Influence of vanadium compound coating on lithium-rich layered oxide cathode for lithium-ion batteries
Shi-Xuan Liaoa,
Chong-Heng Shenc,
Yan-Jun Zhonga,
Wan-Hong Yand,
Xia-Xing Shia,
Shan-Shan Peia,
Xiaodong Guoa,
Ben-He Zhonga,
Xing-Long Wang*a and
Heng Liu*b
aCollege of Chemical Engineering, Sichuan University, Chengdu 610065, China. E-mail: wangxl@scu.edu.cn
bCollege of Material Science and Engineering, Sichuan University, Chengdu 610064, China. E-mail: h_liu@scu.edu.cn
cSchool of Energy Research, State Key Lab of Physical Chemistry of Solid Surfaces, Xiamen University, Xiamen 361005, China
dDepartment of Materials Science and Engineering, National University of Defense Technology, Changsha 410073, China
First published on 21st October 2014
Abstract
A vanadium compound is applied as a coating material to improve the electrochemical performance of the lithium-rich layered oxide Li1.2Mn0.6Ni0.2O2. The physicochemical properties of the material before and after coating are characterized by scanning electron microscopy (SEM), powder X-ray diffraction (XRD), high-resolution transmission electron microscopy (HRTEM), energy dispersive spectrometry (EDS), X-ray photoelectron spectroscopy (XPS), and infrared spectroscopy (FT-IR). Results reveal that LiδV2O5 (δ is very small) is successfully coated on the as-prepared material, and the crystal properties of the powder have been modified after coating. The formation of the LiδV2O5 coating layer is a result of some Li-ions diffusing from the Li1.2Mn0.6Ni0.2O2 particle to the coating layer at the interface. The material before and after coating serve as the cathode for lithium-ion batteries and were investigated by galvanostatic measurements within a voltage range of 2.0–4.8 V (vs. Li/Li+). The initial coulombic efficiency (CE1) of Li1.2Mn0.6Ni0.2O2 is improved from 71.8% to 87.7% due to the LiδV2O5 coating layer, which can act as an insertion host to accept the lithium ions that could not be inserted back into the bulk lattice during the first discharge process. Additionally, the electrochemical performances (cycling performance and rate capability) of the modified Li1.2Mn0.6Ni0.2O2 are very superior to the pristine one. The significantly improved electrochemical performances are attributed primarily to: (i) the modified crystal properties after coating; (ii) the amelioration of the charge-transfer resistance after coating; (iii) the coating layer which can contribute to stabilizing the electrode surface by suppressing the side reactions between electrode and electrolyte.
1. Introduction
Rechargeable lithium-ion batteries are of great significance as power sources to realize energy storage from sustainable sources such as the wind and solar power.1 For such applications, high capacity, low cost and safe cathode materials are needed. Among the positive-electrode candidates used so far, lithium-rich layered oxides have attracted dramatic interest owing to their high discharge capacity (∼250 mA h g−1) and low cost.2–5 Lithium-rich cathode material can be formulated as xLi2MnO3·(1 − x)LiMO2 (0 < x < 1) or Li[MxLi(1/3 − x/3)Mn(2/3 − 2x/3)]O2 (0 < x < 1). (Note, the M in the above formulas is a transition metal element or any combination of transition metal elements, such as Ni, Co, Cr, Mn0.5Ni0.5 and Mn0.333Ni0.333Co0.333). In large-scale applications we should always avoid using the cobalt because cobalt is expensive, toxic and unfriendly to the environment.6 Here we select a cobalt-free lithium-rich composite Li1.2Mn0.6Ni0.2O2 (i.e., 0.5Li2MnO3·0.5LiMn0.5Ni0.5O2) as the research object. As reported previously, Li1.2Mn0.6Ni0.2O2 undergoes a large initial irreversible capacity loss with low coulombic efficiency (CE1) about 70–75% depending on the synthesis method.7–9 The large initial irreversible capacity loss (ICL) is predominantly caused by the irreversible removal of “Li2O” from the electrode in the initial charge process.9 This is an intractable problem that prohibits its use in practical application. Many efforts like surface modification with inert materials (such as, Al2O3,10 AlPO4 (ref. 11)) or with electro-active materials (such as LiNiPO4,12 LiMnPO4 (ref. 13)) have been found to lower the initial ICL and enhance the electrochemical performances of the lithium-rich materials. However, when utilized on Li1.2Mn0.6Ni0.2O2, few strategies (include mentioned above), if any, have meet the request.In the current work, vanadium compound is adopted to modify the surface of the lithium-rich composite Li1.2Mn0.6Ni0.2O2. SEM, TEM, XPS, FT-IR and XRD results demonstrate that LiδV2O5 is coated on the surface of particle successfully. Electrochemical estimations show that the CE1 of the electrode increases from 70.9 to 87.7%, the cycling performance and rate capability of the electrode are also improved manifestly after coating.
2. Experimental
2.1 Preparations of the pristine Li1.2Mn0.6Ni0.2O2 (PL), vanadium compound coated material (VCL) and pure coating material (VP)
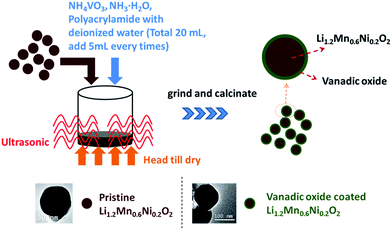
Stoichiometric amounts of nickel acetate tetrahydrate (NiC4H6O4·4H2O, analytical reagent AR) and manganese acetate tetrahydrate (MnC4H6O4·4H2O, AR) were dissolved in distilled water. Then add dropwise oxalic acid (excess 20%) into the mixed solution. The calculated lithium hydroxide (LiOH·H2O, AR, excess 3–5%) was added into the mixture subsequently. The mixture was continuously stirred at 95 °C until dry to obtain the precursor. The precursor was preheated at 400 °C for 1–5 h, and then calcined in air at 500 °C for 3–5 h, 750 °C for 2–5 h, 900 °C for 6–10 h to obtain the pristine powder.The solution (VOsol.) (∼20 mL) that contained ammonium metavanadate (NH4VO3, 0.3274 g), NH3·H2O, H2O and polyacrylamide (0.1260 g) was divided into several parts (e.g., we divided the solution into 4 parts), then, added them into the pristine powder (2.5198 g) and dried completely, again and again (about four cycles). Note that the two-phase mixture was treated under ultrasonic at 80 °C to form homogeneous dispersoid. The homogeneous dispersoid was stirred under ultrasonic until dry again and again, then ground and calcined at 400 °C for 2–4 h to obtain the final vanadium compound coated material (VCL). The approach was introduced in Scheme 1. Here, if the obtained coat layer is V2O5, it is 9.09 wt% of the composite.
The same solution (VOsol.) mentioned above was dried in a drying oven, then ground and calcined at 400 °C for 2–4 h to obtain the final pure coating material (VP).
2.2 Characterizations
The morphologies of PL and VCL were measured by scanning electron microscopy (SEM, S4800). The microstructures of the samples were conducted by mean of transmission electron microscope technique (TEM, JEM-2100). The coating layer elemental analysis of the VCL sample was examined by EDS. X-ray photoelectron spectroscopy (XPS, Quantum 2000 spectrometer, USA) measurements were performed to characterize the information on the surface of the particles. The FTIR spectra of PL and VCL were recorded on an IRAHinity-1 instrument. The crystalline phases of the powders are identified by powder X-ray diffraction (PANalytical X'pert Powder with Cu Kα radiation). The collected intensity data of XRD were analyzed by a Rietveld refinement program, and the refinement was performed using the GSAS software.142.3 Electrochemical measurements
Positive-electrodes were prepared by mixing the active material, carbon (acetylene black), and the binder in a weight ratio of 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 13
13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7. The slurry was then coated on Al-foil current collectors and was dried in a vacuum oven. The dried electrodes were subsequently assembled into CR2032 coin cells in glove box filled with argon using Li metal as counter electrode with a porous polypropylene film (Celgard 2300). The electrolyte consisted of a solution of 1 M LiPF6 in a mixture of ethylene carbonate (EC) and dimethyl carbonate (DMC; EC/DMC, 1
7. The slurry was then coated on Al-foil current collectors and was dried in a vacuum oven. The dried electrodes were subsequently assembled into CR2032 coin cells in glove box filled with argon using Li metal as counter electrode with a porous polypropylene film (Celgard 2300). The electrolyte consisted of a solution of 1 M LiPF6 in a mixture of ethylene carbonate (EC) and dimethyl carbonate (DMC; EC/DMC, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v). The coin cells are tested galvanostatically between 2.0 and 4.8 V (vs. Li/Li+) at room temperature. The electrochemical impedance spectroscopy (EIS) of the sample was recorded on a CHI660B electrochemical workstation over a frequency range from 100 kHz to 10 mHz. Before EIS measurements, both samples are cycled to the same state. Cycled at 0.05 C for once, and cycled at 0.1 C, 0.2 C, 0.5 C, 1 C for twice, respectively. Here, Li foil both served as the counter and reference electrodes during EIS measurements.
1, v/v). The coin cells are tested galvanostatically between 2.0 and 4.8 V (vs. Li/Li+) at room temperature. The electrochemical impedance spectroscopy (EIS) of the sample was recorded on a CHI660B electrochemical workstation over a frequency range from 100 kHz to 10 mHz. Before EIS measurements, both samples are cycled to the same state. Cycled at 0.05 C for once, and cycled at 0.1 C, 0.2 C, 0.5 C, 1 C for twice, respectively. Here, Li foil both served as the counter and reference electrodes during EIS measurements.
3. Results and discussion
The SEM micrographs of the PL particles and the VCL particles are depicted in Fig. 1a and c with the resolution of 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000×, respectively. The particles before and after coating nearly have no difference from the view of morphology, both are kind of polygonal prism with uniform distribution. This indicates that the coating treatment has little effect on the morphology due to the mild mix and low treat temperature. The distribution characteristics of the PL material and the VCL material are observed by the Laser Granulometric Analysis. The cumulative distribution curve and frequency distribution of the samples (Fig. 1b and d) are very similar with each other as we can see from the insets. The particle sizes of PL and VCL are detected with value of D10 = 10.491 μm, D50 = 26.580 μm, D90 = 51.349 μm and D10 = 11.738 μm, D50 = 28.914 μm, D90 = 56.603 μm, respectively. The calculated distribution width (D90 − D10)/D50 of the PL material and the VCL material is respectively 1.55 and 1.54, which suggests the differences about granulometric distribution between the samples are very small. In a nutshell, the coating treatment has very few effects on the morphology and distribution of the particles.
000×, respectively. The particles before and after coating nearly have no difference from the view of morphology, both are kind of polygonal prism with uniform distribution. This indicates that the coating treatment has little effect on the morphology due to the mild mix and low treat temperature. The distribution characteristics of the PL material and the VCL material are observed by the Laser Granulometric Analysis. The cumulative distribution curve and frequency distribution of the samples (Fig. 1b and d) are very similar with each other as we can see from the insets. The particle sizes of PL and VCL are detected with value of D10 = 10.491 μm, D50 = 26.580 μm, D90 = 51.349 μm and D10 = 11.738 μm, D50 = 28.914 μm, D90 = 56.603 μm, respectively. The calculated distribution width (D90 − D10)/D50 of the PL material and the VCL material is respectively 1.55 and 1.54, which suggests the differences about granulometric distribution between the samples are very small. In a nutshell, the coating treatment has very few effects on the morphology and distribution of the particles.
 | ||
| Fig. 1 SEM micrograph of (a) PL calcined at 900 °C and (c) VCL after coating treatment; particle distribution characteristics of (b) PL and (d) VCL. | ||
The microstructures of the PL sample and the VCL sample are displayed in Fig. 2a–f. Compared with PL, the VCL shows an coating layer with a certain thickness from the observation of the edge of the block in the HRTEM images. This implies successfully cover the as-synthetized powder Li1.2Mn0.6Ni0.2O2 with a coating layer. And the coating layer has partial crystalline phases as circled out in the Fig. 2f. The EDS estimation of the coating layer at ‘1#’ shows that the coating layer nearly only has vanadium but no nickel or manganese (V/Mn/Ni = 6.47![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.22
0.22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0).
0).
 | ||
| Fig. 2 HRTEM image of (a), (b) PL and (c), (d) VCL; (e) EDS spectrum of the coated layer at 1#; (f) the enlarged figure for the coating layer. | ||
To further find out the surface specific information of the coating layer, the pure coating material (VP) was prepared as presented in the Experimental section. The VP was characterized by the powder X-ray diffraction. As shown in Fig. 3a, we can find that this powder is V2O5 with well crystallinity, which is consistent with the previously report.15 Similarly, we speculate that the coating layer is V2O5 with well crystallinity. To prove our assumption, XPS was carried out. The prominent appearance of V2p reflections in the VCL material (Fig. 3b) demonstrates the surface with vanadium, and this is consistent with the EDS analysis. Fig. 3c reveals the high-resolution spectrum of the V2p region that observed in the VCL sample. The fitted spectra shows binding energy of V2p3/2 is 515.84, 516.38 and 516.95 eV, and the binding energy of V2p1/2 is 523.01 and 524.21 eV. The binding energy of V2p3/2 and V2p1/2 for V5+ is 517 and 525 eV, respectively.16 These indicative valence state of vanadium on the surface is less than +5. As all the calcinations are carried out in air, the oxygen deficiency will not occur. According to Pauling's rule of electroneutrality, the relatively lower oxidation state of vanadic species is probably a result of some Li-ions diffusing from the Li1.2Mn0.6Ni0.2O2 particle to the coating layer. Hence, the VCL sample can be regarded as Li1.2−δMn0.6Ni0.2O2–LiδV2O5. Here, δ is very small. At first, the coating layer is expected to be V2O5 with well crystallinity. Nevertheless, XPS and HRTEM results show that the coating layer is not really V2O5 and its phase is not complete crystalline phase, which suggest that the layer seems to be much influenced by the interface interaction during the coating treatment. Furthermore, FTIR was carried out to investigate the interface interaction. It is known that the V2O5 displays three major absorption peaks at 617, 827 and 1022 cm−1 in the FTIR spectrum.17 The characteristic peaks of the VCL material at 820, 965 and 1125 cm−1 are very different compare to the pristine sample (Fig. 3d). To a large extent the three peaks are associated with the vibration of the kind of vanadic oxide group, which confirms the significant existence of interface interaction and also indicates successfully covering the surface of the cathode with vanadium compound.
 | ||
| Fig. 3 (a) XRD pattern of the VP; (b) XPS spectra for PL and VCL; (c) V2p spectrum of VCL; (d) FIIR spectra for the PL and VCL. | ||
The experimental XRD patterns of the powder before and after coating are analyzed thanks to a Rietveld refinement. Fig. 4 exhibits the Rieltveld refinement results of the PL and the VCL. There is no great difference in the reflections for the samples. Nearly all of the reflections in XRD patterns of the samples can be well indexed as the α-NaFeO2 layered phase (hexagonal, R![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m).18 This indicates that the main crystal structure of the powder is not affected by coating.
m).18 This indicates that the main crystal structure of the powder is not affected by coating.
The lattice parameters of the powder before and after coating are given in Table 1. Slight changes of lattice parameters after coating are observed. The c/a ratio for the PL and the VCL material is 4.9881 and 4.9929, respectively, which are both higher than 4.899 for ideal cubic close stacking,19 suggesting more clear layered quality of the VCL material. In the Li1.2Mn0.6Ni0.2O2, a partial of Ni ions and Li ions interchange will occur due to the similar radius of Ni2+ and Li+ (rLi+ = 0.76 Å, rNi2+ = 0.69 Å).20 the extent of cation disorder between Li+ and Ni2+ in the structure of the sample can be estimated by intensity ratio of (003)H and (104)H reflections (I(003)/I(104)),21 the greater the ratio is, the less the disordering is. The intensity ratio I(003)/I(104) of VCL (1.55) is greater than that of PL (1.40), indicating that the level of cation disordering is lowered after coating. Distinctly, the crystal properties (lattice parameters, layered quality, cation disordering) have been modified after coating. The modifications are attributed largely to the interface interaction between the particle and coating layer, and these modifications may be of benefits for electrochemical performances of Li1.2Mn0.6Ni0.2O2 cathode.
| Sample | a/Å | c/Å | c/a | V/Å3 | Rwp | Rp |
|---|---|---|---|---|---|---|
| PL | 2.857(3) | 14.252(5) | 4.9881 | 100.77 | 1.74% | 1.39% |
| VCL | 2.858(6) | 14.272(8) | 4.9929 | 101.01 | 2.49% | 1.94% |
The Li1.2Mn0.6Ni0.2O2 undergoes a large initial ICL, which is predominantly related to the irreversible removal of “Li2O” from the electrode in the initial charge process.9 To help understand this, the possible ideal composition changes for Li1.2Mn0.6Ni0.2O2 during initial charge process are given in Fig. 5. When the pristine cathode (E0) is charged at a lower voltage <4.45 V, its composition changes through lithium extraction with the concomitant oxidation of Ni2+ to Ni4+ alone the red arrow from E0 until it reaches the Li2MnO3–Ni0.5Mn0.5O2 tie-line at E2, where the nickel is completely oxidized to Ni4+; this step can be represented by the following reaction (1); when charged to higher potential (>4.45 V), the composition changes from E2 to E4, with the accompanying activation of Li2MnO3-component. In this step, electrons are removed from the oxygen 2p band to form an unstable intermediate configuration with O22−, and followed by loss of O2 to stabilize the structure. In the process, oxygen vacancies are eliminated with transition metal ions and oxide ion rearrangements at the end of the initial charge. The oxygen vacancies elimination will lead to a decrease of lithium sites, and with every two oxygen vacancies eliminating, two lithium ion sites will be eliminated. The irreversible removal can be looked as “Li2O”. The “Li2O” cannot intercalate back into the host in the subsequent discharge process, therefore, huge capacity loss has been observed during the first cycle of Li1.2Mn0.6Ni0.2O2. The second step process can be represented by the reaction (2).
 | ||
| Fig. 5 Possible ideal composition changes for Li1.2Mn0.6Ni0.2O2 in the initial charge process (here, n, z, and x are determined by the xLi2−2zMnO3−z·(1 − x)Li1−nNi0.5Mn0.5O2). | ||
As shown in Fig. 6a, the PL electrode undergoes a large initial ICL of 81 mA h g−1 with coulombic efficiency of 71.81%. To suppress the capacity loss, we have introduced a vanadium compound to modify the surface of Li1.2Mn0.6Ni0.2O2 particle. After coating, the initial ICL and CE1 of the VCL sample are 36 mA h g−1 and 87.69%, respectively, indicating successful suppression of the capacity loss during the first cycle by coating. This improvement is ascribed to the LiδV2O5 coating layer can serve as an insertion host to accommodate the lithium ions that could not be inserted back into the host lattice during the first discharge process. This is further confirmed by the discharge curves of PL and VCL in Fig. 6a. The elliptical circle marked on the discharge curve (at around 3 V) of the VCL electrode, which is different from the discharge curve of the PL, suggests that some lithium ions inserted back into the coating layer. Moreover, the lithiation with LiδV2O5 is reversible. As shown in Fig. 6b, the similar discharge voltage plateau at around 3 V can be still observed after the initial cycle. This is compliant with the XPS studies that the coating layer is not pure V2O5 it is rather LiδV2O5.
The rate capabilities of PL and VCL are depicted in Fig. 7a. The discharge capacities can reach 262, 246, 230, 213 and 197 mA h g−1 for the VCL electrode at the test current rates of C/20 (10 mA g−1), C/10, C/5, C/2, and 1 C, respectively. However, the discharge capacities of the PL electrode are 207, 201, 186, 158 and 149 mA h g−1 at the rates of C/20, C/10, C/5, C/2 and 1 C, respectively. As illustrated in Fig. 7a, the VCL electrode manifests significantly advantages on rate capability. The discharge capacity at 1 C for VCL electrode is almost the same as the capacity at C/10 for the PL electrode (Fig. 7a). If the capacity retention of the electrode at C/20 is defined to 100%, the capacity retention of the VCL (PL) electrode at C/10, C/5, C/2 and 1 C is 93.9% (97.1%), 87.8% (89.8%), 81.3% (76.3%) and 75.2% (71.9%), respectively (Fig. 7b). It is obvious that the capacity retentions of the VCL electrode at high current rates are higher than those of the pristine one. The improved of the rate capability is due to the coating layer, which has reduced the charge-transfer resistance (Rct) as demonstrated in Fig. 7c. Because vanadic oxides are electric conductors, the vanadium compound coating layer can ameliorate the charge transfer from active particles to the current collector, causing a decrease in Rct. By comparing the diameter of the main semicircles, the Rct for PL is larger than that of VCL. The Rct increases greatly for PL after 18 cycles. On the other hand, small increase is observed for the VCL. Fig. 7d presents the cycle stability of the VCL electrode that after rate capability examination. A specific capacity of 181 mA h g−1 at 200 mA g−1 (1 C) after 50 cycles is obtained, which means 91.4% of the initial capacity is remained after cycling. High coulombic efficiency near 100% is reached during these cycles, which reveals good reversibility of the electrode.
There is no doubt that the electrochemical performances of Li1.2Mn0.6Ni0.2O2 have been improved after coating. Superficially, the observed improvements are due to the vanadium compound coating layer because vanadic oxides are known to be very reversible electrochemically with respect to Li/Li+. However, the electrochemical performances of VP are not good (Fig. 7). Consequently, the improvements are actually due to: (i) the modified crystal properties after coating; (ii) the coating layer can play a critical role in suppressing the side reactions by separating electrode from electrolyte.
4. Conclusions
SEM, HRTEM, EDS, XPS, FI-IR and XRD results have demonstrated that LiδV2O5 is well covered on the surface of the pristine powder. The crystal properties of the powder are modified after coating treatment. The electrochemical performances of the Li1.2Mn0.6Ni0.2O2 are successfully enhanced by coating with vanadium compound. As for rate capability, the discharge capacity at 1 C for coated electrode is almost the same as the capacity at C/10 for the pristine one. On the other hand, the specific initial discharge capacity of the coated electrode is approximately 1.32 times higher than that of the pristine one at the current density of 200 mA g−1 (1 C). Moreover, for the modified electrode, a good cycling stability is demonstrated with the initial specific discharge capacity of 197 mA h g−1 without any noticeable capacity fading over 50 cycles at 1 C rate. In summary, we have obtained a modified Li1.2Mn0.6Ni0.2O2 with high initial coulombic efficiency and excellent electrochemical performances.Acknowledgements
This work was supported by the Sichuan University Funds for Young Scientists (no. 2011SCU11081), the Research Fund for the Doctoral Program of Higher Education, the Ministry of Education (no. 20120181120103), and the Open Found of National Engineering Center for Phosphorus Chemical Industry (no. 2013LF1012).Notes and references
- M. Armand and J.-M. Tarascon, Nature, 2008, 451, 652 CrossRef CAS PubMed.
- C. S. Johnson, N. Li, C. Lefief, J. T. Vaghey and M. M. Thackeray, Chem. Mater., 2008, 20, 6095 CrossRef CAS.
- F. Wu, N. Li, Y. Su, H. Shou, L. Bao, W. Yang, L. Zhang, R. An and S. Chen, Adv. Mater., 2013, 25, 3722 CrossRef CAS PubMed.
- Z. Lu and J. R. Dahn, J. Electrochem. Soc., 2012, 149, A815 CrossRef PubMed.
- Y. J. Park, Y.-S. Hong, X. Wu, K. S. Ryu and S. H. Chang, J. Power Sources, 2004, 129, 288 CrossRef CAS PubMed.
- A. R. Armstrong and P. G. Bruce, Nature, 1996, 381, 499 CrossRef CAS.
- F. Cheng, Y. Xin, J. Chen, L. Lu, X. Zhang and H. Zhou, J. Mater. Chem. A, 2013, 1, 5301 CAS.
- Z. Q. Deng and A. Manthiram, J. Phys. Chem. C, 2011, 115, 7097 CAS.
- A. R. Armstrong, M. Holzapfel, P. Novak, C. S. Johnson, S.-H. Kang, M. M. Thackeray and P. G. Bruce, J. Am. Chem. Soc., 2006, 128, 8694 CrossRef CAS PubMed.
- J. Cho, Y. J. Kim, T.-J. Kim and B. Park, Chem. Mater., 2001, 13, 18 CrossRef CAS.
- Q. Y. Wang, J. Liu, A. V. Murugan and A. Manthiram, J. Mater. Chem., 2009, 19, 4965 RSC.
- S.-H. Kang and M. M. Thackeray, Electrochem. Commun., 2009, 11, 748 CrossRef CAS PubMed.
- Q. Q. Qiao, H. Z. Zhang, G. R. Li, S. H. Ye, C. W. Wang and X. P. Gao, J. Mater. Chem. A, 2013, 1, 5262 CAS.
- C.-H. Shen, Q. Wang, F. Fu, L. Huang, Z. Lin, S.-Y. Shen, H. Su, X.-M. Zheng, B.-B. Xu, J.-T. Li and S.-G. Sun, ACS Appl. Mater. Interfaces, 2014, 6, 5516 CAS.
- D. Chen, R. Yi, S. Chen, T. Xu, M. L. Gordin, D. Lv and D. Wang, Mater. Sci. Eng., B, 2014, 185, 7 CrossRef CAS PubMed.
- B. M. Reddy, B. Chowdhury, I. Ganesh, E. P. Reddy, T. C. Rojas and A. Fernández, J. Phys. Chem. B, 1998, 102, 10176 CrossRef CAS.
- W. Chen, L. Q. Mai, J. F. Peng, Q. Xu and Q. Y. Zhu, J. Mater. Sci., 2004, 39, 2625 CrossRef CAS.
- M. Park, J. Lee, W. Choi, D. Im, S. Doo and K. Park, J. Mater. Chem., 2010, 20, 7208 RSC.
- Y. Gao, M. V. Yakovleva and W. B. Ebner, Electrochem. Solid-State Lett., 1998, 1, 117 CrossRef CAS PubMed.
- S.-H. Kang, P. Kempgens, S. Greenbaum, A. J. Kropf, K. Amine and M. M. Thackeray, J. Mater. Chem., 2007, 17, 2069 RSC.
- K. M. Shaju, G. V. Subba Rao and B. V. R. Chowdari, Electrochim. Acta, 2002, 48, 145 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2014 |