp)(OTf)2 (dppp = 1,3-diphenylphosphinopropane) in nitromethane at room temperature (Fig. 2) immediately gave a single major species in solution, as characterised by 1H, 13C and 31P NMR (Fig. 3). This assembly appears quantitative by NMR. The four-fold symmetry of the parent complex (Fig. 3a) is maintained in the product indicating the formation of a highly symmetric species with all tpy ligands being equivalent. Significant changes in NMR chemical shifts and very considerable broadening of signals at room temperature are observed upon Pd(II) coordination. Variable temperature NMR confirmed room temperature to be close to the coalescence temperature (in CD3NO2 or CD3CN). High temperature (348 K, Fig. 3c) 1H and heteronuclear 2D NMR experiments (see ESI,† Fig. S13–S17 for details) were used to unambiguously assign all proton and carbon NMR signals. The signals of the protons of the pendant pyridyl groups are shifted slightly upfield (Δδ HD2 − 0.09; HD3 − 0.17 ppm; Fig. 3a and b) upon coordination to the Pd(II) centre. However, the most significant changes in chemical shifts are for the signals of the terpyridine moiety, which is distant from the Pd(II) coordination site. Signals corresponding to the protons of the terminal pyridine ring (ring A, see Fig. 1 for labelling scheme) are shifted upfield (Δδ ppm: HA3 − 0.46; HA4 − 1.03; HA5 − 1.34; HA6 − 0.54) upon coordination with the Pd(II) corner units. These upfield shifts are consistent with the highly shielding environment expected within a molecular cage.23 The 31P{1H} NMR spectrum (Fig. S8, ESI†) exhibited a single sharp singlet at 8.81 ppm for the dppp ligand, also supporting the high symmetry of the structure, in addition to the expected multiplet for the PF6− anions (−144 ppm). The 13C{1H} spectrum (Fig. S7, ESI†) was fully assigned by 2D experiments (see Fig. S11, S12, S16 and S17, ESI†), including the two non-equivalent phenyl rings of the dppp ligand. The 13C NMR signals showed characteristic peak shifts upon coordination to Pd(II), with signals of the CD2 and CD3 carbons (Δδ CD2 − 0.6; CD3 + 2.7; CD4 − 3.1 ppm) and the carbons on the central phenyl ring (Δδ CC1 + 2.1; CC2 − 0.9; CC4 + 1.2; CC5 − 0.8 ppm) showing the largest shifts relative to Ru(1)2(PF6)2.
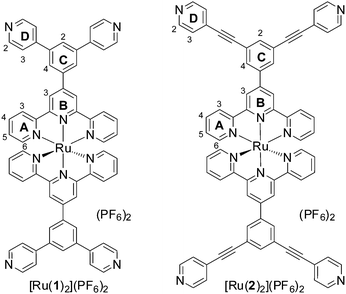 | ||
| Fig. 1 Structure of complexes that are building blocks of more complex supramolecular cages, showing the numbering scheme adopted. | ||
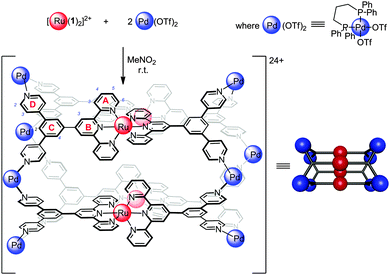 | ||
| Fig. 2 Synthesis of molecular tetrameric cage 324+ from complex [Ru(1)2](PF6)2 and Pd(dppp)(OTf)2 in nitromethane or acetonitrile at room temperature within minutes. | ||
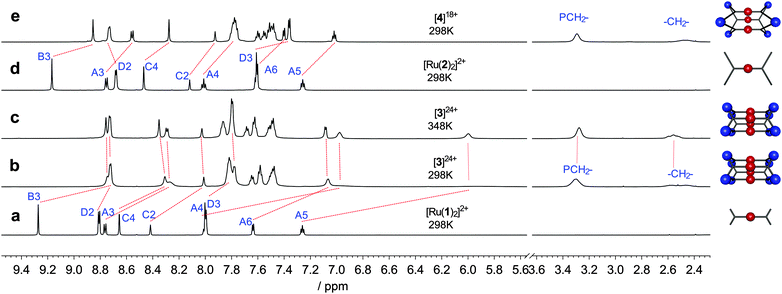 | ||
| Fig. 3 1H-NMR (CD3NO2, 600 MHz) spectra of (a) Ru(1)2(PF6)2 298 K; (b) cage 3 298 K; (c) cage 3 348 K; (d) Ru(2)2(PF6)2 298 K and (e) cage 4 298 K. See Fig. 1 for labelling scheme. Peaks not labelled correspond to the two non-equivalent phenyl rings of the dppp ligand. | ||
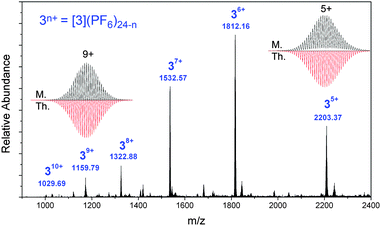
Electrospray ionisation mass spectrometry (ESI-MS) of a nitromethane solution of the product resulted in the formation of a relatively abundant distribution of ions that were charged from +5 to +10 (Fig. 4). The difference in m/z values between adjacent ions in the distribution and the isotopic patterns were assigned to [3(PF6−)24−n]n+ (n = 5 to 10), formed by the sequential loss of PF6 counter anions from [3(PF6)24] (11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 742 Da; Fig. 4). This Ru4Pd8 tetrameric cage structure, [3](PF6)24, is the smallest least-strained structure possible for this system.24 Additional peaks were observed corresponding to the loss of one Pd(dppp)2+ unit which were confirmed by collision-induced dissociation (CID) experiments. This structure is also consistent with the broad 1H NMR signals observed in solution, as the {Ru(tpy)2} units rotate slowly on the NMR timescale and the environment inside and outside of the cage is non-equivalent. This restricted rotation is due to the close contact between the {Ru(tpy)2} groups forming something resembling a poorly assembled gear box (see later X-ray structure discussion). Although stable in acetonitrile at high concentrations (>1 mM), based on 1H NMR, the cage disassembles upon dissolution in acetonitrile (see Fig. S25, ESI†), which is not surprising given this solvent can act as an excellent ligand for Pd(II) centres. However, the complex was observed to be stable in pure nitromethane (Fig. S26, ESI†), a polar but very weakly coordinating solvent, over the same concentration range, confirming this solvent as an excellent choice for this class of metallosupramolecular systems.
742 Da; Fig. 4). This Ru4Pd8 tetrameric cage structure, [3](PF6)24, is the smallest least-strained structure possible for this system.24 Additional peaks were observed corresponding to the loss of one Pd(dppp)2+ unit which were confirmed by collision-induced dissociation (CID) experiments. This structure is also consistent with the broad 1H NMR signals observed in solution, as the {Ru(tpy)2} units rotate slowly on the NMR timescale and the environment inside and outside of the cage is non-equivalent. This restricted rotation is due to the close contact between the {Ru(tpy)2} groups forming something resembling a poorly assembled gear box (see later X-ray structure discussion). Although stable in acetonitrile at high concentrations (>1 mM), based on 1H NMR, the cage disassembles upon dissolution in acetonitrile (see Fig. S25, ESI†), which is not surprising given this solvent can act as an excellent ligand for Pd(II) centres. However, the complex was observed to be stable in pure nitromethane (Fig. S26, ESI†), a polar but very weakly coordinating solvent, over the same concentration range, confirming this solvent as an excellent choice for this class of metallosupramolecular systems.
The analogous reaction with complex [Ru(2)2](PF6)2, which features alkyne spacers between the phenyl and pendant pyridyl rings, and Pd(dppp)(OTf)2 also formed a single major product in solution (Fig. 5). The 31P{1H} spectrum (Fig. S29, ESI†) again revealed a single sharp singlet at 8.91 ppm, effectively identical to that observed for [3]24+, and consistent with the formation of a single major product. However, in contrast to cage 324+, the 1H NMR signals (Fig. 3e) of this new species were not significantly broader than the parent Ru(2)2+ complex, suggesting the rotation of {Ru(tpy)2} units was effectively unhindered in this structure. The 1H NMR signals corresponding to the pendant pyridine protons were shifted relative to [Ru(2)2](PF6)2 (Δδ HD2 + 0.05; HD3 − 0.32), but as was the case for cage 324+, the signals of the terpyridine group were significantly affected, all being shifted upfield (Δδ HB3 − 0.31; HA3 − 0.19; HA4 − 0.26; HA5 − 0.24; HA6 − 0.28) but to a much less extent than in cage 324+. These peak shifts correspond to a cage environment considerably less shielded than in cage 324+. The 13C{1H} NMR spectrum (Fig. S28, ESI†) reveals relatively small changes in peak shifts for most signals, with the notable exceptions of the pendant pyridyl signals (Δδ CD2 − 0.3; CD3 + 2.7; CD4 + 3.6 ppm), the alkynes (Δδ CC–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C + 4.5 ppm; CD–C
C + 4.5 ppm; CD–C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C − 1.5 ppm) and the central phenyl ring (Δδ CC1 − 1.6; CC2 + 0.2; CC4 + 2.0; CC5 + 0.1 ppm). These peak shifts reflect not only the electronic effect of Pd(II) coordination to the pyridyl group, but also the strain introduced to the alkyne upon bending to form a smaller cyclic structure, hence the observed changes are significantly different to that observed for cage 324+.
C − 1.5 ppm) and the central phenyl ring (Δδ CC1 − 1.6; CC2 + 0.2; CC4 + 2.0; CC5 + 0.1 ppm). These peak shifts reflect not only the electronic effect of Pd(II) coordination to the pyridyl group, but also the strain introduced to the alkyne upon bending to form a smaller cyclic structure, hence the observed changes are significantly different to that observed for cage 324+.
 | ||
| Fig. 5 Synthesis of molecular trimeric cage 424+ from complex [Ru(2)2](PF6)2 and Pd(dppp)(OTf)2 in nitromethane or acetonitrile at room temperature within minutes. | ||
The ESI-MS of a solution of this product (Fig. S36, ESI†) revealed a trimeric, rather than tetrameric structure, consistent with the structure of cage 4(PF6)18 (Fig. 5) indicating the additional flexibility of the alkyne spacers was sufficient to allow a smaller structure to form and simple molecular modelling (MMFF, Fig. S40, ESI†) supports this assignment. This structure was found to precipitate from solution (acetonitrile) over time to form an insoluble red powder, presumably a coordination polymer. In nitromethane the cage appears stable over several months in solution.
Slow diffusion of toluene into a nitromethane solution of cage 3(PF6)24 gave red block crystals suitable for X-ray diffraction.‡ The molecule crystallises in the P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) space group with the asymmetric unit containing half of one cage molecule and disordered solvents and anions (Fig. 6). The complex forms a box-like structure approximately 21 × 21 × 32 Å in dimensions with Pd(II) centres at each end forming near perfect squares (Pd–Pd–Pd angles of 86.0–92.8° and Pd⋯Pd distances of 13.2–13.4 Å). The centre of the cage is occupied by {Ru(tpy)2} units with alternating Ru⋯Ru distances of 11.82 and 8.78 Å and inter-{Ru(tpy)2} pyridine⋯pyridine separations of (centroid⋯centroid) 3.86 and 5.34 Å forming portals which are occupied by PF6− counterions. The cavities and each end of the cage are sufficiently large to potentially accommodate large guests such as a C60 molecule. The {Ru(tpy)2} groups form pairs of terpyridine ‘embraces’25 (Fig. S37, ESI†), a type of favourable edge-to-face and face-to-face aromatic interactions, as often seen in solid state packing of simple {M(tpy)2}n+ complexes. These interactions reveal the origin of the restricted rotation of these units in solution. Although not requiring a concerted rotation of all the {Ru(tpy)2} units, it appears this type of favourable π–π stacking interactions are more significant than simply steric crowding and result in the hindered rotation of these units. The cages are assembled together in the crystal structure via additional intermolecular terpyridine embraces to form 1D chain along the crystallographic a axis as well as extensive π–π interactions between the pyridine and phenyl rings along the b axis (Fig. S39, ESI†).
space group with the asymmetric unit containing half of one cage molecule and disordered solvents and anions (Fig. 6). The complex forms a box-like structure approximately 21 × 21 × 32 Å in dimensions with Pd(II) centres at each end forming near perfect squares (Pd–Pd–Pd angles of 86.0–92.8° and Pd⋯Pd distances of 13.2–13.4 Å). The centre of the cage is occupied by {Ru(tpy)2} units with alternating Ru⋯Ru distances of 11.82 and 8.78 Å and inter-{Ru(tpy)2} pyridine⋯pyridine separations of (centroid⋯centroid) 3.86 and 5.34 Å forming portals which are occupied by PF6− counterions. The cavities and each end of the cage are sufficiently large to potentially accommodate large guests such as a C60 molecule. The {Ru(tpy)2} groups form pairs of terpyridine ‘embraces’25 (Fig. S37, ESI†), a type of favourable edge-to-face and face-to-face aromatic interactions, as often seen in solid state packing of simple {M(tpy)2}n+ complexes. These interactions reveal the origin of the restricted rotation of these units in solution. Although not requiring a concerted rotation of all the {Ru(tpy)2} units, it appears this type of favourable π–π stacking interactions are more significant than simply steric crowding and result in the hindered rotation of these units. The cages are assembled together in the crystal structure via additional intermolecular terpyridine embraces to form 1D chain along the crystallographic a axis as well as extensive π–π interactions between the pyridine and phenyl rings along the b axis (Fig. S39, ESI†).
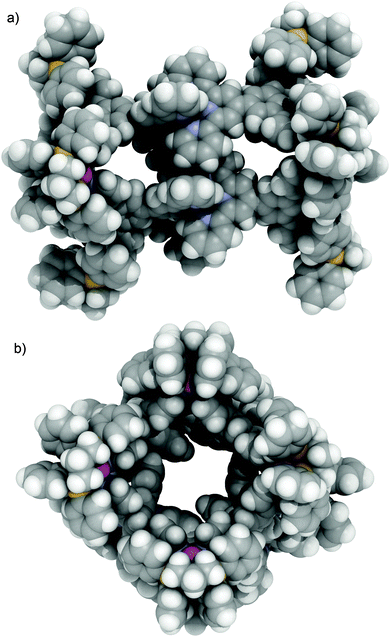 | ||
| Fig. 6 The single crystal X-ray crystal structure of [3](PF6)17.5·CH3NO2. Viewed down the (a) crystallographic a axis, and (b) c* axis. Solvent and anions omitted for clarity. | ||
Preliminary investigations of the photophysical properties of cage [3](PF6)24 indicate the functionality of the parent complex is retained in the cage structure. The 1MLCT absorption maxima of the [Ru(1)2](PF6)2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 complex and cage [3](PF6)24 were both located at 490 nm, while the 3MLCT emission spectra were essentially superimposable centred at 640 nm. The excited state lifetimes (1.26 ± 0.01 ns and 1.21 ± 0.01 ns respectively) were similarly identical, and are comparable to those of related [Ru(4′-tolyl-tpy)(tpytpy)]2+ complexes (see ESI† for details).26
20 complex and cage [3](PF6)24 were both located at 490 nm, while the 3MLCT emission spectra were essentially superimposable centred at 640 nm. The excited state lifetimes (1.26 ± 0.01 ns and 1.21 ± 0.01 ns respectively) were similarly identical, and are comparable to those of related [Ru(4′-tolyl-tpy)(tpytpy)]2+ complexes (see ESI† for details).26
Three dimensional molecular cages containing [Ru(tpy)2]2+ units are reported and characterised in solution and the solid state. The photophysical properties of the parent Ru(II) complex [Ru(1)2]2+ are retained in the cage 324+, suggesting this new class of molecular cages may be potential candidates to act as photosensitizers for bound guest molecules. The introduction of an alkyne spacer, producing a larger ligand, resulted in the formation of a smaller, trimeric cage highlighting the flexibility of these spacer units and the subtlety of the assembly process.
This work was supported by a National Science Foundation of China (NSFC) Research Fund for International Young Scientists Project (No. 21450110060 and 21271102). The Australian Research Council is acknowledged for Future Fellowship (EGM, FT100100795) and Discovery Early Career Research Awards (WAD, DE130100424). Photophysical measurements were undertaken at the Photochemistry and Ultrafast Laser Spectroscopy (PULS ) facility, School of Chemistry and Molecular Biosciences, with financial support from the University of Queensland (MEI-2013000106). Crystallographic data was collected in-house at UNSW, or at the MX1 beamline at the Australian Synchrotron under a Collaborative Access Program (AS143_MXCAP_8503).
) facility, School of Chemistry and Molecular Biosciences, with financial support from the University of Queensland (MEI-2013000106). Crystallographic data was collected in-house at UNSW, or at the MX1 beamline at the Australian Synchrotron under a Collaborative Access Program (AS143_MXCAP_8503).
Notes and references
- S. Campagna, F. Puntoriero, F. Nastasi, G. Bergamini and V. Balzani, in Photochemistry and Photophysics of Coordination Compounds I, ed. V. Balzani and S. Campagna, Springer-Verlag Berlin, Berlin, 2007, vol. 280, pp. 117–214 Search PubMed.
- (a) W. J. Youngblood, S.-H. A. Lee, Y. Kobayashi, E. A. Hernandez-Pagan, P. G. Hoertz, T. A. Moore, A. L. Moore, D. Gust and T. E. Mallouk, J. Am. Chem. Soc., 2009, 131, 926–927 CrossRef CAS PubMed; (b) L. Duan, F. Bozoglian, S. Mandal, B. Stewart, T. Privalov, A. Llobet and L. Sun, Nat. Chem., 2012, 4, 418–423 CrossRef CAS PubMed.
- D. M. Schultz and T. P. Yoon, Science, 2014, 343, 985 CrossRef CAS PubMed.
- V. Fernandez-Moreira, F. L. Thorp-Greenwood and M. P. Coogan, Chem. Commun., 2010, 46, 186–202 RSC.
- Y. Sun, Z. Chen, E. Puodziukynaite, D. M. Jenkins, J. R. Reynolds and K. S. Schanze, Macromolecules, 2012, 45, 2632–2642 CrossRef CAS.
- P. Mahato, S. Saha, S. Choudhury and A. Das, Chem. Commun., 2011, 47, 11074–11076 RSC.
- (a) J. E. Beves, E. C. Constable, C. E. Housecroft, C. J. Kepert and D. J. Price, CrystEngComm, 2007, 9, 456–459 RSC; (b) J. E. Beves, E. C. Constable, C. E. Housecroft, C. J. Kepert, M. Neuburger, D. J. Price, S. Schaffner and J. A. Zampese, Dalton Trans., 2008, 6742–6751 RSC; (c) J. E. Beves, E. C. Constable, S. Decurtins, E. L. Dunphy, C. E. Housecroft, T. D. Keene, M. Neuburger and S. Schaffner, CrystEngComm, 2008, 10, 986–990 RSC; (d) J. E. Beves, D. J. Bray, J. K. Clegg, E. C. Constable, C. E. Housecroft, K. A. Jolliffe, C. J. Kepert, L. F. Lindoy, M. Neuburger, D. J. Price, S. Schaffner and F. Schaper, Inorg. Chim. Acta, 2008, 361, 2582–2590 CrossRef CAS PubMed.
- (a) M. S. Deshpande, A. S. Kumbhar, V. G. Puranik and K. Selvaraj, Cryst. Growth Des., 2006, 6, 743–748 CrossRef CAS; (b) C. A. Kent, D. Liu, L. Ma, J. M. Papanikolas, T. J. Meyer and W. Lin, J. Am. Chem. Soc., 2011, 133, 12940–12943 CrossRef CAS PubMed; (c) R. W. Larsen and L. Wojtas, J. Phys. Chem. A, 2012, 116, 7830–7835 CrossRef CAS PubMed; (d) T. Toyao, M. Saito, S. Dohshi, K. Mochizuki, M. Iwata, H. Higashimura, Y. Horiuchi and M. Matsuoka, Chem. Commun., 2014, 50, 6779–6781 RSC.
- (a) V. Balzani, G. Bergamini, F. Marchioni and P. Ceroni, Coord. Chem. Rev., 2006, 250, 1254–1266 CrossRef CAS PubMed; (b) S. Bonnet and J.-P. Collin, Chem. Soc. Rev., 2008, 37, 1207–1217 RSC.
- (a) S.-S. Sun and A. J. Lees, Inorg. Chem., 2001, 40, 3154–3160 CrossRef CAS PubMed; (b) G. R. Newkome, T. J. Cho, C. N. Moorefield, R. Cush, P. S. Russo, L. A. Godínez, M. J. Saunders and P. Mohapatra, Chem. – Eur. J., 2002, 8, 2946–2954 CrossRef CAS; (c) G. R. Newkome, T. J. Cho, C. N. Moorefield, P. P. Mohapatra and L. A. Godínez, Chem. – Eur. J., 2004, 10, 1493–1500 CrossRef CAS PubMed; (d) A. Schultz, X. Li, B. Barkakaty, C. N. Moorefield, C. Wesdemiotis and G. R. Newkome, J. Am. Chem. Soc., 2012, 134, 7672–7675 CrossRef CAS PubMed.
- M. W. Cooke and G. S. Hanan, Chem. Soc. Rev., 2007, 36, 1466–1476 RSC.
- For a recent example of a molecular cage prepared with Ru(II) polypyridyl complexes with Pd(II) ions, see: K. Li, L.-Y. Zhang, C. Yan, S.-C. Wei, M. Pan, L. Zhang and C.-Y. Su, J. Am. Chem. Soc., 2014, 136, 4456–4459 CrossRef CAS PubMed.
- For examples of cages featuring Ru(II) organometallics, see (a) B. Therrien, G. Süss-Fink, P. Govindaswamy, A. K. Renfrew and P. J. Dyson, Angew. Chem., Int. Ed., 2008, 47, 3773–3776 CrossRef CAS PubMed; (b) A. Granzhan, T. Riis-Johannessen, R. Scopelliti and K. Severin, Angew. Chem., Int. Ed., 2010, 49, 5515–5518 CrossRef CAS PubMed; (c) L. Wang, J.-F. Liu, W. Yang, F.-Y. Yi, S. Dang and Z.-M. Sun, Dalton Trans., 2014, 43, 17244–17247 RSC.
- For leading examples of functional molecular cages, see (a) M. D. Pluth, R. G. Bergman and K. N. Raymond, Science, 2007, 316, 85–88 CrossRef CAS PubMed; (b) P. Mal, B. Breiner, K. Rissanen and J. R. Nitschke, Science, 2009, 324, 1697–1699 CrossRef CAS PubMed; (c) Y. Inokuma, M. Kawano and M. Fujita, Nat. Chem., 2011, 3, 349–358 CrossRef CAS PubMed.
- For recent examples of cages containing Ru(II) and pyridyl ligands, see (a) A. J. Metherell and M. D. Ward, Chem. Commun., 2014, 50, 6330–6332 RSC; (b) A. J. Metherell and M. D. Ward, Chem. Commun., 2014, 50, 10979–10982 RSC.
- E. C. Constable, Chem. Soc. Rev., 2007, 36, 246–253 RSC.
- It should be noted that the excited state properties of [Ru(tpy)2]2+ are limited compared to [Ru(bpy)3]2+, for details see: A. K. Pal and G. S. Hanan, Chem. Soc. Rev., 2014, 43, 6184–6197 RSC.
- For a recent examples of a terpyridine containing molecular cages, see (a) C. Wang, X.-Q. Hao, M. Wang, C. Guo, B. Xu, E. N. Tan, Y.-Y. Zhang, Y. Yu, Z.-Y. Li, H.-B. Yang, M.-P. Song and X. Li, Chem. Sci., 2014, 5, 1221–1226 RSC; (b) X. Lu, X. Li, K. Guo, T.-Z. Xie, C. N. Moorefield, C. Wesdemiotis and G. R. Newkome, J. Am. Chem. Soc., 2014, 136, 18149–18155 CrossRef CAS PubMed.
- For a recent example of a molecular cage containing [Ru(tpy)2]2+ formed under kinetic control and characterised by NMR and ESI-MS, see: T.-Z. Xie, S.-Y. Liao, K. Guo, X. Lu, X. Dong, M. Huang, C. N. Moorefield, S. Z. D. Cheng, X. Liu, C. Wesdemiotis and G. R. Newkome, J. Am. Chem. Soc., 2014, 136, 8165–8168 CrossRef CAS PubMed.
- J. Yang, J. K. Clegg, Q. Jiang, X. Lui, H. Yan, W. Zhong and J. E. Beves, Dalton Trans., 2013, 42, 15625–15636 RSC.
- (a) M. Fujita, J. Yazaki and K. Ogura, J. Am. Chem. Soc., 1990, 112, 5645–5647 CrossRef CAS; (b) M. Fujita, M. Tominaga, A. Hori and B. Therrien, Acc. Chem. Res., 2005, 38, 369–378 CrossRef CAS PubMed.
- (a) P. J. Stang, D. H. Cao, S. Saito and A. M. Arif, J. Am. Chem. Soc., 1995, 117, 6273–6283 CrossRef CAS; (b) R. Chakrabarty, P. S. Mukherjee and P. J. Stang, Chem. Rev., 2011, 111, 6810–6918 CrossRef CAS PubMed.
- W. Meng, B. Breiner, K. Rissanen, J. D. Thoburn, J. K. Clegg and J. R. Nitschke, Angew. Chem., Int. Ed., 2011, 50, 3479–3483 CrossRef CAS PubMed.
- Self-assembled systems tend to form the smallest least-strained species on entropy grounds. For a detailed discussion see C. Piguet, Chem. Commun., 2010, 46, 6209–6231 RSC.
- For a detailed analysis of tpy embraces, see (a) J. McMurtrie and I. Dance, CrystEngComm, 2005, 7, 216–229 RSC; (b) J. McMurtrie and I. Dance, CrystEngComm, 2009, 11, 1141–1149 RSC.
- E. G. Moore, M. Benaglia, G. Bergamini and P. Ceroni, Eur. J. Inorg. Chem., 2015, 414–420 CrossRef CAS.
Footnotes |
| † Electronic supplementary information (ESI) available: Synthetic procedures, 1H, 13C, 31P, COSY, HSQC, HMBC, NOESY and variable temperature and variable concentration NMR spectra, X-ray crystal refinement details, ESI-MS and photophysical data. CCDC 1040284 and 1045515. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c4cc10292d |
‡ Crystal data and refinement: C232H188N20P8Pd4Ru2·8.75(F6P)·0.5(CNO2), M = 5429.01, T = 150(2) K, λ = 0.71073 Å, triclinic, space group P![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) , a = 23.8865 (13), b = 25.4288 (13), C = 31.7818 (17) (Å), α = 96.150 (3), β = 107.106 (3), γ = 113.591 (2) (°), V (Å3) = 16341.8 (16), Z = 2, m (mm−1) = 0.46, F(000) = 5460.5, data/restr/param. 57493/1988/3008. GOOF on F2 = 1.11, R[F2 > 2s(F2)] = 0.113, wR(F2) = 0.320. CCDC reference number 1040284. , a = 23.8865 (13), b = 25.4288 (13), C = 31.7818 (17) (Å), α = 96.150 (3), β = 107.106 (3), γ = 113.591 (2) (°), V (Å3) = 16341.8 (16), Z = 2, m (mm−1) = 0.46, F(000) = 5460.5, data/restr/param. 57493/1988/3008. GOOF on F2 = 1.11, R[F2 > 2s(F2)] = 0.113, wR(F2) = 0.320. CCDC reference number 1040284. |
| This journal is © The Royal Society of Chemistry 2015 |