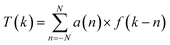Structural, spectroscopic and magnetic properties of a novel copper(II) L-tyrosinato complex†
Agnieszka Wojciechowska*a,
Anna Gągorb,
Julia Jezierskac and
Marek Duczmala
aFaculty of Chemistry, Wrocław University of Technology, Wybrzeże Wyspiańskiego 27, 50-370 Wrocław, Poland. E-mail: agnieszka.wojciechowska@pwr.edu.pl; Fax: +48 71 320 43 60; Tel: +48 71 320 36 66
bInstitute of Low Temperature and Structure Research Polish Academy of Sciences, Okólna 2, 50-422 Wrocław, Poland
cFaculty of Chemistry, University of Wrocław, F. Joliot-Curie 14, 50-383 Wrocław, Poland
First published on 7th November 2014
Abstract
The complex [Cu(L-Tyr)2(H2O)]·H2O (1) (L-Tyr = L-tyrosine) was obtained as crystals and characterized by X-ray, spectroscopic (FT-IR, FT-Raman, NIR-vis-UV, and EPR) and magnetic methods. The monomeric complex crystallized in the monoclinic P21 symmetry with a = 11.967 (1) Å, b = 5.9986 (4) Å, c = 14.936 (1) Å. The amino N and carboxylate O atoms of chelating L-tyrosinate anions together with the O atom of water molecules create a slightly distorted square pyramidal environment around the Cu(II) ions (τ = 0.11). The distance of about 5.998(1) Å [Cu(L-Tyr)2(H2O)] units is involved in a polymeric chain based on N(1)–H⋯O(4)vi and N(2)–H⋯O(5)vi hydrogen bonds. The d–d band found in the polycrystalline-reflectance spectrum at 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 cm−1 is composed of three 2B1(dx2–y2) → 2A1 (dz2), 2B1 → 2B2(dxy) and 2B1 → 2E (dxz ≈ dyz) transitions with energies 14
700 cm−1 is composed of three 2B1(dx2–y2) → 2A1 (dz2), 2B1 → 2B2(dxy) and 2B1 → 2E (dxz ≈ dyz) transitions with energies 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 880, 15
880, 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 and 19
800 and 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 950 cm−1, respectively. In DMSO solution the complex preserves its square pyramidal geometry as evidenced by an intensive band at ca. 16
950 cm−1, respectively. In DMSO solution the complex preserves its square pyramidal geometry as evidenced by an intensive band at ca. 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 cm−1. The EPR spectral parameters for powder (g‖ = 2.236 and g⊥ = 2.063) and DMSO frozen solution (g‖ = 2.250, g⊥ = 2.055, and A‖ = 180 G) correspond to an axial symmetry of the Cu(II) coordination geometry with the dx2–y2 orbital as a ground state of the unpaired electron. Furthermore, the frozen solution spectrum revealed signals corresponding to S = 1 spin states of Cu(II) ions coupled by dipole–dipole interactions with distinctly resolved hyperfine splitting due to two copper nuclei (|D| = 0.0468 cm−1, g‖ = 2.238, g⊥ = 2.06 and A‖ = 85 G). The variable-temperature magnetic susceptibility measurements revealed the existence of a weak ferromagnetic interaction between neighboring copper(II) ions through the N–H⋯O hydrogen bonds.
400 cm−1. The EPR spectral parameters for powder (g‖ = 2.236 and g⊥ = 2.063) and DMSO frozen solution (g‖ = 2.250, g⊥ = 2.055, and A‖ = 180 G) correspond to an axial symmetry of the Cu(II) coordination geometry with the dx2–y2 orbital as a ground state of the unpaired electron. Furthermore, the frozen solution spectrum revealed signals corresponding to S = 1 spin states of Cu(II) ions coupled by dipole–dipole interactions with distinctly resolved hyperfine splitting due to two copper nuclei (|D| = 0.0468 cm−1, g‖ = 2.238, g⊥ = 2.06 and A‖ = 85 G). The variable-temperature magnetic susceptibility measurements revealed the existence of a weak ferromagnetic interaction between neighboring copper(II) ions through the N–H⋯O hydrogen bonds.
Introduction
2-Amino-3-(4-hydroxyphenyl)propionic acid, referred to as L-tyrosine (L-Tyr), is a very important endogenic amino acid and a precursor of thyroid hormones like triiodothyronine (T3) and tyroxine (T4);1 neurotransmitters like dopamine,2 noradrenaline,3 adrenaline;3 alkaloid morphine2 and mescaline4 and, also, the natural pigment – melanin.5 L-Tyrosine, as well as other amino acids, forms coordination bonds with metal ions. These metal ions complexes are structural, spectroscopic, magnetic characterized6–12 and tested against bacterial13,14 or fungal13–15 strains. However, their biological properties are relatively rarely evaluated.First literature report of L-tyrosine interaction with copper(II) ion comes from 1968, when Van der Helm and co-workers published the results of a variation of the square-pyramidal copper(II) surrounded in a complex with glycyl-L-leucyl-L-tyrosine peptide.16 The deprotonated amide nitrogen and carboxyl oxygen atoms of the L-tyrosyl residues together with glycyl nitrogen and oxygen atoms form a base of the square-pyramidal geometry around two copper(II) ions. Further refinement of this copper-peptide dimer crystal structure indicated the occurrence of two peptides and two copper atom in an asymmetric unit.17 But, first crystal structure of copper(II) complex containing pure L-tyrosine was described in 1972 as a structure of bis-(L-tyrosinato)copper(II) complex.18 Later, McAuliffe and co-workers studying the donor properties of this amino acid with Cu2+, Ni2+, Zn2+ ions obtained a powder of [Cu(L-Tyr)2(H2O)] compound.19 After that researchers drew attention to polymeric structure and expected magnetic properties of the bis-(L-tyrosinato)copper(II) complex,18 finally, they described it as a coordination polymer of formula [Cu(L-Tyr)2]n.20–22 In recent years, a narrow group of L-tyrosinato five-coordinated Cu(II) ion complexes has been extended by four new complexes consisting of aromatic diamine and water molecule located in vertices of a square-pyramid, characterized by crystal structure, spectroscopic and magnetic studies: [Cu(L-Tyr)(2,2′-bpy)(H2O)]Cl·3H2O (2,2′-bpy = 2,2′-bipyridine),23 [Cu(L-Tyr)(phen)(H2O)]ClO4·2.5H2O,24 [Cu(L-Tyr)(phen)(H2O)]ClO4 25 and [Cu(phen)(L-Tyr)(EA)]ClO4 26 (phen = 1,10-phenanthroline, EA = ethylamine), [Cu(L-Tyr)(5,6-dmp)(H2O)]ClO4 (5,6-dmp = 5,6-dimethyl-1,10-phenanthroline),27 [Cu(IP)(L-Tyr)(H2O)]ClO4·H2O (IP = imidazol[4,5-f][1,10] phenanthroline).28 Especially, the [Cu(L-Tyr)(5,6-dmp)(H2O)]ClO4 complex is worth to mention, because of the studies of DNA cleavage and anticancer activity of this complex revealed its interaction with DNA of a calf thymic lymphocytes leading to the mitotic catastrophe and apoptotic cell death.27
Further studies on the L-tyrosinato Cu(II) complexes are focused on investigating 3-iodo- and 3,5-diiodo-derivatives. Both bioligands form crystalline complexes of square-pyramid geometry with water molecule in [Cu(2,2′-bpy)(L-Ityr)(H2O)]·NO3·CH3OH·H2O (L-Ityr = 3-iodo-L-tyrosine),29 [Cu(hista)(L-I2tyrO−)(H2O)]2·2H2O (L-I2tyr = 3,5-diiodo-L-tyrosine),29 [Cu(2,2′-bpy)(L-I2tyrO−)(H2O)]·2H2O,30 as well as nitrate(V) ion in [Cu(2,2′-bpy)(L-I2tyrOH)(NO3)]·CH3OH.30 The modification of phenolate ring leads to synthesizing the reduced Schiff base of benzaldehyde and L-tyrosine – Ph-TyrH ligand. This L-Tyr derivative interacts with copper(II) ion giving crystals of [Cu(Ph-Tyr)(L)(ClO4)]31 complex (Ph-Tyr = L-phenylalanine-L-tyrosine, L = phen or dppz = dipyrido[3,2-a:2′,3′-c]phenazine).
L-Tyrosine in combination with other amino acids forms various dipeptides. These compounds chelated Cu2+ ion and with bidentate organic N ligands and water molecule create five-coordinated copper(II) complexes i.e. [Cu(L-Tyr-hist)] (L-Tyr-hist = L-tyrosyl-L-histidine),32 [Cu(L-leucyl-L-tyrosine)(H2O)],33 [Cu(glycyl-L-tyrosine)(H2O)2]·2H2O,34 [Cu(Gly-L-Tyr)(bzp)]34 and [Cu(Gly-L-Tyr)(bzmp)]19 (Gly-L-Tyr = glycyl-L-tyrosinate; bzp = N-benzyl-N-2-pyridylmethylamine; bzmp = N-benzyl-N-6-methyl-2-pyridylmethylamine).35 L-Tyr, also, in combination with the vitamin B6-amino acid Schiff base builds a monomeric [Cu(5′-phosphopyridoxylidene-DL-Tyr)(H2O)] complex.36 The bridging carboxylate mode described as μ-(κ3N, O; O′) binds to the another Cu atom forming Cu–O–C–O–Cu link and causes a polymeric nature of the complex catena-(μ2-N-salicylidene-L-Tyr)-copper(II).37
As it was shown by us, the square pyramid geometry is preferred in Cu(II) complexes with L-tyrosine and its derivatives. It was found that, in [Cu(L-Tyr)2X] and [Cu(L-Tyr)(diamine)X] units the apical position is favored by the water molecule18,23–25 and hardly ever is it replaced by NO3− 30 or perchlorate ion in [Cu(Ph-Tyr)(L)(ClO4)]31 and [Cu(hista)(L-Tyr)(ClO4)]38 (hista = histamine) or ethylamine.26
In most cases, the interaction of L-tyrosine or its derivatives with copper(II) ions leads to the formation of square pyramidal complexes, what seems to be favored. However, four- or hexa- coordinated complexes, consisting of L-Tyr or its modifications, are also reported. Square planar geometry is created by L-tyrosinato O and N atom donors and N atoms of heterocyclic bases in [Cu(2,2′-bpy)(L-Tyr)]ClO4·2H2O39 and [Cu(Fc-Tyr)(L)]·ClO4 31 complexes (Fc = ferrocene, L = phen, dpq = dipyrido[3,2-d:2′,3′-f]quinoxaline, dppz, nip = 2-(naphthalene-1-yl)-1H-imidazo[4,5-f][1,10]phenanthroline). Square planar geometry around Cu2+ ions is found in the salicylidene-L-tyrosinato complex of formulae K[CuL(Ac)]·H2O40,41 and with hydroxyl dipeptide as L-seryl-L-tyrosine,42 which were synthesized as polycrystalline powder and widely physicochemically studied.
Among two six-coordinate crystalline L-tyrosinato complexes of formulae [Cu2(L-Tyr)2(NO3)(4,4′-bpy)(H2O)2]n 43and [Cu((N-pyridin-2-ylmethyl)-L-Tyr)2]·2H2O,44 the first compound is a chiral two-dimensional coordination polymer consisting of [Cu2(L-Tyr)2(NO3)(4,4′-bpy)(H2O)2]n units43 joined by bridging oxygen carboxylate atoms forming μ-(O; O′) mode.
In living organisms, the degradation of pheomelanin gives biomolecule 3-amino-L-tyrosine. Thus, it's protonation as well as the coordination behavior towards Cu(II) ions depending on pH values were performed via spectroscopic and potentiometric methods.45 [Cu(H2A)]2+ complex is formatted at pH = 3.7 in concentration ca. 30 mol dm−3 as a first species. The monomeric [Cu(HA)]+ and the dimeric [Cu2A2H]+ entities are formed as a consequential complexes cations in the lower acidity, 4.3–5.0 pH range. But, the most stable dimeric [Cu2A2] species formation starts with maximum concentration of ca. 90% found between 5.5 to 8.5 pH value. Finally, the anion [Cu(A2)]2− complex is formed at pH = 10 in low concentration.
As we presented above L-tyrosine is a very flexible ligand and its coordination behavior, especially with Cu(II) bio-ions, arouses interest. The incessant studies of L-tyrosinate anions interactions with metal ions lead to the understanding of the process of complex formation and the stability of L-tyrosinato complexes as solid state and, also, in protic and aprotic solvents. We have undertaken the study of the Cu–L-tyrosine system to understand (i) the influence of co-ligands on the physicochemical properties of the complex and (ii) the biological characteristics of the species as it must be underlined that the biological. The biological synthesis pathways of neurotransmitters, hormone of thyroid and melanin are strongly dependent on the L-tyrosine concentration in blood. The complexation of the bio-availability metal ions by this amino acid could be one of the factors decreasing of concentration of free L-tyrosine in cellular fluids.
In the procedure of synthesizing new metal–L-tyrosine complexes we applied Zn2+, Cu2+, Ni2+, Co2+ ions and biomolecules as co-ligands i.e. L-carnitine (L-car), salicylic (Sal) and acetylsalicylic (AcSal) acids, indole (Ind) and 7-azaindole (7-AzInd). Finally, we synthesized copper(II) L-tyrosinato complex as crystals of formula [Cu(L-Tyr)2(H2O)]·H2O (1) by applying various metal ions and co-ligands. In this work we structurally characterized the complex 1 by X-ray diffraction (SCXRD), spectroscopic methods as vibrational FT-IR, FT-Raman, electronic NIR-vis-UV, electron paramagnetic resonance (EPR) together with analysis of the magnetic susceptibility of the metal center interaction.
Experimental section
Chemicals, complex synthesis
All chemicals were of reagent grade and used as received. Hydrates of CuCl2·2H2O, Cu(NO3)2·3H2O, CuSO4·7H2O and L-tyrosine disodium salt hydrate, L-carnitine hydrochloride, salicylic and acetylsalicylic acids, were purchased from Sigma-Aldrich. Dimethyl sulfoxide (DMSO) and methanol (MeOH) for spectroscopy used for recording the electronic and EPR spectra were obtained from Fluka. Dichloromethane (CH2Cl2) comes from Merck.A total of 10 mL of a 0.1 M aqueous solution of Cu2+ ions was slowly mixed with 20 mL 0.1 M aqueous solution of L-carnitine hydrochloride. 10 mL of a 0.1 M aqueous solution of L-tyrosine disodium salt was added dropwisely to the blue clear mixture. 4 mL of dichloromethane was added to the obtained turquoise clear mixture and the mixture was continuously agitated. After 1 day, the resulting solution was filtered and the filtrate was left to evaporate slowly at room temperature. Navy crystals of complex 1 were obtained within 3 days. They were filtered, washed with water and one of them was X-ray characterized. Also, crystals of complex 1 crystallized from the mixture formed with molar ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 by aqueous solution of CuCl2 and methanol solution of salicylic or acetylsalicylic acids and L-tyrosine disodium salt in aqueous solution and 0.1 M concentration of each reagents. Anal. calcd for C18H24N2O8Cu (MW = 459.93): C, 47.01; H, 5.26; N, 6.09; Cu, 13.82. Found: C, 46.85; H, 5.03; N, 6.30; Cu, 13.45%.
1 by aqueous solution of CuCl2 and methanol solution of salicylic or acetylsalicylic acids and L-tyrosine disodium salt in aqueous solution and 0.1 M concentration of each reagents. Anal. calcd for C18H24N2O8Cu (MW = 459.93): C, 47.01; H, 5.26; N, 6.09; Cu, 13.82. Found: C, 46.85; H, 5.03; N, 6.30; Cu, 13.45%.
X-ray crystallographic studies
The crystal structure of [Cu(L-Tyr)2(H2O)]·H2O (1) was determined using a single-crystal X-ray diffraction. The data were collected on a Xcalibur diffractometer operating in κ-geometry and equipped with a two-dimensional CCD detector. Mo Kα radiation (0.71073 Å) was used. The data were collected in ω-scan mode with Δω = 1.0° using CrysAlis CCD program. The CrysAlis. RED software version 1.170.32 (Oxford Diffraction)46 was used for data processing. The structure was solved by direct methods and refined by the full-matrix least-squares method against F2 by means of SHELX-97 program package.47 Anisotropic displacement parameters were applied for all non-hydrogen atoms. The hydrogen atoms from the organic part, excluding those from hydroxyl groups, were generated geometrically and treated as riding atoms. The Uiso(H) were constrained to be 1.2Ueq (carrier atom). Disordered water molecules with occupancy factor of 0.5 were located from the difference Fourier map. The hydrogen atoms from disordered molecules were not identified. Powder diffraction data (XRD) were collected in a reflection mode, in the Bragg–Brentano geometry using X'Pert PRO X-ray diffraction system. Fig. S1† presents XRD patterns for [Cu(L-Tyr)2(H2O)]·H2O (1). All peaks are indexed in the unit cell obtained from the single-crystal X-ray diffraction. The differences in relative intensity for some peaks originate from texture effects. The more disturbed is the intensity of 1 0 0 peak that is much higher compare to other peaks. Obtained powders are homogenous without the traces of different phases.Physicochemical and spectroscopic studies
Elemental analysis for Cu2+ ions was performed by the ICP-AES method and that of CHN using the Kumpan method. The vibrational FT-IR spectra were taken for complex 1 and both ligands (L-tyrosine disodium salt hydrate and L-carnitine hydrochloride). FT-IR and FIR spectra over the range 4000–50 cm−1 were recorded in KBr pellets (0.5 wt% mass) or Nujoll mulls using the Perkin-Elmer FTIR-2000 spectrophotometer with resolutions of 4 cm−1 and 2 cm−1, respectively. The FT-Raman spectrum of 1 was measured on a Bruker MultiRam spectrometer equipped with a Nd:YaG laser and a liquid N2 cooled germanium detector with resolution of 2 cm−1 with co-addition of 256 scans. The NIR-vis-UV electronic spectra were obtained on a Cary 500 Scan Spectrophotometer over the range 5000–50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cm−1 with a measure step of 10 cm−1 at 293 K. Solid-state reflectance spectra were measured for 1, L-tyrosine disodium salt with identical parameters as a baseline of white reference sample. Absorbance spectra were recorded for 1 and ligand in DMSO with concentration of 9.83 × 10−4 M (1) and 1.042 × 10−2 M (L-Tyr salt), respectively. Additionally, the absorbance spectra of 1 in FBS, RPMI-1640 and RPMI-1640 supplemented with 10% PBS were collected.
000 cm−1 with a measure step of 10 cm−1 at 293 K. Solid-state reflectance spectra were measured for 1, L-tyrosine disodium salt with identical parameters as a baseline of white reference sample. Absorbance spectra were recorded for 1 and ligand in DMSO with concentration of 9.83 × 10−4 M (1) and 1.042 × 10−2 M (L-Tyr salt), respectively. Additionally, the absorbance spectra of 1 in FBS, RPMI-1640 and RPMI-1640 supplemented with 10% PBS were collected.
The spectra of complex 1 were enhanced by using the variable digital method.48–52 The method uses a single convolution of the spectral points measured at equal steps with a filter function a(n):
Fluorescence emission spectra of 1 dissolved in DMSO of 0.5, 0.25 and 0.125 mg mol−1 concentrations were carried out on LS50B Perkin Elmer spectrophotometer in the range 250–850 nm. Samples were prepared in 10 mm path length quartz cuvettes to avoid the inner-filter effect. The fluorescence intensity was measured with an excitation wavelength of 303 nm.
EPR spectra of solid state and DMSO solution of complex 1 were measured using a Bruker Elexys E 500 Spectrometer equipped with NMR teslameter (ER 036TM) and frequency counter (E 41 FRC) at X-band at room and 77 K temperatures. The experimental spectra were simulated using computer programs, DoubletExact (S = 1/2) and CuDimer (S = 1), written by Dr Andrew Ozarowski from NHMFL, University of Florida. Magnetic susceptibility of complex 1 in the temperature range from 1.7 to 300 K in a field of 500 mT and magnetization up to 5 T were measured with a Quantum Design SQUID magnetometer. The powder samples were pressed into pellets to avoid magnetic torquing. Diamagnetic correction (−243 × 10−6 emu mol−1) was calculated using Pascal's constants.
Results and discussion
L-Carnitine, salicylic and acetylsalicylic acids are used as reagents in preparation of 1 but, none of them is included in a crystal structure. They play a catalytic function. The synthesis using only Cu2+ and L-tyrosine salts gave crystals of the coordination polymer [Cu(L-Tyr)2]n described previously.20,21Crystal structure
[Cu(L-Tyr)2(H2O)]·H2O (1) crystallizes in the monoclinic P21 symmetry with relatively small unit cell accommodating two molecules of [Cu(L-Tyr)2(H2O)] complex, V = 1046.4 (2) Å3 and Z = 2. The crystal data, data collection and refinement results are presented in Table S1.† The structure of [Cu(L-Tyr)2(H2O)] complex is shown in Fig. 1. Selected bond lengths and angles are listed in Table 1. Copper atom is coordinated by chelating carboxylate oxygen atoms (O(1), O(4)) and the amine nitrogen atoms (N(1), N(2)) from two L-tyrosinate anions and oxygen atom O(1)w of coordinated water molecule. The coordination sphere is described as a square pyramid with the corners of the CuN2O2 basal plane, occupied by chelating atoms and the apical position occupied by a water molecule. The side chain aromatic ring of L-tyr is placed approximately parallel to the basal plane of square pyramid. The coordination sphere may be extended to six-coordinated, taking into account the intramolecular cation-π interaction between the aromatic ring of the coordinated L-Tyr ligand and copper atom (Cu2+⋯π distance equals 3.483(4) Å). Similar configuration of intramolecular stacking with distances 3.49 Å was found in [Cu(L-Tyr)2]n 20–22 and other L-tyrosinato, L-phenolato or tryptophano Pd(II) and Cu(II) ions complexes, which consist of a characteristically bent aromatic ring.20–22,26–31,38,53 For 1 the Cu2+⋯π distance is comparable to complexes listed above. The square planar geometry around Cu2+ is distorted, similar to the geometry around Pd2+ (ref. 53d) and Cu2+ (ref. 38 and 53b) suggesting that aromatic ring in the vicinity of the metal coordination sphere may have an electronic effect on the central metal ion. On the other hand, the reactivity of the aromatic ring may be disturbed by the metal center. Due to this weak bonding aromatic groups of amino acids in proteins may act as important binding sites for the copper ion. It has been shown that L-tyrosine side chain is coordinated to the Cu2+ ion in the active site of the enzyme occupying an axial position in the square-pyramidal geometry around copper center.53g,h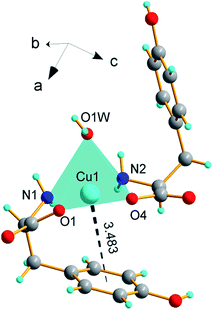 | ||
| Fig. 1 The structure of [Cu(L-Tyr)2(H2O)] complex, dashed line stands for Cu(II)⋯π interaction with distance of 3.483(4) Å. | ||
| Distances | |||
| Cu1–O4 | 1.946(4) | Cu1–N2 | 1.979(5) |
| Cu1–O1 | 1.972(4) | Cu1–N1 | 1.987(5) |
| Cu1–O1W | 2.347(4) | ||
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||
| Angles | |||
| O4–Cu1–O1 | 94.1(2) | N2–Cu1–O1W | 85.9(2) |
| O4–Cu1–N2 | 83.5(2) | N1–Cu1–O1W | 99.8(2) |
| O1–Cu1–N2 | 177.4(2) | C2–O1–Cu1 | 113.0(4) |
| O4–Cu1–N1 | 170.6(2) | C10–O4–Cu1 | 116.9(4) |
| N2–Cu1–N1 | 83.1(2) | C1–N1–Cu1 | 107.6(4) |
| N2–Cu1–N1 | 99.1(2) | C11–N2–Cu1 | 111.5(4) |
| O4–Cu1–O1W | 89.4(2) | C2–C1–C3 | 110.9(5) |
| O1–Cu1–O1W | 95.3(2) | C13–C12–C11 | 116(5) |
The Cu–Namine bonds (N(1)–Cu 1.99(1), N(2)–Cu 1.98(1) Å) are comparable with Cu–Ocarboxylate distances (Cu–O(1) 1.97(1) Cu–O(4) 1.95(1) Å) and are quite similar to those found in [Cu(L-Tyr)2]n and other Cu(II) complexes.18,20–22,53i The distance from Cu(II) to apical O(1)water oxygen is longer 2.347(4) Å but within the range of 2.2–2.9 Å known for the axial Cu–O bond lengths.54 The angular parameter of the degree of trigonality defined as τ = (β − α)/60;55 where α = N(1)–Cu(1)–O(4) (170, 6(2) deg), β = N(2)–Cu(1)–O(1) (177, 4(2) deg.), equals 0.113, and evidences a small distortion of the basal plane. The values of τ parameter, for all five-coordinated copper(II) L-tyrosinato and its derivative complexes deposited in the Cambridge Structural Database (CSD),56 are collected in Table 2. In addition, it is noteworthy that this amino-acid forms five-coordinate complexes with only square planar geometry around Cu(II) ions. The obtained value 0.113 for 1 is nestled between 0.125 and 0.0813 calculated for [Cu(Ph-Tyr)(phen)(ClO4)]31 and [Cu(IP)(L-Tyr)(H2O)]ClO4·H2O,28 respectively (Table 2).
| Complex | Chromophore | Angles: α; β | τ = (β − α)/60 (ref. 55) | |
|---|---|---|---|---|
| a L-Tyr = L-tyrosine; 2,2′-bpy = 2,2′-bipyridine; phen = 1,10-phenanthroline; 5,6-dmp = 5,6-dimethyl-1,10-phenanthroline; IP = imidazol[4,5-f][1,10] phenanthroline; L-Ityr = 3-iodo-L-tyrosine; hista = histamine; I2tyr = 3,5-diiodo-L-tyrosine; L-Tyr-hist = L-tyrosyl-L-histidine; Ph-Tyr = L-phenylalanine-L-tyrosine. | ||||
| 1. | [Cu(2,2′-bpy)(L-Tyr)(H2O)]Cl·3H2O23 | N2NOO′ | 174.0; 151.8 | 0.370 |
| 2. | [Cu(2,2′-bpy)(I2tyrOH)(NO3)]·CH3OH30 | N2NOO′ | 174.8; 156.3 | 0.308 |
| 3. | [Cu(2,2′-bpy)(I2tyrO−)(H2O)]·2H2O30 | N2NOO′ | 177.1; 160.3 | 0.280 |
| 4. | [Cu(2,2′-bpy)(L-Ityr)(H2O)]NO3·CH3OH·H2O29 | N2NOO′ | 173.7; 159.9 | 0.230 |
| 5. | [Cu(L-Tyr)(phen)(H2O)]ClO4·2.5H2O24 | N2NOO′ | Cu(1) 173.7; 161.8 | 0.198 |
| Cu(2) 170.8; 160.3 | 0.175 | |||
| 6. | [Cu(L-Tyr)2]n 18,20,21 | N2O3 | 179.2; 167.5 20 | 0.195 |
| 179.32; 167.75 21 | 0.193 | |||
| 179.3; 168.018 | 0.188 | |||
| 7. | [Cu(hista)(L-I2tyrO−)(H2O)]2·2H2O29 | N2NOO′ | Cu(2) 173.6; 163.6 | 0.166 |
| Cu(1) 165.9; 164.6 | 0.022 | |||
| 8. | [Cu(Ph-Tyr)(phen)(ClO4)]31 | N2NOO′ | 176.08; 168.56 | 0.125 |
| 9. | [Cu(L-Tyr)2(H2O)]·H2O (this work) | N2O2O′ | 170.6; 177.4 | 0.113 |
| 10. | [Cu(IP)(L-Tyr)(H2O)]ClO4·H2O28 | N2NOO′ | 170.63; 165.74 | 0.081 |
| 11. | [Cu(L-Tyr)(5,6-dmp)(H2O)]ClO4 27 | N2NOO′ | Cu(2) 166.62; 163.95 | 0.044 |
| Cu(1) 166.19; 164.30 | 0.032 | |||
| 12. | [Cu(L-Tyr-hist)]32 | N3O2 | 177.0; 174.8 | 0.037 |
| 13. | [Cu(hista)(L-Tyr)(ClO4)]31 | N2NOO′ | 170.1; 168.6 | 0.025 |
| D–H⋯A | d(D–H)/Å | d(H⋯A)/Å | D(D⋯A)/Å | <DHA/deg |
|---|---|---|---|---|
| a Symmetry transformations used to generate the equivalent atoms: (i) −x + 1, y + 1/2, −z + 2; (ii) x − 1, y, z; (iii) −x + 1, y − 1/2, −z + 1; (iv) −x, y + 1/2, −z + 1; (v) −x + 1, y + 1/2, −z + 1; (vi) x, y + 1, z. | ||||
| O3–H31⋯O5i | 0.98(2) | 1.85(3) | 2.713(6) | 145(5) |
| O6–H61⋯O2ii | 0.90(2) | 1.82(3) | 2.692(7) | 161(5) |
| O1W–H1W⋯O2iii | 0.96 | 1.84 | 2.795(6) | 178 |
| O1W–H2W⋯O6iv | 0.89 | 1.91 | 2.804(7) | 180 |
| N1–H1A⋯O2v | 0.90 | 2.25 | 3.117(6) | 161 |
| N1–H1B⋯O4vi | 0.90 | 2.10 | 2.961(6) | 159 |
| N2–H2A⋯O5vi | 0.90 | 2.02 | 2.904(7) | 167 |
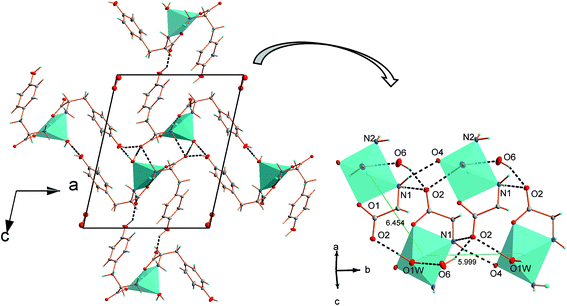
The asymmetric unit contains one symmetrically independent molecule of the complex and water disordered over two positions with probability 0.5. Crystal packing is presented in Fig. 2. The complete list of hydrogen bond interactions is presented in Table 3. Centers separated by the distance of 6.454 Å are joined by N(1)–H⋯O(2)v and O(1)w–H⋯O(2)iii interactions. Additionally, O(1)w atom of coordinated water is a donor atom in O(1)w–H⋯O(6)iv bond to hydroxyl oxygen atom. Both phenolate oxygen atoms O(6) and O(3) act as donors and are involved in hydrogen bonds with uncoordinated carboxylate O(5) and O(2) atoms. Hydroxyl oxygen O(6) atom is a donor in O(6)–H⋯O(2)ii interaction with uncoordinated O(2)ii. Whereas, hydroxyl oxygen O(3) atom from L-Tyr interacts via the O(3)–H⋯O(5)i bond with the uncoordinated oxygen atom O5 from the carboxylate group of second L-Tyr anion joining two distant centers.
| Compound | Topology | d(Cu⋯Cu) (Å) | Hb (Å) | Jc (cm−1) |
|---|---|---|---|---|
| a L1 = 6-amino-1,3-dimethyl-5-((2-carboxyphenyl)azo)uracil; H4egta = 3,12-bis(carboxymethyl)-6,9-dioxa-3,12-diazatetradecanedioic acid; L2 = 2-aminomethylbenzimidazole; L3 = L-isoleucinate; L4 = 8-amino-4-methyl-5-azaoct-3-en-2-one.b The distance between the adjacent basal (equatorial) planes.c The J values were brought to the −JSiSj convention.d Heisenberg dimer.e Ising chain. | ||||
| [CuL1py]2·0.5H2O79a | 4-coord, Td/D4h | 3.993 | −6.5 | |
| [{Cu(en)}2(μ-egta)]·4H2O79b | Equatorial-axial | 5.781 | 1.521 | −1.35 |
| [Cu(L2)(Cl)(L3)]·H2O79c | Basal–basal | 5.776 | 2.381 | +0.21d |
| +0.99e | ||||
| [Cu(L-Tyr)2(H2O)]·H2O (this work) | Basal–basal | 5.999 | 1.024 | +0.30 |
| [CuL4(N3)]2 (ref. 79d) | Basal–basal | 5.700 | 2.104 | +1.02 |
As it is seen in Table 3, carboxylate oxygen atoms act as acceptors in hydrogen bonds formation, O(2) is engaged in three bonds, O(5) in two and O(4) only in one hydrogen bond, whereas, carboxylate O(1) atom does not form any hydrogen interactions. Some hydrogen bonds may also be present between disordered water molecules since the closest O(2)⋯O(2)i distance is equal to 3.06 Å.
Due to several hydrogen bond interactions complex polymorphic structures are formed. Neighboring Cu(II) centers with Cu2+⋯Cu2+ distance equal to 5.998(1) Å are directly connected by N(1)–H⋯O(4)vi hydrogen bonds; donor and acceptor atoms are coordinated to two adjacent centers and together with N(2)–H⋯O(5)vi interactions form tapes of [Cu(L-Tyr)2(H2O)] expanding along b direction, see Fig. 3. It must be emphasized that O(4) and O(5) atoms are from one molecule of amino acid. In the crystal structure of [Cu(L-Tyr)2]n (ref. 21 and 22) hydrogen bonds of similar strength are formed by N(1A)–H(1A1)⋯O(2A) and N(1B)–H(1B2)⋯O(2B) and involve oxygen atoms. However, the acceptors of hydrogen are from two independent L-Tyr anions. The donor to acceptor distances are slightly shorter for 1 compare to distances found for [Cu(L-Tyr)2]n (2.961 Å and 2.904 Å for 1 and 3.072(2) Å and 3.080(3) Å for [Cu(L-Tyr)2]n).21,22
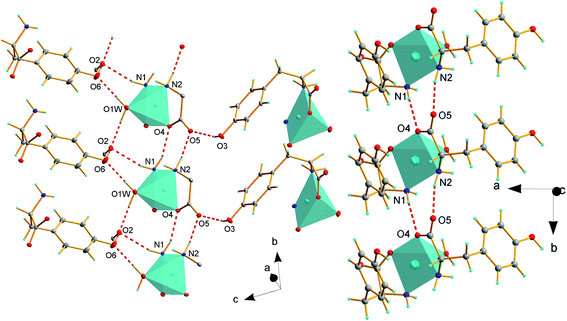 | ||
| Fig. 3 Hydrogen bond interactions in [Cu(L-Tyr)2(H2O)]·H2O, part of the ligands are omitted for picture clarity. | ||
The chains based on the N(1)–H⋯O(4)vi and N(2)–H⋯O(5)vi hydrogen bonds propagate along b axis and join copper centers laying in the distance of 5.998(1) Å. The 5.998(1) Å and 6.454(1) Å distances are the shortest possible immediate contacts between metal centres and it is highly probable that hydrogen bonded chains are pathways for magnetic interactions (vide infra). The other distances between Cu(II) ions are definitely longer and equal to 9.936(1) Å and 11.967(1) Å and should be definitely rejected as pathways of magnetic exchange (Fig. 4).
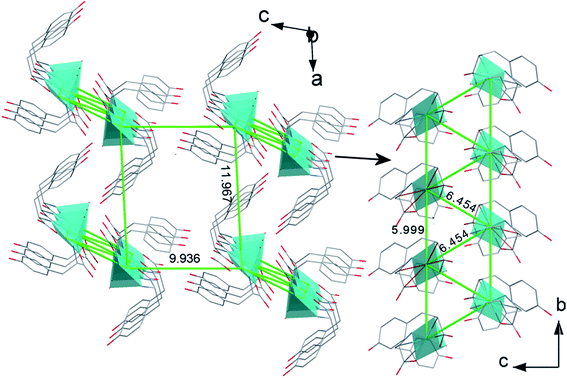 | ||
| Fig. 4 The projection of the crystal structure of [Cu(L-Tyr)2(H2O)]·H2O with marked distances between copper centers. | ||
FT-IR vibrational and FT-Raman spectra
The spectrum of 1 exhibits a well seen band at 3482 cm−1. This band is not found in FT-IR spectrum for [Cu(L-Tyr)2]n complex.21 Therefore, it should be attributed to the OH stretching vibrations and confirm the presence of water molecules in the crystal structure of 1. Whereas, the OH stretching modes of phenolate rings generate a band found at 3372 cm−1 in a spectrum of 1. In the spectral range 3400–2700 cm−1 several modes of νas, νs NH, ν CH and intramolecular O–H⋯N hydrogen bond between COOH and NH2 groups vibrations as overlapped bands are observed.57,58 The νas and νsym (NH2) vibrations generate bands at 3284 cm−1 and 3264 are slightly downshifted in comparison with 3310 and 3295 cm−1 found for [Cu(L-Tyr)2]n.21 The Raman spectra of 1 exhibits bands at 3065, 3042, 3019 and 2967, 2948 and 2928 cm−1.In spectrum 1 regions 1700–1540 cm−1 and 1470–1350 cm−1 are the most interesting from the coordination perspective. In these spectral ranges the bands assigned as νas and νsym vibrations of COO− groups arise. Strong and broad absorption found at 1700–1540 cm−1 consist of 1653, 1620, 1609, 1602 and 1585 cm−1 overlapping bands of νas C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, scissoring NH2 vibrations and, also, C–C vibrations of aromatic L-tyrosinato residues.57,59 In Raman spectra two strong bands were found at 1615 and 1591 cm−1. The ν C–C and bending HC–CC vibrations in ring generate the very strong band at 1502, 1513 and 1515 for L-tyr salt, [Cu(L-Tyr)2]n and complex 1, respectively.57,60 One very weak band has been found at 1513 cm−1 in Raman spectra. The subsequent range 1470–1300 cm−1 presents weakly bands at 1456, 1444, 1406, 1380, 1352, 1329, 1311 cm−1, which have their equivalents in reported previously spectra for [Cu(L-Tyr)2]n (Fig. S1†).21 For both complexes the observed modes between 1470 cm−1 and 1375 cm−1 are the results of splitting band at ca. 1430 cm−1 found for pure L-tyrosine salt and arise by stretching C–C and CO and bending HCC, HCH, HCN vibrations. The spectrum of 1 exhibits four bands, whereas six bands are found for [Cu(L-Tyr)2]n. Especially, the differences in the number and positions of the bands are the results of carboxylate monodentatelly and bridging coordinated carboxylate group in 1 and [Cu(L-Tyr)2]n, respectively. The involvement of oxygen atoms in several hydrogen bonds is an additional factor, what explains the differences in the spectra of these compounds. Whereas, bending HCN, HNC, HC–Cring and HO–Cring and ν C–Cring vibrations appear as weak bands found at 1365, 1352, 1329 cm−1. It stays in a good correlation with quantum chemical calculations for conformers of L-tyrosine.57 The spectral range 1290–1210 cm−1 shows two very intensive and one weak bands at 1270, 1247 and 1212 cm−1, which correspond to the band at 1271, 1242 and 1213 cm−1 observed in Raman spectrum. The energy values of those modes are reduced to ca. 10 cm−1 to lower energy in comparison with L-Tyr salt (Fig. S2†). The bands are the result of several modes combination i.e. ν (CC), τ(HCCC), ϕ (HCC), ν (OC) and ϕ (HNC).57
O, scissoring NH2 vibrations and, also, C–C vibrations of aromatic L-tyrosinato residues.57,59 In Raman spectra two strong bands were found at 1615 and 1591 cm−1. The ν C–C and bending HC–CC vibrations in ring generate the very strong band at 1502, 1513 and 1515 for L-tyr salt, [Cu(L-Tyr)2]n and complex 1, respectively.57,60 One very weak band has been found at 1513 cm−1 in Raman spectra. The subsequent range 1470–1300 cm−1 presents weakly bands at 1456, 1444, 1406, 1380, 1352, 1329, 1311 cm−1, which have their equivalents in reported previously spectra for [Cu(L-Tyr)2]n (Fig. S1†).21 For both complexes the observed modes between 1470 cm−1 and 1375 cm−1 are the results of splitting band at ca. 1430 cm−1 found for pure L-tyrosine salt and arise by stretching C–C and CO and bending HCC, HCH, HCN vibrations. The spectrum of 1 exhibits four bands, whereas six bands are found for [Cu(L-Tyr)2]n. Especially, the differences in the number and positions of the bands are the results of carboxylate monodentatelly and bridging coordinated carboxylate group in 1 and [Cu(L-Tyr)2]n, respectively. The involvement of oxygen atoms in several hydrogen bonds is an additional factor, what explains the differences in the spectra of these compounds. Whereas, bending HCN, HNC, HC–Cring and HO–Cring and ν C–Cring vibrations appear as weak bands found at 1365, 1352, 1329 cm−1. It stays in a good correlation with quantum chemical calculations for conformers of L-tyrosine.57 The spectral range 1290–1210 cm−1 shows two very intensive and one weak bands at 1270, 1247 and 1212 cm−1, which correspond to the band at 1271, 1242 and 1213 cm−1 observed in Raman spectrum. The energy values of those modes are reduced to ca. 10 cm−1 to lower energy in comparison with L-Tyr salt (Fig. S2†). The bands are the result of several modes combination i.e. ν (CC), τ(HCCC), ϕ (HCC), ν (OC) and ϕ (HNC).57
NIR-vis-UV electronic spectroscopy and fluorescence spectra
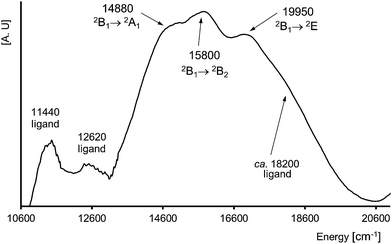
The N and O atoms of L-tyrosine molecules and water O atom create five-coordinated environment around Cu(II) ion (d9 system) and [CuN2O2O′] chromophore. The value of τ parameter 0.113 suggests the very small distortion towards trigonal bipyramid (TB). The single, broad and almost symmetrical band found with maximum at 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 cm−1 is characteristic for square pyramidal (SP) geometry of the crystal field around copper(II) centre (Fig. 5). It is attributable for the d–d transition of the distorted SP and it is very convergent to the spectra for SP environment found for [Cu(L-Arg)2(H2O)]2·(pma)·3H2O (L-Arg = L-arginine, pma4− = pyromellitate)61 and [Cu(L-Tyr)2]n.21 In 1 the distortion is a little bit smaller than in [Cu(L-Arg)2H2O]2+ cation complex (0.1635 (CuA); 0.1483 (CuB)) as well as [Cu(L-Tyr)2]n (0.19) and the maximum is red-shifted ca. 1100 cm−1 for 1. In SP crystal field the one-electron ground state configuration follows as dx2–y2 > dz2 > dxy > dxz ≈ dyz,62 what indicates three d–d electronic transitions. The filtration of 10
700 cm−1 is characteristic for square pyramidal (SP) geometry of the crystal field around copper(II) centre (Fig. 5). It is attributable for the d–d transition of the distorted SP and it is very convergent to the spectra for SP environment found for [Cu(L-Arg)2(H2O)]2·(pma)·3H2O (L-Arg = L-arginine, pma4− = pyromellitate)61 and [Cu(L-Tyr)2]n.21 In 1 the distortion is a little bit smaller than in [Cu(L-Arg)2H2O]2+ cation complex (0.1635 (CuA); 0.1483 (CuB)) as well as [Cu(L-Tyr)2]n (0.19) and the maximum is red-shifted ca. 1100 cm−1 for 1. In SP crystal field the one-electron ground state configuration follows as dx2–y2 > dz2 > dxy > dxz ≈ dyz,62 what indicates three d–d electronic transitions. The filtration of 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000–20
000–20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cm−1 spectral range discloses five bands with energies 11
000 cm−1 spectral range discloses five bands with energies 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 440, 12
440, 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 620, 14
620, 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 880, 15
880, 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 and 19
800 and 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 950 cm−1 (Fig. 6). The first two of them are associated with L-tyrosine overtones (Fig. 5 and 6), whereas, subsequent should be correlated with crystal field transitions. The single d–d band consists of three isolated oneself components. The spectrum is interpreted according to C4v symmetry and those components are ascribed to the transition 2B1(dx2–y2) → 2A1 (dz2), 2B1 → 2B2 (dxy) and 2B1 → 2E (dxz ≈ dyz) (Fig. 6).62–65 As in the case of the reflectance spectrum, the absorbance spectra exhibit intensive, symmetrical single band at ca. 16
950 cm−1 (Fig. 6). The first two of them are associated with L-tyrosine overtones (Fig. 5 and 6), whereas, subsequent should be correlated with crystal field transitions. The single d–d band consists of three isolated oneself components. The spectrum is interpreted according to C4v symmetry and those components are ascribed to the transition 2B1(dx2–y2) → 2A1 (dz2), 2B1 → 2B2 (dxy) and 2B1 → 2E (dxz ≈ dyz) (Fig. 6).62–65 As in the case of the reflectance spectrum, the absorbance spectra exhibit intensive, symmetrical single band at ca. 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 cm−1 (Fig. S3†). This principal absorption with value of ε = 80 L3 mol−1 cm−1 is characteristic for five-coordinated Cu(II) ion complexes as well as for other L-tyrosinato copper(II) spectra recorded in solvents i.e. DMF–Tris–HCl buffer,31 aqueous,24 and also for two other aromatic aminoacids as L-phenylalanine and L-tryptophane.66,67 The spectrum of 1 recorded after 5 months preserves its shape, maximum and intensity. So, complex 1 is stable over time and coordination sphere is unchanged by the DMSO molecules. Preserving the pyramidal geometry around Cu(II) ion in DMSO solution is one of the most important observation in the context of biological research.
400 cm−1 (Fig. S3†). This principal absorption with value of ε = 80 L3 mol−1 cm−1 is characteristic for five-coordinated Cu(II) ion complexes as well as for other L-tyrosinato copper(II) spectra recorded in solvents i.e. DMF–Tris–HCl buffer,31 aqueous,24 and also for two other aromatic aminoacids as L-phenylalanine and L-tryptophane.66,67 The spectrum of 1 recorded after 5 months preserves its shape, maximum and intensity. So, complex 1 is stable over time and coordination sphere is unchanged by the DMSO molecules. Preserving the pyramidal geometry around Cu(II) ion in DMSO solution is one of the most important observation in the context of biological research.
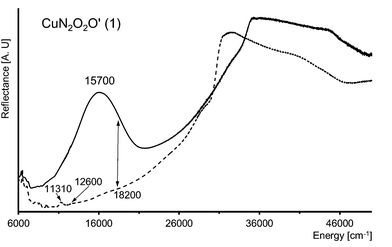 | ||
| Fig. 5 Polycrystalline electronic reflectance spectrum of 1 (–) and pure L-tyrosine disodium salt (- - -). | ||
 | ||
| Fig. 6 The effect of filtration process of reflectance spectrum for 1 (visible region) (step = 20 cm−1, α = 200 and N = 30). | ||
The fluorescence emission spectra show the emission wavelength at 14![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900 and 16
900 and 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 cm−1. The highly sensitive and selective peak at 16
400 cm−1. The highly sensitive and selective peak at 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 cm−1 (620 nm) is characteristic for L-Tyr68,69 (Fig. S4†)).
400 cm−1 (620 nm) is characteristic for L-Tyr68,69 (Fig. S4†)).
EPR spectra
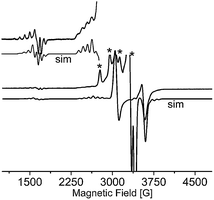
The EPR spectra of 1 were recorded at X-band on powder sample at 295 and 77 K (Fig. 7) and also on glassy DMSO solutions at 77 K (Fig. 8). The solid-state samples clearly indicate an axial S = 1/2 species spectrum. Diagonal components of g tensor, g‖ = 2.236 and g⊥ = 2.063, obtained by simulation of the solid state spectrum, adopt the relation g‖(z) > g⊥(x,y) corresponding to dx2–y2 orbital of unpaired electron ground state, in agreement with the square-pyramidal geometry of Cu(II) complex. The EPR spectral parameters are very close to those obtained on the basis of monocrystal EPR measurements of [Cu(L-Tyr)2]n (g‖ = 2.2414 and g⊥ = 2.0584).22 The EPR parameters of the monomeric species observed in the DMSO frozen solution g‖ = 2.250, g⊥ = 2.055, and A‖ = 180 G, are typical for N2O2 donor set from two bidentate L-tyrosine ligands coordinated in Cu(II) xy plane. Surprisingly, the same spectrum of the DMSO frozen solution exhibits the lines associated with S = 1 species. It is clearly demonstrated by the presence of forbidden (ΔMS = ±2) resonance transition at half-field, displaying a 7-line splitting structure from hyperfine interaction with two copper nuclei (I(Cu) = 3/2) within the dimeric unit. The hyperfine coupling parameter, A‖ = 85 G, is equal to half the value which characterizes the monomeric Cu(II) complex. The same splitting is observed at allowed low field ΔMS = ±1 transition superimposed on the lines of dominant monomeric species. The simulation of EPR spectrum using S = 1 spin Hamiltonian gave |D| = 0.0468 cm−1, g‖ = 2.238, g⊥ = 2.06 and A‖ = 85 G what suggests that Cu(II) pair is involved in a very week exchange coupling.70 According to the crystal structure data, there are two oxygens, phenolic and carboxylic, which are not coordinated to Cu(II) ion and may participate in the bridging of Cu(II) ions at long distance. The Yamauchi and co-workers reported that the copper(II) complexes of Tyr-containing dipeptides dimerized through the phenoxide bridge at pH 8–11.70a,b Assuming dipole–dipole nature of the obtained D parameter the Cu⋯Cu distance may be estimated70c as close to 4 Å.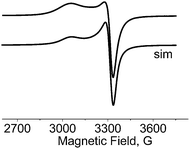 | ||
| Fig. 7 X-band EPR spectra of powdered complex 1 at 77 K simulated using the parameters given in the text. | ||
Magnetic measurements
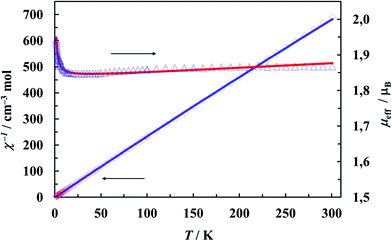
The magnetic susceptibility of 1 (Fig. 9) shows the Curie–Weiss behavior virtually in the whole temperature range with a Weiss constant θ = −0.40 K and a magnetic moment of 1.87μB. The effective magnetic moment is constant at 12–300 K (1.85–1.87μB) and below 12 K increases to 1.94μB at 1.72 K. The negative value of θ usually indicates an antiferromagnetic exchange coupling, but it is important to note that after subtracting temperature independent paramagnetism from the raw experimental data, the parameter changes the sign (see further discussion).The increase of the effective magnetic moment at low temperatures suggests the existence of weak intermolecular ferromagnetic interactions. Taking into account the structural features, structures with two competing interactions cannot be excluded. Although, there is no direct metal–metal bridges, the copper ions are connected through the elaborate system of hydrogen bonds (Fig. 2 and 3). They provide potential magnetic superexchange pathways, often quite effective in copper complexes71 (Fig. 4). Looking for possible magnetic interactions we find Cu(II) double chains running along the b axis. The ions in adjacent chains are shifted from one another by half the shortest Cu⋯Cu distance. Such an arrangement of magnetic ions is called a double triangular chain72 or, in case of antiferromagnetic interactions, a frustrated double chain.73 [Cu(L-Tyr)2(H2O)] units in the chains are linked through the double N–H⋯O hydrogen-bonds involving both the coordinated and free carboxylate-oxygen atoms. Possible interactions transmitted in this way should not be very weak because: (i) N–H⋯O bonds came under the strongest hydrogen bonds,74 (ii) the atoms involved in the bonds (directly or indirectly) belong to the basal planes of the coordination polyhedra. The adjacent planes N1N2O1O4 are strictly parallel, but shifted from each other by 1.024 Å. Each copper ion is linked to two ions in the adjacent chain by single hydrogen bonds O1W–H1W⋯O2. These contacts are probably very weak, attaching the uncoordinated oxygen atom of the carboxylate group with the apical water molecule in the apical-basal mode.
The magnetic susceptibility data were fitted using a high-temperature series expansion (HTSE) derived from the one-dimensional Heisenberg model for S = 1/2, based on the Padé approximant technique:75,76
| χ = (Ng2β2/4kT)[N/D]2/3 + Nα |
Since it is not possible to calculate field dependent magnetization within the above model, we have tried to ignore very weak exchange interactions, treating the system as a set of isolated Cu(II) ions and leaving the rest of the parameters (g, Nα) unchanged. The M(B) dependence calculated for various external fields is drawn in Fig. 10 as a solid line and fits the experimental data pretty well.
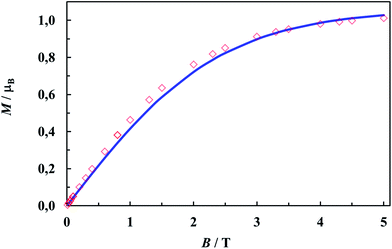 | ||
| Fig. 10 Magnetization as a function of magnetic induction at 1.72 K. The line is the Brillouin function calculated for S = 1/2, g = 2.1207 (EPR average) and Nα = 61 × 10−6 emu mol−1. | ||
Magnetic interactions transmitted via O–H⋯O hydrogen bonds have been detected in dozens of compounds, some magnetostructural relationships were also found.71,78 Much less common are the arrangements with nitrogen donor atoms. Table 4 compiles the Cu(II) compounds with the N–H⋯O bridging mode, where the exchange integrals have been determined from the magnetization data.79 No clear magnetostructural trends have been observed (also among the geometric parameters not listed in the Table) but it is worth to mention that the basal–basal type configuration of the CuN2O2O′ coordination polyhedra favors ferromagnetic interactions. This is rather unexpected, especially in light of the results obtained previously for the compounds with O–H⋯O hydrogen bonds,71,80 where “a such condition has been shown to be crucial for the system to exhibit antiferromagnetic interactions”.80b
Conclusion
In summary, we have presented a five-coordinated copper(II) L-tyrosinato complex, [Cu(L-Tyr)2(H2O)]·H2O (1), and characterization of its crystal structure, spectroscopic as well as magnetic properties. The L-tyrosinate anions chelate copper(II) ions and together with water molecules form square pyramidal (SP) coordination around metal centres. The monomer [Cu(L-Tyr)2(H2O)] complexes are directly connected by N(1)–H⋯O(4)vi and N(2)–H⋯O(5)vi hydrogen bonds. These interactions form tapes of [Cu(L-tyr)2(H2O)] in which joined Cu2+ centers are distanced of 5.998(1) Å. The chain based on N(1)–H⋯O(4)vi and N(2)–H⋯O(5)vi hydrogen bonds establishes a magnetic superexchange pathway of a weak ferromagnetic interaction between neighboring copper(II) ions. The EPR parameters of the monomeric species observed in the DMSO frozen solution are typical for N2O2 donor set. Also, in the DMSO frozen solution the 7-line splitting structure from hyperfine interaction with two copper nuclei (I(Cu) = 3/2) within the dimeric unit were found. In the reflectance spectrum, the single d–d band is composed by 2B1(dx2–y2) → 2A1 (dz2), 2B1 → 2B2 (dxy) and 2B1 → 2E (dxz ≈ dyz) transition with the ground state 2B1(dx2–y2). The SP copper(II) geometry has been preserved in DMSO solution.Acknowledgements
The work was financed by a statutory activity subsidy from the Polish Ministry of Science and Higher Education of the Republic of Poland for the Faculty of Chemistry, Wrocław University of Technology and for Faculty of Chemistry, University of Wrocław. The fluorescence spectra were measured at Chemistry Department, University of California, Berkeley, during the A.W. internship of TOP-500 Innovators Poland Program.References
- S. Meyers, Alternative Med. Rev., 2000, 5, 64 CAS.
- E.-J. Lee and P. J. Facchini, Plant Physiol., 2011, 1067 CrossRef CAS PubMed.
- (a) A. Nakagawa, H. Minami, J.-S. Kim, T. Koyanagi, T. Katyama, F. Sato and H. Kumagai, Nat. Commun., 2011, 2:326, 1 Search PubMed; (b) A. Nakagawa, H. Minami, J.-S. Kim, T. Koyanagi, T. Katayama, F. Sato and H. Kumagai, Nat. Commun., 2012, 3:819, 9 Search PubMed.
- P. Kovacic and R. Somanathan, Oxid. Med. Cell. Longevity, 2009, 2, 181 CrossRef PubMed.
- A. Slominski, M. A. Zmijewski and J. Pawelek, Pigm. Cell Melanoma Res., 2012, 25, 14 CrossRef CAS PubMed.
- S.-Q. Li and N.-H. Hu, Acta Crystallogr., Sect. E: Struct. Rep. Online, 2011, 67, m884 CAS.
- S. Zhang and N.-H. Hu, Acta Crystallogr., Sect. C: Cryst. Struct. Commun., 2009, 65, m7 CAS.
- D. U. Miodragovic, G. A. Bogdanovic, S. M. Milosavljevic, M. J. Malinar, M. B. Celap, A. Spasojevic-de Bire, S. Macura and N. Juranic, Enantiomer, 2001, 6, 299 CAS.
- R. Dreos, G. Nardin, L. Randaccio, P. Siega and G. Tauzher, Inorg. Chem., 2004, 43, 3433 CrossRef CAS PubMed.
- E. Gao, H.-Y. Li and Q.-T. Liu, Acta Chim. Sin., 2005, 63, 1225 CAS.
- D.-Q. Li, J. Zhou and X. Liu, Acta Crystallogr., Sect. C: Cryst. Struct. Commun., 2007, 63, m371 CAS.
- P. Kumar, A. K. Singh, J. K. Saxena and D. S. Pandey, J. Organomet. Chem., 2009, 694, 3570 CrossRef CAS PubMed.
- M. S. Refat, S. A. El-Korashy and A. S. Ahmed, J. Mol. Struct., 2008, 881, 28 CrossRef CAS PubMed.
- G. A. Thakur and M. M. Shaikih, Acta Poloniae Pharmaceutica Drug Research, 2006, 63, 95 CAS.
- Md. R. Islam, S. M. R. Islam, A. S. M. Noman, J. A. Khanam, S. M. M. Ali, S. Alam and M. Lee, Mycobiology, 2007, 35, 25 CrossRef CAS PubMed.
- D. Van der Helm and W. A. Franks, J. Am. Chem. Soc., 1968, 90, 5627 CrossRef CAS.
- W. A. Franks and D. Van der Helm, Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem., 1970, 27, 1299 CrossRef.
- D. Van der Helm and C. E. Tatsch, Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem., 1972, 28, 2307 CrossRef CAS.
- C. A. McAuliffe and S. G. Murray, Inorg. Chim. Acta, 1973, 7, 171 CrossRef CAS.
- J. Weng, M. Hong, Q. Shi, R. Cao and A. S. C. Chan, Eur. J. Inorg. Chem., 2002, 2553 CrossRef CAS.
- A. Wojciechowska, M. Daszkiewicz and A. Bienko, Polyhedron, 2009, 28, 1481 CrossRef CAS PubMed.
- V. Padares-Garcia, R. C. Santana, R. Madrid, B. Baldo, A. Vega and E. Spodine, J. Inorg. Biochem., 2012, 114, 75 CrossRef PubMed.
- X. Solans, L. Ruiz-Ramirez, A. Martinez, L. Gasque and R. Moreno-Esparza, Acta Crystallogr., Sect. C: Cryst. Struct. Commun., 1992, 48, 1785 CrossRef.
- T. Sugimori, H. Masuda, N. Ohata, K. Koiwai, A. Odani and O. Yamauchi, Inorg. Chem., 1997, 36, 576 CrossRef CAS.
- P. R. Reddy and P. Manjula, Chem. Biodiversity, 2009, 6, 71 CAS.
- D. Kannan and M. N. Arumugham, International Journal of Inorganic and Bioinorganic Chemistry, 2012, 2, 50 Search PubMed.
- S. Ramakrishna, V. Rajendiran, M. Palaniandavar, V. S. Periasamy, B. S. Srinag, H. Krishnamurthy and M. A. Akbarsha, Inorg. Chem., 2009, 48, 1309 CrossRef PubMed.
- G. Qin, L. Xue-Yi, L. Qing-Bin, L. Sheng-Rong, M. Xue-Dan and F. Xiao-Long, Chin. J. Chem., 2007, 25, 791 CrossRef.
- F. Zhang, T. Yajima, H. Masuda, A. Odani and O. Yamauchi, Inorg. Chem., 1997, 36, 5777 CrossRef CAS PubMed.
- F. Zhang, A. Odani, H. Masuda and O. Yamauchi, Inorg. Chem., 1996, 35, 7148 CrossRef CAS PubMed.
- T. K. Goswami, S. Gadadhar, A. A. Karande and A. R. Chakravarty, Polyhedron, 2013, 52, 1287 CrossRef CAS PubMed.
- H. Masuda, A. Odani and O. Yamauchi, Inorg. Chem., 1989, 28, 624 CrossRef CAS.
- D. Van der Helm, S. E. Ealick and J. Burks, Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem., 1975, 31, 1013 CrossRef.
- A. Mosset and J. J. Bonnet, Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem., 1977, 33, 2807 CrossRef.
- T. Yajima, M. Okajima, A. Odani and O. Yamauchi, Inorg. Chim. Acta, 2002, 339, 445 CrossRef CAS.
- I. I. Mathews and H. Manohar, Polyhedron, 1991, 10, 2163 CrossRef.
- R. Hamalainen, M. Ahlgren, U. Turpeinen and M. Rantala, Acta Chem. Scand., Ser. A, 1978, 32, 235 CrossRef PubMed.
- O. Yamauchi, T. Kohzuma, H. Masuda, K. Toriumi and K. Sato, Inorg. Chem., 1989, 28, 4066 CrossRef CAS.
- O. Yamauchi, A. Odani and H. Masuda, Inorg. Chim. Acta, 1992, 198, 749 CrossRef.
- W. Shi, D. Chen, G. Wang and Y. Xu, Appl. Magn. Reson., 2001, 20, 289 CrossRef CAS.
- W.-L. Shi and D.-Y. Chen, Acta Phys.-Chim. Sin., 2001, 17, 181 CAS.
- T. Kolev, B. B. Koleva and M. Spiteller, J. Coord. Chem., 2008, 61, 1897 CrossRef CAS.
- S. Zhang and N.-H. Hu, Acta Crystallogr., Sect. C: Cryst. Struct. Commun., 2009, 65, m7 CAS.
- X.-F. Li and R. Cao, Chin. J. Struct. Chem., 2009, 28, 1439 CAS.
- P. Sipos and T. Kiss, J. Chem. Soc., Dalton Trans., 1990, 2909 RSC.
- CrysAlisCCD CrysAlis RED, Oxford Diffraction Ltd., Version 1.171.33.42, release 29-05-2009 CrysAlis171, 2009.
- G. M. Sheldrick, Acta Crystallogr., Sect. A: Found. Crystallogr., 2008, 64, 112 CrossRef CAS PubMed.
- G. Bierman and H. Ziegler, Anal. Chem., 1986, 58, 536 CrossRef.
- J. Myrczek, Spectrosc. Lett., 1990, 23, 1027 CrossRef CAS.
- A. Wojciechowska, Z. Staszak, W. Bronowska, A. Pietraszko and M. Cieślak-Golonka, Polyhedron, 2001, 20, 2063 CrossRef CAS.
- A. Wojciechowska, D. Dobrzyńska and J. Janczak, Polyhedron, 2012, 47, 118 CrossRef CAS PubMed.
- D. Dobrzyńska, L. B. Jerzykiewicz, M. Duczmal and A. Wojciechowska, Polyhedron, 2011, 30, 2684 CrossRef PubMed.
- (a) O. Yamauchi, A. Odani and M. Takani, J. Chem. Soc., Dalton Trans., 2002, 3411 RSC; (b) T. Yajima, R. Takamido, Y. Shimazaki, A. Odani, Y. Nakabayashi and O. Yamauchi, Dalton Trans., 2007, 299 RSC; (c) O. Yamauchi, A. Odani and S. Hirota, Bull. Chem. Soc. Jpn., 2001, 74, 1525 CrossRef CAS; (d) M. Sabat, M. Jeżowska and H. Kozłowski, Inorg. Chim. Acta, 1979, 37, L511 CrossRef CAS; (e) H. Masuda, T. Sugimori, A. Odani and O. Yamauchi, Inorg. Chim. Acta, 1991, 180, 73 CrossRef CAS; (f) M. C. J. Wilce, D. M. Dooley, H. C. Freeman, J. M. Guss, H. Matsunami, W. S. McIntire, C. E. Ruggiero, K. Tanizawa and H. Yamaguchi, Biochemistry, 1997, 36, 16116 CrossRef CAS PubMed; (g) N. Ito, S. E. V. Phillips, C. Stevens, Z. B. Ogel, M. J. McPherson, J. N. S. Keen, K. D. Yadev and P. F. Knowles, Nature, 1991, 350, 87 CrossRef CAS PubMed; (h) H. C. Freeman, Adv. Protein Chem., 1967, 22, 257 CrossRef CAS.
- A. F. Wells, Structural Inorganic Chemistry, Oxford University Press, Oxford, U.K., 1975, p. 259 Search PubMed.
- A. W. Addison, T. N. Rao, J. Reedijk, J. Van Rijn and G. C. Verschoor, J. Chem. Soc., Dalton Trans., 1984, 1349 RSC.
- I. J. Bruno, J. C. Cole, P. R. Edgington, M. Kessler, C. F. Macrae, P. McCabe, S. Hem, J. Pearson and R. Taylor, Acta Crystallogr., Sect. B: Struct. Sci., 2002, 58, 389 CrossRef PubMed.
- S. A. Siddiqui, A. K. Pandey, A. Dwivedi, S. Jain and M. Neeraj, J. Chem. Pharm. Res., 2010, 2, 835 CAS.
- Y. Inokuchi, Y. Kobayashi, T. Ito and T. J. Ebata, J. Phys. Chem. A, 2007, 111, 3209 CrossRef CAS PubMed.
- J. Garcia-Tojal, A. Barcia-Orad, J. Serra, J. Pizarro, L. Lezama, M. Arriortua and T. J. Rojo, Inorg. Biochem., 1999, 75, 45 CrossRef CAS.
- A. Barth, Prog. Biophys. Mol. Biol., 2000, 74, 141 CrossRef CAS.
- N. Ohada, H. Masuda and O. Yamauchi, Inorg. Chim. Acta, 1999, 286, 37 CrossRef.
- D. N. Sathyanarayana, Electronic Absorption Spectroscopy and Related Techniques, Universities Press, 2001, p. 1 Search PubMed.
- G. Murphy, C. O'Sullivan, B. Murphy and B. Hathaway, Inorg. Chem., 1998, 37, 240 CrossRef CAS.
- G. Ondrejovic, A. Kotocova and D. Valigura, Chem. Pap., 2002, 56, 169 Search PubMed.
- D. Dobrzynska, J. Janczak, A. Wojciechowska and K. Helios, J. Mol. Struct., 2010, 973, 62 CrossRef CAS PubMed.
- A. K. Patra, T. Bhowmick, S. Ramakumar, M. Nethaji and A. R. Chakravarty, Dalton Trans., 2008, 6966 RSC.
- S. Y. New, X. Wu, S.-Q. Bai, L. L. Koh, T. S. A. Hor and F. Xue, CrystEngComm, 2011, 13, 4228 RSC.
- A. G. Griesbeck, J. Neudorfl and A. Kiff, Beilstein J. Org. Chem., 2011, 7, 518 CrossRef CAS PubMed.
- M. S. Attia, A. O. Youssef and A. A. Essawy, Anal. Methods, 2012, 4, 2323 RSC.
- (a) O. Yamauchi, K. Tsujide and A. Odani, J. Am. Chem. Soc., 1985, 107, 659 CrossRef CAS; (b) R. J. W. Hefford and L. D. Pettit, J. Chem. Soc., Dalton Trans., 1981, 1331 RSC; (c) D. L. Reger, A. Debreczeni, M. D. Smith, J. Jezierska and A. Ożarowski, Inorg. Chem., 2012, 51, 1068 CrossRef CAS PubMed.
- C. Desplanches, E. Ruiz, A. Rodriguez-Fortea and S. Alvarez, J. Am. Chem. Soc., 2002, 124, 5197 CrossRef CAS PubMed.
- S. Angelov, M. Drillon, E. Zhecheva, R. Stoyanova, M. Belaiche, A. Derory and A. Herr, Inorg. Chem., 1992, 31, 1514 CrossRef CAS.
- R. Georges, J. J. Borras-Almenar, E. Coronado, J. Curely and M. Drillon, Magnetic chains. An overview of the models, in Magnetism: Molecules to Materials. Models and Experiments, ed. J. S. Miller and M. Drillon, Wiley-VCH, 2001, p. 1 Search PubMed.
- J. D. Dunitz and A. Gavezzotti, Cryst. Growth Des., 2012, 12, 5873 CAS.
- G. A. Baker Jr, G. S. Rushbrooke and H. E. Gilbert, Phys. Rev., 1964, 135, A1272 CrossRef.
- O. Kahn, Molecular Magnetism, VCH Publishers, Inc., New York, 1993 Search PubMed.
- D. C. Johnston, M. Troyer, S. Miyahara, D. Lidsky, K. Ueda, M. Azuma, Z. Hiroi, M. Takano, M. Isobe, Y. Ueda, M. A. Korotin, V. I. Anisimov, A. V. Mahajan and J. L. Miller, 2000, arXiv:cond-mat/0001147.
- (a) B. Le Guennic, N. Ben Amor, D. Maynau and V. Robert, J. Chem. Theory Comput., 2009, 5, 1506 CrossRef CAS; (b) N. A. G. Bandeira and B. Le Guennic, J. Phys. Chem. A, 2012, 116, 3465 CrossRef CAS PubMed.
- (a) E. Colacio, J. P. Costes, R. Kivekäs, J. P. Laurent, J. Ruiz and M. Sundberg, Inorg. Chem., 1991, 30, 1475 CrossRef CAS; (b) E. Escriva, J. Server-Carrio, J. Garcia-Lozano, J. V. Folgado, F. Sapina and L. Lezama, Inorg. Chim. Acta, 1998, 279, 58 CrossRef CAS; (c) G. Carpinteyro-López, J. L. Alcántara-Flores, D. Ramírez-Rosales, R. Escudero, B. M. Cabrera-Vivas, S. Bernès, R. Zamorano-Ulloa and Y. Reyes-Ortega, ARKIVOC, 2008, 31 Search PubMed; (d) M. S. Ray, A. Ghosh, R. Battacharya, G. Mukhopadhyay, M. G. B. Drew and J. Ribas, Dalton Trans., 2004, 252 RSC.
- (a) W. Plass, A. Pohlmann and J. Rautengarten, Angew. Chem., Int. Ed., 2001, 40, 4207 CrossRef CAS; (b) J. Tang, J. Sánchez Costa, A. Golobič, B. Kozlečvar, A. Robertazzi, A. V. Vargiu, P. Gamez and J. Reedijk, Inorg. Chem., 2009, 48, 5473 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: X-ray crystallographic data in CIF format for 1, tables and supplementary figures. CCDC 980662. For ESI and crystallographic data in CIF or other electronic format see DOI: 10.1039/c4ra10717a |
| This journal is © The Royal Society of Chemistry 2014 |