Cocrystal explosive hydrate of a powerful explosive, HNIW, with enhanced safety†
Abstract
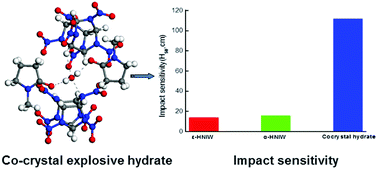
A novel cocrystal explosive hydrate containing HNIW (hexanitrohexaazaisowurtzitane), NMP (N-methyl-2-pyrrolidone) and water was synthesized through cocrystallization. The crystal structure was characterized by power X-ray diffraction (PXRD) and single crystal X-ray diffraction (SXRD). This crystalline structure belongs to the monoclinic crystal system with space group P2(1)/c. The properties of the cocrystal hydrate including thermal behavior, impact sensitivity and detonation performance were also evaluated. The cocrystal hydrate shows a unique thermal behavior with an endothermic peak and a decomposition peak at 91 and 252 °C, respectively. Besides, the cocrystal hydrate displays an impact height with a 50% ignition probability of 112 cm, indicating a substantial reduction in impact sensitivity compared to pure α-HNIW and ε-HNIW. Furthermore, although the power is diluted, the cocrystal hydrate is predicted to have better detonation performance than pure DNAN (2,4-dinitroanisole) and TNT (trinitrotoluene). Therefore, the cocrystal hydrate may be a promising low sensitivity explosive.

 Please wait while we load your content...
Please wait while we load your content...