Synthesis, crystal structure and photoluminescence of novel blue-emitting Eu2+-doped (SiC)x–(AlN)1−x phosphors by a nitriding combustion reaction†
Zhongqi Shi*a,
Zhilei Weia,
Cheng He*a,
Ruifeng Jinga,
Hongjie Wanga and
Guanjun Qiaoab
aState Key Laboratory for Mechanical Behavior of Materials, School of Materials Science and Engineering, Xi'an Jiaotong University, Xi'an 710049, China. E-mail: zhongqishi@mail.xjtu.edu.cn; hecheng@mail.xjtu.edu.cn; Fax: +86 29 82663453; Tel: +86 29 82667942
bSchool of Materials Science and Engineering, Jiangsu University, Zhenjiang 212013, China
First published on 14th November 2014
Abstract
Novel blue-emitting phosphors with the chemical composition of (SiC)x–(AlN)1−x:yEu2+ (x = 0.06–0.50, y = 0.001–0.01) were synthesized by a nitriding combustion reaction route, and the crystal structure, luminescence properties and thermal stability of the (SiC)x–(AlN)1−x:yEu2+ phosphors were investigated by theoretical and experimental approaches. First-principles calculation results prove that the solid solution of SiC with AlN promotes the doping of Eu2+ ions into the (SiC)x–(AlN)1−x host lattice, and Eu2+ ions tend to occupy Al sites of the host. The synthesized (SiC)x–(AlN)1−x:yEu2+ phosphors absorb light in the region of 250–425 nm and show a single and symmetric broadband emission centered at about 470 nm due to the 4f65d–4f7 transitions of Eu2+. The luminescence intensity increases with the SiC content and reaches its maximum at x = 0.20. The critical quenching concentration of Eu2+ in the (SiC)0.20–(AlN)0.80:yEu2+ phosphor is about y = 0.006. The composition-optimized (SiC)0.20–(AlN)0.80:0.006Eu2+ phosphor shows a small thermal quenching, retaining the luminance of 91.1% at 150 °C. The CIE coordinates were measured as (0.135, 0.167) with high color purity. The above results indicate that (SiC)x–(AlN)1−x:yEu2+ is a promising candidate as a blue-emitting ultraviolet convertible phosphor for white LEDs, and the combustion reaction route is expected to be applicable to the synthesis of other kinds of nitride phosphors.
1. Introduction
White light emitting diodes (LEDs) have attracted increasing attention as a promising light source for solid state lighting due to their long lifetime, high luminous efficiency, energy saving, and environmentally friendly characteristics.1–3 Typically, commercial white LEDs can be generated by the combination of a blue LED chip and the famous yellow phosphor, YAG:Ce3+.4 However, the disadvantages of this method are a low color-rendering index (CRI) and high correlated color temperature (CCT) due to red emission deficiency in the visible spectrum. During the last several years, white LEDs fabricated by using ultraviolet (UV)/near-ultraviolet (n-UV) chips coupled with blue, green and red phosphors have attracted much attention because of the advantageous of color stability and excellent color rending.5–7 Therefore, the exploration of novel phosphors with strong absorption, desirable emission/excitation, and high efficiency under UV/n-UV excitation plays an important role in the development of white LEDs. Furthermore, low fabrication cost, low thermal quenching as well as high chemical stability are also required for the phosphors.As a highly efficient activator with allowed 4f–5d transitions, Eu2+ ion has been widely investigated for the rare-earth-doped phosphors. Because the 4f–5d transition of Eu2+ is sensitive to the crystal field and covalency, the Eu2+-doped phosphors have strong absorption in the UV to the visible spectral region and exhibit broad emission bands from blue to red.8 Recently, Eu2+-doped nitride phosphors have gained great interest due to several merits, such as wide band gap, significant luminescence properties with high efficiency, and excellent thermal and chemical stabilities.7 Examples of these known nitride phosphors are, α-SiAlON:Eu2+,9,10 β-SiAlON:Eu2+,11,12 AlN:Eu2+,13–15 MSi2O2N2:Eu2+ (M = Ca, Sr, Ba),16,17 CaAlSiN3:Eu2+,8,18,19 M2Si5N8:Eu2+,20–22 α-SrNCN:Eu2+,23 and Ba3Ga3N5:Eu2+.24 In 1978, Cutler et al.25 first reported that SiC and AlN can form a 2H solid solution (SiC–AlN ss) in overall compositions. Subsequently, Zangvil and Ruh26 presented a tentative SiC–AlN phase diagram. Several researchers also reported that SiC–AlN ss has excellent mechanical properties (especially in high temperature), good chemical stability, high thermal conductivity, and adjustable semiconducting properties,27–33 which indicates that SiC–AlN ss has great opportunities to be an excellent host for the phosphors. However, to the best of our knowledge, no details regarding the luminescence properties of rare earth-doped (SiC)x–(AlN)1−x crystalline material have been reported in the literature.
In this paper, we report the preparation of blue-emitting (SiC)x–(AlN)1−x:yEu2+ (x = 0.06–0.50, y = 0.001–0.01) phosphors by a combustion synthesis method, which involves the merits of simple processing, high energy efficiency and time saving. The crystal structure, luminescence properties and thermal stability of the (SiC)x–(AlN)1−x:yEu2+ phosphors were investigated in detail. It is believed that the novel (SiC)x–(AlN)1−x:yEu2+ phosphors can act as blue-emitting UV convertible phosphors for white LEDs.
2. Experimental
The powders with the composition of (SiC)x–(AlN)1−x:yEu2+ (0 ≤ x ≤ 0.50, 0.001 ≤ y ≤ 0.01) were fabricated by nitriding combustion synthesis method. The powder mixture of high-purity Al (>99.0%, 2–3 μm, Henan yuanyang aluminum industry Co. Ltd., Xinxiang, China), AlN (type H, >99.9%, ∼0.5 μm, Tokuyama K.K., Hino, Tokyo, Japan), β-SiC (99.09%, 40 nm, Kaier Nano. Co. Ltd., Hefei, China) and Eu2O3 (99.99%, Yuelong Nonferrous Metals Ltd., Shanghai, China) were ball-milled in alcohol using a planetary mill for 2 h. In the powder mixture, Al powder was used as a fuel, and both AlN and SiC powders can be seen as diluent for the complete nitridation of Al powder. After drying and sieving, 20 g of the mixed powder was put into a cylindrical porous graphite crucible (Ø 42 mm × 90 mm H) and ignited from the bottom by passing an electric current of 60 V × 24 A for 20 s through a carbon ribbon under 1.0 MPa N2 atmosphere. Once the combustion reaction was triggered, the temperature increased drastically. A W-Re thermocouple protected by alumina tubes was inserted into the center of the mixture to record the temperature–time pattern of the combustion reaction. The whole combustion synthesis process completed within a few minutes (see ESI Fig. S1†). The products were ground into a powder for subsequent analysis.The phase purity of the synthesized powders were examined by X-ray diffraction (XRD, JDX-3530, JEOL, Japan) with Cu Kα radiation at a scanning rate of 0.2° min−1. Morphologies of the powders were observed using a field emission scanning electron microscopy (FESEM, S4800, Hitachi, Japan). High-resolution transmission electron microscopy (HRTEM; JEOL JEM-2100F) images were obtained on a JEM-2100F electron microscope (JEOL, Japan). Fourier-transform infrared spectra (FTIR) were recorded using a spectrophotometer (AVATAR 360 FTIR, Nicolet, USA) in the wavenumber range of 2000–400 cm−1. The samples' spectra were recorded by transmission in dry air atmosphere through a pastille made of a few milligrams of sample materials mixed with KBr. Photoluminescence (PL) spectra were measured using a Hitachi F-7000 fluorescence spectrophotometer (Hitachi, Japan) at room temperature with a 200 W Xe lamp as the excitation source. The temperature-dependence PL properties were measured on the same spectrophotometer, which was combined with a self-made heating attachment and a computer-controlled electric furnace.
3. Results and discussion
Fig. 1 shows the typical SEM images for the microstructure of the (SiC)x–(AlN)1−x:0.006Eu2+ phosphors (x = 0.06, 0.20 and 0.50) synthesized at Al/(AlN + SiC) mole ratio of 5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5. The mole ratio of choice is the optimized parameter to synthesize the phosphor with higher purity and better luminescence properties according to the XRD patterns and PL spectra of the samples synthesized with the mole ratio ranging from 4
5. The mole ratio of choice is the optimized parameter to synthesize the phosphor with higher purity and better luminescence properties according to the XRD patterns and PL spectra of the samples synthesized with the mole ratio ranging from 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 6 to 6
6 to 6![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 (see ESI Fig. S2 and S3,† respectively). It is observed from Fig. 1 that the particles coarsen with the x value increases. The particle size increases from 1–3 μm for (SiC)0.06–(AlN)0.94:0.006Eu2+ phosphor to 2–5 μm for (SiC)0.20–(AlN)0.80:0.006Eu2+ phosphor, and finally to 2–7 μm for (SiC)0.50–(AlN)0.50:0.006Eu2+ phosphor. In addition, the SiC content obviously affects the particle morphology of the phosphors. The particles exhibit an irregular morphology with x ≤ 0.20, whereas they show a plate-like shape and smooth surface when x = 0.50.
4 (see ESI Fig. S2 and S3,† respectively). It is observed from Fig. 1 that the particles coarsen with the x value increases. The particle size increases from 1–3 μm for (SiC)0.06–(AlN)0.94:0.006Eu2+ phosphor to 2–5 μm for (SiC)0.20–(AlN)0.80:0.006Eu2+ phosphor, and finally to 2–7 μm for (SiC)0.50–(AlN)0.50:0.006Eu2+ phosphor. In addition, the SiC content obviously affects the particle morphology of the phosphors. The particles exhibit an irregular morphology with x ≤ 0.20, whereas they show a plate-like shape and smooth surface when x = 0.50.
FTIR spectra are adopted to investigate the bonds and the inner fine microstructural changes of (SiC)x–(AlN)1−x:0.006Eu2+ phosphors. The results are shown in Fig. 2. In the spectrum of pure AlN powder (Fig. 2(a)), the peak centered at 720 cm−1 corresponds to the Al–N bond of AlN crystal,34 while the absorption bands of the phosphors become broader and weaker, indicating that lattice distortion and lattice imperfection were caused by Eu and/or SiC dissolved into the AlN lattice.35–37 As shown in the FTIR spectrum of AlN:0.006Eu2+ phosphor (Fig. 2(b)), only one broader peak at 720 cm−1 corresponds to the Al–N bond. For the (SiC)0.20–(AlN)0.80:0.006Eu2+ phosphor, three peaks at 740 cm−1, 660 cm−1 and 600 cm−1 are observed, corresponding to Si–C, Al–N and Al–C lattice vibrations, respectively. These peaks are significantly shift to a low energy from those of pure Si–C (800 cm−1) and pure Al–N,37 implying the formation of SiC–AlN ss phase.
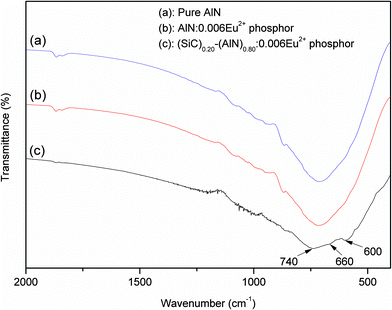 | ||
| Fig. 2 FTIR spectra of (SiC)x–(AlN)1−x:0.006Eu2+ with x = 0 (b) and 0.20 (c). Pure AlN without dopants (a) is made as a reference. | ||
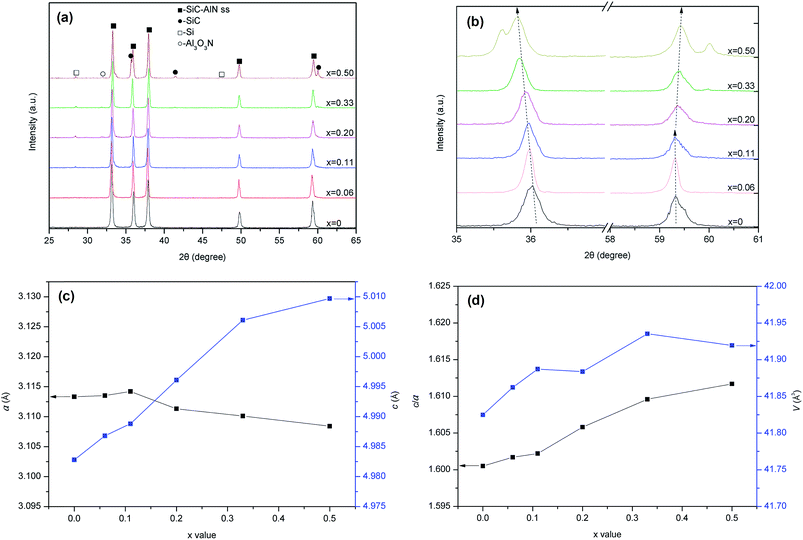
Fig. 3(a) shows the XRD patterns for the (SiC)x–(AlN)1−x:0.006Eu2+ phosphors with the x value of 0, 0.06, 0.11, 0.20, 0.33 and 0.50. As seen, a single SiC–AlN ss phase with a 2H wurzite structure is obtained when x ≤ 0.06. The formation of SiC–AlN ss phase can be described as the following reaction:38
| (1 − x)Al(s) + (1 − x)/2N2(g) + xSiC(s) → (SiC)x–(AlN)1−x (s) | (1) |
The impurities of Si and Al3O3N appear when x ≥ 0.11 and their content rise with a further increase of x value, indicating that more and more Si is displaced from the oxide layer of SiC powder by Al according to the reaction (2), and the formed Al2O3 reacts with AlN to form Al3O3N by the reaction (3):
| 4Al(s) + 3SiO2(s) → 3Si(s) + 2Al2O3(s) | (2) |
| Al2O3(s) + AlN(s) → Al3O3N(s) | (3) |
When the x value further increases >0.20, another impurity of SiC is also detected, demonstrating that the SiC cannot dissolve in the AlN totally with it content >0.20 in present condition.
In order to investigate the effect of SiC content on the crystal cell parameters (a and c) of the SiC–AlN ss phase, the fine scans were carried out from 58 to 61° around the (110) peak for a,38,39 and from 35 to 37° around the (002) peak for c.40 The diffraction peaks at ∼36° shifts to a lower angle, and the peaks at ∼59.5° first decreases and then increase in a narrow range with the increase of SiC content, as shown in Fig. 3(b). The a and c were calculated by using a least-squares procedure with an XRD pattern processing computer program (JADE 5.0, Materials Data) for SiC–AlN ss phase. The results show that the c increases from 4.9748 Å to 5.0097 Å, and the a first increases from 3.1113 Å to 3.1142 Å (x = 0.11) and then decreases to 3.1084 Å, upon the increase of SiC content, as shown in Fig. 3(c). Therefore, the c/a and unit cell volume (V) values of the phosphors with different SiC content can be calculated, and the results are shown in Fig. 3(d). It can be clearly seen that the c/a and V values increase from 1.5989 to 1.6117 and from 41.7052 Å3 to 41.9195 Å3 respectively with the increasing of SiC content, indicating that the SiC–AlN unit cell becomes longer and larger when more and more SiC is solid-soluted in the AlN.
To observe the local crystalline structure of the (SiC)x–(AlN)1−x:yEu2+ phosphors, HRTEM measurements were employed. Fig. 4 shows the TEM and local HRTEM images of the selected samples of (SiC)0.06–(AlN)0.94:0.006Eu2+ and (SiC)0.20–(AlN)0.80:0.006Eu2+. In the HRTEM image of (SiC)0.06–(AlN)0.94:0.006Eu2+ (Fig. 4(b)), the spacing between the neighboring parallel fringes is measured to be 0.272 nm, corresponding to the (100) planes of 2H wurtzite SiC–AlN ss phase. In addition, some indistinct fringes parallel to the (100) planes (in the white line areas) are observed, indicating that the long range order has been lost. This should be caused by the large lattice deformation due to the dissolution of SiC and/or large Eu2+ ions in the host lattice. For the (SiC)0.20–(AlN)0.80:0.006Eu2+ sample, several lattice stripe areas and amorphous areas coexist in the HRTEM image (Fig. 4(d)), exhibiting much higher quantity of grain boundary and defect density in the sample with high content of SiC. However, it is difficult to observe the local environment of the Eu2+ occupation within the SiC–AlN host lattice due to the low resolution for accurate structural determination. In fact, an understanding of the doped Eu2+ in the structure of SiC–AlN host lattice is necessary to elucidate the luminescent behaviors.
 | ||
| Fig. 4 (a) TEM image of (SiC)0.06–(AlN)0.94:0.006Eu2+ and (b) HRTEM image of the selected area in (a); (c) TEM image of (SiC)0.20–(AlN)0.80:0.006Eu2+ and (d) HRTEM image of the selected area in (c). | ||
In order to further clarify the Eu2+ ion site, we perform first-principles calculations of (SiC)x–(AlN)1−x:yEu2+ and investigate the site of the Eu2+ ion by comparing the theoretical and experimental results. Simulations are performed by the CASTEP software package41 based on density functional theory (DFT) with the generalized gradient approximation (GGA) of Perdew, Burke, and Ernzerhof (PBE) exchange–correlation functionals.42 Similar functions have been successfully used to study the structural and electronic properties of AlN (wurtzite structure).43,44 We use a plane wave basis set for the expansion of the wave functions where the electron-ion interaction is described by ultrasoft pseudopotential, which is as soft as possible within the core region. Thus, the cutoff energy is dramatically reduced. With a cutoff energy of 480 eV and the k-points of 5 × 5 × 3, sufficient numbers of wave functions are included so as to obtain precise information about structure and electronic structure of the SiC–AlN host. The convergence tolerances are in energy of 1.0 × 10−5 eV per atom, maximum force of 0.03 eV per atom and maximum displacement of 0.05 eV per atom.
According to the results of FTIR and XRD analyses as shown in Fig. 2 and 3(a), the structure of (SiC)x–(AlN)1−x can be built up by the random substitution of Al–N pair by Si–C pair respectively in the hexagonal AlN (P63mc, space group 186),37 and the unit cell containing 8 atoms exhibits a lattice constant a = 3.1113 Å, c = 4.9805 Å. In order that the Eu2+ concentration approaches close to the experimental results, we prepared the (SiC)x–(AlN)1−x host lattices with the k-points of 5 × 5 × 3 which contain 32, 64, and 128 atoms with a single Eu2+ ion. Considering the radius and charge properties of Eu2+ ions, three possible locations of Eu2+ ions in the (SiC)x–(AlN)1−x host lattice can be existed: (Structure 1) Eu2+ doped in the octahedral interstice of (SiC)x–(AlN)1−x; (Structure 2) Eu2+ occupied an Al site of (SiC)x–(AlN)1−x; (Structure 3) Eu2+ occupied a Si site of (SiC)x–(AlN)1−x, as shown in Fig. 5.
Table 1 shows the forming energies and lattice constants of the above doping structures ((SiC)x–(AlN)1−x:0.06Eu2+) from simulation, and the lattice constants of (SiC)x–(AlN)1−x:0.006Eu2+ obtained from the XRD results (Fig. 3(d)) are listed for comparation. It can be seen from Table 1 that the forming energy of Structures (2) and (3) are much lower than that of Structures (1), indicating that the Structure (2) and (3) are easier to form than the Structure (1). In other words, the Eu2+ ion is apt to occupy a cation site in the (SiC)x–(AlN)1−x host lattice. And according to the data of vacancy forming energy, we can find the vacancy forming energy of Al is lower than that of Si when x = 0–0.50, which means the formation of Al vacancy is easier than that of Si. At the same time, according to the forming energy for Structures (2) and (3), we can find the same results. Therefore, the Eu2+ ion tends to occupy an Al site of (SiC)x–(AlN)1−x to form Structure (2). Moreover, we can find the vacancy forming energy of Al decreases when x value increases, which indicates as the SiC content increases, the Eu2+ ion is easier doped into (SiC)x–(AlN)1−x host lattice. This phenomenon is identical to the experimental results reported by the fabrication of AlN:Eu2+ phosphors with SiC as an effective flux.15,46 The same trendy has also been found from the results of calculation for the (SiC)x–(AlN)1−x:0.03Eu2+ and (SiC)x–(AlN)1−x:0.015Eu2+. Duo to simulation of (SiC)x–(AlN)1−x:0.006Eu2+ needs more than three hundreds atoms, it costs so much computation resource. We believe the prepared (SiC)x–(AlN)1−x:0.006Eu2+ has the same results with the simulated (SiC)x–(AlN)1−x:0.06Eu2+, (SiC)x–(AlN)1−x:0.03Eu2+ and (SiC)x–(AlN)1−x:0.015Eu2+ according to the simulations above.
| x = 0 | x = 0.06 | x = 0.11 | x = 0.20 | x = 0.33 | x = 0.50 | |
|---|---|---|---|---|---|---|
| a Data of lattice constants (AlN: c/a = 1.6003, V = 41.7448; SiC: c/a = 1.6409, V = 41.3721) are obtained from ref. 45. | ||||||
| Forming energy for Structure 1 (Eu doped in interstice site) (eV per atom) | 14.0617 | — | 6.5961 | 6.8619 | 6.9114 | 7.1730 |
| Vacancy forming energy (Al) (eV per atom) | 8.6468 | — | 2.0445 | 2.0260 | 2.0154 | 1.9409 |
| Vacancy forming energy (Si) (eV per atom) | — | — | 3.6382 | 3.5744 | 3.5485 | 3.5539 |
| Forming energy for Structure 2 (Eu occupied Al site) (eV per atom) | 10.2764 | — | 1.8286 | 1.7985 | 1.7951 | 1.7902 |
| Forming energy for Structure 3 (Eu occupied Si site) (eV per atom) | — | — | 3.4630 | 3.4473 | 3.4327 | 3.4087 |
| Simulated c/a of Structure 1 | 1.6007 | — | 1.6051 | 1.6083 | 1.6126 | 1.6144 |
| Simulated V of Structure 1 | 41.9132 | — | 42.2625 | 42.3838 | 42.4254 | 42.4225 |
| Simulated c/a of Structure 2 (Eu occupied Al site) | 1.6008 | — | 1.6026 | 1.6060 | 1.6101 | 1.6124 |
| Simulated V of Structure 2 (Eu occupied Al site) | 41.7530 | — | 41.8927 | 41.8981 | 41.9383 | 41.9405 |
| Simulated c/a of Structure 3 (Eu occupied Si site) | — | — | 1.6018 | 1.6049 | 1.6085 | 1.6105 |
| Simulated V of Structure 3 (Eu occupied Si site) | — | — | 41.7628 | 41.7837 | 41.8258 | 41.8725 |
| Experimental c/a for (SiC)x–(AlN)1−x:0.006Eu2+ | 1.6005 | 1.6017 | 1.6022 | 1.6058 | 1.6096 | 1.6117 |
| Experimental V for (SiC)x–(AlN)1−x:0.006Eu2+ | 41.8247 | 41.8622 | 41.8871 | 41.8837 | 41.9352 | 41.9195 |
The lattice constants are also considered to further prove the Eu2+ ion site. According to the Table 1, we can find that both of the c/a and V of the prepared (SiC)x–(AlN)1−x:0.006Eu2+ phosphors increase as the x value increases, which is because the c/a of SiC is more than that of AlN45 and the large Eu2+ ions are doped into the host lattice. Furthermore, based on the results of simulations, it can be found that the data of both c/a and V of the (SiC)x–(AlN)1−x:yEu2+ with different structures have similar varying tendency as those of the experimental results. However, the simulated results of Structure (2) are almost the same as the experimental results, which is agreement with the simulated results of forming energies above. Hence, we can understand that the Structure (2) is the most likely structure for the (SiC)x–(AlN)1−x:yEu2+ phosphors.
Fig. 6 shows the excitation and emission spectra of the (SiC)x–(AlN)1−x:0.006Eu2+ phosphors with different SiC content. The excitation spectrum for the sample without SiC has only one weak and broad band centered at 310 nm. However, for the samples added with SiC, the shapes of the excitation spectra have changed and show a broad band in the range of 250–425 nm, which consist of three bands centered at 285 nm, 330 nm and 360 nm, respectively. These three excitation bands correspond to the crystal-field splitting of the 5d level of the Eu2+ ions. The emission spectrum of the phosphor without SiC additive has two weak emission bands at 470 nm and 505 nm. The former band is attributed to 4f65d–4f7 transitions of Eu2+,13 and the later should be ascribed to the higher electronegativity and nephelauxetic effect of N3− ion than O2− ion.47 The green-yellow emission was also observed in the AlN:Eu2+ phosphor prepared by carbothermal reduction method,14 where the low oxygen content was gained by adding excess carbon. When the SiC is added, the phosphors show only one emission band centered at about 470 nm. The luminescence intensity increases with the SiC content and reaches its maximum at x = 0.20. The decrease of luminescence intensity with higher SiC content should be caused by the existence of impurity phases and incomplete solid-solution of SiC into the AlN matrix (see Fig. 3(a)).
 | ||
| Fig. 6 Excitation (λem = 470 nm) and emission spectra (λex = 330 nm) of (SiC)x–(AlN)1−x:0.006Eu2+ (x = 0, 0.06, 0.11, 0.20, 0.33 and 0.50). | ||
Fig. 7 exhibits the emission spectra of several (SiC)0.20–(AlN)0.80:yEu2+ (y = 0.001, 0.0024, 0.0048, 0.006, 0.0072, 0.0084 and 0.01) phosphors. We can observe clearly that all the phosphors show only one emission band centered at about 470 nm under 330 nm excitation. Meanwhile, the PL intensity exhibits an enhancement initially until y = 0.006, beyond which its intensity shows a drastic decrease (see the inset of Fig. 7). The decrease should be ascribed to the concentration quenching of activator ions. The appearance of concentration quenching is caused by nonradiative energy transfer between neighboring doped Eu2+ ions. The critical distance Rc can be calculated by utilizing the following equation by Blasse:48
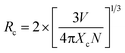 | (4) |
The dependence of the highest relative emission intensity and peak position of (SiC)x–(AlN)1−x:yEu2+ phosphors on the SiC content (x value) and corresponding Eu2+ concentration (y value) was investigated, as shown in Fig. S4.† It can be seen that the Eu2+ concentration for the highest emission intensity of the phosphors increases gradually from 0.0048 to 0.0072 with the increase of x values from 0.06 to 0.50, illustrating that the solid solution of SiC with AlN improves the solubility of Eu2+ ions in the SiC–AlN host lattice, which is in accordance with the results of first-principles calculations (Table 1). In addition, the relative intensity rapidly increases with the increase of x values from 0.06 to 0.20 (corresponding Eu2+ concentration increasing from 0.0048 to 0.006), and reaches a maximum at about x = 0.20. Above this content, the relative intensity decreases seriously. The reason for the decrease can be ascribed to the impurity phases, as has been explained in Fig. 6. Meanwhile, the peak position of (SiC)x–(AlN)1−x:yEu2+ phosphors shifts slightly from 470 to 478 nm by increasing the x value up to 0.50. As can be seen from Fig. 6, the shapes of the excitation spectra remains unchanged for the SiC added phosphors, indicating that the local environment of Eu2+ is similar. The redshifting behavior is therefore attributed to the increased Stokes shifts with the addition of SiC.
For the application of high power LEDs, the thermal stability of phosphor is one of important issues to be considered. Temperature dependence of emission spectra for the composition-optimized (SiC)0.20–(AlN)0.80:0.006Eu2+ phosphor under excitation at 330 nm is shown in Fig. 8(a). With increasing temperature, the PL intensities decrease while the peak positions almost keep still. Fig. 8(b) shows the temperature dependence of PL intensity of (SiC)0.20–(AlN)0.80:0.006Eu2+ together with the AlN:Eu2+,13 and the commercial BaMgAl10O17:Eu2+ (BAM:Eu2+) phosphors. It can be observed that the thermal stability of (SiC)0.20–(AlN)0.80:0.006Eu2+ phosphor is similar to that of the AlN:Eu2+ phosphor, and much more obvious than that of the BAM phosphor. The intensity at 150 °C (423 K) remains at about 91.1% of the initial intensity. The activation energy (ΔE) can be expressed by eqn (5):50
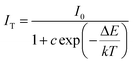 | (5) |
 | ||
| Fig. 8 (a) The temperature dependence of the emission spectra of (SiC)0.20–(AlN)0.80:0.006Eu2+ excited at 330 nm. (b) Temperature dependence of PL intensity of (SiC)0.20–(AlN)0.80:0.006Eu2+ together with the AlN:Eu2+ phosphor13 and the commercial BAM:Eu2+. The inset shows the relationship between ln(I0/IT − 1) and 1/(kT) for the (SiC)0.20–(AlN)0.80:0.006Eu2+ phosphor in the measured temperature range and calculated ΔE. | ||
 | ||
| Fig. 9 Color coordinates of (SiC)0.20–(AlN)0.80:0.006Eu2+ in the CIE chromaticity diagram, and the inset shows (SiC)0.20–(AlN)0.80:0.006Eu2+ under 365 nm excitation in the UV box. | ||
4. Conclusions
In summary, we have synthesized novel blue-emitting nitride phosphors with chemical composition (SiC)x–(AlN)1−x:yEu2+ (x = 0.06–0.50, y = 0.001–0.01) via a simple, facile nitriding combustion reaction method. First-principles calculations of (SiC)x–(AlN)1−x:yEu2+ were performed to investigate the forming energies and possible doping sites of the Eu2+ ions in the host lattice. The results prove that the solid solution of SiC with AlN promotes the doping of Eu2+ ions into the (SiC)x–(AlN)1−x host, and Eu2+ ions tend to occupy Al sites of the host. The composition-optimized (SiC)0.20–(AlN)0.80:0.006Eu2+ phosphor shows broad absorption bands in the UV range, and bright blue emissions peaking at 470 nm are obtained upon 330 nm excitation. Studies of the temperature-dependent luminescence properties indicate that the composition-optimized phosphor has a small thermal quenching, retaining its luminance of 91.1% at 150 °C. The CIE coordinates are (0.135, 0.167) with high color purity. This indicates a prospective for application of the present novel phosphors in white LEDs.Acknowledgements
This work is supported by the National Natural Science Foundation of China (51002115 and 51471124), Program for New Century Excellent Talents in University (NCET–12–0454) and Program for Young Excellent Talents in Shaanxi Province (2013KJXX–50).References
- E. F. Schubert and J. K. Kim, Science, 2005, 308, 1274–1278 CrossRef CAS PubMed.
- T. Hashimoto, F. Wu, J. S. Speck and S. Nakamura, Nat. Mater., 2007, 6, 568–571 CrossRef CAS PubMed.
- T. Nishida, T. Ban and N. Kobayashi, Appl. Phys. Lett., 2003, 82, 3817–3819 CrossRef CAS PubMed.
- V. Bachmann, C. Ronda and A. Meijerink, Chem. Mater., 2009, 21, 2077–2084 CrossRef CAS.
- J. Y. Han, W. B. Im, G. Y. Lee and D. Y. Jeon, J. Mater. Chem., 2012, 22, 8793–8798 RSC.
- C. C. Lin and R. S. Liu, J. Phys. Chem. Lett., 2011, 2, 1268–1277 CrossRef CAS.
- R. J. Xie and N. Hirosaki, Sci. Technol. Adv. Mater., 2007, 8, 588–600 CrossRef CAS PubMed.
- X. Piao, K. Machida, T. Horikawa, H. Hanzawa, Y. Shimomura and N. Kijima, Chem. Mater., 2007, 19, 4592–4599 CrossRef CAS.
- X. Xu, J. Tang, T. Nishimura and L. Hao, Acta Mater., 2011, 59, 1570–1576 CrossRef CAS PubMed.
- T. Suehiro, H. Onuma, N. Hirosaki, R. J. Xie, T. Sato and A. Miyamoto, J. Phys. Chem. C, 2010, 114, 1337–1342 CAS.
- R. J. Xie, N. Hirosaki, H. L. Li, Y. Q. Li and M. Mitomo, J. Electrochem. Soc., 2007, 154, J314–J319 CrossRef CAS PubMed.
- S. W. Choi and S. H. Hong, J. Electrochem. Soc., 2010, 157, J297–J300 CrossRef CAS PubMed.
- K. Inoue, N. Hirosaki, R. J. Xie and T. Takeda, J. Phys. Chem. C, 2009, 113, 9393–9397 Search PubMed.
- L. J. Yin, X. Xu, W. Yu, J. G. Yang, L. X. Yang, X. F. Yang, L. Y. Hao and X. J. Liu, J. Am. Ceram. Soc., 2010, 93, 1702–1707 CAS.
- Z. Shi, W. Yang, S. Bai, G. Qiao and Z. Jin, Ceram. Int., 2011, 37, 2051–2054 CrossRef CAS PubMed.
- V. Bachmann, C. Ronda, O. Oeckler, W. Schnick and A. Meijerink, Chem. Mater., 2009, 21, 316–325 CrossRef CAS.
- Y. Q. Li, A. C. A. Delsing, G. De With and H. T. Hintzen, Chem. Mater., 2005, 17, 3242–3248 CrossRef CAS.
- J. Cho, B. K. Bang, S. J. Jeong and C. H. Kim, RSC Adv., 2014, 4, 23218–23222 RSC.
- B. Lei, K. I. Machida, T. Horikawa and H. Hanzawa, Chem. Lett., 2010, 39, 104–105 CrossRef CAS.
- M. Zeuner, P. J. Schmidt and W. Schnick, Chem. Mater., 2009, 21, 2467–2473 CrossRef CAS.
- B. Lei, K. I. Machida, T. Horikawa, H. Hanzawa, N. Kijima, Y. Shimomura and H. Yamamoto, J. Electrochem. Soc., 2010, 157, J196–J201 CrossRef CAS PubMed.
- R. J. Xie, N. Hirosaki, T. Suehiro, F. F. Xu and M. Mitomo, Chem. Mater., 2006, 18, 5578–5583 CrossRef CAS.
- M. Krings, G. Montana, R. Dronskowski and C. Wickleder, Chem. Mater., 2011, 23, 1694–1699 CrossRef CAS.
- F. Hintze, F. Hummel, P. J. Schmidt, D. Wiechert and W. Schnick, Chem. Mater., 2012, 24, 402–407 CrossRef CAS.
- I. B. Cutler, P. D. Miller, W. Rafaniello, H. K. Park, D. P. Thompson and K. H. Jack, Nature, 1978, 275, 434–435 CrossRef CAS.
- A. Zangvil and R. Ruh, J. Am. Ceram. Soc., 1988, 71, 884–890 CrossRef CAS PubMed.
- Y. B. Pan, J. H. Qui, M. Kawagoe, M. Morita, S. H. Tan and D. L. Jiang, J. Eur. Ceram. Soc., 1999, 19, 1789–1793 CrossRef CAS.
- W. Rafaniello, K. Cho and A. V. Virkar, J. Mater. Sci., 1981, 16, 3479–3488 CrossRef CAS.
- G. Magnani and L. Beaulardi, J. Eur. Ceram. Soc., 2006, 26, 3407–3413 CrossRef CAS PubMed.
- R. Roucka, J. Tolle, A. V. G. Chizmeshya, P. A. Crozier, C. D. Poweleit, D. J. Smith, I. S. T. Tsong and J. Kouvetakis, Phys. Rev. Lett., 2002, 88, 206102 CrossRef CAS.
- R. Kobayashi, J. Tatami, T. Wakihara, T. Meguro and K. Komeya, J. Am. Ceram. Soc., 2006, 89, 1295–1299 CrossRef CAS PubMed.
- R. Kobayashi, J. Tatami, T. Wakihara, K. Komeya, T. Meguro, R. Tu and T. Goto, J. Am. Ceram. Soc., 2010, 93, 4026–4029 CrossRef CAS PubMed.
- N. B. Singh, B. Wagner, A. Berghmans, D. J. Knuteson, S. McLaughlin, D. Kahler, D. Thomson and M. King, Cryst. Growth Des., 2010, 10, 3508–3514 CAS.
- K. Li, Z. G. Liu, M. Y. Yu, S. Y. Dong, Q. L. Wang, X. P. Hao and D. L. Cui, Acta Chim. Sin., 2004, 62, 1144–1147 CAS.
- J. Liu, M. Kang, R. Sun and W. Y. Hu, China Powder Sci. Technol., 2008, 14, 28–31 CAS.
- X. B. Yu, X. L. Xu, C. L. Zhou, J. F. Tang, X. D. Peng and S. P. Yang, Mater. Res. Bull., 2006, 41, 1578–1583 CrossRef CAS PubMed.
- J. Tolle, R. Roucka, A. V. G. Chizmeshya, P. A. Crozier, D. J. Smith, I. S. T. Tsong and J. Kouvetakis, Solid State Sci., 2002, 4, 1509–1519 CrossRef CAS.
- M. Ohyanagi, K. Shirai, N. Balandina, M. Hisa and Z. A. Munir, J. Am. Ceram. Soc., 2000, 83, 1108–1112 CrossRef CAS PubMed.
- R. C. Juang, C. C. Chen, J. C. Kuo, T. Y. Huang and Y. Y. Li, J. Alloys Compd., 2009, 480, 928–933 CrossRef CAS PubMed.
- X. Su, W. Zhou, J. Xu, J. Wang, X. He, C. Fu and Z. Li, J. Am. Ceram. Soc., 2012, 95, 1388–1393 CrossRef CAS PubMed.
- M. C. Payne, M. P. Teter, D. C. Allan, T. A. Arias and J. D. Joannopoulos, Rev. Mod. Phys., 1992, 64, 1045–1097 CrossRef CAS.
- J. P. Perdew, K. Burke and M. Ernzerhof, Phys. Rev. Lett., 1996, 77, 3865–3868 CrossRef CAS.
- L. J. Yin, Q. Q. Zhu, W. Yu, L. Y. Hao, X. Xu, F. C. Hu and M. H. Lee, J. Appl. Phys., 2012, 111, 053534 CrossRef PubMed.
- W. Lei, D. Liu, X. Chen, P. Zhu, Q. Cui and G. Zou, J. Phys. Chem. C, 2010, 114, 15574–15577 CAS.
- Y. Pan, S. Tan and D. Jiang, J. Inorg. Mater., 1995, 10, 189–192 CAS.
- B. Dierre, X. L. Yuan, K. Inoue, N. Hirosaki, R. J. Xie and T. Sekiguchi, J. Am. Ceram. Soc., 2009, 92, 1272–1275 CrossRef CAS PubMed.
- R. J. Xie, N. Hirosaki, K. Sakuma, Y. Yamamoto and M. Mitomo, Appl. Phys. Lett., 2004, 84, 5404–5406 CrossRef CAS PubMed.
- G. Blasse, Philips Res. Rep., 1969, 24, 131–144 CAS.
- T. C. Liu, H. Kominami, H. F. Greer, W. Zhou, Y. Nakanishi and R. S. Liu, Chem. Mater., 2012, 24, 3486–3492 CrossRef CAS.
- S. Bhushan and M. V. Chukichev, J. Mater. Sci. Lett., 1988, 9, 319–321 CrossRef.
Footnote |
| † Electronic supplementary information (ESI) available: See DOI: 10.1039/c4ra11277f |
| This journal is © The Royal Society of Chemistry 2014 |