Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Dye sensitized solar cells with cobalt and iodine-based electrolyte: the role of thiocyanate-free ruthenium sensitizers†
Kuan-Lin
Wu‡
a,
Yue
Hu‡
b,
Chun-Tien
Chao
a,
Ya-Wen
Yang
a,
Ting-Yun
Hsiao
c,
Neil
Robertson
*b and
Yun
Chi
*a
aDepartment of Chemistry and Low Carbon Energy Research Center, National Tsing Hua University, Hsinchu 30013, Taiwan. E-mail: ychi@mx.nthu.edu.tw
bEaStCHEM, School of Chemistry, University of Edinburgh, King's Buildings, West Mains Road, Edinburgh EH9 3 JJ, UK. E-mail: Neil.Robertson@ed.ac.uk
cDepartment of Engineering and System Science, National Tsing Hua University, Hsinchu 30013, Taiwan
First published on 7th October 2014
Abstract
Three isomeric Ru(II) metal complexes with distinctively oriented tpiq ancillary chelates, TFRS-80a, 80b and 80c, were prepared from the condensation of Ru(4,4′-diethoxycarbonyl-2,2′-bipyridine) (p-cymene)Cl with tpiqH, i.e. 6-(5-(2,6-bis(hexyloxy)phenyl)thiophen-2-yl)-1-(3-(trifluoromethyl)-1H-pyrazol-5-yl)isoquinoline. Photophysical and electrochemical investigations, together with DFT and TD-DFT calculations, allowed a comprehensive understanding of their basic properties in both solution state and on TiO2 surface. DSC cells with both an ultra-thin layer of transparent TiO2 (3.6 μm) and I−/I3− electrolyte were fabricated, for which the symmetric sensitizers TFRS-80a and 80c showed better performances (η = 8.37 and 8.26%) over that of the asymmetric counterpart TFRS-80b (η = 5.55%), the latter suffered from poor dye loading and consequently lowered JSC and VOC. In sharp contrast, all DSC cells with [Co(phen)3]2+/3+ electrolyte gave superior efficiencies (η = 8.36–9.06%), for which the thiocyanate-free architecture, the improved light harvesting capability, and the possession of conjugated and bulky 5-(2,6-bis(hexyloxy)phenyl)thiophen-2-yl functional moieties are three primary factors governing the observed results.
Introduction
Photovoltaic technologies are pivotal to the future progression of human societies, among which dye-sensitized solar cells (DSCs)1 have already achieved a highest conversion efficiency of ∼13%, using Zn(II) porphyrin sensitizers together with Co2+/3+ electrolytes.2 A typical DSC consists of a dye that self-assembles onto a mesoporous TiO2 photoanode. The sensitizers can rapidly inject electrons into the conduction band of the TiO2, following excitation by incident solar irradiation. The oxidized sensitizers are then regenerated by redox couples in the electrolyte and become available for the next round of light harvesting, electron injection and reduction. It is believed that better sensitizers would offer a leap in improvement in the overall efficiency of DSC devices.Organic donor–acceptor dyes with cyanoacrylic anchor are highly competitive due to their potentially simple design, synthetic flexibility and scalability.3 However, their disadvantage is the poor stability under the combined effects of UV irradiation and water content in the electrolyte, which triggers both the reversion of cyanoacrylic acid to aldehyde,4 and photoisomerization of acrylic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C bond.5 Recently, a class of organic dyes bearing benzoic acid have ingeniously avoided these degradation pathways and, in combination with filling defects on the TiO2 surface to attenuate interfacial charge recombination, showed excellent efficiency of >12% with Co2+/3+ electrolytes.6
C bond.5 Recently, a class of organic dyes bearing benzoic acid have ingeniously avoided these degradation pathways and, in combination with filling defects on the TiO2 surface to attenuate interfacial charge recombination, showed excellent efficiency of >12% with Co2+/3+ electrolytes.6
Ru(II) complexes with thiocyanate ancillaries are known to be both efficient and relatively stable;7 hence, they have been subjected to advanced studies aimed towards possible commercialization.8 On the other hand, there are growing studies on Ru(II) sensitizers devoid of thiocyanate ligand, among which van Koten and coworkers have utilized cyclometalate ancillaries to construct the first class of thiocyanate-free Ru(II) sensitizers, albeit of lower efficiency.9 However, their true potential was only realized after Grätzel, who employed the electron deficient 2,4-difluorophenyl pyridinato chelate to construct the sensitizer YE05, showed a prominent conversion efficiency (η) of 10.1% at standard AM 1.5 sunlight.10 Later, cyclometalates and other ancillaries were systematically employed by Berlinguette in attempts to expand this class of Ru(II) sensitizers.11
In this content, our group has carried out studies using N-donor ancillaries, such as pyridyl azolate,12 2,6-diazolyl pyridine13 and 2-azolyl-6-phenylpyridine,14 to replace the aforementioned cyclometalates. Scheme 1 depicts three Ru(II) sensitizers with trans-substituted pyrazolate fragments, all derived from their parent complex TFRS-1, showing respectable η of ≥10% using I−/I3− based electrolyte for TFRS-52,15 and η of 8.71% using [Co(bpy)3]2+/3+ electrolyte for TFRS-42, respectively.16 It is notable that TFRS-42 exhibited the highest η for the Co2+/3+ electrolytic system due to its charge neutrality, greater spatial congestion and absence of thiocyanate ligands, all of which are essential for reducing the recombination across the interface of TiO2 and electrolyte.
These TFRS sensitizers were prepared by coupling of Ru(diethyl 2,2′-bipyridine-4,4′-dicarboxylate) (p-cymene)Cl with two equiv. of chelating pyrazole, followed by hydrolysis in basic media. In the absence of any regioselectivity, a maximum of three isomers would be expected, for which the other two structures would differ from all the trans-substituted TFRS dyes shown in Scheme 1 by reshuffling the orientation of the azolate chelates.17 Herein, we wish to report the detailed study on the system where all three isomeric sensitizers, TFRS-80a, 80b and 80c, have been isolated and characterized. These sensitizers are derived from a π-conjugated tpiqH chelate, i.e. 6-(5-(2,6-bis(hexyloxy)phenyl)thiophen-2-yl)-1-(3-(trifluoromethyl)-1H-pyrazol-5-yl)isoquinoline, such that their higher absorptivity and potential for fabrication of DSCs with high η of 9.06% triggered the full determination of their photophysical and electrochemical properties and structure-efficiency relationship of both I−/I3− and Co2+/3+ based dye-sensitized solar cells (Scheme 2).
Results and discussion
Syntheses
The pyrazolate ancillary, 5-(2,6-bis(hexyloxy)phenyl)thiophen-2-yl substituted tpiqH chelate, was selected for this investigation due to the large π-conjugation expected for both the isoquinolinyl fragment and thienyl appendage at the C6 position, resulting in a combined hyperchromic effect and bathochromic shift in the recorded UV/Vis spectra. Moreover, the main function of the 2,6-dihexyloxylphenyl group is to provide both further increased π-conjugation on the whole molecule and tailored steric encumbrance over the thienyl fragment, such that the formation of intermolecular π–π-stacking interactions around the isoquinolinyl entities can be effectively suppressed. This knowledge is obtained from the molecular design of several efficient DSC sensitizers, on which similar 2,6-dialkoxyphenyl groups were incorporated for suppressing aggregation, improving the solubility in organic solvents, and enhancing the overall efficiency of the fabricated DSC devices.18This chelating ancillary is treated with the metal reagent Ru(4,4′-diethoxycarbonyl-2,2′-bipyridine) (p-cymene)Cl and the basic promoter KOAc in xylenes, according to the established protocol. All three isomeric products were separated using SiO2 column chromatography, after executing two consecutive elutions using a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 mixture of ethyl acetate and hexane, followed by employment of a 1
4 mixture of ethyl acetate and hexane, followed by employment of a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 mixture of ethyl acetate and CH2Cl2. Subsequent hydrolysis of each compound in NaOH–water–acetone mixture afford the carboxylic sensitizers TFRS-80a, 80b and 80c in 32%, 11% and 15% yields, respectively. It is notable that these isomers represent formation of all three possible structural isomeric Ru(II) based complexes, for which their structural identification can be achieved according to their 1H and 19F NMR spectral pattern. Of particular importance is the 19F NMR spectral data, which unambiguous confirmed the symmetric or asymmetric nature of TFRS-80a/80c and TFRS-80b by revealing a single 19F NMR signal and two signals of equal intensity, respectively.
20 mixture of ethyl acetate and CH2Cl2. Subsequent hydrolysis of each compound in NaOH–water–acetone mixture afford the carboxylic sensitizers TFRS-80a, 80b and 80c in 32%, 11% and 15% yields, respectively. It is notable that these isomers represent formation of all three possible structural isomeric Ru(II) based complexes, for which their structural identification can be achieved according to their 1H and 19F NMR spectral pattern. Of particular importance is the 19F NMR spectral data, which unambiguous confirmed the symmetric or asymmetric nature of TFRS-80a/80c and TFRS-80b by revealing a single 19F NMR signal and two signals of equal intensity, respectively.
Photophysical and electrochemical behavior
The absorption spectra of TFRS-80a, 80b and 80c were measured in DMF and are depicted in Fig. 1, while pertinent numerical data are summarized in Table 1. As can be seen, all sensitizers show strong absorption at ∼360 nm with extinction coefficient (ε) falling in the range of 5.1–6.2 × 104 M−1 cm−1, which is apparently due to the ligand centered π–π* transition. In addition, there is another lower energy, broad transition centered at ∼525 nm, with a slightly lower extinction coefficient of 3.2–3.9 × 104 M−1 cm−1. Furthermore, the recorded extinction coefficients are all markedly higher than the corresponding band of N719 (1.4 × 104 M−1 cm−1) and relevant MLCT band of C101 (1.75 × 104 M−1 cm−1)19 and TG6 (2.3 × 104 M−1 cm−1),20 confirming the excellent light harvesting capability in the visible region. The comparisons of experimental UV-visible spectra with the computational simulated oscillator strengths are presented in Fig. S1–S3 of ESI.†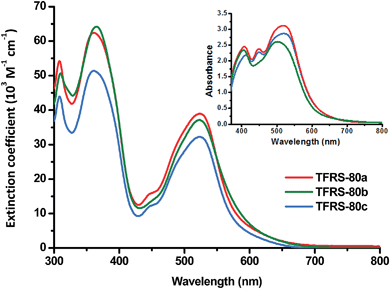 | ||
| Fig. 1 UV/Vis absorption spectra of various TFRS-80 sensitizers (1 × 10−5 M) in DMF. Inset: spectra of samples adsorbed on 5 μm transparent TiO2 thin film. | ||
| Dye | λ abs [nm] (ε × 10−3 [M−1 cm−1]) | E °′ox | E 0–0 | E°′*c |
|---|---|---|---|---|
| a Oxidation potential of dye was measured in DMF with 0.1 M [TBA][PF6] and with a scan rate of 50 mV s−1. It was calibrated with Fc/Fc+ reference and converted to NHE by addition of 0.63 V. b E 0–0 was determined from the intersection of the absorption and the tangent of emission peak in DMF. c E°′* = E°′ox − E0–0. | ||||
| TFRS-80a | 309 (54), 360 (62), 523 (39) | 0.87 | 1.89 | −1.00 |
| TFRS-80b | 310 (51), 366 (64), 524 (37) | 0.83 | 1.90 | −1.07 |
| TFRS-80c | 308 (44), 362 (51), 527 (32) | 0.78 | 1.87 | −1.09 |
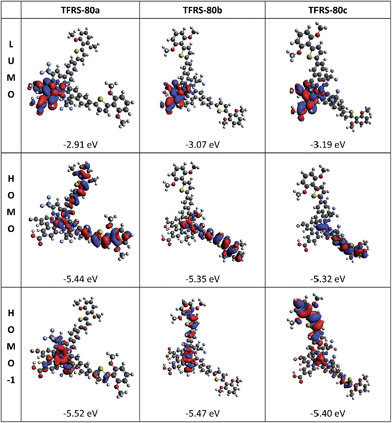
DFT and TD-DFT calculations using a DMF polarizable continuum model were carried out, showing that in each case the LUMO is based upon the dicarboxy bipyridine as expected, and that the HOMO is distributed across the Ru orbitals and the entire tpiq ligand (Fig. 2). In particular, we note that this conjugation extends to the thienyl and 2,6-dihexyloxylphenyl fragments in keeping with the intended ligand design and ensuring effective charge separation between electrons injected into the TiO2 and the positive charge on the oxidized dye. It is also apparent that the delocalization of the HOMO differs across the isomer series, with only that of TFRS-80a spread across both tpiq ligands, presumably accounting for the lower HOMO energy for this dye. The TD-DFT calculations reproduced the transition energies of the charge-transfer bands moderately well, with the results sufficient to give insight into the orbital origins of transitions. The 525 nm charge transfer band in each case (calculated at around 480 nm) originates from a mixture of the Ru(II) metal core and the pyrazolate to dicarboxy bipyridine, i.e. a mixing of MLCT and LLCT transitions, in a way analogous to the assignment made for other TFRS sensitizers.12b
In the context of DSC application, further enhancements in absorption were observed upon depositing these sensitizers on the TiO2 surface, such that all of the absorptions broadened substantially, the recorded spectra showed an absence of the semi-transparent region centered at 430 nm recorded in DMF solution, as well as red-shifting of the lowest energy absorption peak maxima to ∼700 nm. We speculate that such a broadened spectral profile is beneficial to the competitive harvesting of shorter wavelength irradiation,21 particular for DSC devices that utilize the I−/I3− based electrolytes.
Cyclic voltammetry was conducted to reveal the variation in electrochemical potentials among the three isomers, and to verify whether the oxidation potential of the ground state (E°′ox) matches the redox potential of the redox mediators. As shown in Table 1, the oxidation potential of the isomers follows the trend of TFRS-80c < 80b < 80a which are reproduced well by the calculated HOMO energies shown by the computational results (see Tables S1–S3†). The E°′ox is attributed to the Ru(II) metal oxidation, and appeared in the range of 0.78–0.87 V (vs. NHE, normal hydrogen electrode), all more positive than that of I−/I3− redox couple (E° = ca. 0.4 V) and the redox potential of the [Co(phen)3]2+/3+ (E° = ca. 0.62 V vs. NHE). The limited variation in E°′ox potentials in these studies can be attributed to the identical local coordination environment around the central Ru(II) atom, with the small differences attributed to different amounts of delocalization and stabilization of the HOMO. In addition, the zero–zero transition energy (E0–0) or band gap was determined from the intersection of the absorption and normalized emission spectra. From this, the excited-state oxidation potential (E°′*) is estimated from the difference of E°′ox and E0–0, from which the calculated values of −1.00 to −1.09 V (vs. NHE) were obtained. Since all of the E°′* are significantly more negative than the conduction band edge of the TiO2 electrode (ECB ∼ −0.2 and −0.5 V vs. NHE),22 this confirms that efficient electron injection from the excited sensitizer to the conduction band of TiO2 should occur.
To probe the longer-term stability of the dye oxidized state we carried out spectroelectrochemical studies of each dye in solution upon oxidation (Fig. S4–S6†). It was apparent that TFRS-80c showed the best isosbestic points during the oxidation and was more fully returned to the starting spectrum upon reduction in comparison with either TFRS-80a or TFRS-80b. We have previously suggested the possibility of some isomerization within this type of dye series upon oxidation,17 which may also provide an explanation in this case. We note however, that as for previous TFRS-2 and TFRS-52 dyes, the oxidative stability was much higher than we have observed for thiocyanate-containing dyes such as N3.17
To further clarify the influence of the dye structure on the solar cell performance (described in the next section), we carried out electrochemical studies of the dyes bound to mesoporous TiO2 films. Since the electrochemical window used lies entirely within the band gap of the TiO2, the TiO2 remains insulating and the redox occurs via a hole-diffusion process starting from the base of the film where dye is in contact with the FTO electrode. Firstly, we observe excellent reversibility of the redox process for all three isomers (Fig. S7–S9†) with little change between redox cycles 1 to 51 confirming these as stable sensitizers. This observation is different from that of TFRS-2 and 52, for which the asymmetric isomer b is found to be more stable versus the respective isomer a.17 Moreover, the quantity of dye uptake was observed to be TFRS-80a > c > b, (Table S4†) consistent with that observed during DSC fabrication (see below). Furthermore, following previously-described procedures and equations listed in ESI,† the maximum observed current and dye concentration were used to calculate a hole-diffusion coefficient for the case of each dye and these were observed to be in the order TFRS-80a > 80b > 80c (4.21, 1.28 and 0.203 × 10−10 cm2 s−1 respectively). These values are all 1–3 orders of magnitude lower than typical values reported for other Ru and organic dyes.23 It seems likely that this arises due to success of the design strategy whereby the 2,6-dihexyloxylphenyl on the tpiq ligand can suppress π–π interactions and avoid aggregation. In addition, the order of the values among the isomers further supports this conclusion, since TFRS-80a, 80b and 80c have respectively zero, one and two tpiq arms in the plane of the surface to minimize dye–dye electronic interactions.
Device characteristics
The photovoltaic properties of these sensitizers were examined, for which the details of cell fabrication and data measurements are depicted in the Experimental section. All cells were fabricated using 3.6 μm (20 nm) + 3.5 μm (400 nm) of mesoporous TiO2 thin film. The photocurrent–voltage characteristics were summarized in Tables 2 and 3. Three electrolyte solutions were employed for these studies; the first (i.e. electrolyte I–A) consisted of 0.6 M 1,2-dimethyl-3-propylimidazolium iodide (DMPII), 0.05 M I2, and 0.5 M t-butylpyridine (TBP) in acetonitrile, while the second (i.e. Co(phen)3]2+/3+ electrolyte Co-phen) and the third (i.e. electrolyte I–B) contained 0.45 M [Co(phen)3][TFSI]2, 0.15 M [Co(phen)3][TFSI]3, 0.15 M LiTFSI, 0.8 M TBP in acetonitrile, as well as 0.45 M DMPII, 0.05 M I2, 0.15 M LiI, 0.8 M TBP in acetonitrile, respectively. The electrolyte I–A possesses no Li+, while both Co-phen and I–B contain 0.15 M of Li+ cation in electrolyte, such that they can provide an intimate comparison between cell characteristics of the cobalt and iodine based redox couples. Comparative studies on cells with both I−/I3− and Co2+/3+ electrolytes are starting to gain momentum, inspired by the recent report that the Co2+/3+ cells have shown promising photostability under full sun solar illumination.24| Dye | J SC [mA cm−2] | V OC [mV] | FF | η [%] | Dye loadingb |
|---|---|---|---|---|---|
a All devices were fabricated using methods depicted in the Experimental section.
b Dye desorption experiment was performed using 1 M TBAOH in water–MeOH (v/v, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1). The dye loading is in unit of 10−7 mol cm−2. 1). The dye loading is in unit of 10−7 mol cm−2.
|
|||||
| TFRS-80a | 12.93 | 890 | 0.727 | 8.37 | 1.72 |
| TFRS-80b | 9.81 | 780 | 0.725 | 5.55 | 1.03 |
| TFRS-80c | 12.41 | 880 | 0.756 | 8.26 | 1.21 |
| TFRS-80ac | 13.12 | 870 | 0.731 | 8.34 | |
| Dye | Electrolyte | J SC [mA cm−2] | V OC [mV] | FF | η [%] |
|---|---|---|---|---|---|
| TFRS-80a | Co-phen | 13.44 | 840 | 0.757 | 8.55 |
| I–B | 14.49 | 780 | 0.668 | 7.55 | |
| TFRS-80b | Co-phen | 13.30 | 820 | 0.766 | 8.36 |
| I–B | 10.39 | 680 | 0.681 | 4.80 | |
| TFRS-80c | Co-phen | 14.32 | 840 | 0.754 | 9.06 |
| I–B | 14.84 | 730 | 0.651 | 7.06 |
For the DSCs using electrolyte I–A, TFRS-80a exhibited a JSC of 12.93 mA cm−2, a VOC of 890 mV, and a fill factor (FF) of 0.727, while TFRS-80c gave performance data of 12.41 mA cm−2, 880 mV and 0.756, respectively. Their overall conversion efficiencies (η) were calculated to be 8.37% and 8.26%, hence both are superior to that obtained for the asymmetric sensitizer TFRS-80b, with JSC = 9.81 mA cm−2, VOC of 780 mV, FF = 0.725 and η = 5.55%. Since the mixture of isomers 80a and 80c can be easily separated from 80b, but separation of 80a and 80c required repeated column chromatography, from the point view of practical application we therefore attempted DSC fabrication using the naturally occurring mixture of TFRS-80a and 80c, for which the recorded characteristics were JSC = 13.12 mA cm−2, VOC of 870 mV, FF = 0.731, and η = 8.34%, respectively. These data (cf.TFRS-80ac) showed no difference from cells fabricated using each of the pure samples, suggesting the retention of all device performances.
Fig. 3a exhibits the incident photon-to-current conversion efficiency (IPCE) action spectra recorded using I−/I3− electrolyte I–A. The onset of the IPCE spectra of TFRS-80a, 80c, and the mixture of TFRS-80a and 80c are all close to ∼780 nm, and with excellent IPCE performance in the range from 400 to 560 nm, among which the highest IPCE of 78% is recorded at around 530 nm, while TFRS-80b showed a notably lowered IPCE of only 51% at the same position. Apparently, the symmetrical TFRS-80a and 80c exhibit much better IPCE action spectra as well as superior J–V characteristics, versus those exhibited by the asymmetric stereoisomer 80b. It appears to us that the significantly reduced dye loading of 80b on the TiO2 surface, which not only reduced the absorptivity of 80b on TiO2 (Fig. 1) but also increased the charge recombination at the TiO2/electrolyte interface, is the major contributing factor for the poor overall conversion efficiency detected.
 | ||
| Fig. 3 IPCE action spectra for DSC cells fabricated using (a) electrolyte I–A and (b) electrolyte Co-phen and another I−/I3− electrolyte I–B under AM1.5 solar irradiation. | ||
Next, the photovoltaic performance of these sensitizers was evaluated by using Co(phen)3]2+/3+ based redox mediator in acetonitrile solution (i.e. Co-phen). A TiO2 blocking layer was pre-deposited on FTO glass using an aqueous TiCl4 solution. This measure is for retarding charge recombination between FTO and the Co2+/3+ mediator in electrolyte.25 Interestingly, the DSC device fabricated using TFRS-80c and [Co(phen)3]2+/3+ redox couple afforded the highest performance characteristics of JSC = 14.32 mA cm−2, VOC = 840 mV, and FF = 0.754, corresponding to an overall η = 9.06% under AM 1.5G one sun irradiation. We attribute this to the diaxial arrangement of the isoquinolinyl substituents on the Ru(II) metal complex, on which the bulky 2,6-dialkoxyphenyl group is expected to form a closely packed insulation layer, as they are now lying directly on top of the TiO2 electrode surface.26 This spatial arrangement is expected to be very effective in preventing the oxidized Co3+ species from approaching close to the TiO2 surface in comparison with the other isomers 80a and 80b, for which there is at least one 2,6-dialkoxyphenyl group per molecule orientated further away from the TiO2 surface, and cannot be effective in suppressing the charge recombination against the accumulated, oxidized Co3+ metal species.
For a further comparison, all sensitizers were subjected to DSC fabrication using the I−/I3− reference electrolyte (i.e. electrolyte I–B) under identical cell parameters. As can be seen, the overall efficiencies span the range 4.80–7.55%, among which the best one is that fabricated using TFRS-80a. In comparison across the different electrolytes, these data remain 1.0% lower than those documented for the cell fabricated using the corresponding sensitizer and the [Co(phen)3]2+/3+ electrolyte Co-phen. For TFRS-80c, the difference increases to approx. 2.0%, which is even greater. Moreover, the DSC device fabricated using TFRS-80b and I−/I3− electrolyte I–B showed the worst overall η of 4.80%. These data are much inferior to those observed for the Co2+/3+ based cell, with performance data of JSC = 13.30 mA cm−2, VOC = 820 mV, and FF = 0.766, corresponding to an overall η = 8.36%. The latter result is probably due to the effective insulating power of this sensitizer against the bulky Co3+ metal species, leading to the more effective suppression of charge recombination. In addition, the lowered efficiencies of all I−/I3− based DSCs can be traced to the inferior VOC, mainly caused by the greater loss-in-potential for the I−/I3− redox couple versus the Co2+/3+ electrolyte.
Moreover, the cell efficiency of TFRS-80b is ∼2.5% lower than those of the symmetric counterparts, i.e.TFRS-80a and TFRS-80c, upon using the I−/I3− based electrolytes I–A and I–B. On the other hand, the TFRS-80b sensitizer showed a much smaller difference of 0.7% in efficiency upon switching to Co2+/3+ electrolyte, compared with the best TFRS-80c. Since all of these sensitizers have essentially identical spectroscopic and electrochemical properties, this large variation can only be explained by the inferior dye-loading for TFRS-80b that generated a larger number of voids on the TiO2 surface. Accordingly, the I3− ion is much smaller and can penetrate much deeper into the dye layer versus that of Co2+/3+ electrolyte, giving much greater charge recombination and the greater difference in efficiencies.
Fig. 3b exhibits the incident photon-to-current conversion efficiency (IPCE) action spectra recorded using the electrolytes Co-phen and I–B. Integration of the IPCE spectra yields the calculated JSC data which are in good agreement with the experimental values. It is also notable that the Co2+/3+ electrolyte exhibited the higher photocurrent response from 370 to 440 nm versus that of the I−/I3− electrolyte, which is ascribed to the lower molar absorption coefficients of the Co2+/3+ complexes in the high energy region compared with the I−/I3− redox couple. Concurrently, similar to other reported Ru(II) sensitizers, we also observed a degradation of the photocurrent response in the lower energy region when switching to the Co2+/3+ electrolyte.27 This phenomenon is probably due to the poor dye regeneration efficiencies caused by the diminishing of the overpotential for dye regeneration.
For a closer comparison, the best recorded efficiency of TFRS-80 series obtained in this study (9.06%) is slightly higher than that of recently reported tris-heteroleptic Ru(II) sensitizers with 2′,6′-dimethoxy-2,3′-bipyridine cyclometalate (η = 8.6%).27a In turn, both data are superior to the thiocyanate-free Ru(II) sensitizer with ppy-(CF3)2 cyclometalate (η = 5.5%),11a and traditional thiocyanate-containing Ru(II) sensitizers, such as: N719 (η = 1.8%) and Z907 (η = 6.5%),27cZ907 with co-grafting phosphonic acid (η = 8.4%),27b and C101 (η = 3.6%) and TT-230 (η = 1.8%).28 It is notable that the TT-230 dye was even functionalized with the cyclopenta(2,1-b:3,4-b′)dithiophene moieties,29 which were widely used in organic push–pull dyes for extending the optical response, retarding charge recombination and suppressing dark current, but is still unable to boost its performances. On the other hand, DSCs with Co2+/3+ electrolytes are known to display higher solar cell efficiency, if the employed organic sensitizers were decorated with adequate bulky and electron donating appendages30 and with rigidified skeletal structure,31 to bring forth the anticipated enhancement in both JSC and VOC, by avoiding the aggregation and facilitating the photo-induced electron transfer process.
To gain further insight into the rates of interfacial recombination of electrons from the TiO2 conduction band to the redox mediators in the electrolyte, variation of the TiO2 conduction band potential was accessed by measuring the capacitance for three DSC devices at each VOC using the charge extraction (CE) method and intensity-modulated photovoltage spectroscopy (IMVS) measurement. Comparing that of TFRS-80a and 80c, a lower VOC for TFRS-80b is noticed (see Table 3). As shown in Fig. 4a, the CE results indicate that the TiO2 conduction band potential of the devices with the Co-phen electrolyte showed a systematic upward shift in the order TFRS-80b < 80c < 80a, consistent with the variation of their VOC.32Fig. 4b and c show plots of electron lifetime under five different light intensities. The results indicate a systematic trend with the electron lifetime showing an order of TFRS-80c > 80a > 80b for the Co-phen electrolyte and the order of TFRS-80a > 80c > 80b for the electrolyte I–B, respectively. These trends correspond to the degree of charge recombination, and are also consistent with the variation of VOC for these devices. Normally, the electron lifetime has the opposite trend versus charge recombination. Longer electron lifetime would correspond to smaller charge recombination loss and higher VOC in solar cells. Additionally, the electron lifetime of TFRS-80c is the highest for all the cells using Co2+/3+ based electrolyte. Thus, this proves the non-accumulation of Co3+ species in the proximity of TiO2 surface and the decrease in charge recombination due to the efficient blocking effect of TFRS sensitizers.
Conclusion
To sum up, we have designed and prepared a series of thiocyanate-free Ru(II) sensitizers, i.e.TFRS-80a, 80b and 80c, all with similar electrochemical properties and identical optical response under simulated one sun illumination, but showing varied steric impediment upon depositing on TiO2 surface according to the coordination orientation of the tpiq ancillaries. Among the photophysical and electrochemical parameters collected, the hole-diffusion coefficients are the most important, which follow the trend of TFRS-80a > 80b > 80c (4.21, 1.28 and 0.203 × 10−10 cm2 s−1). This trend supports the effective blockage of π–π interactions by 2,6-dihexyloxylphenyl substituents, since TFRS-80a, 80b and 80c have respectively zero, one and two tpiq arms in position to minimize dye–dye interactions on the TiO2 surface.DSCs with I−/I3− electrolyte were first fabricated, among which the TFRS-80a and TFRS-80b showed the highest and the lowest efficiencies of η = 8.37 and 5.55%, for which the large variation was mainly determined by the amount of dye uptake and hence, give decreased light harvesting capability and enhanced charge recombination across the TiO2-dye–electrolyte interface for the asymmetric TFRS-80b. In sharp contrast, DSCs with [Co(phen)3]2+/3+ electrolyte showed much superior efficiencies for all TFRS-80 sensitizers and, most importantly, the detected efficiency increased to η = 9.06% in the symmetrical TFRS-80a. Their advantages are apparently due to the combination of several factors, namely: (i) charge neutrality, (ii) absence of thiocyanate ligands, (iii) enhanced dye loading, and (iv) adequate spatial impediment upon depositing on TiO2 surface. All these contributing factors are essential for preventing the strong association to the Co2+/3+ mediator, which therefore reduces the charge recombination across the interface of TiO2 and electrolyte. The knowledge gained in this study should be of help to the future optimization of Ru(II) metal based sensitizers for DSC cells employing various Co2+/3+ based mediators.
Experimental section
General procedures
All reactions were performed under nitrogen. Solvents were distilled from appropriate drying agents prior to use. Commercially available reagents were used without further purification. All reactions were monitored by TLC with pre-coated silica gel plates (Merck, 0.20 mm with fluorescent indicator UV254). Compounds were visualized with UV irradiation at 254 or 365 nm. Flash column chromatography was carried out using silica gel obtained from Merck (230–400 mesh). Mass spectra were obtained on a JEOL SX-102A instrument operating in electron impact (EI) or fast atom bombardment (FAB) mode. 1H and 19F NMR spectra were recorded on a Bruker-400 instrument. Photophysical data were obtained using an Edinburgh Fluorescence spectrometer FLS928P. Details of the synthetic protocols for the tri-dentate ancillary chelates and the procedures for the DFT calculations are all given in the ESI.†Synthesis of TFRS-80a, 80b and 80c
A xylene solution of 6-(5-(2,6-bis(hexyloxy)phenyl)thiophen-2-yl)-1-(3-(trifluoromethyl)-1H-pyrazol-5-yl)isoquinoline (113 mg, 0.209 mmol), Ru(diethyl-2,2′-bipyridine-4,4′-dicarboxylate) (p-cymene)Cl (60 mg, 0.104 mmol), and KOAc (52 mg, 0.531 mmol) was heated at reflux under N2 for 6 h. After the removal of solvent under vacuum, the residue was dissolved in CH2Cl2 and washed with water (3 × 20 mL). Concentration of this CH2Cl2 solution gave a dark-brown oily solid. It was then purified by silica gel column chromatography eluting with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 mixture of ethyl acetate and hexane to afford a mixture of isomers a and c and analytically pure isomer b. Then, analytically pure a and c were separated by a second silica gel column chromatography using a 1
4 mixture of ethyl acetate and hexane to afford a mixture of isomers a and c and analytically pure isomer b. Then, analytically pure a and c were separated by a second silica gel column chromatography using a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 mixture of ethyl acetate and CH2Cl2.
20 mixture of ethyl acetate and CH2Cl2.
For hydrolysis, each of the samples was dissolved in a mixed acetone (20 mL) and 1 M NaOH solution (0.1 mL), and heated to reflux under N2 for 3 h. After this, the solution was diluted with water (10 mL) and, then, acidified with 2 M HCl to pH 3 to afford a brown precipitate. This was collected and washed with water, acetone, and diethylether in sequence, yield: 52 mg, 32% for TFRS-80a, 18 mg, 11% for TFRS-80b, and 25 mg, 15% for TFRS-80c.
Device fabrication
The cells consisted of a 3.6 μm transparent layer of 20 nm TiO2 nanoparticles, on which was superimposed a second layer, 3.5 μm of 400 nm TiO2 nanoparticles for enhancing light scattering. This double layer film was heated to 500 °C, sintered for 30 min, cooled to 80 °C, and then immersed into the dye solution (0.3 mM) containing 10 vol.% DMSO and 2 eq. of tetrabutylammonium deoxycholate [TBA][DOC] in anhydrous ethanol for 12 h. The iodine electrolyte I–A contains 0.6 M DMPII (1,2-dimethyl-3-propyl-imidazolium iodide), 0.05 M I2, 0.5 M TBP (4-tert-butylpyridine) in acetonitrile. The cobalt electrolyte Co-phen contains 0.45 M [Co(phen)3][TFSI]2, 0.15 M [Co(phen)3][TFSI]3, 0.15 M LiTFSI, and 0.8 M TBP in acetonitrile. The iodine electrolyte I–B contains 0.45 M DMPII, 0.05 M I2, 0.15 M LiI, and 0.8 M TBP in acetonitrile. The counter electrodes were coated with an ultra-thin layer of the PVP capped platinum nanoclusters (PVP–Pt) in aqueous solution via a so-called “two-step dip coating” process on FTO glass (7 Ω/TEC7, 2.2 mm thick, Pilkington), followed by a post heating at 325 °C for 10 min.33 The dye sensitized TiO2 electrodes were assembled with Pt counter electrodes by inserting a hot-melt Surlyn film (Meltonix 1170–25, 25 μm, Solaronix) as spacer, and then heated at 130 °C. The electrolyte was injected into the cell through a predrilled hole at the counter electrode. The hole was sealed with a Surlyn sheet and a thin glass to avoid leakage. All fabricated DSC cells consist of an active area of 5 × 5 mm2, and the performances were measured using a black metal mask with an aperture area of 4 × 4 mm2.Electrochemical characterization of dyes on TiO2
Conductive FTO glass slides were cleaned with soap, water, deionized water, acetone and ethanol, dried under hot air and TiO2 paste (Dyesol 18 NR-T) was deposited on the FTO glass via doctor blading. The films were sintered at 450 °C for 30 min; the resulting film thicknesses were ∼6 μm. After cooling, the films were left in the dye bath (0.3 mM in DMSO–EtOH (v/v, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9)) for 30 h, then rinsed in acetonitrile for 1 min. Cyclic voltammetry (CV) measurements were performed using a three-electrode cell with the FTO/dyed-TiO2 film as the working electrode, a Pt rod counter electrode and an Ag/AgCl reference electrode. The electrolyte composition was 0.1 M TBAPF6 dissolved in acetonitrile bubbled with nitrogen for 15 min before the experiment.
9)) for 30 h, then rinsed in acetonitrile for 1 min. Cyclic voltammetry (CV) measurements were performed using a three-electrode cell with the FTO/dyed-TiO2 film as the working electrode, a Pt rod counter electrode and an Ag/AgCl reference electrode. The electrolyte composition was 0.1 M TBAPF6 dissolved in acetonitrile bubbled with nitrogen for 15 min before the experiment.
Photovoltaic characterization
Photovoltaic measurements were carried out under a class-AAA solar simulator (Model 11016A, Sun 3000, ABET Technologies) equipped with a 550 W xenon light source and water-cooling stage (25 °C). The output power density was calibrated to be 100 mW cm−2 using a certificated KG-5 Si reference cell and with a circular aperture of 8 mm. The current–voltage characteristic of each cell was obtained with a 4-wire sense mode, delay time set as 100 ms and bias scan from short-circuit to open-circuit by using a Keithley digital source meter (Model 2400). The spectra of the incident photon-to-current conversion efficiency (IPCE) were calculated with the equation 1240 JSC(λ)/(λPin(λ)) where JSC is the short-circuit current density under each monochromatic illumination in units of A cm−2, λ is the wavelength of incident monochromatic light in units of nanometers, and Pin is the monochromatic light intensity in units of W cm−2, plotted as a function of incident wavelength with an increment of 10 nm. The current was pre-amplified by a current amplifier (SR570) and measured by Keithley 2400 source meter. It should be noted that 10 sets of JSC (interval 50 ms) were collected sequentially after illuminating the device for 3 seconds and then averaged for calculation of IPCE. A 300 W Xe lamp (Model 6258, Newport Oriel) combined with an Oriel cornerstone 260 1/4 m monochromator (Model 74100) provided a monochromatic beam (dc mode) for the device under test conditions. The beam power intensity was calibrated with a power meter (Model 1936-C, Newport) equipped with a Newport 818-UV photodetector.Charge extraction and intensity-modulated photovoltage spectroscopy
Charge extraction was measured with the PGSTAT302N electrochemical workstation (Autolab) at open-circuit condition for the photovoltage of the device to attain a steady state. A red light-emitting diode (LED, 627 nm) was attenuated while the device simultaneously switched to a short-circuit condition to measure the excess charges generated in the film. Intensity-modulated photovoltage spectroscopy (IMVS) measurement was conducted using the same electrochemical workstation equipped with a frequency response analyzer (FRA) to drive the red LED. Photovoltage response of the cells was analyzed in the frequency range of 1–104 Hz and LED supplied the AC (modulation depth 10%) perturbation current superimposed on the DC current.Acknowledgements
This research was supported by Ministry of Science and technology of Taiwan, MOST 100-2119-M-002-008, and the UK EPSRC Global Grant EP/K004468. YH thanks the China Scholarship Council and the University of Edinburgh for a studentship.References
- (a) L. M. Goncalves, V. de Zea Bermudez, H. A. Ribeiro and A. M. Mendes, Energy Environ. Sci., 2008, 1, 655 RSC; (b) A. Hagfeldt, G. Boschloo, L. Sun, L. Kloo and H. Pettersson, Chem. Rev., 2010, 110, 6595 CrossRef CAS PubMed; (c) J.-H. Yum, E. Baranoff, S. Wenger, M. K. Nazeeruddin and M. Grätzel, Energy Environ. Sci., 2011, 4, 842 RSC; (d) Y. Bai, I. Mora-Seró, F. De Angelis, J. Bisquert and P. Wang, Chem. Rev., 2014, 114, 10095 CrossRef CAS PubMed.
- (a) S. Mathew, A. Yella, P. Gao, R. Humphry-Baker, F. E. CurchodBasile, N. Ashari-Astani, I. Tavernelli, U. Rothlisberger, M. K. Nazeeruddin and M. Grätzel, Nat. Chem., 2014, 6, 242 CrossRef CAS PubMed; (b) A. Yella, C.-L. Mai, S. M. Zakeeruddin, S.-N. Chang, C.-H. Hsieh, C.-Y. Yeh and M. Grätzel, Angew. Chem., Int. Ed., 2014, 53, 2973 CrossRef CAS PubMed.
- (a) A. Mishra, M. K. R. Fischer and P. Bäuerle, Angew. Chem., Int. Ed., 2009, 48, 2474 CrossRef CAS PubMed; (b) Z. Ning, Y. Fu and H. Tian, Energy Environ. Sci., 2010, 3, 1170 RSC; (c) Y. Wu and W. Zhu, Chem. Soc. Rev., 2013, 42, 2039 RSC; (d) Y.-S. Yen, H.-H. Chou, Y.-C. Chen, C.-Y. Hsu and J. T. Lin, J. Mater. Chem., 2012, 22, 8734 RSC; (e) Y. Ooyama and Y. Harima, ChemPhysChem, 2012, 13, 4032 CrossRef CAS PubMed; (f) S. Ito, H. Miura, S. Uchida, M. Takata, K. Sumioka, P. Liska, P. Comte, P. Pechy and M. Grätzel, Chem. Commun., 2008, 5194 RSC; (g) G. Zhang, H. Bala, Y. Cheng, D. Shi, X. Lv, Q. Yu and P. Wang, Chem. Commun., 2009, 2198 RSC; (h) Q. Yu, D. Zhou, Y. Shi, X. Si, Y. Wang and P. Wang, Energy Environ. Sci., 2010, 3, 1722 RSC; (i) J.-H. Yum, E. Baranoff, F. Kessler, T. Moehl, S. Ahmad, T. Bessho, A. Marchioro, E. Ghadiri, J.-E. Moser, C. Yi, M. K. Nazeeruddin and M. Grätzel, Nat. Commun., 2012, 3, 631 CrossRef PubMed.
- C. Chen, X. Yang, M. Cheng, F. Zhang and L. Sun, ChemSusChem, 2013, 6, 1270 CrossRef CAS PubMed.
- B. Zietz, E. Gabrielsson, V. Johansson, A. M. El-Zohry, L. Sun and L. Kloo, Phys. Chem. Chem. Phys., 2014, 16, 2251 RSC.
- (a) M. Zhang, J. Zhang, Y. Fan, L. Yang, Y. Wang, R. Li and P. Wang, Energy Environ. Sci, 2013, 6, 2939 RSC; (b) M. Zhang, Y. Wang, M. Xu, W. Ma, R. Li and P. Wang, Energy Environ. Sci., 2013, 6, 2944 RSC.
- (a) M. K. Nazeeruddin, P. Péchy, T. Renouard, S. M. Zakeeruddin, R. Humphry-Baker, P. Comte, P. Liska, L. Cevey, E. Costa, V. Shklover, L. Spiccia, G. B. Deacon, C. A. Bignozzi and M. Grätzel, J. Am. Chem. Soc., 2001, 123, 1613 CrossRef CAS PubMed; (b) J.-H. Yum, I. Jung, C. Baik, J. Ko, M. K. Nazeeruddin and M. Grätzel, Energy Environ. Sci., 2009, 2, 100 RSC; (c) Y. Cao, Y. Bai, Q. Yu, Y. Cheng, S. Liu, D. Shi, F. Gao and P. Wang, J. Phys. Chem. C, 2009, 113, 6290 CrossRef CAS; (d) L. H. Nguyen, H. K. Mulmudi, D. Sabba, S. A. Kulkarni, S. K. Batabyal, K. Nonomura, M. Grätzel and S. G. Mhaisalkar, Phys. Chem. Chem. Phys., 2012, 14, 16182 RSC; (e) S.-W. Wang, C.-C. Chou, F.-C. Hu, K.-L. Wu, Y. Chi, J. N. Clifford, E. J. Palomares, S.-H. Liun, P.-T. Chou, T. C. Wei and T. Y. Hsiao, J. Mater. Chem. A, 2014, 2, 17618 RSC.
- A. Hinsch, W. Veurman, H. Brandt, K. Flarup Jensen and S. Mastroianni
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) , ChemPhysChem, 2014, 15, 1076 CrossRef CAS PubMed.
, ChemPhysChem, 2014, 15, 1076 CrossRef CAS PubMed. - (a) S. H. Wadman, J. M. Kroon, K. Bakker, M. Lutz, A. L. Spek, G. P. M. van Klink and G. van Koten, Chem. Commun., 2007, 1907 RSC; (b) S. H. Wadman, J. M. Kroon, K. Bakker, R. W. A. Havenith, G. P. M. van Klink and G. van Koten, Organometallics, 2010, 29, 1569 CrossRef CAS.
- (a) T. Bessho, E. Yoneda, J.-H. Yum, M. Guglielmi, I. Tavernelli, H. Imai, U. Rothlisberger, M. K. Nazeeruddin and M. Grätzel, J. Am. Chem. Soc., 2009, 131, 5930 CrossRef CAS PubMed; (b) F. De Angelis, S. Fantacci, A. Selloni, M. K. Nazeeruddin and M. Grätzel, J. Phys. Chem. C, 2010, 114, 6054 CrossRef CAS.
- (a) P. G. Bomben, T. J. Gordon, E. Schott and C. P. Berlinguette, Angew. Chem., Int. Ed., 2011, 50, 10682 CrossRef CAS PubMed; (b) P. G. Bomben, K. C. D. Robson, B. D. Koivisto and C. P. Berlinguette, Coord. Chem. Rev., 2012, 256, 1438 CrossRef CAS PubMed; (c) K. C. D. Robson, P. G. Bomben and C. P. Berlinguette, Dalton Trans., 2012, 41, 7814 RSC; (d) S. Sinn, B. Schulze, C. Friebe, D. G. Brown, M. Jäger, E. Altuntaş, J. Kübel, O. Guntner, C. P. Berlinguette, B. Dietzek and U. S. Schubert, Inorg. Chem., 2014, 53, 2083 CrossRef CAS PubMed.
- (a) B.-S. Chen, K. Chen, Y.-H. Hong, W.-H. Liu, T.-H. Li, C.-H. Lai, P.-T. Chou, Y. Chi and G.-H. Lee, Chem. Commun., 2009, 5844 RSC; (b) K.-L. Wu, H.-C. Hsu, K. Chen, Y. Chi, M.-W. Chung, W.-H. Liu and P.-T. Chou, Chem. Commun., 2010, 46, 5124 RSC; (c) Y. Chi, B. Tong and P.-T. Chou, Coord. Chem. Rev., 2014, 281, 1 CrossRef CAS PubMed.
- (a) C.-C. Chou, K.-L. Wu, Y. Chi, W.-P. Hu, S. J. Yu, G.-H. Lee, C.-L. Lin and P.-T. Chou, Angew. Chem., Int. Ed., 2011, 50, 2054 CrossRef CAS PubMed; (b) K.-L. Wu, S.-T. Ho, C.-C. Chou, Y.-C. Chang, H.-A. Pan, Y. Chi and P.-T. Chou, Angew. Chem., Int. Ed., 2012, 51, 5642 CrossRef CAS PubMed; (c) C.-C. Chou, F.-C. Hu, H.-H. Yeh, H.-P. Wu, Y. Chi, J. N. Clifford, E. Palomares, S.-H. Liu, P.-T. Chou and G.-H. Lee, Angew. Chem., Int. Ed., 2014, 53, 178 CrossRef CAS PubMed.
- (a) C.-W. Hsu, S.-T. Ho, K.-L. Wu, Y. Chi, S.-H. Liu and P.-T. Chou, Energy Environ. Sci., 2012, 5, 7549 RSC; (b) C.-C. Chou, P.-H. Chen, F.-C. Hu, Y. Chi, S.-T. Ho, J.-J. Kai, S.-H. Liu and P.-T. Chou, J. Mater. Chem. A, 2014, 2, 5418 RSC.
- (a) K.-L. Wu, W.-P. Ku, J. N. Clifford, E. Palomares, S.-T. Ho, Y. Chi, S.-H. Liu, P.-T. Chou, M. K. Nazeeruddin and M. Grätzel, Energy Environ. Sci., 2013, 6, 859 RSC; (b) S.-W. Wang, K.-L. Wu, E. Ghadiri, M. G. Lobello, S.-T. Ho, Y. Chi, J.-E. Moser, F. De Angelis, M. Grätzel and M. K. Nazeeruddin, Chem. Sci., 2013, 4, 2423 RSC.
- K.-L. Wu, J. N. Clifford, S.-W. Wang, Y. Aswani, E. Palomares, M. G. Lobello, E. Mosconi, F. De Angelis, W.-P. Ku, Y. Chi, M. K. Nazeeruddin and M. Grätzel, ChemSusChem, 2014, 7, 2930 CrossRef CAS PubMed.
- F.-C. Hu, S.-W. Wang, Y. Chi, N. Robertson, T. Hewat, Y. Hu, S.-H. Liu, P.-T. Chou, P.-F. Yang and H.-W. Lin, ChemPhysChem, 2014, 15, 1207 CrossRef CAS PubMed.
- (a) C.-L. Wang, J.-Y. Hu, C.-H. Wu, H.-H. Kuo, Y.-C. Chang, Z.-J. Lan, H.-P. Wu, E. Wei-Guang Diau and C.-Y. Lin, Energy Environ. Sci., 2014, 7, 1392 RSC; (b) Z. Wang, H. Wang, M. Liang, Y. Tan, F. Cheng, Z. Sun and S. Xue, ACS Appl. Mater. Interfaces, 2014, 6, 5768 CrossRef CAS PubMed; (c) J. Yang, P. Ganesan, J. Teuscher, T. Moehl, Y. J. Kim, C. Yi, P. Comte, K. Pei, T. W. Holcombe, M. K. Nazeeruddin, J. Hua, S. M. Zakeeruddin, H. Tian and M. Grätzel, J. Am. Chem. Soc., 2014, 136, 5722 CrossRef CAS PubMed.
- F. Gao, Y. Wang, D. Shi, J. Zhang, M. Wang, X. Jing, R. Humphry-Baker, P. Wang, S. M. Zakeeruddin and M. Grätzel, J. Am. Chem. Soc., 2008, 130, 10720 CrossRef CAS PubMed.
- F. Matar, T. H. Ghaddar, K. Walley, T. DosSantos, J. R. Durrant and B. O'Regan, J. Mater. Chem., 2008, 18, 4246 RSC.
- (a) S.-Q. Fan, C. Kim, B. Fang, K.-X. Liao, G.-J. Yang, C.-J. Li, J.-J. Kim and J. Ko, J. Phys. Chem. C, 2011, 115, 7747 CrossRef CAS; (b) H. Ozawa, R. Shimizu and H. Arakawa, RSC Adv., 2012, 2, 3198 RSC; (c) L. Han, A. Islam, H. Chen, C. Malapaka, B. Chiranjeevi, S. Zhang, X. Yang and M. Yanagida, Energy Environ. Sci., 2012, 5, 6057 RSC.
- (a) M. Liang and J. Chen, Chem. Soc. Rev., 2013, 42, 3453 RSC; (b) R. Stalder, D. Xie, A. Islam, L. Han, J. R. Reynolds and K. S. Schanze, ACS Appl. Mater. Interfaces, 2014, 6, 8715 CrossRef CAS PubMed.
- (a) X. Li, M. K. Nazeeruddin, M. Thelakkat, P. R. F. Barnes, R. Vilar and J. R. Durrant, Phys. Chem. Chem. Phys., 2011, 13, 1575 RSC; (b) D. Moia, V. Vaissier, I. Lopez-Duarte, T. Torres, M. K. Nazeeruddin, B. C. O'Regan, J. Nelson and P. R. F. Barnes, Chem. Sci., 2014, 5, 281 RSC.
- R. Jiang, A. Anderson, P. R. F. Barnes, L. Xiaoe, C. Law and B. C. O'Regan, J. Mater. Chem. A, 2014, 2, 4751 CAS.
- J.-H. Yum, T. Moehl, J. Yoon, A. K. Chandiran, F. Kessler, P. Gratia and M. Grätzel, J. Phys. Chem. C, 2014, 118, 16799 CAS.
- (a) S. Archer and J. A. Weinstein, Coord. Chem. Rev., 2012, 256, 2530 CrossRef CAS PubMed; (b) L.-L. Li and E. W.-G. Diau, Chem. Soc. Rev., 2013, 42, 291 RSC.
- (a) L. E. Polander, A. Yella, B. F. E. Curchod, A. N. Ashari, J. Teuscher, R. Scopelliti, P. Gao, S. Mathew, J.-E. Moser, I. Tavernelli, U. Rothlisberger, M. Gratzel, M. K. Nazeeruddin and J. Frey, Angew. Chem., Int. Ed., 2013, 52, 8731 CrossRef CAS PubMed; (b) Y. Liu, J. R. Jennings, X. Wang and Q. Wang, Phys. Chem. Chem. Phys., 2013, 15, 6170 RSC; (c) Y. Liu, J. R. Jennings, Y. Huang, Q. Wang, S. M. Zakeeruddin and M. Grätzel, J. Phys. Chem. C, 2011, 115, 18847 CrossRef CAS.
- S. A. Kumar, M. Urbani, M. Medel, M. Ince, D. González-Rodríguez, A. K. Chandiran, A. N. Bhaskarwar, T. Torres, M. K. Nazeeruddin and M. Grätzel, J. Phys. Chem. Lett., 2014, 5, 501 CrossRef.
- (a) Q. Feng, X. Jia, G. Zhou and Z.-S. Wang, Chem. Commun., 2013, 49, 7445 RSC; (b) H. Ellis, S. K. Eriksson, S. M. Feldt, E. Gabrielsson, P. W. Lohse, R. Lindblad, L. Sun, H. Rensmo, G. Boschloo and A. Hagfeldt, J. Phys. Chem. C, 2013, 117, 21029 CrossRef CAS; (c) L. Cabau, L. Pelleja, J. N. Clifford, C. V. Kumar and E. Palomares, J. Mater. Chem. A, 2013, 1, 8994 RSC.
- M. Xu, M. Zhang, M. Pastore, R. Li, F. De Angelis and P. Wang, Chem. Sci., 2012, 3, 976 RSC.
- N. Cai, J. Zhang, M. Xu, M. Zhang and P. Wang, Adv. Funct. Mater., 2013, 23, 3539 CrossRef CAS.
- T. Moehl, H. N. Tsao, K.-L. Wu, H.-C. Hsu, Y. Chi, E. Ronca, F. De Angelis, M. K. Nazeeruddin and M. Grätzel, Chem. Mater., 2013, 25, 4497 CrossRef CAS.
- T. C. Wei, C. C. Wan and Y. Y. Wang, Appl. Phys. Lett., 2006, 88, 103122 CrossRef PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: 10.1039/c4ta04208e |
| ‡ K.-L. W. and Y. H. contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2014 |