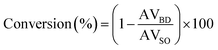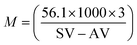Efficient synthesis of biodiesel over a recyclable catalyst comprising a monolacunary silicotungstate and zeolite Hβ
Nilesh Narkhede and
Anjali Patel*
Polyoxometalate and Catalysis Laboratory, Department of Chemistry, Faculty of Science, The M. S. University of Baroda, Vadodara, 390002, India. E-mail: aupatel_chem@yahoo.com; Tel: +91 265 2795552
First published on 19th November 2014
Abstract
This paper presents a study of esterification of oleic acid as well as transesterification of soybean oil with methanol over a solid acid catalyst comprising a monolacunary silicotungstate anchored to zeolite Hβ. The optimization of the reaction parameters were carried out to get the maximum conversion. The 3.5 wt% catalyst charge at 60 °C showed 82% conversion of oleic acid with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 mole ratio of acid to alcohol in 10 h whereas the 4 wt% catalyst charge at 65 °C showed 96% conversion of soybean oil with a ratio of oil to alcohol of 1
20 mole ratio of acid to alcohol in 10 h whereas the 4 wt% catalyst charge at 65 °C showed 96% conversion of soybean oil with a ratio of oil to alcohol of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 (w/w) in 8 h. The catalyst was found to be readily reusable and exhibited consistency in activity upon reuse four times without appreciable loss in the conversion.
4 (w/w) in 8 h. The catalyst was found to be readily reusable and exhibited consistency in activity upon reuse four times without appreciable loss in the conversion.
1. Introduction
Polyoxometalates (POMS) are polymeric oxoanions formed by condensation of metal oxide anions of early transition metals (W6+, V5+, Mo6+) under acidic conditions via dehydration of water. Recently, POM based catalysts have played an important role in the field of acid as well as oxidation catalysis due to their high Bronsted acidity as well as their tendency to exhibit fast reversible multi-electron redox transformations.1,2The modification of parent POMs can lead to the development of new generation catalysts with enhanced properties of acidity, redox potential and stability. The modification of properties can be basically done by tuning the structural properties at the atomic or molecular level. One of the ways to modify the structural properties is by creating defect in the parent POMs leading to the formation of lacunary polyoxometalates (LPOMs). Due to the structural diversity as well as the unique electronic properties,3,4 LPOMs are of potential importance in catalysis.
Among LPOMs, many reports on synthesis and characterization of monolacunary silicotungstate are available in literature.5–8 It is well known that silicotungstates are environmentally benign as they lack side reactions unlike in the case of mineral acids and are easy to handle.9 As they are soluble in polar media, in the present work SiW11 is made heterogeneous by heterogenizing on to Hβ in order to make it reusable environmentally benign catalyst. Reports on use of lacunary silicotungstate as homogeneous catalysts are also available.10–13 However, reports on supported lacunary silicotungstate as heterogeneous catalyst are very scanty.14–17 We have reported synthesis, characterization and catalytic activity of SiW11 supported on to MCM-41 for esterification and transesterification reaction.18 The obtained results encouraged us to extend our work to zeolite support. In this work, for the first time, we are reporting the synthesis, characterization and catalytic application of SiW11 anchored to zeolite Hβ.
Zeolite beta is a high silica zeolite with large pores and is a good candidate for catalysis as it possesses high thermal and chemical stability, surface silanol groups and hydrophobic character which are the typical characteristics of ideal support.19
Soybean oil is a vegetable oil extracted from the seeds of the soybean (Glycine max). It is one of the most widely consumed cooking oils. As a drying oil, processed soybean oil is also used as a base for printing inks (soy ink) and oil paints. The main FFA constituents of soybean oil are palmitic acid (11%), stearic acid (4%), oleic acid (23%), linoleic acid (54%) and linolenic acid (8%).20 This suggests that soybean oil possesses triglyceride esters of both saturated and unsaturated FFA and is perfect feedstock to study the catalytic behaviour.
Considering all these aspects, in the present work, we report the synthesis and characterization of a catalyst comprising zeolite beta (Hβ) and SiW11 by different physicochemical techniques and its application for esterification of free fatty acid, oleic acid and transesterification of soybean oil with methanol. Kinetic study was also carried out for esterification of oleic acid to determine activation energy (Ea) and Arrhenius constant (A). Catalyst was recycled up to four cycles and reused catalyst was characterized by surface area measurement (BET method), Fourier transform infrared spectroscopy (FT-IR), and Powder X-ray diffraction (XRD) in order to check sustainability of reused catalyst.
2. Experimental
2.1. Materials
The chemicals used were of A.R. grade. Oleic acid (A.R.), methanol, sodium tungstate, sodium silicate, toluene, trichloroacetic acid, n-butylamine and acetone were purchased from Merck. Soybean oil was purchased from local market. Sodium form of zeolite beta (β) with Si/Al ratio 10 was procured from Zeolites and Allied Products, Bombay, India, and used as received.2.2. Treatment of the support (Hβ)
The zeolite support (Naβ) was converted in to NH4+ form by conventional ion exchange method using a 10 wt%, 1 M NH4Cl aqueous solution.21 The resulted NH4+ type zeolite was further converted to H+ type by calcination in air at 550 °C for 6 h.2.3. Synthesis of monolacunary silicotungstate (SiW11)
Sodium tungstate (0.22 mol, 7.2 g) and sodium silicate (0.02 mol, 0.56 g) were dissolved in 150–200 mL of distilled water and heated to 80 °C followed by the addition of dilute nitric acid to adjust the pH to 4.8. The volume was then reduced to half by evaporation and the heteropoly anion was separated by liquid–liquid extraction with 80–100 mL of acetone. The extraction was repeated until the acetone extract showed the absence of NO3− (test performed by adding iron(II) sulfate to a solution of a nitrate, then slowly adding concentrated sulphuric acid such that the sulphuric acid forms a layer below the aqueous solution. An absence of brown ring at the junction of the two layers indicating absence of the nitrate ions). The extracted sodium salt was dried in air at room temperature. The resulting material was designated as SiW11.182.4. Synthesis of the catalysts (SiW11 anchored to Hβ)
A series of catalysts containing 10–40% of SiW11 anchored to Hβ were synthesized by impregnation method. Hβ (1 g) was impregnated with an aqueous solution of SiW11 (0.1/10–0.4/40 g mL−1 of double distilled water) and dried at 100 °C for 10 h. The obtained materials were treated with 10%, 0.1 N HCl, filtered, washed with double distilled water and dried at 100 °C. The resulting materials were designated as 10% SiW11/Hβ, 20% SiW11/Hβ, 30% SiW11/Hβ and 40% SiW11/Hβ, respectively.2.5. Catalyst characterization
EDX analysis was carried out using JSM 5910 LV joint with an INCA instrument for EDX-SEM. TGA analysis of the samples was carried out using a Mettler Toledo Star SW 7.01 instrument under nitrogen atmosphere. The BET surface area measurements were performed in a Micromeritics ASAP 2010 volumetric static adsorption instrument with N2 adsorption at 77 K. For FT-IR spectra, samples pressed with dried KBr into discs were recorded in the range of wave numbers 4000–400 cm−1 by using a Perkin-Elmer spectrometer. The X-ray powder diffraction (XRD) patterns of the support and the catalyst were measured using a Philips X'pert MPD system in the 2θ range of 1–60° using CuKα radiation (λ = 1.54056 Å). Scanning electron microscope depictions were recorded on a JEOL-SEM instrument (model-JSM-5610LV).2.6. n-Butylamine acidity determination
The total surface acidity was determined by n-butylamine titration. A 0.025 M solution of n-butylamine in toluene was used for estimation. The catalyst weighing 0.5 g was suspended in this solution for 24 h and excess base was titrated against trichloroacetic acid in toluene using neutral red as an indicator. This gives total surface acidity of the material.2.7. Determination of types of acidic sites (potentiometric titration method)
A small quantity (0.1 mL) of 0.05 N, n-butylamine in acetonitrile was added to a suspension of 0.5 g of the catalyst in 50 mL of acetonitrile and the system was stirred at 25 °C. Then, the suspension was potentiometrically titrated against 0.05 N, n-butylamine in acetonitrile. The electrode potential variation was measured with a digital pH meter.The acidity of the catalyst measured by this technique allows us to evaluate the total number of acid sites as well as their acidic strength. In order to interpret the results, it is suggested that the initial electrode potential (Ei) indicates the maximum acid strength of the surface sites and the range where the plateau is reached (meq. per g solid) indicates the total number of acid sites.21 The acidic strength of surface sites can be assigned according to the following ranges: very strong site, Ei > 100 mV; strong site, 0 < Ei < 100 mV; weak site, −100 < Ei < 0 mV and very weak site, Ei < −100 mV.
2.8. Reaction procedure
where, AVBD is acid value of biodiesel (oil layer) and AVSO refers to acid value of soybean oil.22–25
3. Results and discussion
3.1. Catalyst characterization
The n-butylamine acidity values for all the catalysts are presented in Table 1. The results indicate that Hβ is fairly acidic. As the % SiW11 content increases, acidity values also increases. The variation in the number of acidic sites is dependent on the amount of the SiW11 on the support. The catalyst, 30% SiW11/Hβ showed the highest amount of total acidity with more strong acidic sites than other catalysts. The observed decrease in the acidity of the 40% SiW11/Hβ was due to the blocking of acidic sites at higher loadings due to multilayer formation. Considering this aspect we have selected 30% SiW11/Hβ for further detailed characterization by EDS, TGA, BET surface area, FT-IR, SEM and XRD.The plots of the electrode potential as a function of meq. amine per g of the catalyst is shown in Fig. 1. It is observed that, both the support and catalyst contain strong acid sites. The distribution of types as well as strength of acidic sites in terms of initial electrode potential is shown in Table 2. It is clear that on inclusion of SiW11 on to the support, there is a drastic increase in the strength of acidic sites as well as amount of strong acidic sites.
Elemental analysis performed on 30% SiW11/Hβ was consistent with theoretical expected values (theoretical: W = 16.7%; Si = 26.9%, Al = 2.9; observed: W = 16.5%; Si = 27.6%, Al = 2.2). This confirms that synthesized catalyst contains 30% SiW11 i.e. 0.3 g of SiW11/1 g of Hβ.
The TGA of Hβ (Fig. 2a) shows a unique weight loss of 13–15% up to 250 °C for, which is attributed to desorption of physically adsorbed water. No further weight loss was observed beyond 250 °C which indicates zeolite Hβ retains its framework structure up to 500 °C. The TGA of SiW11 (Fig. 2b) shows the preliminary weight loss of 7% from 30–130 °C. This is due to the removal of adsorbed water molecules. Second weight loss of 2.5% at 230 °C may be due to loss of water of crystallization. The steady weight loss after 330 °C indicates the decomposition of SiW11 species. The TGA of 30% SiW11/Hβ (Fig. 2c) shows initial weight of 6% up to 170 °C may be due to the removal of adsorbed water molecules. Second weight loss of 2% has been observed up to 240 °C which is due to loss of water of crystallization. No notable loss up to 410 °C indicates the stability of the catalyst up to 410 °C. An increase in thermal stability indicates the presence of chemical interaction between SiW11 and the support.18
The TGA data shows that anchoring of SiW11 to Hβ increases the thermal stability of SiW11. An increase in thermal stability indicates the presence strong interaction between SiW11 and Hβ.
The specific surface area and pore diameter of Hβ was found to be 587.2 m2 g−1 and 2.48 nm and that for 30% SiW11/Hβ was found to be 439.8 m2 g−1 and 1.71 nm, respectively. The incorporation of SiW11 into the framework of the support is apparent here, as it leads to 25% reduction in surface area as well as 31% reduction in pore width. This gives first indication of chemical interaction between SiW11 and support.
The N2 adsorption isotherms of support and the catalyst are displayed in Fig. 3a and b, respectively. Both isotherm showed type I pattern with three stages: monolayer adsorption of nitrogen on the walls of pores at P/Po < 0.4, the part characterized by a sharp increase in adsorption due to capillary condensation in mesopores with hysteresis at P/Po = 0.4–0.8, and multilayer adsorption on the outer surface of the particles. The decrease in pore diameter of the catalyst (Fig. 3b) is due to the presence of SiW11 inside the zeolite framework, as anticipated.
The FT-IR spectra of SiW11 (Fig. 4) shows bands at 987 cm−1 (W![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) Od), 948 cm−1 (Si–Oa), 886 and 795 cm−1 (W–Ob–W) and 727 cm−1 (W–Oc–W).26 Where Oa, Ob, Oc, and Od attributed to the oxygen atoms connected to silicon, to oxygen atoms bridging to two tungsten (from two different triads for Ob and from the same triad for Oc), and to the terminal oxygen W
Od), 948 cm−1 (Si–Oa), 886 and 795 cm−1 (W–Ob–W) and 727 cm−1 (W–Oc–W).26 Where Oa, Ob, Oc, and Od attributed to the oxygen atoms connected to silicon, to oxygen atoms bridging to two tungsten (from two different triads for Ob and from the same triad for Oc), and to the terminal oxygen W![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, respectively. FT-IR spectra for Hβ and 30% SiW11/Hβ (Fig. 4) shows large and broad peak appearing in range of 1000–1300 cm−1 is due to asymmetric stretching vibration O–T–O (νasym), which is sensitive to the silicon and aluminium contents in the zeolite Hβ framework. A broad band between 3700 and 3200 cm−1 is assigned as hydrogen bonds of silanol groups. The typical band for SiW11, at 942 cm−1 (Si–Oa), 860 cm−1 (W–Ob–W) and 715 cm−1 (W–Oc–W) are clearly observed for 30% SiW11/Hβ (Fig. 4b).
O, respectively. FT-IR spectra for Hβ and 30% SiW11/Hβ (Fig. 4) shows large and broad peak appearing in range of 1000–1300 cm−1 is due to asymmetric stretching vibration O–T–O (νasym), which is sensitive to the silicon and aluminium contents in the zeolite Hβ framework. A broad band between 3700 and 3200 cm−1 is assigned as hydrogen bonds of silanol groups. The typical band for SiW11, at 942 cm−1 (Si–Oa), 860 cm−1 (W–Ob–W) and 715 cm−1 (W–Oc–W) are clearly observed for 30% SiW11/Hβ (Fig. 4b).
FT-IR spectra indicate that the SiW11 preserves its Keggin ion unit even after anchoring to the support. The significant shift in the bands indicates the strong chemical interaction between SiW11 and Hβ.
BET and FT-IR analyses confirm the retention of SiW11 species inside the channels of the support as well as strong hydrogen bonding between SiW11 and H of the Si–OH group. To study the dispersion of SiW11 species inside the zeolite framework, the catalyst was further characterized by XRD and SEM.
Crystalline peaks for 30% SiW11/Hβ (Fig. 5) suggests that zeolite framework has been retained after incorporation of SiW11. The reflection at 2θ = 29° indicate the presence of the crystalline phase of SiW11 on to the support.27 The catalyst shows reflections of SiW11 with very low intensity indicating a well dispersion of SiW11 inside the channels of the support.28
The SEM images show that the surface morphology of 30% SiW11/Hβ (Fig. 6b) is identical to that of Hβ (Fig. 6a). The absence of aggregates of SiW11 species in the catalyst suggests that SiW11 species are well dispersed inside the zeolite channels.28 The results are in good agreement with the XRD data.
Thus, XRD and SEM confirm the uniform dispersion of SiW11 inside the channels of the support.
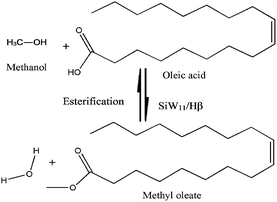
3.2. Esterification of oleic acid with methanol
The esterification of oleic acid with methanol over the present catalyst is shown in Scheme 1. In order to overcome the equilibrium limitation, the reaction was carried out by taking methanol in excess.The effect of different reaction variables such as % SiW11 content, acid/alcohol molar ratio, % catalyst charge, reaction time and temperature were studied to optimize the conditions for maximum conversion of oleic acid and the data are presented in the Fig. 7.
 | ||
| Fig. 7 Effect of % SiW11 content, oleic acid/methanol ratio, % catalyst charge, reaction time and temperature on esterification of oleic acid over 30% SiW11/Hβ. | ||
As shown in Fig. 7, the optimized conditions for maximum conversion (82%) are: SiW11 content = 30%; mole ratio of acid to alcohol = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20; % catalyst charge = 3.53 wt%; reaction temperature = 60 °C and reaction time = 10 h.
20; % catalyst charge = 3.53 wt%; reaction temperature = 60 °C and reaction time = 10 h.
The control experiment using SiW11 and Hβ was carried out under the optimized conditions. It is seen from the Table 3 that Hβ is not much active towards the esterification of oleic acid indicating catalytic activity is truly due to SiW11 only. This is due to the presence of high concentration of very strong acid sites in the catalyst than support (Table 2). The % conversion values show that catalytic activity of SiW11 is retained after introduction in to the channels of Hβ. Thus, we were successful in synthesizing a heterogeneous catalyst and in overcoming the traditional problems of homogeneous catalyst.
| Catalyst | % Conversion | TON |
|---|---|---|
| a % catalyst charge, 3.53 wt% (100 mg); mole ratio of oleic acid/alcohol, 1/20; reaction temperature, 60 °C; reaction time, 10 h.b Catalyst quantity of 23 mg.c TON was calculated from the formula, TON = moles of product/moles of catalyst. | ||
| Hβ | 23 | — |
| 30% SiW11/Hβa | 82 | 1008 |
| SiW11b | 79 | 971 |
3.3. Heterogeneity test
A rigorous proof of heterogeneity can be obtained by filtering the catalyst at the reaction temperature before completion of the reaction and testing the filtrate for activity.29 A test was performed by filtering the catalyst from the reaction mixture at 60 °C after 6 h, and the filtrate was allowed to react up to 10 h. The product of 10 h and the filtrate were analysed by gas chromatogram. The oleic acid conversion of both the samples was 64%, indicating the present catalyst falls into the category C, i.e. the metal does not leach and the observed catalysis is truly heterogeneous in nature.29 On the basis of these results, it can be concluded that there is no leaching of SiW11 species from the support, and the present catalyst is truly heterogeneous in nature.3.4. Recycling of the catalyst
Reusability of the heterogeneous acid catalyst is an important aspect which makes it economical and preferable to homogeneous ones. After completion of reaction, the catalyst was separated from the reaction mixture by simple centrifugation. The separated catalyst was then treated with 5 mL methanol. The subsequent three washings were carried out using distilled water (5 mL) and then dried at 100 °C for 5 h. The recovered catalyst was charged for the further run. The obtained conversions for four successive runs are 82%, 81%, 79% and 79% respectively.The reused catalyst was further characterized by FT-IR, XRD and surface area measurement in order to see any structural change. FT-IR spectra of the used catalyst (Fig. 8a) shows retention of bands at 942 cm−1 (Si–Oa), 860 cm−1 (W–Ob–W) and 715 cm−1 (W–Oc–W) suggesting that the structure of SiW11 in regenerated catalyst is intact. An XRD pattern of the regenerated catalyst (Fig. 8b) shows retention of typical reflections in the regenerated catalyst suggesting the retention of the zeolite framework. BET surface area and pore diameter of the reused catalyst were found to be 430.2 m2 g−1 and 1.68 nm, respectively, which are comparable with the fresh catalyst (surface area: 439.8 m2 g−1; pore diameter: 1.71 nm).
3.5. Kinetic study
A detailed study on the kinetic behaviour was tested for esterification of oleic acid over 30% SiW11/Hβ. In all the experiments, reaction mixtures were analysed at a fixed interval of time. Esterification of oleic acid with methanol was carried with a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 molar ratio; since methanol was taken in large excess, the rate law is expected to follow first-order dependence.18
20 molar ratio; since methanol was taken in large excess, the rate law is expected to follow first-order dependence.18
The plot of ln(Co/C) versus time (Fig. 9) shows a linear association of oleic acid consumption with respect to time. With an increase in the reaction time, there is a gradual increase in the oleic acid conversion over the present catalyst. This observation indicates that esterification of oleic acid follows first-order dependence with respect to time.
This was further supported by the study of the effect of catalyst amount on the rate of esterification of oleic acid. The catalyst amount was varied from 17 × 10−3 to 53 × 10−3 mmol at a fixed substrate concentration of 10 mmol and at a temperature of 60 °C. It can be observed from Fig. 10 that the rate of reaction increases linearly with an increase in the catalyst amount.
3.6. Estimation of activation energy (Ea)
The graph of ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) k versus 1/T was plotted (Fig. 11) and the value of activation energy (Ea) and the pre-exponential factor (A) was calculated from the plot using the Arrhenius equation. The rate constant, k for the esterification of oleic acid with methanol by using 30% SiW11/Hβ at 60 °C was found to be 2.6 × 10−3 min−1. The pre-exponential factor (A) and activation energy (Ea) were found to be 1.486 × 104 min−1 and 43 kJ mol−1, respectively.
k versus 1/T was plotted (Fig. 11) and the value of activation energy (Ea) and the pre-exponential factor (A) was calculated from the plot using the Arrhenius equation. The rate constant, k for the esterification of oleic acid with methanol by using 30% SiW11/Hβ at 60 °C was found to be 2.6 × 10−3 min−1. The pre-exponential factor (A) and activation energy (Ea) were found to be 1.486 × 104 min−1 and 43 kJ mol−1, respectively.
It is important to know whether the reaction rate is diffusion limited/mass transfer limited or it is truly directed by the chemical step where the catalyst is being used at its maximum capacity. It is reported that Ea for diffusion limited process is as low as 10–15 kJ mol−1, and reactions whose rate is governed by a truly chemical step show activation energy higher than 25 kJ mol−1.30 In the present system, the activation energy is 43 kJ mol−1, and therefore, the rate is truly governed by the chemical step.
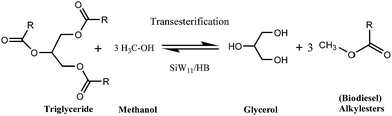
3.7. Transesterification of soybean oil with methanol
Transesterification is a universal and established method for biodiesel production from vegetable and other fatty oils (Scheme 2). Application of the present catalyst was extended to soybean oil with methanol. The properties such as acid value, saponification value and average molecular weight of soybean oil are given in Table 4. Acid values and saponification values were determined by acid–base titration and from these values average molecular weight was determined using following equation,where, SV denotes saponification value and AV denotes to acid value.
| Acid value, mg KOH per g | Saponification value, mg KOH per g | Average molecular weight, g mol−1 |
|---|---|---|
| 0.467 | 178.3 | 946.5 |
The effect of reaction parameters such as % SiW11 content, oil/alcohol ratio, % catalyst charge, reaction time and temperature were studied to optimize the conditions for maximum soybean oil conversion.
To study the effect of % SiW11 content, reaction was performed with 10% SiW11/Hβ, 20% SiW11/Hβ, 30% SiW11/Hβ and 40% SiW11/Hβ (Fig. 12). The conversion of soybean oil increases on increasing SiW11 content. The enhanced activity could be assigned to the increase in the active sites. The 40% SiW11 loaded catalyst showed no significant increase in the conversion. Therefore, 30% SiW11/Hβ was used for further studies.
 | ||
Fig. 12 Effect of % SiW11 content. Reaction conditions: weight ratio of oil/methanol, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4; % catalyst charge, 4 wt%; reaction temperature, 65 °C; reaction time, 8 h. 4; % catalyst charge, 4 wt%; reaction temperature, 65 °C; reaction time, 8 h. | ||
To see the effect of the oil/alcohol ratio, the transesterification reaction was performed by varying the ratio of oil to methanol from 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 to 1
2 to 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 (w/w) using 4 wt% of the catalyst for 8 h at 65 °C. It was observed from Fig. 13 that the soybean oil conversion increases with an increase in the oil/methanol ratio and reaches to 96% at the oil/methanol ratio of 1
10 (w/w) using 4 wt% of the catalyst for 8 h at 65 °C. It was observed from Fig. 13 that the soybean oil conversion increases with an increase in the oil/methanol ratio and reaches to 96% at the oil/methanol ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4. However, the further increase in ratio beyond 1
4. However, the further increase in ratio beyond 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 resulted in a small decrease in the conversion of soybean oil. When too much excess methanol is used for the transesterification reaction, the alcohol will tend to increase the solubility of glycerol. This can lead to unfavorable glycerolysis reaction of biodiesel to occur and thereby decreases the conversion. Hence, the ratio of 1
4 resulted in a small decrease in the conversion of soybean oil. When too much excess methanol is used for the transesterification reaction, the alcohol will tend to increase the solubility of glycerol. This can lead to unfavorable glycerolysis reaction of biodiesel to occur and thereby decreases the conversion. Hence, the ratio of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 is optimum for obtaining high conversion.
4 is optimum for obtaining high conversion.
 | ||
| Fig. 13 Effect of oil/methanol weight ratio. Reaction conditions: % catalyst charge, 4 wt%; reaction temperature, 65 °C; reaction time, 8 h. | ||
The effect of the % catalyst charge on soybean oil conversion was studied by varying catalyst amount in the range 1–6 wt%. As shown in Fig. 14, the conversion increases with increase in amount of 30% SiW11/Hβ and touches optimum of 96%. The increase in the conversion can be attributed to an increase in the number of available catalytically active sites. As a result 4 wt% of the catalyst was considered to be optimum for the maximum conversion.
 | ||
Fig. 14 Effect of % catalyst charge. Reaction conditions: weight ratio of oil/methanol, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4; reaction temperature, 65 °C; reaction time, 8 h. 4; reaction temperature, 65 °C; reaction time, 8 h. | ||
The reaction time was varied within a range from 2 to 10 h. As observed from Fig. 15, when the reaction time was extended to 8 h, the conversion increased to 96%, as an equilibrium conversion. However, no significant increase in the conversion was observed with prolongation of the reaction beyond 8 h. These results indicated that the optimum reaction time was 8 h for maximum conversion.
 | ||
Fig. 15 Effect of reaction time. Reaction conditions: ratio of oil/methanol, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4; % catalyst charge, 4 wt%, 200 mg; reaction temperature, 65 °C. 4; % catalyst charge, 4 wt%, 200 mg; reaction temperature, 65 °C. | ||
The effect of reaction temperature on the conversion was investigated. As illustrated in Fig. 16, the reaction temperature strongly affected the conversion. There was a gradual increase in the conversion upon increasing reaction temperature from 40 to 70 °C. The maximum of 96% conversion was achieved at 65 °C. The high reaction temperature results in high reaction rate. However, the further increase in reaction temperature from 65 to 70 °C results no significant increase in the conversion because of attaining the boiling point of methanol. It might be the reason for decrease in the conversion of transesterification.
 | ||
Fig. 16 Effect of reaction temperature. Reaction conditions: ratio of oil/methanol, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4; % catalyst charge, 4 wt%; reaction time, 8 h. 4; % catalyst charge, 4 wt%; reaction time, 8 h. | ||
The optimized conditions for maximum % conversion (96%) are: weight ratio of oil to alcohol 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4; % catalyst charge, 4 wt%; reaction temperature 65 °C, and reaction time 8 h.
4; % catalyst charge, 4 wt%; reaction temperature 65 °C, and reaction time 8 h.
The control experiments were carried out under optimized conditions with Hβ (31% conversion) and SiW11 (93% conversion). Almost the same activity was obtained for 30% SiW11/Hβ catalyst (96% conversion) using same concentration of active species indicating that SiW11 is the real active species.
Heterogeneity test was carried out as described previously and it was found that catalyst falls in category C which designates truly heterogeneous nature.
Further, the catalyst was recycled up to four cycles and conversions for successive four runs are 96%, 96%, 94% and 93% respectively. The results indicate that catalyst can be recycled up to four cycles without any significant loss in the conversion.
The catalytic activity of the present system for biodiesel synthesis using real high FFA feedstock, jatropha oil was carried out under optimized conditions of transesterification reaction. The conversion of FFA was found to be 97%. Further the activity of the catalyst for simultaneous esterification and transesterication of acid oil was checked. The acid oil used was prepared in the laboratory by mixing measured amounts of oleic acid (10 wt% of soybean oil) and soybean oil and 94% conversion was found under optimized conditions. This suggests that the present catalytic system works efficiently for simultaneous esterification as well as transesterification.
4. Conclusions
In conclusion, we have come up with a novel heterogeneous catalyst comprising of SiW11 and Hβ. The characterization methods confirm that SiW11 remains intact inside the framework of the zeolite support with strong hydrogen bonding.The present catalyst exhibits excellent activity for esterification of oleic acid (82% conversion) with 43 kJ mol−1 of activation energy. The present catalyst has been found to be suitable for the synthesis of biodiesel from soybean oil under very mild conditions. The catalyst was efficient and it gives 96% conversion. The catalyst was reused with consistency in activity up to four times. The catalyst was also evaluated for simultaneous esterification and transesterification. The observed 94% conversion shows that the present catalyst can be employed as an efficient environmentally benign catalyst, for the feedstocks that are rich in FFAs and where simultaneous esterification and transesterification of triglycerides takes place. The present catalytic system comprises properties like heterogeneous nature, recyclable, highly active, thermally stable, and non-toxic which make it environmentally benign.
Acknowledgements
We are thankful to UGC (39-837/2010 (SR)) for the financial support. AP is thankful to DST (SR/S5/GC-01/2009), Delhi, for financing a part of this work. One of the authors, Mr Nilesh Narkhede, is also thankful to UGC, New Delhi for the award of Research Fellowship.Notes and references
- P. Dupont and F. Lefebvre, J. Mol. Catal. A: Chem., 1996, 114, 299 CrossRef CAS.
- C. De Castro, J. Primo and A. Corma, J. Mol. Catal. A: Chem., 1998, 134, 215 CrossRef CAS.
- M. T. Pope, Heteropoly and Isopoly Oxometalates, Springer-Verlag, Berlin, 1983 Search PubMed.
- M. T. Pope and A. Muller, Angew. Chem., Int. Ed. Engl., 1991, 30, 34 CrossRef.
- K. Kamata, K. Yohehara, Y. Sumida, K. Yamaguchi, S. Hikichi and N. Mizuno, Science, 2003, 300, 964 CrossRef CAS PubMed.
- H. Chermette and F. Lefebvre, C. R. Chim., 2012, 15, 143 CrossRef CAS PubMed.
- M. Harrup and C. L. Hill, Inorg. Chem., 1994, 33, 5448 CrossRef CAS.
- N. Mizuno, K. Kamata and K. Yamaguchi, Top. Organomet. Chem., 2011, 37, 127 CrossRef CAS.
- N. Mizuno and M. Misono, Chem. Rev., 1998, 98, 199 CrossRef CAS PubMed.
- K. Kamata, M. Kotani, K. Yamaguchi, S. Hikichi and N. Mizuno, Chem.–Eur. J., 2007, 13, 639 CrossRef CAS PubMed.
- B. S. Bassil and U. Kortz, Dalton Trans., 2011, 40, 9649 RSC.
- T. D. Phan, M. A. Kinch, J. E. Barker and T. Ren, Tetrahedron Lett., 2005, 46, 397 CrossRef CAS PubMed.
- A. Sartorel, M. Carraro, A. Bagno, G. Scorrano and M. Bonchio, Angew. Chem., Int. Ed., 2007, 46, 3255 CrossRef CAS PubMed.
- D. Li, Y. Guo, C. Hu, L. Mao and E. Wang, Appl. Catal., A, 2002, 235, 11 CrossRef CAS.
- X. Luo and C. Yang, Phys. Chem. Chem. Phys., 2011, 13, 7892 RSC.
- Y. Guo, Y. Yang, C. Hu, C. Guo, E. Wang, Y. Zou and S. Feng, J. Mater. Chem., 2002, 12, 3046 RSC.
- C. M. Granadeiroa, A. D. S. Barbosaa, P. Silva, F. A. Almeida Pazb, V. K. Sainic, J. Piresc, B. de Castroa, S. S. Balulaa and L. Cunha-Silvaa, Appl. Catal., A, 2013, 453, 316 CrossRef PubMed.
- A. Patel and N. Narkhede, Catal. Sci. Technol., 2013, 3, 3317 CAS.
- R. M. Barrer, Hydrothermal Chemistry of Zeolites, Academic Press, London, 1982 Search PubMed.
- A. K. Endalew, Y. Kiros and R. Zanzi, Biomass Bioenergy, 2011, 35, 3787 CrossRef CAS PubMed.
- M. Chidambaram, C. Venkatesan, P. Moreaub, A. Finiels, A. V. Ramaswamy and A. P. Singh, Appl. Catal. A, 2002, 224, 129 CrossRef CAS.
- A. Talebian-Kiakalaieh, N. A. S. Amin, A. Zarei and I. Noshadi, Appl. Energy, 2013, 102, 283 CrossRef CAS PubMed.
- J. M. Marchetti, V. U. Miguel and A. F. Errazu, Fuel, 2007, 86, 906 CrossRef CAS PubMed.
- G. Corro, U. Pal and N. Tellez, Appl. Catal., B, 2013, 129, 39 CrossRef CAS PubMed.
- V. Brahmkhatri and A. Patel, Appl. Catal., A, 2011, 403, 161 CrossRef CAS PubMed.
- Y. Yang, Y. Guo, C. Hu and E. Wang, Appl. Catal., A, 2003, 252, 305 CrossRef CAS.
- R. Wei, M. Guo and J. Wang, Chin. J. Chem. Eng., 2009, 17, 58 CrossRef CAS.
- O. da Silva Lacerda Jr, R. Marinho Cavalcanti, T. Moreira de Matos, R. Simões Angélica, G. eraldo Narciso da Rocha Filho and I. de Carvalho Lopes Barros, Fuel, 2013, 108, 604 CrossRef PubMed.
- R. A. Sheldon, M. Walau, I. W. C. E. Arends and U. Schuchurdt, Acc. Chem. Res., 1998, 31, 485 CrossRef CAS.
- G. C. Bond, Heterogeneous catalysis: principles and applications, Oxford Chemistry Series, 1974, ch. 3, p. 49 Search PubMed.
- L. Xu, W. Li, J. Hu, K. Li, X. Yang, F. Ma, Y. Guo, X. Yu and Y. Guo, J. Mater. Chem., 2009, 19, 8571 RSC.
- Y. Kuwahara, K. Tsuji, T. Ohmichi, T. Kamegawa, K. Mori and H. Yamashita, Catal. Sci. Technol., 2012, 2, 1842 CAS.
- W. Xie, H. Wang and H. Li, Ind. Eng. Chem. Res., 2012, 51, 225 CrossRef CAS.
- Q. Shu, B. Yang, H. Yuan, S. Qing and G. Zhu, Catal. Commun., 2007, 8, 2159 CrossRef CAS PubMed.
- A. P. S. Dias, J. Puna, M. J. N. Correia, I. Nogueira, J. Gomes and J. Bordado, Fuel Process. Technol., 2013, 116, 94 CrossRef PubMed.
- J. F. Puna, M. J. N. Correia, A. P. S. Dias, J. Gomes and J. Bordado, React. Kinet., Mech. Catal., 2013, 109, 405 CrossRef CAS.
- J. F. Puna, J. F. Gomes, J. C. Bordado, M. J. N. Correia and A. P. S. Dias, Appl. Catal., A, 2014, 470, 451 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2014 |